Exploring the Correlation Between PEMF Therapy and Biomodulation
AUG 11, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEMF Therapy Background and Objectives
Pulsed Electromagnetic Field (PEMF) therapy has emerged as a promising non-invasive treatment modality in recent years, garnering attention for its potential to modulate biological processes. The technology harnesses the power of electromagnetic fields to influence cellular activity, offering a wide range of therapeutic applications. PEMF therapy's roots can be traced back to the mid-20th century, with early research focusing on bone healing and tissue regeneration.
As the field has evolved, researchers have uncovered a broader spectrum of potential benefits, including pain management, improved circulation, and enhanced cellular repair. The underlying principle of PEMF therapy is based on the concept that electromagnetic fields can interact with the body's own electromagnetic nature, influencing cellular behavior and physiological processes.
The development of PEMF technology has been marked by significant milestones, from rudimentary devices used in early experiments to sophisticated, precisely calibrated systems employed in modern clinical settings. Advancements in electronics and our understanding of bioelectromagnetics have led to more targeted and effective PEMF applications.
Current research aims to elucidate the mechanisms by which PEMF therapy influences biological systems at the molecular and cellular levels. Scientists are exploring how different frequencies, intensities, and waveforms of electromagnetic fields can be optimized for specific therapeutic outcomes. The goal is to develop more personalized and efficient PEMF protocols that can address a wide range of health conditions.
The correlation between PEMF therapy and biomodulation is a central focus of ongoing investigations. Biomodulation refers to the ability to modify biological processes through external stimuli, and PEMF therapy shows promise in this regard. Researchers are working to uncover how electromagnetic fields can modulate gene expression, protein synthesis, and cellular signaling pathways to promote healing and restore homeostasis.
As we look to the future, the objectives of PEMF therapy research are multifaceted. There is a push to establish standardized protocols and dosing guidelines to ensure consistent and reproducible results across different applications. Additionally, efforts are being made to integrate PEMF technology with other treatment modalities to create synergistic therapeutic approaches.
The ultimate aim is to position PEMF therapy as a mainstream treatment option, supported by robust scientific evidence and clinical validation. This involves conducting large-scale clinical trials, refining the technology for home use, and exploring novel applications in fields such as neurology, oncology, and regenerative medicine. By understanding the intricate relationship between electromagnetic fields and biological systems, researchers hope to unlock new possibilities for non-invasive, drug-free treatments that can improve quality of life for patients across a spectrum of health conditions.
As the field has evolved, researchers have uncovered a broader spectrum of potential benefits, including pain management, improved circulation, and enhanced cellular repair. The underlying principle of PEMF therapy is based on the concept that electromagnetic fields can interact with the body's own electromagnetic nature, influencing cellular behavior and physiological processes.
The development of PEMF technology has been marked by significant milestones, from rudimentary devices used in early experiments to sophisticated, precisely calibrated systems employed in modern clinical settings. Advancements in electronics and our understanding of bioelectromagnetics have led to more targeted and effective PEMF applications.
Current research aims to elucidate the mechanisms by which PEMF therapy influences biological systems at the molecular and cellular levels. Scientists are exploring how different frequencies, intensities, and waveforms of electromagnetic fields can be optimized for specific therapeutic outcomes. The goal is to develop more personalized and efficient PEMF protocols that can address a wide range of health conditions.
The correlation between PEMF therapy and biomodulation is a central focus of ongoing investigations. Biomodulation refers to the ability to modify biological processes through external stimuli, and PEMF therapy shows promise in this regard. Researchers are working to uncover how electromagnetic fields can modulate gene expression, protein synthesis, and cellular signaling pathways to promote healing and restore homeostasis.
As we look to the future, the objectives of PEMF therapy research are multifaceted. There is a push to establish standardized protocols and dosing guidelines to ensure consistent and reproducible results across different applications. Additionally, efforts are being made to integrate PEMF technology with other treatment modalities to create synergistic therapeutic approaches.
The ultimate aim is to position PEMF therapy as a mainstream treatment option, supported by robust scientific evidence and clinical validation. This involves conducting large-scale clinical trials, refining the technology for home use, and exploring novel applications in fields such as neurology, oncology, and regenerative medicine. By understanding the intricate relationship between electromagnetic fields and biological systems, researchers hope to unlock new possibilities for non-invasive, drug-free treatments that can improve quality of life for patients across a spectrum of health conditions.
Market Analysis for PEMF Devices
The PEMF (Pulsed Electromagnetic Field) therapy device market has shown significant growth in recent years, driven by increasing awareness of non-invasive treatment options and the rising prevalence of chronic diseases. The global PEMF therapy device market was valued at approximately $500 million in 2020 and is projected to reach $1.3 billion by 2028, growing at a CAGR of 12.5% during the forecast period.
The market for PEMF devices is segmented based on application, end-user, and region. In terms of application, the market is divided into pain management, bone growth, neurological disorders, and others. Pain management currently holds the largest market share due to the high prevalence of chronic pain conditions and the growing demand for non-pharmacological treatment options.
Geographically, North America dominates the PEMF device market, followed by Europe and Asia-Pacific. The United States, in particular, accounts for a significant portion of the market share due to its advanced healthcare infrastructure and high adoption rate of innovative medical technologies. However, the Asia-Pacific region is expected to witness the fastest growth during the forecast period, driven by increasing healthcare expenditure, growing awareness of alternative therapies, and improving access to medical devices in emerging economies like China and India.
The market is characterized by the presence of both established players and new entrants. Key companies in the PEMF device market include OMI PLC, Orthofix Medical Inc., Bemer Group, and Curatronic Ltd. These companies are focusing on product innovation, strategic partnerships, and geographical expansion to strengthen their market position.
Factors driving the growth of the PEMF device market include the increasing prevalence of chronic diseases, growing geriatric population, rising demand for non-invasive treatment options, and advancements in PEMF technology. Additionally, the expanding applications of PEMF therapy in sports medicine and rehabilitation are opening up new opportunities for market growth.
However, the market also faces challenges such as the high cost of PEMF devices, lack of awareness among patients and healthcare providers in some regions, and stringent regulatory requirements for medical device approval. Despite these challenges, the overall outlook for the PEMF device market remains positive, with ongoing research and development efforts expected to further expand the therapeutic applications of PEMF technology and drive market growth in the coming years.
The market for PEMF devices is segmented based on application, end-user, and region. In terms of application, the market is divided into pain management, bone growth, neurological disorders, and others. Pain management currently holds the largest market share due to the high prevalence of chronic pain conditions and the growing demand for non-pharmacological treatment options.
Geographically, North America dominates the PEMF device market, followed by Europe and Asia-Pacific. The United States, in particular, accounts for a significant portion of the market share due to its advanced healthcare infrastructure and high adoption rate of innovative medical technologies. However, the Asia-Pacific region is expected to witness the fastest growth during the forecast period, driven by increasing healthcare expenditure, growing awareness of alternative therapies, and improving access to medical devices in emerging economies like China and India.
The market is characterized by the presence of both established players and new entrants. Key companies in the PEMF device market include OMI PLC, Orthofix Medical Inc., Bemer Group, and Curatronic Ltd. These companies are focusing on product innovation, strategic partnerships, and geographical expansion to strengthen their market position.
Factors driving the growth of the PEMF device market include the increasing prevalence of chronic diseases, growing geriatric population, rising demand for non-invasive treatment options, and advancements in PEMF technology. Additionally, the expanding applications of PEMF therapy in sports medicine and rehabilitation are opening up new opportunities for market growth.
However, the market also faces challenges such as the high cost of PEMF devices, lack of awareness among patients and healthcare providers in some regions, and stringent regulatory requirements for medical device approval. Despite these challenges, the overall outlook for the PEMF device market remains positive, with ongoing research and development efforts expected to further expand the therapeutic applications of PEMF technology and drive market growth in the coming years.
Current PEMF Technology Landscape
The current PEMF (Pulsed Electromagnetic Field) technology landscape is characterized by a diverse range of devices and applications, reflecting the growing interest in this therapeutic approach. PEMF devices typically generate low-frequency electromagnetic fields, ranging from 1 Hz to 10,000 Hz, with varying intensities and waveforms. These devices are designed to deliver pulsed electromagnetic fields to specific areas of the body, aiming to promote cellular repair and regeneration.
In the medical field, PEMF technology has gained significant traction, particularly in orthopedics and pain management. FDA-approved PEMF devices are now available for treating non-union fractures and depression. These devices often utilize specific frequencies and intensities tailored to their intended therapeutic applications. For instance, bone healing applications typically use lower frequencies (15-30 Hz), while pain management may employ higher frequencies (60-100 Hz).
The consumer market has also seen a proliferation of PEMF devices, ranging from full-body mats to localized applicators. These devices often offer adjustable frequencies and intensities, allowing users to customize their treatments. Many of these consumer-grade devices operate at lower intensities compared to medical-grade equipment, focusing on general wellness and relaxation rather than specific medical conditions.
Recent technological advancements have led to the development of more sophisticated PEMF systems. These include devices with multiple coils for targeted therapy, programmable treatment protocols, and integration with mobile applications for user tracking and customization. Some advanced systems incorporate biofeedback mechanisms, adjusting the electromagnetic field in real-time based on the user's physiological responses.
The integration of PEMF technology with other therapeutic modalities is an emerging trend. Combination devices that incorporate PEMF with infrared therapy, vibration, or even virtual reality for pain management are entering the market. This convergence of technologies aims to enhance the overall therapeutic effect and provide a more comprehensive treatment approach.
In terms of form factors, there is a trend towards more portable and user-friendly designs. Wearable PEMF devices, such as braces and patches, allow for continuous treatment during daily activities. These devices often utilize rechargeable batteries and can be controlled via smartphone apps, improving accessibility and treatment adherence.
Research and development in PEMF technology are ongoing, with a focus on optimizing field parameters for specific conditions and understanding the underlying mechanisms of action. Studies are exploring the potential of PEMF in new areas such as neurological disorders, wound healing, and even cancer treatment, although many of these applications are still in experimental stages.
In the medical field, PEMF technology has gained significant traction, particularly in orthopedics and pain management. FDA-approved PEMF devices are now available for treating non-union fractures and depression. These devices often utilize specific frequencies and intensities tailored to their intended therapeutic applications. For instance, bone healing applications typically use lower frequencies (15-30 Hz), while pain management may employ higher frequencies (60-100 Hz).
The consumer market has also seen a proliferation of PEMF devices, ranging from full-body mats to localized applicators. These devices often offer adjustable frequencies and intensities, allowing users to customize their treatments. Many of these consumer-grade devices operate at lower intensities compared to medical-grade equipment, focusing on general wellness and relaxation rather than specific medical conditions.
Recent technological advancements have led to the development of more sophisticated PEMF systems. These include devices with multiple coils for targeted therapy, programmable treatment protocols, and integration with mobile applications for user tracking and customization. Some advanced systems incorporate biofeedback mechanisms, adjusting the electromagnetic field in real-time based on the user's physiological responses.
The integration of PEMF technology with other therapeutic modalities is an emerging trend. Combination devices that incorporate PEMF with infrared therapy, vibration, or even virtual reality for pain management are entering the market. This convergence of technologies aims to enhance the overall therapeutic effect and provide a more comprehensive treatment approach.
In terms of form factors, there is a trend towards more portable and user-friendly designs. Wearable PEMF devices, such as braces and patches, allow for continuous treatment during daily activities. These devices often utilize rechargeable batteries and can be controlled via smartphone apps, improving accessibility and treatment adherence.
Research and development in PEMF technology are ongoing, with a focus on optimizing field parameters for specific conditions and understanding the underlying mechanisms of action. Studies are exploring the potential of PEMF in new areas such as neurological disorders, wound healing, and even cancer treatment, although many of these applications are still in experimental stages.
Existing PEMF-Biomodulation Solutions
01 PEMF therapy for pain management and tissue healing
Pulsed Electromagnetic Field (PEMF) therapy is used for managing pain and promoting tissue healing. It involves applying electromagnetic fields to the body, which can stimulate cellular repair, reduce inflammation, and improve blood circulation. This non-invasive treatment is effective for various conditions, including musculoskeletal disorders and chronic pain.- PEMF therapy for pain management and tissue healing: Pulsed Electromagnetic Field (PEMF) therapy is used for managing pain and promoting tissue healing. It involves applying electromagnetic fields to the body, which can stimulate cellular repair, reduce inflammation, and improve blood circulation. This non-invasive treatment is effective for various conditions, including musculoskeletal disorders and chronic pain.
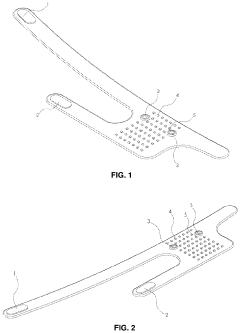
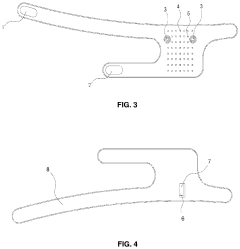
- PEMF devices for targeted biomodulation: Specialized PEMF devices are designed for targeted biomodulation of specific body areas or conditions. These devices can be customized to deliver precise electromagnetic pulses at varying frequencies and intensities. They may include wearable applicators, portable units, or larger clinical systems, allowing for flexible treatment options in different settings.
- PEMF therapy for neurological and psychiatric disorders: PEMF therapy shows promise in treating neurological and psychiatric disorders. The electromagnetic fields can modulate neural activity, potentially benefiting conditions such as depression, anxiety, and neurodegenerative diseases. This approach offers a non-pharmacological alternative or complement to traditional treatments, with fewer side effects.
- Combination of PEMF with other therapies: PEMF therapy is often combined with other treatment modalities to enhance overall therapeutic effects. This may include integration with physical therapy, acupuncture, or other forms of energy medicine. The synergistic approach can potentially improve outcomes for various health conditions and accelerate recovery processes.
- Advanced PEMF technology and control systems: Recent advancements in PEMF technology focus on improving control systems and treatment precision. This includes developing smart devices with programmable settings, real-time monitoring capabilities, and integration with mobile applications. These innovations aim to enhance treatment efficacy, user experience, and the ability to personalize therapy protocols.
02 PEMF devices for targeted biomodulation
Specialized PEMF devices are designed for targeted biomodulation of specific body areas or conditions. These devices can be customized to deliver precise electromagnetic pulses, optimizing the therapeutic effects for different applications such as wound healing, bone regeneration, and neurological disorders. The devices may include wearable or portable options for convenient use.Expand Specific Solutions03 Combination of PEMF with other therapies
PEMF therapy is often combined with other treatment modalities to enhance overall therapeutic outcomes. This may include integration with light therapy, ultrasound, or traditional medical treatments. The synergistic effects of combined therapies can lead to improved results in various medical applications, such as accelerated healing and enhanced pain relief.Expand Specific Solutions04 PEMF for cellular and molecular biomodulation
PEMF therapy is utilized for its effects on cellular and molecular levels. It can influence cell signaling pathways, gene expression, and protein synthesis. This biomodulation can lead to enhanced cellular function, improved metabolism, and increased production of beneficial substances like growth factors and neurotransmitters, contributing to overall health and well-being.Expand Specific Solutions05 Advanced PEMF systems with feedback and control mechanisms
Modern PEMF therapy systems incorporate advanced feedback and control mechanisms. These systems can adjust the electromagnetic field parameters based on real-time physiological data from the patient. This adaptive approach allows for personalized treatment protocols, optimizing the therapy's effectiveness and safety for individual needs and responses.Expand Specific Solutions
Key PEMF Industry Players
The field of PEMF therapy and biomodulation is in a growth phase, with increasing market size and technological advancements. The global market for PEMF devices is expanding, driven by growing applications in pain management, bone healing, and neurological disorders. Technologically, the sector is progressing from early-stage research to more sophisticated clinical applications. Companies like Medtronic, Neuroenhancement Lab, and SofPulse are at the forefront, developing innovative PEMF devices and therapies. Academic institutions such as the National University of Singapore and ETH Zurich are contributing to the fundamental research, while pharmaceutical giants like Novartis and Roche are exploring potential synergies with their existing product lines. The competitive landscape is diverse, with both established medical device companies and emerging biotech firms vying for market share.
Medtronic AF Luxembourg SARL
Technical Solution: Medtronic, through its subsidiary Medtronic AF Luxembourg SARL, has been exploring PEMF therapy in the context of neuromodulation and pain management. Their research focuses on integrating PEMF technology with existing neurostimulation devices to enhance therapeutic outcomes. Medtronic's approach involves using precisely controlled electromagnetic fields to modulate neural activity and influence pain signaling pathways. The company has developed prototype devices that combine traditional spinal cord stimulation with PEMF therapy, aiming to provide more comprehensive pain relief for patients with chronic conditions[8]. Preliminary studies have shown promising results in reducing pain scores and improving quality of life for patients with conditions such as failed back surgery syndrome and complex regional pain syndrome[9].
Strengths: Integration with established neurostimulation technologies, potential for synergistic effects in pain management. Weaknesses: Complex regulatory pathway for combined technologies, may require surgical implantation.
SofPulse, Inc.
Technical Solution: SofPulse, Inc. has developed an innovative PEMF therapy device that focuses on post-surgical pain management and accelerated healing. Their technology utilizes a unique waveform and frequency combination designed to optimize cellular response. The SofPulse device emits low-frequency pulsed electromagnetic fields in the range of 1-30 Hz, which have been shown to modulate inflammatory responses and promote tissue repair[4]. The device is compact and portable, allowing for continuous treatment during the critical post-operative period. Clinical studies have demonstrated significant reductions in pain medication usage and faster recovery times in patients using SofPulse therapy after various surgical procedures[5].
Strengths: Portable design for continuous treatment, specific focus on post-surgical applications. Weaknesses: Limited to low-frequency range, may not be as versatile for other conditions.
Core PEMF-Biomodulation Research
Flexible Photobiomodulation and Pulsed Electromagnetic Field Therapy Device
PatentPendingUS20230001222A1
Innovation
- A flexible wearable device that combines PEMF and PBM therapies, featuring a flexible substrate with electromagnetic coils and light-emitting diodes, controlled by a single module that can switch between pre-set frequency sequences, and is wirelessly enabled for remote control.
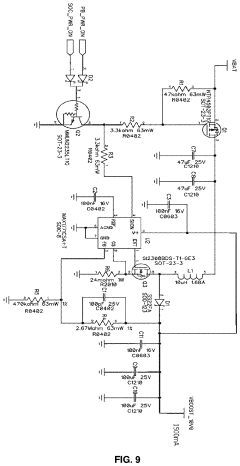
A pulsed electromagnetic field apparatus and method for generating frequencies
PatentWO2024127242A1
Innovation
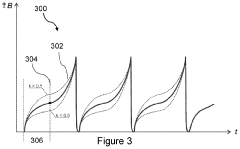
- A PEMF apparatus with a pulse generator and electromagnetic field generation means that uses modified sawtooth waveforms with pre-stress and relaxation periods, and quasi-sine signals with pulse width modulation, along with a feedback circuit for frequency stability and precision, and a bifilar antenna for scalar wave generation.
Regulatory Framework for PEMF Devices
The regulatory framework for PEMF (Pulsed Electromagnetic Field) devices is a complex and evolving landscape that plays a crucial role in ensuring the safety and efficacy of these therapeutic tools. In the United States, the Food and Drug Administration (FDA) is the primary regulatory body overseeing PEMF devices. These devices are generally classified as Class II medical devices, requiring a 510(k) premarket notification submission to demonstrate substantial equivalence to a legally marketed predicate device.
The FDA's regulatory approach to PEMF devices is based on their intended use and the level of risk associated with the device. For devices intended to treat specific medical conditions, manufacturers must provide clinical data demonstrating safety and effectiveness. However, for general wellness claims, such as promoting relaxation or improving sleep quality, the FDA typically exercises enforcement discretion, allowing these devices to be marketed without premarket review.
In the European Union, PEMF devices fall under the Medical Device Regulation (MDR), which came into full effect in May 2021. The MDR has introduced more stringent requirements for clinical evidence, post-market surveillance, and traceability. Manufacturers must obtain CE marking to market their PEMF devices in the EU, which involves demonstrating compliance with essential safety and performance requirements.
Globally, regulatory frameworks for PEMF devices vary significantly. Some countries have adopted regulations similar to those in the US or EU, while others have less developed regulatory systems. This variability creates challenges for manufacturers seeking to market their devices internationally, as they must navigate different regulatory requirements in each market.
The regulatory landscape for PEMF devices is also influenced by ongoing scientific research into the correlation between PEMF therapy and biomodulation. As new evidence emerges regarding the mechanisms of action and potential therapeutic applications of PEMF, regulatory bodies may update their guidelines and requirements. This dynamic relationship between scientific advancement and regulatory oversight underscores the importance of continued research in this field.
Manufacturers of PEMF devices must stay abreast of these regulatory developments and ensure ongoing compliance. This includes maintaining robust quality management systems, conducting post-market surveillance, and promptly reporting any adverse events. As the field of PEMF therapy continues to evolve, it is likely that regulatory frameworks will adapt to address new technologies, applications, and safety considerations, further shaping the landscape of this promising therapeutic modality.
The FDA's regulatory approach to PEMF devices is based on their intended use and the level of risk associated with the device. For devices intended to treat specific medical conditions, manufacturers must provide clinical data demonstrating safety and effectiveness. However, for general wellness claims, such as promoting relaxation or improving sleep quality, the FDA typically exercises enforcement discretion, allowing these devices to be marketed without premarket review.
In the European Union, PEMF devices fall under the Medical Device Regulation (MDR), which came into full effect in May 2021. The MDR has introduced more stringent requirements for clinical evidence, post-market surveillance, and traceability. Manufacturers must obtain CE marking to market their PEMF devices in the EU, which involves demonstrating compliance with essential safety and performance requirements.
Globally, regulatory frameworks for PEMF devices vary significantly. Some countries have adopted regulations similar to those in the US or EU, while others have less developed regulatory systems. This variability creates challenges for manufacturers seeking to market their devices internationally, as they must navigate different regulatory requirements in each market.
The regulatory landscape for PEMF devices is also influenced by ongoing scientific research into the correlation between PEMF therapy and biomodulation. As new evidence emerges regarding the mechanisms of action and potential therapeutic applications of PEMF, regulatory bodies may update their guidelines and requirements. This dynamic relationship between scientific advancement and regulatory oversight underscores the importance of continued research in this field.
Manufacturers of PEMF devices must stay abreast of these regulatory developments and ensure ongoing compliance. This includes maintaining robust quality management systems, conducting post-market surveillance, and promptly reporting any adverse events. As the field of PEMF therapy continues to evolve, it is likely that regulatory frameworks will adapt to address new technologies, applications, and safety considerations, further shaping the landscape of this promising therapeutic modality.
Safety and Efficacy Considerations
Safety and efficacy considerations are paramount when exploring the correlation between Pulsed Electromagnetic Field (PEMF) therapy and biomodulation. The application of electromagnetic fields for therapeutic purposes necessitates a thorough evaluation of potential risks and benefits to ensure patient well-being and treatment effectiveness.
One of the primary safety concerns in PEMF therapy is the potential for electromagnetic interference with implanted medical devices, such as pacemakers or insulin pumps. Patients with such devices may require special precautions or may be advised against PEMF therapy altogether. Additionally, the long-term effects of repeated exposure to electromagnetic fields, particularly at higher intensities, warrant careful investigation to rule out any adverse health impacts.
Dosage and exposure duration are critical factors in both safety and efficacy. While low-intensity PEMF therapy is generally considered safe for most individuals, higher intensities may pose increased risks. Establishing optimal treatment protocols that balance therapeutic effects with minimal side effects is essential for widespread clinical adoption.
The efficacy of PEMF therapy in biomodulation has shown promise in various applications, including pain management, bone healing, and tissue regeneration. However, the heterogeneity in study designs, treatment parameters, and outcome measures has led to inconsistent results across different research efforts. Standardization of protocols and rigorous, large-scale clinical trials are necessary to establish the true efficacy of PEMF therapy for specific conditions.
Another consideration is the potential for placebo effects in PEMF therapy studies. Double-blind, sham-controlled trials are crucial to differentiate between the physiological effects of electromagnetic fields and psychological factors that may influence perceived outcomes.
The mechanism of action by which PEMF therapy influences cellular processes and tissue function is not fully understood. Further research into the molecular and cellular responses to electromagnetic fields is needed to optimize treatment parameters and predict potential interactions with other therapies or medications.
Regulatory oversight and quality control in the manufacturing of PEMF devices are essential to ensure consistent and safe delivery of electromagnetic fields. Adherence to international safety standards and regular device calibration are necessary to maintain treatment integrity and patient safety.
In conclusion, while PEMF therapy shows promise in various biomodulation applications, careful consideration of safety protocols, standardized efficacy assessments, and ongoing research into mechanisms of action are crucial for its responsible development and integration into clinical practice.
One of the primary safety concerns in PEMF therapy is the potential for electromagnetic interference with implanted medical devices, such as pacemakers or insulin pumps. Patients with such devices may require special precautions or may be advised against PEMF therapy altogether. Additionally, the long-term effects of repeated exposure to electromagnetic fields, particularly at higher intensities, warrant careful investigation to rule out any adverse health impacts.
Dosage and exposure duration are critical factors in both safety and efficacy. While low-intensity PEMF therapy is generally considered safe for most individuals, higher intensities may pose increased risks. Establishing optimal treatment protocols that balance therapeutic effects with minimal side effects is essential for widespread clinical adoption.
The efficacy of PEMF therapy in biomodulation has shown promise in various applications, including pain management, bone healing, and tissue regeneration. However, the heterogeneity in study designs, treatment parameters, and outcome measures has led to inconsistent results across different research efforts. Standardization of protocols and rigorous, large-scale clinical trials are necessary to establish the true efficacy of PEMF therapy for specific conditions.
Another consideration is the potential for placebo effects in PEMF therapy studies. Double-blind, sham-controlled trials are crucial to differentiate between the physiological effects of electromagnetic fields and psychological factors that may influence perceived outcomes.
The mechanism of action by which PEMF therapy influences cellular processes and tissue function is not fully understood. Further research into the molecular and cellular responses to electromagnetic fields is needed to optimize treatment parameters and predict potential interactions with other therapies or medications.
Regulatory oversight and quality control in the manufacturing of PEMF devices are essential to ensure consistent and safe delivery of electromagnetic fields. Adherence to international safety standards and regular device calibration are necessary to maintain treatment integrity and patient safety.
In conclusion, while PEMF therapy shows promise in various biomodulation applications, careful consideration of safety protocols, standardized efficacy assessments, and ongoing research into mechanisms of action are crucial for its responsible development and integration into clinical practice.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!