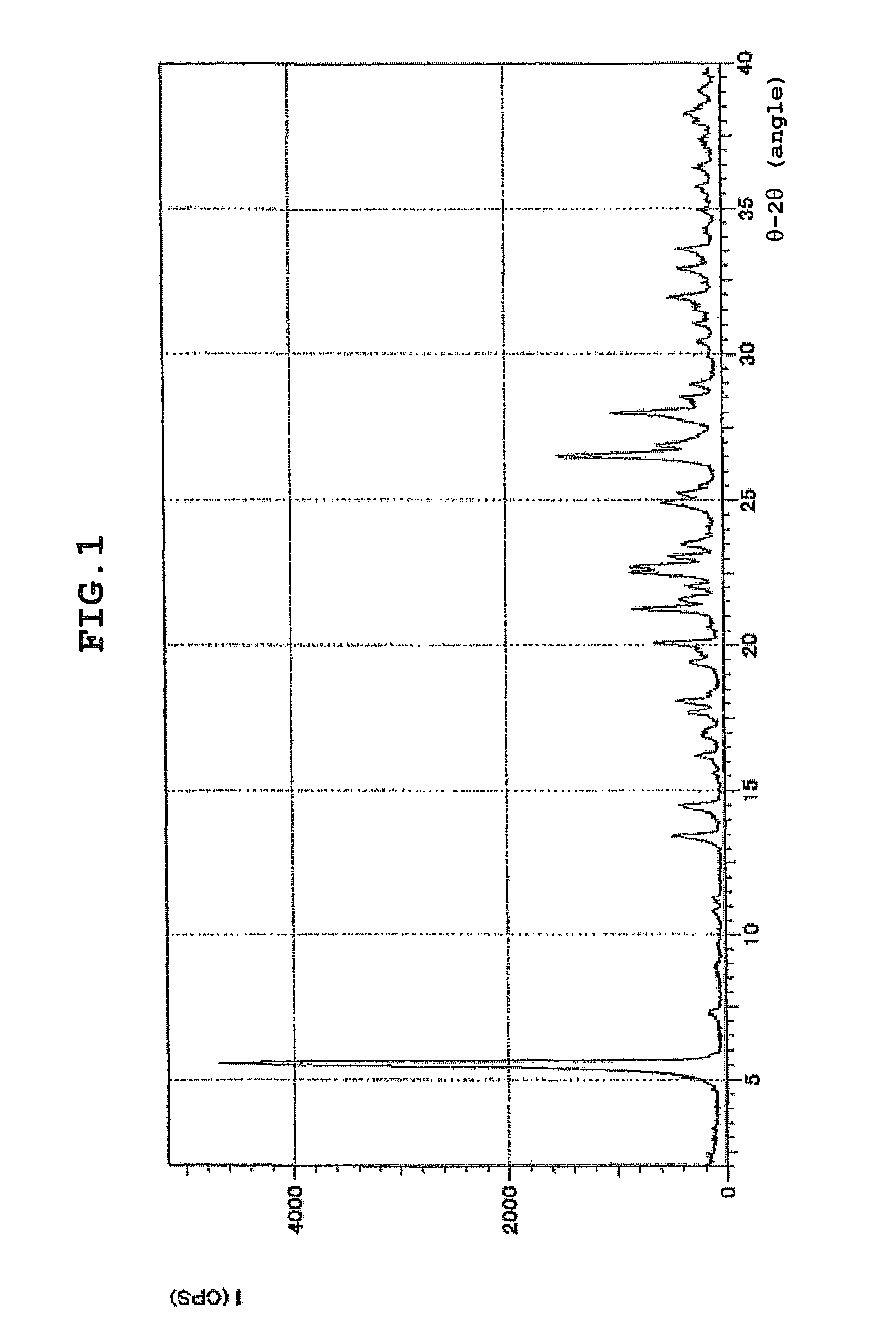
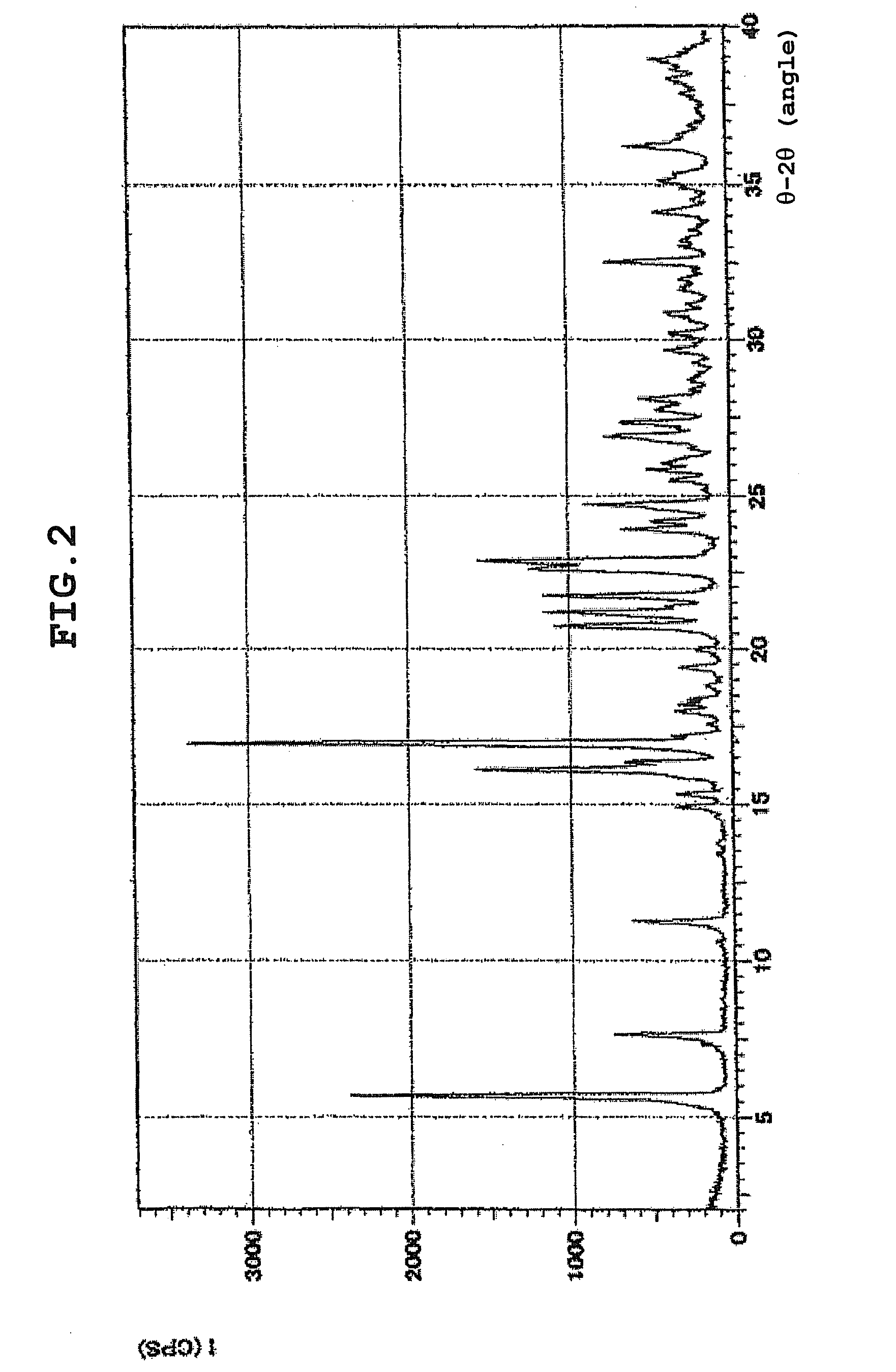
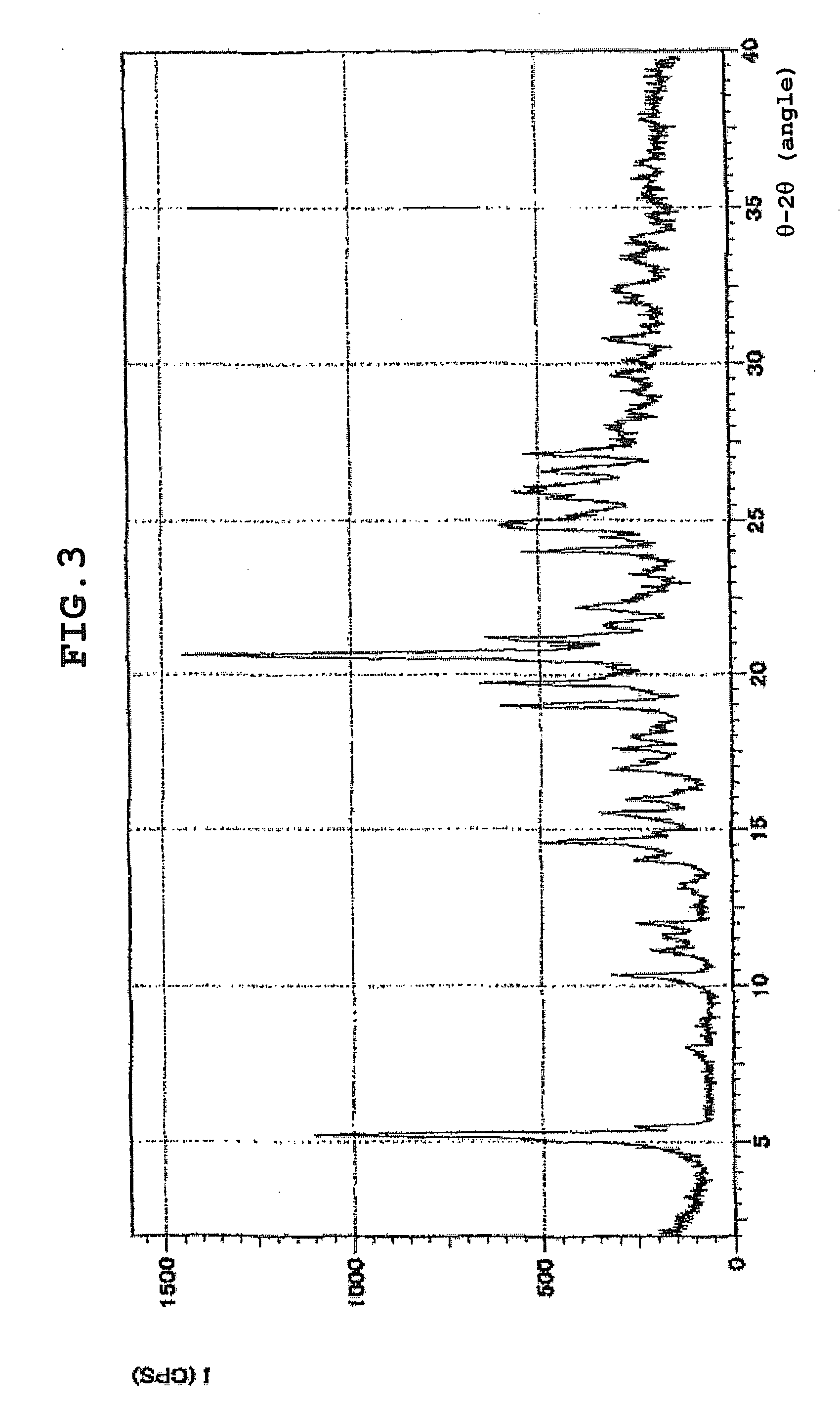
How To Identify Hydrate Structures Using X-ray Diffraction
FEB 27, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydrate Structure Analysis Background and XRD Objectives
Hydrate structures represent a critical class of crystalline materials where water molecules are incorporated into the crystal lattice alongside the host compound, fundamentally altering the physical, chemical, and pharmaceutical properties of the material. These structures have gained unprecedented importance across multiple industries, particularly in pharmaceutical development where hydrate formation can significantly impact drug solubility, bioavailability, stability, and manufacturing processes. The energy sector also recognizes hydrates as both opportunities and challenges, with natural gas hydrates representing vast energy reserves while simultaneously posing pipeline flow assurance risks.
The complexity of hydrate systems stems from their diverse structural arrangements and formation mechanisms. Water molecules can occupy specific crystallographic sites, form hydrogen-bonded networks, or exist in channels and cavities within the host structure. This structural diversity creates multiple hydrate phases for a single compound, each exhibiting distinct thermodynamic stability ranges and transformation behaviors under varying temperature and humidity conditions.
Traditional analytical approaches for hydrate characterization often fall short in providing comprehensive structural information. Thermal analysis techniques, while useful for quantifying water content, cannot distinguish between different hydrate phases or provide detailed structural arrangements. Spectroscopic methods offer molecular-level insights but lack the spatial resolution necessary for complete structural determination. Microscopy techniques provide morphological information but cannot reveal the internal crystal structure critical for understanding hydrate behavior.
X-ray diffraction emerges as the definitive analytical technique for hydrate structure identification due to its unique capability to probe crystalline arrangements at the atomic level. XRD provides unambiguous fingerprint patterns that enable precise phase identification, structural parameter determination, and quantitative analysis of hydrate systems. The technique's sensitivity to subtle structural differences allows discrimination between closely related hydrate phases that may appear identical using other analytical methods.
The primary objectives of employing XRD for hydrate structure analysis encompass several critical aspects. Phase identification represents the fundamental goal, enabling researchers to determine the specific hydrate form present and distinguish it from anhydrous phases or other hydrate stoichiometries. Structural characterization objectives include determining unit cell parameters, space group symmetry, and atomic positions, providing complete three-dimensional structural models essential for understanding property-structure relationships.
Quantitative analysis objectives focus on determining phase purity, hydrate stoichiometry, and transformation kinetics under controlled environmental conditions. These measurements are crucial for pharmaceutical development, where regulatory requirements demand precise characterization of drug substance polymorphic and hydrate forms. Additionally, XRD enables monitoring of hydrate stability and transformation processes, supporting formulation development and storage condition optimization.
The complexity of hydrate systems stems from their diverse structural arrangements and formation mechanisms. Water molecules can occupy specific crystallographic sites, form hydrogen-bonded networks, or exist in channels and cavities within the host structure. This structural diversity creates multiple hydrate phases for a single compound, each exhibiting distinct thermodynamic stability ranges and transformation behaviors under varying temperature and humidity conditions.
Traditional analytical approaches for hydrate characterization often fall short in providing comprehensive structural information. Thermal analysis techniques, while useful for quantifying water content, cannot distinguish between different hydrate phases or provide detailed structural arrangements. Spectroscopic methods offer molecular-level insights but lack the spatial resolution necessary for complete structural determination. Microscopy techniques provide morphological information but cannot reveal the internal crystal structure critical for understanding hydrate behavior.
X-ray diffraction emerges as the definitive analytical technique for hydrate structure identification due to its unique capability to probe crystalline arrangements at the atomic level. XRD provides unambiguous fingerprint patterns that enable precise phase identification, structural parameter determination, and quantitative analysis of hydrate systems. The technique's sensitivity to subtle structural differences allows discrimination between closely related hydrate phases that may appear identical using other analytical methods.
The primary objectives of employing XRD for hydrate structure analysis encompass several critical aspects. Phase identification represents the fundamental goal, enabling researchers to determine the specific hydrate form present and distinguish it from anhydrous phases or other hydrate stoichiometries. Structural characterization objectives include determining unit cell parameters, space group symmetry, and atomic positions, providing complete three-dimensional structural models essential for understanding property-structure relationships.
Quantitative analysis objectives focus on determining phase purity, hydrate stoichiometry, and transformation kinetics under controlled environmental conditions. These measurements are crucial for pharmaceutical development, where regulatory requirements demand precise characterization of drug substance polymorphic and hydrate forms. Additionally, XRD enables monitoring of hydrate stability and transformation processes, supporting formulation development and storage condition optimization.
Market Demand for Hydrate Structure Identification
The pharmaceutical industry represents the largest market segment for hydrate structure identification services, driven by the critical need to understand polymorphic forms of drug compounds. Pharmaceutical companies require precise hydrate characterization during drug development phases to ensure product stability, bioavailability, and regulatory compliance. The increasing complexity of modern drug molecules and the growing emphasis on personalized medicine have intensified the demand for advanced structural analysis capabilities.
Chemical manufacturing sectors, particularly specialty chemicals and materials science companies, constitute another significant market driver. These industries rely on hydrate structure identification to optimize production processes, control product quality, and develop new materials with enhanced properties. The expansion of advanced materials research, including metal-organic frameworks and crystalline porous materials, has created substantial opportunities for X-ray diffraction-based hydrate analysis services.
Academic and research institutions generate consistent demand for hydrate structure identification technologies, particularly in crystallography, materials science, and pharmaceutical research programs. Government-funded research initiatives and collaborative industry-academia projects continue to expand the market base, with increasing emphasis on fundamental research into hydrate formation mechanisms and structure-property relationships.
The growing regulatory requirements across multiple industries have significantly amplified market demand. Regulatory agencies increasingly require comprehensive structural characterization data for new chemical entities, pharmaceutical formulations, and industrial materials. This regulatory pressure has transformed hydrate structure identification from an optional analytical technique into a mandatory requirement for many product development pathways.
Emerging applications in energy storage, gas separation technologies, and environmental remediation are creating new market opportunities. The development of gas hydrates for energy applications and the need to understand hydrate behavior in industrial processes have expanded the traditional market boundaries beyond pharmaceutical and chemical sectors.
Geographic market expansion is evident in developing economies where pharmaceutical manufacturing and chemical industries are experiencing rapid growth. The establishment of new research facilities and quality control laboratories in these regions has created additional demand for hydrate structure identification capabilities, supported by increasing investment in analytical infrastructure and technical expertise development.
Chemical manufacturing sectors, particularly specialty chemicals and materials science companies, constitute another significant market driver. These industries rely on hydrate structure identification to optimize production processes, control product quality, and develop new materials with enhanced properties. The expansion of advanced materials research, including metal-organic frameworks and crystalline porous materials, has created substantial opportunities for X-ray diffraction-based hydrate analysis services.
Academic and research institutions generate consistent demand for hydrate structure identification technologies, particularly in crystallography, materials science, and pharmaceutical research programs. Government-funded research initiatives and collaborative industry-academia projects continue to expand the market base, with increasing emphasis on fundamental research into hydrate formation mechanisms and structure-property relationships.
The growing regulatory requirements across multiple industries have significantly amplified market demand. Regulatory agencies increasingly require comprehensive structural characterization data for new chemical entities, pharmaceutical formulations, and industrial materials. This regulatory pressure has transformed hydrate structure identification from an optional analytical technique into a mandatory requirement for many product development pathways.
Emerging applications in energy storage, gas separation technologies, and environmental remediation are creating new market opportunities. The development of gas hydrates for energy applications and the need to understand hydrate behavior in industrial processes have expanded the traditional market boundaries beyond pharmaceutical and chemical sectors.
Geographic market expansion is evident in developing economies where pharmaceutical manufacturing and chemical industries are experiencing rapid growth. The establishment of new research facilities and quality control laboratories in these regions has created additional demand for hydrate structure identification capabilities, supported by increasing investment in analytical infrastructure and technical expertise development.
Current XRD Limitations in Hydrate Characterization
X-ray diffraction faces significant resolution limitations when characterizing hydrate structures, particularly in distinguishing between closely related polymorphic forms. The inherent peak broadening effects in powder diffraction patterns often obscure subtle structural differences between hydrate phases, making it challenging to differentiate between monohydrates, dihydrates, and higher hydrates with similar lattice parameters. This limitation becomes particularly pronounced when analyzing pharmaceutical compounds where multiple hydrate forms may coexist.
Sample preparation presents another critical constraint in XRD-based hydrate characterization. Hydrated crystals are inherently sensitive to environmental conditions, and the standard grinding procedures required for powder diffraction can induce dehydration or phase transitions. The mechanical stress applied during sample preparation may convert stable hydrates to anhydrous forms or trigger polymorphic transformations, leading to misidentification of the original hydrate structure.
Temperature and humidity control during measurement represents a persistent challenge for accurate hydrate characterization. Most conventional XRD systems lack sophisticated environmental chambers capable of maintaining precise moisture levels throughout the measurement period. Hydrate samples can undergo dehydration under the X-ray beam due to heating effects or exposure to ambient laboratory conditions, resulting in time-dependent changes in diffraction patterns that complicate structural identification.
The quantitative analysis of hydrate mixtures poses additional difficulties due to overlapping diffraction peaks and varying scattering factors between hydrated and anhydrous phases. Standard Rietveld refinement methods often struggle to accurately determine the relative proportions of different hydrate forms, particularly when amorphous content is present or when dealing with poorly crystalline hydrate phases that produce weak, broad diffraction features.
Peak indexing and unit cell determination become problematic for complex hydrate structures with large unit cells or low symmetry space groups. The increased number of reflections and potential peak overlap in such systems can lead to ambiguous indexing solutions, making it difficult to establish definitive structural models. This limitation is exacerbated when dealing with channel hydrates or framework structures where water molecules occupy multiple crystallographic sites with varying occupancies.
Dynamic hydration processes present temporal challenges that conventional XRD measurements cannot adequately address. The kinetics of hydration and dehydration reactions often occur on timescales that require specialized in-situ measurement capabilities, which are not readily available in standard laboratory XRD setups, limiting the ability to monitor real-time structural changes during phase transitions.
Sample preparation presents another critical constraint in XRD-based hydrate characterization. Hydrated crystals are inherently sensitive to environmental conditions, and the standard grinding procedures required for powder diffraction can induce dehydration or phase transitions. The mechanical stress applied during sample preparation may convert stable hydrates to anhydrous forms or trigger polymorphic transformations, leading to misidentification of the original hydrate structure.
Temperature and humidity control during measurement represents a persistent challenge for accurate hydrate characterization. Most conventional XRD systems lack sophisticated environmental chambers capable of maintaining precise moisture levels throughout the measurement period. Hydrate samples can undergo dehydration under the X-ray beam due to heating effects or exposure to ambient laboratory conditions, resulting in time-dependent changes in diffraction patterns that complicate structural identification.
The quantitative analysis of hydrate mixtures poses additional difficulties due to overlapping diffraction peaks and varying scattering factors between hydrated and anhydrous phases. Standard Rietveld refinement methods often struggle to accurately determine the relative proportions of different hydrate forms, particularly when amorphous content is present or when dealing with poorly crystalline hydrate phases that produce weak, broad diffraction features.
Peak indexing and unit cell determination become problematic for complex hydrate structures with large unit cells or low symmetry space groups. The increased number of reflections and potential peak overlap in such systems can lead to ambiguous indexing solutions, making it difficult to establish definitive structural models. This limitation is exacerbated when dealing with channel hydrates or framework structures where water molecules occupy multiple crystallographic sites with varying occupancies.
Dynamic hydration processes present temporal challenges that conventional XRD measurements cannot adequately address. The kinetics of hydration and dehydration reactions often occur on timescales that require specialized in-situ measurement capabilities, which are not readily available in standard laboratory XRD setups, limiting the ability to monitor real-time structural changes during phase transitions.
Existing XRD Methods for Hydrate Structure Determination
01 Characterization of pharmaceutical hydrate polymorphs using X-ray diffraction
X-ray diffraction techniques are employed to identify and characterize different hydrate forms of pharmaceutical compounds. This method allows for the determination of crystal structures, unit cell parameters, and the degree of hydration in pharmaceutical materials. The technique is essential for understanding the solid-state properties of drug substances and ensuring consistent product quality.- Characterization of pharmaceutical hydrate polymorphs using X-ray diffraction: X-ray diffraction techniques are employed to identify and characterize different hydrate forms of pharmaceutical compounds. This method allows for the determination of crystal structures, unit cell parameters, and the degree of hydration in pharmaceutical materials. The technique is essential for understanding the solid-state properties of drug substances and ensuring consistent product quality.
- X-ray powder diffraction patterns for hydrate identification: Powder X-ray diffraction patterns serve as fingerprints for identifying specific hydrate forms of compounds. The characteristic diffraction peaks at specific angles enable differentiation between anhydrous forms, various hydrate states, and polymorphic variations. This analytical approach is crucial for quality control and regulatory compliance in pharmaceutical development.
- Single crystal X-ray diffraction for detailed hydrate structure determination: Single crystal X-ray diffraction provides comprehensive three-dimensional structural information about hydrate crystals, including the precise positions of water molecules within the crystal lattice. This technique reveals hydrogen bonding networks, molecular conformations, and the spatial arrangement of components in hydrated crystalline materials.
- Temperature-dependent X-ray diffraction studies of hydrate stability: Variable temperature X-ray diffraction experiments are used to investigate the thermal stability and dehydration behavior of hydrate structures. These studies track structural changes as hydrates lose water molecules, helping to determine storage conditions and predict shelf-life stability of hydrated pharmaceutical products.
- X-ray diffraction analysis of multi-component hydrate systems: Advanced X-ray diffraction methods are applied to analyze complex hydrate systems containing multiple components, including co-crystals, solvates, and mixed hydrate phases. These techniques enable the identification of phase compositions, structural relationships, and the interactions between different molecular species in hydrated crystalline materials.
02 Analysis of crystalline hydrate structures in active pharmaceutical ingredients
X-ray diffraction is utilized to analyze the crystalline structure of hydrated active pharmaceutical ingredients. This includes determining the water content, molecular arrangement, and stability of various hydrate forms. The structural information obtained helps in selecting the most suitable form for drug development and manufacturing processes.Expand Specific Solutions03 Determination of hydrate formation conditions and stability
X-ray diffraction methods are applied to study the conditions under which hydrates form and their stability profiles. This involves monitoring phase transitions, dehydration processes, and the effects of temperature and humidity on hydrate structures. Understanding these parameters is crucial for proper storage and formulation of pharmaceutical products.Expand Specific Solutions04 Identification of novel hydrate crystal forms
X-ray diffraction techniques enable the discovery and identification of new hydrate crystal forms of compounds. This includes solving crystal structures from powder or single crystal diffraction data, determining space groups, and establishing unique diffraction patterns that serve as fingerprints for each hydrate form. Such identification is important for patent protection and regulatory submissions.Expand Specific Solutions05 Quality control and batch consistency verification of hydrate materials
X-ray diffraction serves as a quality control tool to verify the consistency of hydrate forms across different production batches. The technique provides rapid, non-destructive analysis to confirm that materials maintain the desired crystalline structure and hydration state. This ensures product uniformity and compliance with pharmaceutical specifications.Expand Specific Solutions
Key Players in XRD Equipment and Hydrate Research
The X-ray diffraction hydrate structure identification field represents a mature analytical technology sector experiencing steady growth driven by pharmaceutical and materials science applications. The market demonstrates robust expansion, particularly in drug development where hydrate polymorphism critically impacts bioavailability and stability. Technology maturity varies significantly across players, with established analytical instrument manufacturers like Bruker AXS, Thermo Fisher Scientific, and Sigray leading advanced X-ray diffraction capabilities. Pharmaceutical giants including F. Hoffmann-La Roche, GlaxoSmithKline, and Merck Sharp & Dohme leverage these technologies for drug characterization, while specialized research institutions such as Japan Synchrotron Radiation Research Institute and Paul Scherrer Institut provide cutting-edge synchrotron facilities. The competitive landscape features a clear division between technology providers offering sophisticated instrumentation and end-users in pharmaceutical development, with emerging players like Chinese Academy of Science Guangzhou Energy Research Institute expanding applications into energy materials research, indicating growing diversification beyond traditional pharmaceutical applications.
Thermo Fisher Scientific (Ecublens) SARL
Technical Solution: Thermo Fisher Scientific offers integrated X-ray diffraction solutions through their ARL X'TRA and Equinox series for hydrate structure identification. Their approach combines powder diffraction analysis with advanced pattern matching algorithms and crystallographic databases specifically curated for hydrate phases. The company provides comprehensive workflow solutions including sample preparation protocols, automated measurement sequences, and data interpretation software that can identify hydrate structures by comparing experimental diffraction patterns against reference databases containing known hydrate polymorphs and their characteristic peak positions.
Strengths: Comprehensive analytical workflow solutions with robust database support and automated analysis capabilities. Weaknesses: Limited customization options for specialized hydrate research applications compared to dedicated research instruments.
Bruker AXS LLC
Technical Solution: Bruker AXS specializes in advanced X-ray diffraction systems specifically designed for structural analysis including hydrate identification. Their D8 ADVANCE series provides high-resolution powder diffraction capabilities with specialized software for phase identification and quantitative analysis of crystalline hydrates. The company's DIFFRAC.SUITE software package includes comprehensive databases and analytical tools for automated hydrate structure determination, enabling researchers to distinguish between different hydration states through characteristic diffraction patterns and lattice parameter variations.
Strengths: Industry-leading XRD instrumentation with specialized hydrate analysis capabilities and comprehensive software solutions. Weaknesses: High equipment costs and requires specialized training for optimal operation.
Core XRD Innovations in Hydrate Pattern Analysis
Crystalline form of a beta-lactamase inhibitor
PatentInactiveEP2970338A1
Innovation
- Development of crystalline solid forms, specifically hydrates like Hydrate 1, which exhibit low hygroscopicity and improved stability, characterized by specific X-ray powder diffraction patterns and thermal stability profiles, are obtained through controlled temperature and humidity conditions or solvent-based crystallization methods.
Salt of proline derivative, solvate thereof, and production method thereof
PatentInactiveUS8003790B2
Innovation
- Systematic characterization of multiple hydrate forms of proline derivative salts using specific X-ray diffraction peak patterns, providing distinct fingerprint identification for different hydrate stoichiometries (1.0 to 2.0 hydrates).
- Establishment of precise diffraction angle ranges (±0.2°) for reliable identification of different hydrate polymorphs, enabling robust quality control and form selection in pharmaceutical development.
- Development of standardized X-ray diffraction reference patterns for specific proline derivative hydrobromide hydrates, facilitating reproducible identification across different laboratories and instruments.
Sample Preparation Standards for Hydrate XRD Analysis
Sample preparation represents the most critical phase in hydrate structure identification through X-ray diffraction analysis, as improper handling can lead to phase transitions, dehydration, or structural degradation that compromises analytical accuracy. The establishment of rigorous preparation standards ensures reproducible results and maintains the integrity of hydrate crystal structures throughout the analytical process.
Temperature control during sample preparation constitutes the primary consideration for hydrate stability preservation. Most pharmaceutical and chemical hydrates exhibit thermal sensitivity, requiring preparation environments maintained between 15-25°C with relative humidity levels carefully controlled to prevent dehydration or over-hydration. Sample grinding, when necessary, should be performed using cryogenic conditions or minimal mechanical stress to avoid inducing phase transformations through frictional heating.
Particle size optimization plays a crucial role in obtaining high-quality diffraction patterns while preserving hydrate integrity. The recommended particle size range of 10-50 micrometers provides optimal balance between diffraction intensity and structural preservation. Excessive grinding can generate amorphous content or trigger dehydration through mechanical stress, while oversized particles may cause preferred orientation effects that distort peak intensities and complicate structural identification.
Sample mounting techniques must minimize exposure time to ambient conditions and prevent moisture exchange during analysis. Zero-background silicon holders or low-background quartz plates are preferred substrates, with sample thickness maintained at 0.5-1.0 mm to ensure adequate diffraction intensity while avoiding absorption effects. For highly moisture-sensitive hydrates, sealed capillary mounting or environmental chambers with controlled humidity become essential.
Contamination prevention protocols require dedicated sample handling tools and clean preparation environments to avoid introducing foreign crystalline phases that could interfere with hydrate identification. Cross-contamination between different hydrate forms or polymorphs can create complex diffraction patterns that obscure structural determination, necessitating thorough cleaning procedures between sample preparations.
Quality control measures include visual inspection for color changes or morphological alterations that may indicate phase transitions during preparation. Reference standard preparation under identical conditions enables validation of preparation protocols and ensures consistency across multiple analyses. Documentation of preparation conditions, including temperature, humidity, and timing, provides essential metadata for result interpretation and method validation.
Temperature control during sample preparation constitutes the primary consideration for hydrate stability preservation. Most pharmaceutical and chemical hydrates exhibit thermal sensitivity, requiring preparation environments maintained between 15-25°C with relative humidity levels carefully controlled to prevent dehydration or over-hydration. Sample grinding, when necessary, should be performed using cryogenic conditions or minimal mechanical stress to avoid inducing phase transformations through frictional heating.
Particle size optimization plays a crucial role in obtaining high-quality diffraction patterns while preserving hydrate integrity. The recommended particle size range of 10-50 micrometers provides optimal balance between diffraction intensity and structural preservation. Excessive grinding can generate amorphous content or trigger dehydration through mechanical stress, while oversized particles may cause preferred orientation effects that distort peak intensities and complicate structural identification.
Sample mounting techniques must minimize exposure time to ambient conditions and prevent moisture exchange during analysis. Zero-background silicon holders or low-background quartz plates are preferred substrates, with sample thickness maintained at 0.5-1.0 mm to ensure adequate diffraction intensity while avoiding absorption effects. For highly moisture-sensitive hydrates, sealed capillary mounting or environmental chambers with controlled humidity become essential.
Contamination prevention protocols require dedicated sample handling tools and clean preparation environments to avoid introducing foreign crystalline phases that could interfere with hydrate identification. Cross-contamination between different hydrate forms or polymorphs can create complex diffraction patterns that obscure structural determination, necessitating thorough cleaning procedures between sample preparations.
Quality control measures include visual inspection for color changes or morphological alterations that may indicate phase transitions during preparation. Reference standard preparation under identical conditions enables validation of preparation protocols and ensures consistency across multiple analyses. Documentation of preparation conditions, including temperature, humidity, and timing, provides essential metadata for result interpretation and method validation.
Database Development for Hydrate XRD Pattern Matching
The development of comprehensive databases for hydrate XRD pattern matching represents a critical infrastructure component for advancing automated hydrate structure identification. Current database initiatives focus on creating standardized repositories that contain reference diffraction patterns for known hydrate structures, enabling rapid comparison and identification of unknown samples through pattern matching algorithms.
Existing database frameworks primarily rely on the International Centre for Diffraction Data (ICDD) Powder Diffraction File (PDF) database, which contains limited hydrate-specific entries. Specialized hydrate databases are being developed to address this gap, incorporating crystallographic parameters, unit cell dimensions, space group information, and characteristic peak positions for various hydrate types including gas hydrates, pharmaceutical hydrates, and mineral hydrates.
The database architecture typically employs relational database management systems that store both experimental and calculated XRD patterns. Machine learning algorithms are increasingly integrated to enhance pattern recognition capabilities, utilizing neural networks and support vector machines to improve matching accuracy even when dealing with peak shifts, preferred orientation effects, or partial pattern matches.
Data standardization protocols ensure consistency across different measurement conditions and instruments. These protocols define peak intensity normalization methods, angular resolution requirements, and metadata standards including temperature, pressure, and sample preparation conditions. Quality control measures involve cross-validation with multiple experimental datasets and theoretical calculations using density functional theory.
Advanced database systems incorporate fuzzy matching algorithms that account for experimental variations and structural polymorphism in hydrates. These systems can handle peak broadening, background subtraction artifacts, and minor impurity phases that commonly occur in hydrate samples. Integration with crystallographic databases such as the Cambridge Structural Database enhances the predictive capabilities for novel hydrate structures.
Cloud-based database platforms are emerging to facilitate global data sharing and collaborative research efforts. These platforms support real-time updates, version control, and user contribution mechanisms that continuously expand the reference pattern library, ultimately improving the reliability and scope of hydrate identification capabilities.
Existing database frameworks primarily rely on the International Centre for Diffraction Data (ICDD) Powder Diffraction File (PDF) database, which contains limited hydrate-specific entries. Specialized hydrate databases are being developed to address this gap, incorporating crystallographic parameters, unit cell dimensions, space group information, and characteristic peak positions for various hydrate types including gas hydrates, pharmaceutical hydrates, and mineral hydrates.
The database architecture typically employs relational database management systems that store both experimental and calculated XRD patterns. Machine learning algorithms are increasingly integrated to enhance pattern recognition capabilities, utilizing neural networks and support vector machines to improve matching accuracy even when dealing with peak shifts, preferred orientation effects, or partial pattern matches.
Data standardization protocols ensure consistency across different measurement conditions and instruments. These protocols define peak intensity normalization methods, angular resolution requirements, and metadata standards including temperature, pressure, and sample preparation conditions. Quality control measures involve cross-validation with multiple experimental datasets and theoretical calculations using density functional theory.
Advanced database systems incorporate fuzzy matching algorithms that account for experimental variations and structural polymorphism in hydrates. These systems can handle peak broadening, background subtraction artifacts, and minor impurity phases that commonly occur in hydrate samples. Integration with crystallographic databases such as the Cambridge Structural Database enhances the predictive capabilities for novel hydrate structures.
Cloud-based database platforms are emerging to facilitate global data sharing and collaborative research efforts. These platforms support real-time updates, version control, and user contribution mechanisms that continuously expand the reference pattern library, ultimately improving the reliability and scope of hydrate identification capabilities.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!