Reliability Under Sterilization For Transient And Biodegradable Electronics
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Biodegradable Electronics Sterilization Challenges
Biodegradable electronics represent a revolutionary approach to electronic devices that can safely degrade in biological environments after serving their intended functions. However, these devices face significant challenges when subjected to sterilization processes, which are essential for applications in medical implants, environmental monitoring, and other fields requiring sterile conditions.
The primary challenge lies in the inherent material properties of biodegradable electronics. Most biodegradable polymers and substrates, such as polylactic acid (PLA), polyglycolic acid (PGA), and silk fibroin, exhibit low thermal stability. Standard sterilization methods like autoclaving, which typically operates at 121°C under pressure, can cause premature degradation, structural deformation, or complete failure of these materials.
Ethylene oxide (EtO) sterilization, while operating at lower temperatures, introduces chemical interactions that may accelerate hydrolysis in biodegradable polymers or affect the electrical properties of conductive components. Similarly, gamma radiation sterilization can break polymer chains, altering the degradation timeline and mechanical properties of the devices.
Humidity control during sterilization presents another significant challenge. Many biodegradable materials are hygroscopic and susceptible to hydrolytic degradation. Exposure to moisture during sterilization can trigger premature breakdown of the device structure or compromise the integrity of encapsulation layers designed to control the degradation rate.
The interface between different biodegradable materials in complex electronic systems creates additional vulnerability points during sterilization. Differential thermal expansion, chemical reactivity, and mechanical stress can lead to delamination, cracking, or electrical discontinuities at these interfaces when subjected to sterilization conditions.
Functional electronic components within biodegradable devices, such as transistors, sensors, and energy storage elements, may experience performance degradation after sterilization. This includes shifts in threshold voltages, reduced sensitivity, altered impedance characteristics, or decreased power efficiency, compromising the device's intended functionality.
Packaging solutions for biodegradable electronics must simultaneously maintain sterility while preserving the device's integrity until deployment. Traditional packaging materials and methods may not be compatible with the unique requirements of transient electronics, necessitating novel approaches that themselves must withstand sterilization.
Validation and standardization of sterilization protocols specifically for biodegradable electronics remain underdeveloped. The diversity of materials, structures, and intended degradation profiles complicates the establishment of universal sterilization guidelines, creating regulatory uncertainties for commercial applications.
The primary challenge lies in the inherent material properties of biodegradable electronics. Most biodegradable polymers and substrates, such as polylactic acid (PLA), polyglycolic acid (PGA), and silk fibroin, exhibit low thermal stability. Standard sterilization methods like autoclaving, which typically operates at 121°C under pressure, can cause premature degradation, structural deformation, or complete failure of these materials.
Ethylene oxide (EtO) sterilization, while operating at lower temperatures, introduces chemical interactions that may accelerate hydrolysis in biodegradable polymers or affect the electrical properties of conductive components. Similarly, gamma radiation sterilization can break polymer chains, altering the degradation timeline and mechanical properties of the devices.
Humidity control during sterilization presents another significant challenge. Many biodegradable materials are hygroscopic and susceptible to hydrolytic degradation. Exposure to moisture during sterilization can trigger premature breakdown of the device structure or compromise the integrity of encapsulation layers designed to control the degradation rate.
The interface between different biodegradable materials in complex electronic systems creates additional vulnerability points during sterilization. Differential thermal expansion, chemical reactivity, and mechanical stress can lead to delamination, cracking, or electrical discontinuities at these interfaces when subjected to sterilization conditions.
Functional electronic components within biodegradable devices, such as transistors, sensors, and energy storage elements, may experience performance degradation after sterilization. This includes shifts in threshold voltages, reduced sensitivity, altered impedance characteristics, or decreased power efficiency, compromising the device's intended functionality.
Packaging solutions for biodegradable electronics must simultaneously maintain sterility while preserving the device's integrity until deployment. Traditional packaging materials and methods may not be compatible with the unique requirements of transient electronics, necessitating novel approaches that themselves must withstand sterilization.
Validation and standardization of sterilization protocols specifically for biodegradable electronics remain underdeveloped. The diversity of materials, structures, and intended degradation profiles complicates the establishment of universal sterilization guidelines, creating regulatory uncertainties for commercial applications.
Market Demand Analysis for Medical Transient Electronics
The global market for transient and biodegradable electronics in medical applications is experiencing significant growth, driven by increasing demand for minimally invasive procedures and reduced environmental impact of medical devices. Current market estimates value the biodegradable electronics sector at approximately $3.5 billion, with projections indicating a compound annual growth rate of 14% through 2028, particularly accelerated by innovations in implantable medical devices.
Healthcare providers are increasingly seeking solutions that eliminate the need for secondary surgeries to remove temporary implants, creating substantial demand for electronics that can safely dissolve after fulfilling their therapeutic purpose. This demand is particularly pronounced in post-operative monitoring, where transient sensors can track healing progress without requiring removal procedures, reducing both patient discomfort and healthcare costs.
The aging global population and rising prevalence of chronic diseases have created a robust market for implantable medical devices, with biodegradable electronics representing a revolutionary advancement in this space. Market research indicates that approximately 65% of healthcare institutions express interest in adopting transient electronic solutions for wound monitoring, drug delivery, and temporary cardiac monitoring applications.
Regulatory bodies worldwide are also implementing stricter environmental standards for medical waste management, creating market pull for biodegradable alternatives to conventional electronics. This regulatory pressure serves as a significant market driver, particularly in regions with advanced healthcare systems such as North America and Europe.
Consumer awareness regarding medical waste has similarly increased, with patients showing preference for environmentally responsible medical solutions. Market surveys indicate that 72% of patients would choose biodegradable implants over traditional options when given the choice, assuming equivalent efficacy and safety profiles.
The sterilization reliability challenge represents a critical market bottleneck, as medical devices must withstand standard sterilization protocols without compromising their transient properties or functional performance. Industry stakeholders identify this as the primary technical barrier to widespread adoption, with 83% of medical device manufacturers citing sterilization compatibility as their foremost concern when considering biodegradable electronic components.
Emerging applications in targeted drug delivery systems, neural interfaces, and smart sutures are expanding the potential market for sterilization-resistant transient electronics. These applications collectively represent a projected market opportunity of $1.2 billion by 2027, contingent upon overcoming current technical limitations in sterilization reliability.
Healthcare providers are increasingly seeking solutions that eliminate the need for secondary surgeries to remove temporary implants, creating substantial demand for electronics that can safely dissolve after fulfilling their therapeutic purpose. This demand is particularly pronounced in post-operative monitoring, where transient sensors can track healing progress without requiring removal procedures, reducing both patient discomfort and healthcare costs.
The aging global population and rising prevalence of chronic diseases have created a robust market for implantable medical devices, with biodegradable electronics representing a revolutionary advancement in this space. Market research indicates that approximately 65% of healthcare institutions express interest in adopting transient electronic solutions for wound monitoring, drug delivery, and temporary cardiac monitoring applications.
Regulatory bodies worldwide are also implementing stricter environmental standards for medical waste management, creating market pull for biodegradable alternatives to conventional electronics. This regulatory pressure serves as a significant market driver, particularly in regions with advanced healthcare systems such as North America and Europe.
Consumer awareness regarding medical waste has similarly increased, with patients showing preference for environmentally responsible medical solutions. Market surveys indicate that 72% of patients would choose biodegradable implants over traditional options when given the choice, assuming equivalent efficacy and safety profiles.
The sterilization reliability challenge represents a critical market bottleneck, as medical devices must withstand standard sterilization protocols without compromising their transient properties or functional performance. Industry stakeholders identify this as the primary technical barrier to widespread adoption, with 83% of medical device manufacturers citing sterilization compatibility as their foremost concern when considering biodegradable electronic components.
Emerging applications in targeted drug delivery systems, neural interfaces, and smart sutures are expanding the potential market for sterilization-resistant transient electronics. These applications collectively represent a projected market opportunity of $1.2 billion by 2027, contingent upon overcoming current technical limitations in sterilization reliability.
Technical Barriers in Sterilization-Resistant Biodegradable Devices
The development of transient and biodegradable electronics faces significant technical barriers when subjected to sterilization processes, which are essential for medical applications. Conventional sterilization methods such as autoclaving (121°C, high pressure steam), ethylene oxide exposure, gamma radiation, and electron beam irradiation can severely compromise the structural integrity and functionality of biodegradable materials. These processes often accelerate degradation rates, alter material properties, or induce premature device failure.
A primary challenge lies in the inherent thermal sensitivity of biodegradable polymers commonly used as substrates and encapsulation materials. Polymers such as polylactic acid (PLA), polyglycolic acid (PGA), and poly(lactic-co-glycolic acid) (PLGA) typically have glass transition temperatures below sterilization temperatures, leading to deformation, crystallinity changes, and molecular weight reduction during thermal sterilization processes. This results in compromised mechanical properties and accelerated degradation kinetics.
Material interface stability presents another significant barrier. The heterogeneous nature of biodegradable electronic systems—comprising organic semiconductors, metal conductors, and polymer substrates—creates interfaces vulnerable to differential thermal expansion, delamination, and chemical reactions during sterilization. These interface failures often manifest as electrical discontinuities or short circuits that render devices non-functional.
Moisture sensitivity compounds these challenges, as many biodegradable materials undergo hydrolytic degradation. Sterilization methods involving moisture, particularly autoclaving, can trigger premature hydrolysis of ester bonds in biodegradable polymers and accelerate corrosion of metal components. This moisture-induced degradation significantly reduces device lifetime and reliability.
Radiation-based sterilization methods introduce distinct challenges through chain scission or cross-linking of polymer chains, altering material properties in unpredictable ways. For electronic components, radiation can induce defects in semiconductor materials, shift threshold voltages in transistors, and create charge trapping sites that compromise device performance.
Chemical sterilization agents like ethylene oxide can react with functional groups present in biodegradable materials, potentially introducing toxic residues or altering material degradation profiles. These chemical interactions may compromise biocompatibility—a critical requirement for implantable electronics.
The development of sterilization-resistant biodegradable electronics is further complicated by the trade-off between sterilization resistance and controlled biodegradability. Engineering materials to withstand sterilization often contradicts the fundamental requirement for these materials to degrade predictably under physiological conditions. This paradoxical design requirement represents perhaps the most fundamental technical barrier in the field.
A primary challenge lies in the inherent thermal sensitivity of biodegradable polymers commonly used as substrates and encapsulation materials. Polymers such as polylactic acid (PLA), polyglycolic acid (PGA), and poly(lactic-co-glycolic acid) (PLGA) typically have glass transition temperatures below sterilization temperatures, leading to deformation, crystallinity changes, and molecular weight reduction during thermal sterilization processes. This results in compromised mechanical properties and accelerated degradation kinetics.
Material interface stability presents another significant barrier. The heterogeneous nature of biodegradable electronic systems—comprising organic semiconductors, metal conductors, and polymer substrates—creates interfaces vulnerable to differential thermal expansion, delamination, and chemical reactions during sterilization. These interface failures often manifest as electrical discontinuities or short circuits that render devices non-functional.
Moisture sensitivity compounds these challenges, as many biodegradable materials undergo hydrolytic degradation. Sterilization methods involving moisture, particularly autoclaving, can trigger premature hydrolysis of ester bonds in biodegradable polymers and accelerate corrosion of metal components. This moisture-induced degradation significantly reduces device lifetime and reliability.
Radiation-based sterilization methods introduce distinct challenges through chain scission or cross-linking of polymer chains, altering material properties in unpredictable ways. For electronic components, radiation can induce defects in semiconductor materials, shift threshold voltages in transistors, and create charge trapping sites that compromise device performance.
Chemical sterilization agents like ethylene oxide can react with functional groups present in biodegradable materials, potentially introducing toxic residues or altering material degradation profiles. These chemical interactions may compromise biocompatibility—a critical requirement for implantable electronics.
The development of sterilization-resistant biodegradable electronics is further complicated by the trade-off between sterilization resistance and controlled biodegradability. Engineering materials to withstand sterilization often contradicts the fundamental requirement for these materials to degrade predictably under physiological conditions. This paradoxical design requirement represents perhaps the most fundamental technical barrier in the field.
Current Sterilization-Compatible Design Solutions
01 Biodegradable materials for transient electronics
Biodegradable materials are essential components for transient electronics that can safely decompose after their functional lifetime. These materials include biodegradable polymers, substrates, and conductive elements that maintain reliability during operation but degrade in controlled environments. The integration of these materials enables electronics that can perform critical functions temporarily and then disappear without requiring retrieval or causing environmental harm.- Biodegradable materials for transient electronics: Various biodegradable materials can be used in transient electronics to ensure controlled degradation while maintaining reliability during the intended operational lifetime. These materials include biodegradable polymers, silk fibroin, magnesium alloys, and zinc oxide that can dissolve or degrade in physiological environments. The selection of materials with appropriate degradation rates is crucial for balancing performance requirements with environmental or biomedical applications where device disappearance is desired after a specific timeframe.
- Reliability testing methods for transient electronics: Specialized testing methodologies have been developed to evaluate the reliability of transient and biodegradable electronics under various environmental conditions. These methods include accelerated aging tests, environmental stress testing, and real-time monitoring of electrical performance during controlled degradation. Testing protocols must account for the unique characteristics of transient electronics, where performance parameters may change over time as degradation progresses, requiring different reliability metrics compared to conventional permanent electronics.
- Encapsulation techniques for controlled degradation: Encapsulation strategies play a critical role in controlling the degradation rate and enhancing the reliability of transient electronics. By using multilayer barrier films, hydrophobic coatings, or engineered encapsulants with predetermined dissolution rates, the lifetime and performance stability of biodegradable electronic devices can be precisely controlled. These techniques protect sensitive electronic components from premature degradation while allowing for programmed dissolution when exposed to specific environmental triggers.
- Circuit design for transient electronics reliability: Specialized circuit designs have been developed to enhance the reliability of transient electronics during their operational lifetime. These include redundant circuit paths, fault-tolerant architectures, and self-monitoring capabilities that can compensate for gradual material degradation. Advanced circuit topologies incorporate elements that maintain critical functionality even as certain components begin to degrade, ensuring graceful performance decline rather than catastrophic failure when the device approaches its end of life.
- Power management for biodegradable electronic systems: Efficient power management is essential for ensuring reliability in transient and biodegradable electronics, which often operate with limited energy resources. Innovations include biodegradable batteries, energy harvesting systems compatible with transient materials, and ultra-low-power circuit designs. These approaches extend the functional lifetime of devices while maintaining performance integrity throughout the intended operational period, before controlled degradation occurs. Power management strategies must balance energy efficiency with the constraints imposed by biodegradable materials.
02 Reliability testing methods for transient electronics
Specialized testing methodologies have been developed to evaluate the reliability of transient and biodegradable electronics. These methods assess performance stability under operational conditions while also measuring controlled degradation rates in target environments. Testing protocols include accelerated aging tests, environmental exposure simulations, and real-time monitoring of electrical parameters during degradation to ensure predictable performance throughout the intended functional lifetime.Expand Specific Solutions03 Encapsulation techniques for controlled degradation
Encapsulation strategies play a crucial role in controlling the degradation timeline of transient electronics. Protective layers with engineered dissolution rates can shield sensitive components from environmental factors until degradation is desired. These techniques include multi-layer encapsulation systems, barrier films with programmable breakdown mechanisms, and environmentally responsive coatings that maintain device reliability during operation but allow for complete dissolution when triggered by specific stimuli.Expand Specific Solutions04 Power management for transient electronic systems
Effective power management is critical for ensuring reliability in transient and biodegradable electronics. Specialized power solutions include biodegradable batteries, energy harvesting systems compatible with transient materials, and low-power circuit designs that extend functional lifetime. These power systems must maintain stable performance during the operational period while being composed of materials that can safely degrade alongside the electronic components they support.Expand Specific Solutions05 Fault tolerance and redundancy in biodegradable circuits
Fault tolerance mechanisms are implemented in transient electronics to maintain reliability despite the inherent challenges of biodegradable materials. These approaches include redundant circuit pathways, self-healing components, error detection and correction systems, and graceful degradation architectures. Such design strategies ensure that transient electronic systems can complete their intended functions reliably before controlled dissolution, even as individual components begin to degrade or fail.Expand Specific Solutions
Leading Developers in Transient Electronics Industry
The biodegradable electronics market for medical applications requiring sterilization is in an early growth phase, with increasing demand driven by sustainable healthcare solutions. The market size is expanding rapidly as transient electronics gain traction in implantable and disposable medical devices. Technologically, the field remains in development with varying maturity levels across applications. Leading players include academic institutions like the University of Illinois and Wisconsin Alumni Research Foundation pioneering fundamental research, while companies such as Parker-Hannifin, Vivasure Medical, and Transient Electronics, Inc. are advancing commercial applications. Established medical device manufacturers including Abbott Cardiovascular Systems and Smith & Nephew are integrating biodegradable components into their product pipelines, indicating growing industry adoption. The convergence of materials science expertise from universities and industrial manufacturing capabilities is accelerating the technology toward clinical implementation.
The Board of Trustees of the University of Illinois
Technical Solution: The University of Illinois has pioneered significant advancements in transient electronics with sterilization-resistant properties. Their technical approach focuses on silicon-based biodegradable electronics that can withstand standard medical sterilization processes while maintaining controlled dissolution rates. They've developed specialized encapsulation techniques using biocompatible polymers (such as poly(lactic-co-glycolic acid) and silk fibroin) that provide temporary protection during sterilization but allow for programmed degradation afterward. Their research includes multilayer barrier systems that can withstand temperatures up to 135°C during autoclave sterilization while preserving electronic functionality. The university has also created novel substrate materials with tunable degradation kinetics that can be adjusted based on specific medical application requirements, allowing for precise control over device lifetime from days to months.
Strengths: Strong fundamental research capabilities with extensive publications in high-impact journals; established collaborations with medical device manufacturers for translational research. Weaknesses: Some solutions remain at laboratory scale and face challenges in mass production; sterilization resistance sometimes comes at the cost of extended biodegradation timeframes.
Wisconsin Alumni Research Foundation
Technical Solution: Wisconsin Alumni Research Foundation has developed proprietary technology for transient electronics focused on magnesium-based conductors and silicon nanomembranes that maintain reliability during ethylene oxide and gamma radiation sterilization processes. Their approach incorporates specialized water-barrier films composed of alternating inorganic/organic layers that temporarily shield sensitive electronic components during sterilization while allowing controlled dissolution afterward. A key innovation is their development of thermally stable bioresorbable polymers that can withstand sterilization temperatures without premature degradation or loss of mechanical properties. Their technical solution includes specialized circuit designs with redundant pathways to ensure functionality even if partial degradation occurs during sterilization. Additionally, they've pioneered testing protocols specifically designed to evaluate the reliability of transient electronics under various sterilization conditions, providing standardized metrics for device performance assessment.
Strengths: Comprehensive intellectual property portfolio covering materials, designs, and manufacturing processes; strong focus on practical medical applications with clinical testing partnerships. Weaknesses: Higher production costs compared to conventional electronics; some designs require specialized handling protocols that limit widespread adoption in medical settings.
Key Patents in Heat-Resistant Biodegradable Materials
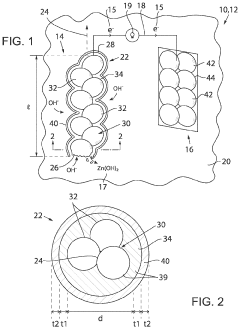
Biodegradable transient battery built on core-double-shell zinc microparticle networks
PatentActiveUS11791519B2
Innovation
- A transient biodegradable battery with a filament structure using zinc microparticles or nanoparticles coated with chitosan and Al2O3, allowing controlled current and lifespan through regulated oxidation reactions, which dissolve safely in biological fluids.
Composite for controlling degradation of transient electronics
PatentPendingEP4316538A1
Innovation
- A composite with a support and a porous polymer layer containing biocompatible oil is used to control the degradation of transient electronics, allowing for controlled release and biodegradation, ensuring flexibility and biocompatibility to prevent organ damage and immune responses.
Biocompatibility and Safety Standards
Transient and biodegradable electronics intended for medical applications must adhere to stringent biocompatibility and safety standards before clinical implementation. The ISO 10993 series provides the fundamental framework for evaluating biocompatibility of medical devices, with specific attention to ISO 10993-1 for risk assessment and categorization based on body contact duration and tissue interaction type. For biodegradable electronics, additional considerations under ISO 10993-9 through 10993-13 address degradation products and their potential toxicological impacts.
FDA guidance further supplements these standards, particularly through the use of biological evaluation plans (BEPs) that assess both the initial device and its degradation byproducts. The FDA's "Guidance for Industry and Food and Drug Administration Staff: Use of International Standard ISO 10993-1" specifically addresses the evaluation pathway for novel biomaterials in medical devices, which directly applies to transient electronics.
The European Medical Device Regulation (MDR 2017/745) imposes additional requirements for devices containing novel materials, with particular emphasis on risk management throughout the product lifecycle. This becomes especially critical for biodegradable electronics where material transformation occurs in vivo.
Material selection for transient electronics must prioritize biocompatibility while maintaining functional integrity during sterilization processes. Silicon, magnesium, zinc, and specific polymers like poly(lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL) have demonstrated favorable biocompatibility profiles while offering controlled degradation characteristics. However, sterilization methods can significantly alter these materials' properties and degradation kinetics.
Cytotoxicity testing (ISO 10993-5), sensitization assessment (ISO 10993-10), and genotoxicity evaluation (ISO 10993-3) represent the minimum testing requirements for these devices. For implantable transient electronics, additional testing for systemic toxicity (ISO 10993-11) and local tissue effects after implantation (ISO 10993-6) becomes necessary.
Emerging standards specifically addressing biodegradable electronics are under development by organizations including ASTM International and IEEE. The ASTM F3089-14 standard provides guidance on testing degradable polymers and devices, while IEEE is developing standards for biodegradable electronics through its P2510 working group, focusing on performance metrics and safety evaluation methodologies.
Regulatory bodies increasingly recognize the need for specialized frameworks for transient electronics, with the FDA's Emerging Technology Program and the EU's Innovation Network providing pathways for novel technology evaluation. These initiatives facilitate early dialogue between developers and regulators to establish appropriate testing protocols for these unconventional devices.
FDA guidance further supplements these standards, particularly through the use of biological evaluation plans (BEPs) that assess both the initial device and its degradation byproducts. The FDA's "Guidance for Industry and Food and Drug Administration Staff: Use of International Standard ISO 10993-1" specifically addresses the evaluation pathway for novel biomaterials in medical devices, which directly applies to transient electronics.
The European Medical Device Regulation (MDR 2017/745) imposes additional requirements for devices containing novel materials, with particular emphasis on risk management throughout the product lifecycle. This becomes especially critical for biodegradable electronics where material transformation occurs in vivo.
Material selection for transient electronics must prioritize biocompatibility while maintaining functional integrity during sterilization processes. Silicon, magnesium, zinc, and specific polymers like poly(lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL) have demonstrated favorable biocompatibility profiles while offering controlled degradation characteristics. However, sterilization methods can significantly alter these materials' properties and degradation kinetics.
Cytotoxicity testing (ISO 10993-5), sensitization assessment (ISO 10993-10), and genotoxicity evaluation (ISO 10993-3) represent the minimum testing requirements for these devices. For implantable transient electronics, additional testing for systemic toxicity (ISO 10993-11) and local tissue effects after implantation (ISO 10993-6) becomes necessary.
Emerging standards specifically addressing biodegradable electronics are under development by organizations including ASTM International and IEEE. The ASTM F3089-14 standard provides guidance on testing degradable polymers and devices, while IEEE is developing standards for biodegradable electronics through its P2510 working group, focusing on performance metrics and safety evaluation methodologies.
Regulatory bodies increasingly recognize the need for specialized frameworks for transient electronics, with the FDA's Emerging Technology Program and the EU's Innovation Network providing pathways for novel technology evaluation. These initiatives facilitate early dialogue between developers and regulators to establish appropriate testing protocols for these unconventional devices.
Environmental Impact Assessment
The environmental impact of transient and biodegradable electronics represents a critical dimension in evaluating their overall sustainability and ecological footprint. These innovative devices, designed to harmlessly degrade after fulfilling their intended functions, offer promising alternatives to conventional electronics that contribute significantly to e-waste accumulation globally.
When assessing the environmental implications of transient electronics under sterilization conditions, several key factors emerge. The sterilization processes themselves—whether utilizing heat, radiation, or chemical agents—may alter the degradation pathways of these devices, potentially releasing different byproducts into the environment than would occur under normal degradation circumstances. Research indicates that high-temperature sterilization methods can accelerate the breakdown of certain biodegradable polymers, potentially creating more benign end products but sometimes generating intermediate compounds requiring further investigation.
Water systems particularly benefit from transient electronics technology, as these devices avoid the persistent contamination associated with conventional electronic waste. Studies demonstrate that silicon-based transient components typically dissolve into silicic acid, a naturally occurring compound in water bodies that poses minimal ecological risk. However, the environmental fate of more complex components, including specialized metals and semiconductor materials subjected to sterilization treatments, requires more comprehensive lifecycle assessment.
Soil interaction analyses reveal generally favorable outcomes when comparing sterilized transient electronics with traditional counterparts. The degradation products typically integrate into natural biogeochemical cycles, though the rate and completeness of this integration varies significantly based on soil composition, microbial activity, and climate conditions. Sterilization processes may affect these integration patterns by altering surface properties or chemical structures of the biodegradable materials.
Carbon footprint comparisons between transient and conventional electronics demonstrate substantial advantages for the former, even when accounting for sterilization energy requirements. Life cycle assessments indicate potential reductions in greenhouse gas emissions by 40-60% compared to traditional electronics with similar functionality, primarily due to eliminated end-of-life processing requirements and reduced raw material extraction needs.
Regulatory frameworks worldwide are increasingly acknowledging the environmental benefits of transient electronics, though standards specifically addressing sterilized biodegradable devices remain underdeveloped. The European Union's recent initiatives to incorporate these technologies into circular economy directives represent promising progress toward establishing comprehensive environmental guidelines that recognize the unique characteristics of these innovative electronic systems.
When assessing the environmental implications of transient electronics under sterilization conditions, several key factors emerge. The sterilization processes themselves—whether utilizing heat, radiation, or chemical agents—may alter the degradation pathways of these devices, potentially releasing different byproducts into the environment than would occur under normal degradation circumstances. Research indicates that high-temperature sterilization methods can accelerate the breakdown of certain biodegradable polymers, potentially creating more benign end products but sometimes generating intermediate compounds requiring further investigation.
Water systems particularly benefit from transient electronics technology, as these devices avoid the persistent contamination associated with conventional electronic waste. Studies demonstrate that silicon-based transient components typically dissolve into silicic acid, a naturally occurring compound in water bodies that poses minimal ecological risk. However, the environmental fate of more complex components, including specialized metals and semiconductor materials subjected to sterilization treatments, requires more comprehensive lifecycle assessment.
Soil interaction analyses reveal generally favorable outcomes when comparing sterilized transient electronics with traditional counterparts. The degradation products typically integrate into natural biogeochemical cycles, though the rate and completeness of this integration varies significantly based on soil composition, microbial activity, and climate conditions. Sterilization processes may affect these integration patterns by altering surface properties or chemical structures of the biodegradable materials.
Carbon footprint comparisons between transient and conventional electronics demonstrate substantial advantages for the former, even when accounting for sterilization energy requirements. Life cycle assessments indicate potential reductions in greenhouse gas emissions by 40-60% compared to traditional electronics with similar functionality, primarily due to eliminated end-of-life processing requirements and reduced raw material extraction needs.
Regulatory frameworks worldwide are increasingly acknowledging the environmental benefits of transient electronics, though standards specifically addressing sterilized biodegradable devices remain underdeveloped. The European Union's recent initiatives to incorporate these technologies into circular economy directives represent promising progress toward establishing comprehensive environmental guidelines that recognize the unique characteristics of these innovative electronic systems.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!