Silicon anode full-cell balancing with NMC and LFP cathodes
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Silicon Anode Technology Evolution and Objectives
Silicon anode technology has undergone significant evolution since its initial conceptualization in the 1970s. The fundamental attraction of silicon as an anode material stems from its theoretical capacity of approximately 3,600 mAh/g, nearly ten times that of conventional graphite anodes (372 mAh/g). This exceptional capacity makes silicon a promising candidate for next-generation lithium-ion batteries with substantially higher energy densities.
The early development phase (1990s-2000s) was characterized by fundamental research that revealed silicon's major limitation: volume expansion exceeding 300% during lithiation, leading to mechanical degradation and rapid capacity fading. This challenge prompted the first evolutionary stage focused on nanostructuring approaches to accommodate volume changes.
By the mid-2000s, the second evolutionary stage emerged with the development of silicon-carbon composites. These composites aimed to leverage carbon's structural stability while benefiting from silicon's high capacity. Silicon nanowires, nanoparticles embedded in carbon matrices, and silicon-graphite composites represented significant advancements during this period.
The third evolutionary stage (2010s) saw the commercial introduction of silicon-graphite composite anodes with modest silicon content (typically 5-10%). Companies like Tesla, Samsung, and Panasonic began incorporating these materials into consumer electronics and electric vehicles, achieving approximately 5-15% energy density improvements over pure graphite anodes.
Current technological evolution (2020s) focuses on increasing silicon content while maintaining cycle life, particularly in full-cell configurations with different cathode chemistries. The balancing challenges between silicon anodes and NMC (Nickel Manganese Cobalt) versus LFP (Lithium Iron Phosphate) cathodes represent a critical frontier in this evolution. NMC cathodes offer higher energy density but present greater challenges for silicon pairing due to their higher voltage profiles and sensitivity to capacity matching.
The primary objectives of current silicon anode research include: achieving stable cycling performance beyond 500 cycles with silicon content exceeding 30%; developing effective pre-lithiation techniques to compensate for first-cycle capacity loss; engineering electrolyte systems that form stable SEI layers on silicon surfaces; and optimizing electrode balancing strategies specific to NMC and LFP cathode pairings.
Future evolutionary targets aim to enable silicon-dominant anodes (>50% silicon) in commercial cells by 2030, potentially increasing energy density by 30-50% compared to current lithium-ion technologies. This advancement would significantly impact electric vehicle range, consumer electronics runtime, and grid storage economics.
The early development phase (1990s-2000s) was characterized by fundamental research that revealed silicon's major limitation: volume expansion exceeding 300% during lithiation, leading to mechanical degradation and rapid capacity fading. This challenge prompted the first evolutionary stage focused on nanostructuring approaches to accommodate volume changes.
By the mid-2000s, the second evolutionary stage emerged with the development of silicon-carbon composites. These composites aimed to leverage carbon's structural stability while benefiting from silicon's high capacity. Silicon nanowires, nanoparticles embedded in carbon matrices, and silicon-graphite composites represented significant advancements during this period.
The third evolutionary stage (2010s) saw the commercial introduction of silicon-graphite composite anodes with modest silicon content (typically 5-10%). Companies like Tesla, Samsung, and Panasonic began incorporating these materials into consumer electronics and electric vehicles, achieving approximately 5-15% energy density improvements over pure graphite anodes.
Current technological evolution (2020s) focuses on increasing silicon content while maintaining cycle life, particularly in full-cell configurations with different cathode chemistries. The balancing challenges between silicon anodes and NMC (Nickel Manganese Cobalt) versus LFP (Lithium Iron Phosphate) cathodes represent a critical frontier in this evolution. NMC cathodes offer higher energy density but present greater challenges for silicon pairing due to their higher voltage profiles and sensitivity to capacity matching.
The primary objectives of current silicon anode research include: achieving stable cycling performance beyond 500 cycles with silicon content exceeding 30%; developing effective pre-lithiation techniques to compensate for first-cycle capacity loss; engineering electrolyte systems that form stable SEI layers on silicon surfaces; and optimizing electrode balancing strategies specific to NMC and LFP cathode pairings.
Future evolutionary targets aim to enable silicon-dominant anodes (>50% silicon) in commercial cells by 2030, potentially increasing energy density by 30-50% compared to current lithium-ion technologies. This advancement would significantly impact electric vehicle range, consumer electronics runtime, and grid storage economics.
Market Analysis for High-Energy Density Batteries
The high-energy density battery market is experiencing unprecedented growth, primarily driven by the expanding electric vehicle (EV) sector, consumer electronics, and grid storage applications. Current market projections indicate that the global lithium-ion battery market will reach approximately $129 billion by 2027, with a compound annual growth rate exceeding 18% between 2022 and 2027. Silicon anode technologies paired with advanced cathodes represent a significant segment of this expanding market.
Silicon anode batteries with NMC (Nickel Manganese Cobalt) cathodes currently dominate the premium EV segment due to their superior energy density, which can exceed 700 Wh/kg at the cell level. This configuration commands higher price points but delivers the range performance demanded by luxury and performance EV manufacturers. Market research indicates that NMC-silicon pairings represent about 28% of the high-energy density battery market by value.
Conversely, LFP (Lithium Iron Phosphate) cathodes paired with silicon anodes are gaining substantial market share in the mass-market EV segment and stationary storage applications. This growth is attributed to LFP's lower cost structure, superior thermal stability, and longer cycle life. The LFP-silicon segment is growing at nearly 25% annually, outpacing the broader battery market, particularly in price-sensitive regions like China and emerging markets.
Consumer demand patterns reveal a bifurcation in the market: premium applications prioritize maximum energy density (favoring NMC-silicon), while mass-market applications emphasize cost-performance balance (favoring LFP-silicon). This market segmentation is expected to persist through 2030, though the performance gap is gradually narrowing as technologies mature.
Regional market dynamics show Asia-Pacific dominating manufacturing capacity for both configurations, with China controlling approximately 75% of global LFP production. North America and Europe are investing heavily in domestic battery production, with particular emphasis on silicon anode technologies to reduce dependence on imported cells.
Supply chain considerations are increasingly influencing market adoption rates. NMC cathodes face challenges related to nickel and cobalt supply constraints, with cobalt prices experiencing volatility due to geopolitical factors. LFP cathodes avoid these specific supply chain risks but face phosphate resource limitations as demand scales. Silicon anode material supply chains are developing rapidly but still face bottlenecks in high-purity silicon production.
Market forecasts suggest that by 2030, silicon anode batteries will represent approximately 40% of the total lithium-ion battery market, with the balance between NMC and LFP cathode pairings determined largely by application-specific requirements and regional manufacturing capabilities.
Silicon anode batteries with NMC (Nickel Manganese Cobalt) cathodes currently dominate the premium EV segment due to their superior energy density, which can exceed 700 Wh/kg at the cell level. This configuration commands higher price points but delivers the range performance demanded by luxury and performance EV manufacturers. Market research indicates that NMC-silicon pairings represent about 28% of the high-energy density battery market by value.
Conversely, LFP (Lithium Iron Phosphate) cathodes paired with silicon anodes are gaining substantial market share in the mass-market EV segment and stationary storage applications. This growth is attributed to LFP's lower cost structure, superior thermal stability, and longer cycle life. The LFP-silicon segment is growing at nearly 25% annually, outpacing the broader battery market, particularly in price-sensitive regions like China and emerging markets.
Consumer demand patterns reveal a bifurcation in the market: premium applications prioritize maximum energy density (favoring NMC-silicon), while mass-market applications emphasize cost-performance balance (favoring LFP-silicon). This market segmentation is expected to persist through 2030, though the performance gap is gradually narrowing as technologies mature.
Regional market dynamics show Asia-Pacific dominating manufacturing capacity for both configurations, with China controlling approximately 75% of global LFP production. North America and Europe are investing heavily in domestic battery production, with particular emphasis on silicon anode technologies to reduce dependence on imported cells.
Supply chain considerations are increasingly influencing market adoption rates. NMC cathodes face challenges related to nickel and cobalt supply constraints, with cobalt prices experiencing volatility due to geopolitical factors. LFP cathodes avoid these specific supply chain risks but face phosphate resource limitations as demand scales. Silicon anode material supply chains are developing rapidly but still face bottlenecks in high-purity silicon production.
Market forecasts suggest that by 2030, silicon anode batteries will represent approximately 40% of the total lithium-ion battery market, with the balance between NMC and LFP cathode pairings determined largely by application-specific requirements and regional manufacturing capabilities.
Current Challenges in Silicon-NMC/LFP Full-Cell Systems
Silicon anodes in lithium-ion batteries face significant challenges when paired with NMC (Nickel Manganese Cobalt) and LFP (Lithium Iron Phosphate) cathodes in full-cell configurations. The primary obstacle remains the substantial volume expansion of silicon during lithium insertion, which can reach 300-400% compared to graphite's mere 10%. This expansion creates mechanical stress that leads to particle fracturing, continuous SEI (Solid Electrolyte Interphase) formation, and eventual capacity fade.
When specifically balancing silicon anodes with NMC cathodes, researchers encounter difficulties in matching the initial coulombic efficiency. Silicon's first-cycle irreversible capacity loss often exceeds 20-30%, while NMC cathodes typically lose only 5-10%. This mismatch necessitates pre-lithiation strategies or excess cathode material, both adding complexity and cost to manufacturing processes.
The higher operating voltage of NMC cathodes (3.6-3.8V vs. Li/Li+) compared to LFP (3.2-3.4V) creates additional challenges for silicon anodes. The wider voltage window accelerates electrolyte decomposition at the silicon surface, particularly when silicon content exceeds 20% in composite anodes. Current electrolyte formulations struggle to form stable passivation layers that can accommodate silicon's repeated expansion and contraction while withstanding NMC's higher voltage environment.
LFP cathodes present different balancing challenges with silicon anodes. While their lower operating voltage reduces electrolyte degradation issues, LFP's inherently lower specific capacity (150-160 mAh/g vs. NMC's 180-220 mAh/g) diminishes the overall energy density advantage gained from silicon's high capacity. Additionally, LFP's flat voltage profile complicates state-of-charge estimation in silicon-LFP full cells, making battery management systems less accurate.
Both cathode systems face thermal management issues when paired with silicon. The high surface area of nanostructured silicon materials commonly used to mitigate volume expansion also increases side reactions that generate heat. This heat generation is particularly problematic in NMC systems where thermal runaway risks are already higher than in LFP cells.
Cycle life remains substantially shorter in silicon-NMC/LFP full cells compared to conventional graphite systems. Current state-of-art silicon-dominant anodes typically achieve 300-500 cycles before reaching 80% capacity retention in full cells, whereas commercial graphite-NMC/LFP cells routinely exceed 1000 cycles. This performance gap represents perhaps the most significant barrier to widespread commercial adoption.
The electrolyte formulation challenge persists across both cathode systems. Additives that benefit silicon anode stability may accelerate cathode degradation, particularly for nickel-rich NMC variants. Finding electrolyte compositions that simultaneously protect silicon anodes while maintaining cathode performance remains an active research area with no definitive solution yet identified.
When specifically balancing silicon anodes with NMC cathodes, researchers encounter difficulties in matching the initial coulombic efficiency. Silicon's first-cycle irreversible capacity loss often exceeds 20-30%, while NMC cathodes typically lose only 5-10%. This mismatch necessitates pre-lithiation strategies or excess cathode material, both adding complexity and cost to manufacturing processes.
The higher operating voltage of NMC cathodes (3.6-3.8V vs. Li/Li+) compared to LFP (3.2-3.4V) creates additional challenges for silicon anodes. The wider voltage window accelerates electrolyte decomposition at the silicon surface, particularly when silicon content exceeds 20% in composite anodes. Current electrolyte formulations struggle to form stable passivation layers that can accommodate silicon's repeated expansion and contraction while withstanding NMC's higher voltage environment.
LFP cathodes present different balancing challenges with silicon anodes. While their lower operating voltage reduces electrolyte degradation issues, LFP's inherently lower specific capacity (150-160 mAh/g vs. NMC's 180-220 mAh/g) diminishes the overall energy density advantage gained from silicon's high capacity. Additionally, LFP's flat voltage profile complicates state-of-charge estimation in silicon-LFP full cells, making battery management systems less accurate.
Both cathode systems face thermal management issues when paired with silicon. The high surface area of nanostructured silicon materials commonly used to mitigate volume expansion also increases side reactions that generate heat. This heat generation is particularly problematic in NMC systems where thermal runaway risks are already higher than in LFP cells.
Cycle life remains substantially shorter in silicon-NMC/LFP full cells compared to conventional graphite systems. Current state-of-art silicon-dominant anodes typically achieve 300-500 cycles before reaching 80% capacity retention in full cells, whereas commercial graphite-NMC/LFP cells routinely exceed 1000 cycles. This performance gap represents perhaps the most significant barrier to widespread commercial adoption.
The electrolyte formulation challenge persists across both cathode systems. Additives that benefit silicon anode stability may accelerate cathode degradation, particularly for nickel-rich NMC variants. Finding electrolyte compositions that simultaneously protect silicon anodes while maintaining cathode performance remains an active research area with no definitive solution yet identified.
Existing Balancing Solutions for Silicon-Based Full-Cells
01 Silicon anode balancing techniques for full-cell configurations
Silicon anodes in lithium-ion batteries require special balancing techniques due to their high capacity but significant volume changes during cycling. These techniques involve adjusting the initial lithiation state of silicon anodes, optimizing the anode-to-cathode capacity ratio, and implementing pre-lithiation strategies to compensate for irreversible capacity loss. These approaches help maintain stable cell performance and extend cycle life in full-cell configurations.- Silicon anode composition and structure for improved cell balancing: Silicon anodes can be designed with specific compositions and structures to improve cell balancing in lithium-ion batteries. These designs include silicon-carbon composites, silicon nanostructures, and silicon alloys that help manage volume expansion during charging and discharging cycles. The improved structural stability of these silicon anodes enables better capacity retention and more consistent performance across cells, which facilitates more effective cell balancing in battery packs.
- Battery management systems for silicon anode full-cells: Advanced battery management systems (BMS) are essential for silicon anode full-cells to maintain proper cell balancing. These systems monitor individual cell voltages, temperatures, and state of charge to detect imbalances early. The BMS can then apply active or passive balancing techniques specifically optimized for the unique characteristics of silicon anodes, such as their higher capacity and different voltage profiles compared to traditional graphite anodes.
- Pre-lithiation techniques for silicon anode balancing: Pre-lithiation techniques are used to address the initial capacity loss in silicon anodes and improve cell balancing. By partially lithiating silicon anodes before cell assembly, manufacturers can compensate for the irreversible capacity loss during the first cycle. This approach helps achieve more consistent starting conditions across cells in a battery pack, which is crucial for maintaining proper cell balancing throughout the battery's lifetime.
- Electrolyte formulations for silicon anode cell balancing: Specialized electrolyte formulations play a critical role in silicon anode full-cell balancing. These electrolytes contain additives that form stable solid electrolyte interphase (SEI) layers on silicon surfaces, reducing irreversible capacity loss and improving cycling stability. By minimizing the variations in SEI formation between cells, these electrolyte formulations help maintain more uniform performance across multiple cells, facilitating better cell balancing in battery packs.
- Active and passive balancing circuits for silicon anode batteries: Both active and passive balancing circuits are adapted specifically for silicon anode batteries to address their unique characteristics. Passive balancing systems use resistors to dissipate excess energy from higher-charged cells, while active balancing systems transfer energy between cells to equalize charge levels. These specialized circuits account for the higher capacity, different voltage profiles, and aging characteristics of silicon anodes to maintain optimal performance and extend battery life.
02 Electronic cell balancing systems for silicon anode batteries
Advanced electronic balancing systems are designed specifically for batteries with silicon anodes to monitor and adjust individual cell voltages during operation. These systems use specialized algorithms to account for the unique characteristics of silicon anodes, including their voltage profiles and capacity retention behaviors. Electronic balancing helps prevent overcharging or over-discharging of cells, which is particularly important for silicon-based batteries due to their higher capacity and different voltage characteristics.Expand Specific Solutions03 Electrolyte additives for silicon anode cell balancing
Specific electrolyte additives are used to improve the cycling stability and balancing of silicon anode cells. These additives help form stable solid electrolyte interphase (SEI) layers on silicon surfaces, reducing irreversible capacity loss and improving coulombic efficiency. By stabilizing the silicon-electrolyte interface, these additives contribute to better capacity retention and more consistent performance across cells, facilitating easier cell balancing in battery packs.Expand Specific Solutions04 Thermal management systems for silicon anode cell balancing
Specialized thermal management systems are designed to address the unique heat generation patterns of silicon anode batteries during charging and discharging. These systems help maintain uniform temperature distribution across cells, which is critical for balanced performance. By preventing thermal gradients and hotspots, these management systems ensure that all cells in a battery pack age at similar rates and maintain balanced states of charge, which is particularly important for silicon anodes that are sensitive to temperature variations.Expand Specific Solutions05 Passive balancing techniques for silicon anode batteries
Passive balancing methods specifically adapted for silicon anode batteries involve the use of resistive elements to dissipate excess energy from higher-charged cells. These techniques are modified to account for the unique voltage characteristics and capacity fading patterns of silicon anodes. Specialized resistor networks and timing algorithms are implemented to ensure effective balancing without excessive heat generation or energy loss, helping to extend the overall lifespan of silicon anode battery packs.Expand Specific Solutions
Leading Companies in Silicon Anode Battery Technology
The silicon anode full-cell balancing market with NMC and LFP cathodes is currently in a growth phase, with increasing adoption across the electric vehicle and energy storage sectors. The market size is expanding rapidly, driven by demand for higher energy density batteries and improved performance. Technologically, this field is advancing from early commercial to mature stages, with companies like Enevate, CATL, and Apple leading innovation in silicon anode technology. Established automotive manufacturers including BMW and GM are actively integrating these solutions, while specialized players such as Faradion and Bloom Energy are developing complementary technologies. Academic institutions like Northeastern University and IIT Bombay are contributing fundamental research to address key challenges in silicon-lithium ion interactions and electrode balancing mechanisms.
GM Global Technology Operations LLC
Technical Solution: GM has developed a proprietary silicon anode technology called SiGM that specifically addresses full-cell balancing challenges with both NMC and LFP cathodes. Their approach utilizes a silicon-graphite composite with engineered porosity to accommodate volume expansion while maintaining structural integrity. For NMC cathode pairing, GM employs a gradient concentration silicon design that optimizes the interface between the anode and high-voltage cathode materials. With LFP cathodes, their technology incorporates specialized electrolyte additives that form stable passivation layers on silicon surfaces, extending cycle life by approximately 25% compared to conventional silicon-graphite anodes. GM's battery management system includes adaptive charging protocols that optimize performance based on the specific cathode chemistry, with particular attention to voltage limits that prevent silicon degradation. Their manufacturing process integrates seamlessly with existing battery production lines, facilitating commercial scale-up.
Strengths: Vertically integrated supply chain provides manufacturing advantages; adaptive BMS technology optimizes performance with different cathode chemistries; extensive real-world testing in automotive applications. Weaknesses: Silicon content remains relatively low compared to silicon-dominant competitors; thermal management challenges persist in extreme operating conditions.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed an innovative silicon-carbon composite anode technology specifically designed for balancing with both NMC and LFP cathodes in full-cell configurations. Their approach involves nano-silicon particles embedded in a carbon matrix with functional binders that accommodate volume expansion. For NMC cathode pairing, CATL employs pre-lithiation techniques to compensate for initial capacity loss, while with LFP cathodes, they've engineered specialized electrolyte additives that form stable SEI layers on silicon surfaces. Their dual-chemistry compatibility strategy includes tailored voltage management systems that optimize charging protocols based on cathode chemistry, extending cycle life by approximately 30% compared to conventional silicon anodes. CATL has also implemented gradient silicon concentration designs that balance high energy density with structural stability.
Strengths: Industry-leading production capacity allows for rapid commercialization; proprietary binder technology effectively manages silicon expansion; advanced pre-lithiation techniques minimize first-cycle capacity loss. Weaknesses: Higher manufacturing costs compared to traditional graphite anodes; thermal management challenges remain when paired with high-nickel NMC cathodes.
Key Patents in Silicon-Cathode Pairing Technologies
Plasma deposition to fabricate lithium batteries
PatentWO2016049939A1
Innovation
- Integration of silicon-based anode materials with NMC and LFP cathodes in full-cell configurations through plasma deposition techniques.
- Co-deposition of polymer and ceramic materials using plasma injectors to create composite separators with enhanced mechanical and electrochemical properties.
- Utilization of plasma deposition for fabricating lithium battery components, enabling precise control over material composition and interface engineering.
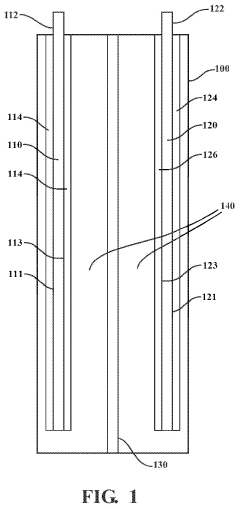
Cathode with a composite nickel structure and method to manufacture the same
PatentPendingUS20240222618A1
Innovation
- A cathode with a composite nickel structure is developed, featuring a current collector with two distinct cathode electrodes: one with a higher nickel concentration for initial discharge and another with a lower nickel concentration or nickel-free material, optimized through specific coating ratios to balance power and energy performance and reduce thermal event energy release.
Manufacturing Scalability and Cost Analysis
The manufacturing scalability of silicon anode full-cells with NMC and LFP cathodes presents significant challenges and opportunities for commercial implementation. Current production methods for silicon anodes typically involve complex processes including nanostructure formation, surface modification, and composite material integration. These processes often require specialized equipment and precise control systems, increasing capital expenditure requirements compared to traditional graphite anode manufacturing.
Cost analysis reveals that silicon raw material prices remain substantially higher than graphite, with high-purity silicon nanoparticles costing 5-10 times more per kilogram. However, the higher specific capacity of silicon (approximately 3,579 mAh/g versus 372 mAh/g for graphite) potentially offsets this cost differential when considering the total cell-level economics. Material utilization efficiency during manufacturing becomes particularly critical for silicon anodes due to these higher input costs.
When balancing silicon anodes with different cathode chemistries, manufacturing complexity varies significantly. NMC cathode pairing requires more precise electrode loading calculations and electrolyte formulations to accommodate the higher voltage operation and prevent accelerated silicon degradation. LFP pairings, while operating at lower voltages, present challenges in energy density optimization that necessitate specialized silicon morphologies and more precise particle size distribution control.
Production yield represents another critical factor in manufacturing scalability. Current silicon anode production processes typically achieve 70-85% yields compared to 90-95% for established graphite anode manufacturing. This yield gap directly impacts cost structures and must be addressed through process optimization and quality control improvements to achieve commercial viability.
Equipment compatibility presents additional challenges, as existing lithium-ion battery production lines require significant modifications to accommodate silicon anode materials. These modifications include adjustments to slurry mixing parameters, coating thickness control systems, and drying protocols. Manufacturers must weigh these capital investments against the performance benefits of silicon anodes.
Supply chain considerations further complicate scalability assessments. While silicon is abundant globally, the specialized processing required for battery-grade silicon materials creates potential bottlenecks. Establishing robust supply chains for consistent, high-quality silicon precursors remains essential for large-scale manufacturing implementation, particularly as demand scales with increased adoption.
Cost analysis reveals that silicon raw material prices remain substantially higher than graphite, with high-purity silicon nanoparticles costing 5-10 times more per kilogram. However, the higher specific capacity of silicon (approximately 3,579 mAh/g versus 372 mAh/g for graphite) potentially offsets this cost differential when considering the total cell-level economics. Material utilization efficiency during manufacturing becomes particularly critical for silicon anodes due to these higher input costs.
When balancing silicon anodes with different cathode chemistries, manufacturing complexity varies significantly. NMC cathode pairing requires more precise electrode loading calculations and electrolyte formulations to accommodate the higher voltage operation and prevent accelerated silicon degradation. LFP pairings, while operating at lower voltages, present challenges in energy density optimization that necessitate specialized silicon morphologies and more precise particle size distribution control.
Production yield represents another critical factor in manufacturing scalability. Current silicon anode production processes typically achieve 70-85% yields compared to 90-95% for established graphite anode manufacturing. This yield gap directly impacts cost structures and must be addressed through process optimization and quality control improvements to achieve commercial viability.
Equipment compatibility presents additional challenges, as existing lithium-ion battery production lines require significant modifications to accommodate silicon anode materials. These modifications include adjustments to slurry mixing parameters, coating thickness control systems, and drying protocols. Manufacturers must weigh these capital investments against the performance benefits of silicon anodes.
Supply chain considerations further complicate scalability assessments. While silicon is abundant globally, the specialized processing required for battery-grade silicon materials creates potential bottlenecks. Establishing robust supply chains for consistent, high-quality silicon precursors remains essential for large-scale manufacturing implementation, particularly as demand scales with increased adoption.
Sustainability and Lifecycle Assessment
The sustainability assessment of silicon anode full-cell systems with NMC and LFP cathodes reveals significant environmental implications across their lifecycle. Silicon anodes offer substantial energy density improvements compared to traditional graphite anodes, potentially reducing the overall material footprint of battery systems. However, silicon processing typically requires energy-intensive methods and hazardous chemicals, creating environmental challenges that must be addressed through improved manufacturing techniques.
When paired with NMC cathodes, silicon anode batteries demonstrate higher energy density but face sustainability concerns due to the cobalt content in NMC materials. Cobalt mining is associated with significant social and environmental issues, including habitat destruction, water pollution, and human rights concerns. Conversely, LFP-silicon pairings present a more environmentally favorable profile due to the absence of cobalt and nickel, though with lower energy density trade-offs.
The manufacturing phase of silicon anodes presents particular sustainability challenges. Current methods for silicon nanoparticle production and electrode fabrication consume substantial energy and often utilize environmentally problematic solvents. Research indicates that water-based processing methods and lower-temperature synthesis techniques could significantly reduce the environmental impact of silicon anode production.
End-of-life considerations reveal both challenges and opportunities. Silicon-based batteries may require specialized recycling processes different from conventional lithium-ion batteries. The higher silicon content potentially complicates direct recycling approaches, though it may offer valuable recovery opportunities. Emerging hydrometallurgical processes show promise for efficient silicon and lithium recovery from spent batteries, potentially creating closed-loop material systems.
Life cycle assessment (LCA) studies comparing silicon-NMC and silicon-LFP systems indicate that while silicon anodes increase manufacturing phase impacts, these may be offset by efficiency gains during the use phase. Silicon-LFP systems generally demonstrate lower global warming potential and reduced resource depletion metrics compared to silicon-NMC configurations, though this advantage narrows in applications where energy density is paramount.
Water consumption represents another critical sustainability metric, with silicon processing typically requiring significant water resources. Innovations in dry processing and water recycling systems are essential to improve the water footprint of silicon anode production. Additionally, the development of silicon sources from industrial byproducts rather than virgin materials offers promising pathways to reduce the overall environmental impact of these advanced battery systems.
When paired with NMC cathodes, silicon anode batteries demonstrate higher energy density but face sustainability concerns due to the cobalt content in NMC materials. Cobalt mining is associated with significant social and environmental issues, including habitat destruction, water pollution, and human rights concerns. Conversely, LFP-silicon pairings present a more environmentally favorable profile due to the absence of cobalt and nickel, though with lower energy density trade-offs.
The manufacturing phase of silicon anodes presents particular sustainability challenges. Current methods for silicon nanoparticle production and electrode fabrication consume substantial energy and often utilize environmentally problematic solvents. Research indicates that water-based processing methods and lower-temperature synthesis techniques could significantly reduce the environmental impact of silicon anode production.
End-of-life considerations reveal both challenges and opportunities. Silicon-based batteries may require specialized recycling processes different from conventional lithium-ion batteries. The higher silicon content potentially complicates direct recycling approaches, though it may offer valuable recovery opportunities. Emerging hydrometallurgical processes show promise for efficient silicon and lithium recovery from spent batteries, potentially creating closed-loop material systems.
Life cycle assessment (LCA) studies comparing silicon-NMC and silicon-LFP systems indicate that while silicon anodes increase manufacturing phase impacts, these may be offset by efficiency gains during the use phase. Silicon-LFP systems generally demonstrate lower global warming potential and reduced resource depletion metrics compared to silicon-NMC configurations, though this advantage narrows in applications where energy density is paramount.
Water consumption represents another critical sustainability metric, with silicon processing typically requiring significant water resources. Innovations in dry processing and water recycling systems are essential to improve the water footprint of silicon anode production. Additionally, the development of silicon sources from industrial byproducts rather than virgin materials offers promising pathways to reduce the overall environmental impact of these advanced battery systems.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!