Advances in Hydrochloric Acid-Based Catalysis
JUL 1, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HCl Catalysis Background and Objectives
Hydrochloric acid-based catalysis has emerged as a pivotal field in chemical engineering, with its roots tracing back to the early 20th century. The evolution of this technology has been driven by the increasing demand for more efficient and environmentally friendly catalytic processes in various industries, including petrochemicals, pharmaceuticals, and fine chemicals manufacturing.
The journey of HCl catalysis began with simple acid-catalyzed reactions and has progressed to sophisticated systems involving complex metal chloride catalysts. This advancement has been marked by significant milestones, such as the development of the Friedel-Crafts reaction in the 1930s and the introduction of zeolite-based catalysts in the 1960s. These breakthroughs have paved the way for more selective and efficient catalytic processes.
In recent years, the focus has shifted towards developing greener and more sustainable HCl-based catalytic systems. This trend is driven by the growing environmental concerns and the need for more atom-efficient processes. Researchers are exploring novel approaches, including the use of ionic liquids, supported catalysts, and recyclable systems, to minimize waste and improve overall process efficiency.
The primary objective of current research in HCl catalysis is to enhance catalytic activity while reducing environmental impact. This involves developing catalysts with higher selectivity, improved stability, and the ability to operate under milder conditions. Additionally, there is a strong emphasis on creating catalytic systems that can be easily recovered and reused, thereby reducing the overall environmental footprint of chemical processes.
Another key goal is to expand the application scope of HCl-based catalysis. While traditionally used in organic synthesis and petrochemical processes, researchers are now exploring its potential in emerging fields such as biomass conversion, CO2 utilization, and the production of advanced materials. This expansion is crucial for addressing global challenges related to sustainability and resource efficiency.
The technological trajectory of HCl catalysis is closely aligned with broader trends in the chemical industry, including the push for process intensification and the integration of catalytic processes with other technologies such as flow chemistry and microreactor systems. These synergies are expected to lead to more compact, efficient, and flexible production processes, capable of meeting the evolving needs of various industries.
As we look to the future, the field of HCl-based catalysis is poised for significant advancements. The integration of computational modeling, high-throughput experimentation, and advanced characterization techniques is expected to accelerate the discovery and optimization of new catalytic systems. This multidisciplinary approach will be crucial in addressing the complex challenges facing the field and in realizing its full potential in driving sustainable chemical transformations.
The journey of HCl catalysis began with simple acid-catalyzed reactions and has progressed to sophisticated systems involving complex metal chloride catalysts. This advancement has been marked by significant milestones, such as the development of the Friedel-Crafts reaction in the 1930s and the introduction of zeolite-based catalysts in the 1960s. These breakthroughs have paved the way for more selective and efficient catalytic processes.
In recent years, the focus has shifted towards developing greener and more sustainable HCl-based catalytic systems. This trend is driven by the growing environmental concerns and the need for more atom-efficient processes. Researchers are exploring novel approaches, including the use of ionic liquids, supported catalysts, and recyclable systems, to minimize waste and improve overall process efficiency.
The primary objective of current research in HCl catalysis is to enhance catalytic activity while reducing environmental impact. This involves developing catalysts with higher selectivity, improved stability, and the ability to operate under milder conditions. Additionally, there is a strong emphasis on creating catalytic systems that can be easily recovered and reused, thereby reducing the overall environmental footprint of chemical processes.
Another key goal is to expand the application scope of HCl-based catalysis. While traditionally used in organic synthesis and petrochemical processes, researchers are now exploring its potential in emerging fields such as biomass conversion, CO2 utilization, and the production of advanced materials. This expansion is crucial for addressing global challenges related to sustainability and resource efficiency.
The technological trajectory of HCl catalysis is closely aligned with broader trends in the chemical industry, including the push for process intensification and the integration of catalytic processes with other technologies such as flow chemistry and microreactor systems. These synergies are expected to lead to more compact, efficient, and flexible production processes, capable of meeting the evolving needs of various industries.
As we look to the future, the field of HCl-based catalysis is poised for significant advancements. The integration of computational modeling, high-throughput experimentation, and advanced characterization techniques is expected to accelerate the discovery and optimization of new catalytic systems. This multidisciplinary approach will be crucial in addressing the complex challenges facing the field and in realizing its full potential in driving sustainable chemical transformations.
Market Demand Analysis for HCl Catalysts
The market demand for hydrochloric acid-based catalysts has been experiencing significant growth in recent years, driven by the increasing need for efficient and sustainable chemical processes across various industries. The global market for HCl catalysts is projected to expand at a steady rate, with key sectors such as petrochemicals, pharmaceuticals, and fine chemicals leading the demand.
In the petrochemical industry, HCl catalysts play a crucial role in processes such as alkylation, isomerization, and cracking. The growing demand for high-octane fuels and specialty chemicals has been a major factor in boosting the market for these catalysts. Additionally, the shift towards cleaner and more efficient refining processes has further accelerated the adoption of advanced HCl-based catalytic systems.
The pharmaceutical sector represents another significant market for HCl catalysts, particularly in the synthesis of active pharmaceutical ingredients (APIs) and intermediates. As the global population ages and healthcare needs increase, the demand for pharmaceutical products continues to rise, consequently driving the market for catalysts used in their production.
Environmental regulations and sustainability concerns have also been shaping the market demand for HCl catalysts. Industries are increasingly seeking catalytic solutions that can reduce waste, improve energy efficiency, and minimize environmental impact. This trend has led to the development of more selective and recyclable HCl-based catalysts, which are gaining traction in the market.
The fine chemicals industry, including the production of flavors, fragrances, and specialty polymers, has also contributed to the growing demand for HCl catalysts. These catalysts enable the synthesis of complex molecules with high selectivity and yield, making them indispensable in the production of high-value chemicals.
Geographically, Asia-Pacific has emerged as a key market for HCl catalysts, driven by rapid industrialization and the expansion of chemical manufacturing capabilities in countries like China and India. North America and Europe continue to be significant markets, with a focus on innovation and the development of more advanced catalytic systems.
The market demand is further influenced by technological advancements in catalyst design and synthesis. Researchers and manufacturers are continuously working on improving catalyst performance, stability, and reusability, which in turn is expected to open up new applications and market opportunities.
In conclusion, the market demand for HCl-based catalysts is robust and diverse, spanning multiple industries and geographical regions. The ongoing trends in sustainability, process efficiency, and technological innovation are likely to sustain and potentially accelerate this demand in the coming years.
In the petrochemical industry, HCl catalysts play a crucial role in processes such as alkylation, isomerization, and cracking. The growing demand for high-octane fuels and specialty chemicals has been a major factor in boosting the market for these catalysts. Additionally, the shift towards cleaner and more efficient refining processes has further accelerated the adoption of advanced HCl-based catalytic systems.
The pharmaceutical sector represents another significant market for HCl catalysts, particularly in the synthesis of active pharmaceutical ingredients (APIs) and intermediates. As the global population ages and healthcare needs increase, the demand for pharmaceutical products continues to rise, consequently driving the market for catalysts used in their production.
Environmental regulations and sustainability concerns have also been shaping the market demand for HCl catalysts. Industries are increasingly seeking catalytic solutions that can reduce waste, improve energy efficiency, and minimize environmental impact. This trend has led to the development of more selective and recyclable HCl-based catalysts, which are gaining traction in the market.
The fine chemicals industry, including the production of flavors, fragrances, and specialty polymers, has also contributed to the growing demand for HCl catalysts. These catalysts enable the synthesis of complex molecules with high selectivity and yield, making them indispensable in the production of high-value chemicals.
Geographically, Asia-Pacific has emerged as a key market for HCl catalysts, driven by rapid industrialization and the expansion of chemical manufacturing capabilities in countries like China and India. North America and Europe continue to be significant markets, with a focus on innovation and the development of more advanced catalytic systems.
The market demand is further influenced by technological advancements in catalyst design and synthesis. Researchers and manufacturers are continuously working on improving catalyst performance, stability, and reusability, which in turn is expected to open up new applications and market opportunities.
In conclusion, the market demand for HCl-based catalysts is robust and diverse, spanning multiple industries and geographical regions. The ongoing trends in sustainability, process efficiency, and technological innovation are likely to sustain and potentially accelerate this demand in the coming years.
Current State and Challenges in HCl Catalysis
Hydrochloric acid-based catalysis has made significant strides in recent years, positioning itself as a crucial area in chemical engineering and industrial processes. The current state of HCl catalysis is characterized by a blend of established techniques and emerging innovations, with researchers and industry professionals continually pushing the boundaries of its applications.
One of the primary advancements in HCl catalysis lies in the development of more efficient and selective catalytic systems. Researchers have successfully engineered catalysts that exhibit enhanced activity and stability under acidic conditions, allowing for improved reaction rates and product yields. These developments have been particularly impactful in petrochemical industries, where HCl-based catalysts play a vital role in various processes, including hydrochlorination and alkylation reactions.
Despite these advancements, several challenges persist in the field of HCl catalysis. Corrosion remains a significant concern, as the highly acidic environment can lead to rapid degradation of reactor materials and catalysts. This necessitates the use of expensive corrosion-resistant materials, which can substantially increase production costs. Additionally, the handling and disposal of HCl present environmental and safety challenges that require careful management and stringent regulatory compliance.
Another notable challenge is the optimization of catalyst performance under varying reaction conditions. The activity and selectivity of HCl-based catalysts can be highly sensitive to factors such as temperature, pressure, and reactant concentrations. Researchers are actively working on developing more robust catalytic systems that maintain high performance across a broader range of operating conditions, thereby enhancing process flexibility and efficiency.
The recovery and recycling of HCl catalysts pose additional challenges. As catalysts deactivate over time, efficient regeneration methods are crucial for maintaining long-term process viability. Current research efforts are focused on developing novel regeneration techniques that can extend catalyst lifetimes and reduce the frequency of replacement, ultimately lowering operational costs and minimizing waste generation.
In terms of geographical distribution, HCl catalysis research and development are primarily concentrated in regions with strong chemical and petrochemical industries. Countries such as the United States, Germany, China, and Japan are at the forefront of innovation in this field, with significant investments in both academic research and industrial applications. This global distribution of expertise has led to a rich ecosystem of collaboration and knowledge exchange, driving further advancements in the field.
Looking ahead, the future of HCl catalysis lies in addressing these challenges while expanding its applications to new areas. Emerging trends include the development of green HCl catalysis processes that minimize environmental impact, the integration of HCl catalysis with other technologies such as flow chemistry and process intensification, and the exploration of novel applications in fine chemical synthesis and pharmaceutical manufacturing.
One of the primary advancements in HCl catalysis lies in the development of more efficient and selective catalytic systems. Researchers have successfully engineered catalysts that exhibit enhanced activity and stability under acidic conditions, allowing for improved reaction rates and product yields. These developments have been particularly impactful in petrochemical industries, where HCl-based catalysts play a vital role in various processes, including hydrochlorination and alkylation reactions.
Despite these advancements, several challenges persist in the field of HCl catalysis. Corrosion remains a significant concern, as the highly acidic environment can lead to rapid degradation of reactor materials and catalysts. This necessitates the use of expensive corrosion-resistant materials, which can substantially increase production costs. Additionally, the handling and disposal of HCl present environmental and safety challenges that require careful management and stringent regulatory compliance.
Another notable challenge is the optimization of catalyst performance under varying reaction conditions. The activity and selectivity of HCl-based catalysts can be highly sensitive to factors such as temperature, pressure, and reactant concentrations. Researchers are actively working on developing more robust catalytic systems that maintain high performance across a broader range of operating conditions, thereby enhancing process flexibility and efficiency.
The recovery and recycling of HCl catalysts pose additional challenges. As catalysts deactivate over time, efficient regeneration methods are crucial for maintaining long-term process viability. Current research efforts are focused on developing novel regeneration techniques that can extend catalyst lifetimes and reduce the frequency of replacement, ultimately lowering operational costs and minimizing waste generation.
In terms of geographical distribution, HCl catalysis research and development are primarily concentrated in regions with strong chemical and petrochemical industries. Countries such as the United States, Germany, China, and Japan are at the forefront of innovation in this field, with significant investments in both academic research and industrial applications. This global distribution of expertise has led to a rich ecosystem of collaboration and knowledge exchange, driving further advancements in the field.
Looking ahead, the future of HCl catalysis lies in addressing these challenges while expanding its applications to new areas. Emerging trends include the development of green HCl catalysis processes that minimize environmental impact, the integration of HCl catalysis with other technologies such as flow chemistry and process intensification, and the exploration of novel applications in fine chemical synthesis and pharmaceutical manufacturing.
Existing HCl Catalysis Solutions
01 Hydrochloric acid as a catalyst in chemical reactions
Hydrochloric acid is widely used as a catalyst in various chemical reactions, particularly in organic synthesis and industrial processes. It acts as a proton donor, facilitating reactions such as hydrolysis, dehydration, and esterification. The acid's strong acidity and ability to form chloride ions make it an effective catalyst for many transformations.- Hydrochloric acid as a catalyst in chemical reactions: Hydrochloric acid is widely used as a catalyst in various chemical reactions, particularly in organic synthesis and industrial processes. It acts as a proton donor, facilitating reactions such as hydrolysis, dehydration, and esterification. The strong acidity of HCl makes it effective in catalyzing reactions that require acidic conditions.
- Supported hydrochloric acid catalysts: Hydrochloric acid can be supported on various materials to create heterogeneous catalysts. These supported catalysts offer advantages such as easier separation from reaction mixtures, improved stability, and potential for reuse. Common support materials include silica, alumina, and polymeric resins. The supported HCl catalysts find applications in petrochemical processes and fine chemical synthesis.
- Hydrochloric acid in combination with metal catalysts: Hydrochloric acid is often used in combination with metal catalysts to enhance catalytic activity or selectivity. The acid can act as a co-catalyst or promoter, modifying the electronic properties of the metal catalyst or assisting in the activation of reactants. This synergistic effect is utilized in various industrial processes, including hydrogenation and isomerization reactions.
- Hydrochloric acid-based catalysts in polymerization reactions: Hydrochloric acid and its derivatives play a crucial role as catalysts in polymerization reactions. They are particularly effective in cationic polymerization processes, initiating the formation of carbocations that propagate the polymer chain. These catalysts are used in the production of various polymers and resins, including polyvinyl chloride (PVC) and phenolic resins.
- Regeneration and recycling of hydrochloric acid catalysts: The regeneration and recycling of hydrochloric acid-based catalysts are important for improving process economics and reducing environmental impact. Various methods have been developed to recover and reactivate spent HCl catalysts, including thermal treatment, chemical regeneration, and membrane separation techniques. These processes aim to restore catalytic activity and extend the catalyst's lifespan.
02 Hydrochloric acid-based catalysts in petrochemical industry
In the petrochemical industry, hydrochloric acid-based catalysts play a crucial role in various processes, including alkylation, isomerization, and cracking of hydrocarbons. These catalysts are often used in combination with metal chlorides or supported on solid materials to enhance their catalytic activity and selectivity in refining and petrochemical production.Expand Specific Solutions03 Hydrochloric acid catalysts in polymer production
Hydrochloric acid-based catalysts are employed in the production of various polymers and resins. They are particularly useful in polymerization reactions, such as the production of polyvinyl chloride (PVC) and other chlorinated polymers. These catalysts can initiate or accelerate polymerization processes, control molecular weight, and influence the properties of the final polymer products.Expand Specific Solutions04 Hydrochloric acid in metal surface treatment and etching
Hydrochloric acid-based solutions are widely used in metal surface treatment and etching processes. These applications include pickling of steel, cleaning and activating metal surfaces prior to plating or coating, and etching of electronic components. The acid's ability to dissolve metal oxides and form soluble metal chlorides makes it effective for these purposes.Expand Specific Solutions05 Hydrochloric acid catalysts in environmental applications
Hydrochloric acid-based catalysts find applications in environmental technologies, such as flue gas treatment, water purification, and waste management. These catalysts can be used for the removal of pollutants, decomposition of organic compounds, and in catalytic processes for the treatment of industrial effluents and emissions.Expand Specific Solutions
Key Players in HCl Catalyst Industry
The field of hydrochloric acid-based catalysis is experiencing significant growth, driven by increasing demand in various industrial applications. The market is in a mature stage but continues to expand due to ongoing research and development efforts. Key players like Chinese Academy of Science Institute of Chemistry, Celanese International Corp., and BASF Corp. are at the forefront of innovation, leveraging their extensive research capabilities and industry experience. The technology's maturity is evident in the diverse applications across sectors, with companies like Mitsui Chemicals, Inc. and ExxonMobil Chemical Patents, Inc. contributing to its advancement. Academic institutions such as the University of Kansas and Tianjin University are also playing crucial roles in pushing the boundaries of this technology, fostering collaborations between industry and academia.
Chinese Academy of Science Institute of Chemistry
Technical Solution: The Chinese Academy of Science Institute of Chemistry has made significant contributions to the field of hydrochloric acid-based catalysis. Their research has focused on developing novel heterogeneous catalysts for HCl oxidation and utilization. One of their key achievements is the design of highly efficient ruthenium-based catalysts supported on cerium oxide for the Deacon process, which demonstrates exceptional stability and activity even at lower temperatures[13]. The institute has also explored the use of metal-organic frameworks (MOFs) as catalysts and catalyst supports in HCl-mediated reactions, showing promising results in terms of catalytic activity and selectivity[14]. Additionally, they have made progress in the development of photocatalytic systems for HCl splitting, aiming to produce hydrogen as a clean energy carrier[15].
Strengths: Cutting-edge research in heterogeneous catalysis, innovative use of MOFs, and exploration of sustainable hydrogen production. Weaknesses: Some technologies may be at early stages of development, requiring further optimization for industrial application.
Celanese International Corp.
Technical Solution: Celanese has made significant strides in hydrochloric acid-based catalysis, particularly in the field of acetic acid production. Their proprietary AO Plus 3 technology utilizes a rhodium-based catalyst system in a methanol carbonylation process, which efficiently incorporates HCl as a promoter[4]. This advanced catalytic system allows for lower water concentrations in the reaction medium, resulting in reduced energy consumption and improved product yield[5]. Celanese has also developed novel reactor designs that optimize mass transfer and reaction kinetics, further enhancing the efficiency of their HCl-promoted catalytic processes[6].
Strengths: High efficiency in acetic acid production, reduced energy consumption, and improved product yield. Proprietary technology gives a competitive edge. Weaknesses: Dependence on precious metal catalysts may lead to higher costs, potential sensitivity to catalyst poisoning.
Core Innovations in HCl-Based Catalysts
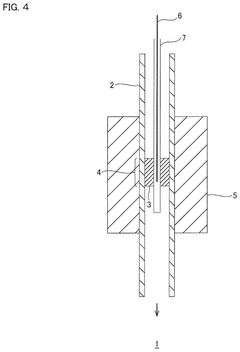
Device and method for producing hypochlorous acid by electrolysis
PatentPendingEP4053306A1
Innovation
- A device design where the cathode is surrounded by a cavity for efficient heat management and dilution, using a static mixing element to combine the diluent and product, ensuring uniform flow and high pressure resistance, thus enhancing product quality and throughput.
Hydrochloric acid oxidation catalyst and method for producing chlorine
PatentPendingUS20240359165A1
Innovation
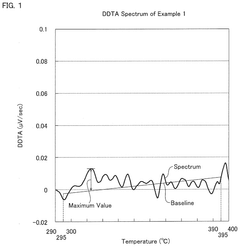
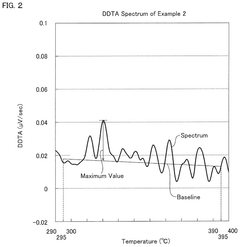
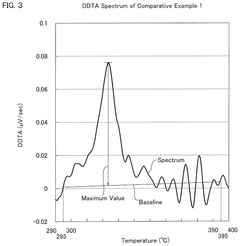
- A hydrochloric acid oxidation catalyst comprising a carrier, copper, an alkali metal, and a rare earth element, with specific properties such as an average pore diameter of 5 nm to 30 nm and a differential thermal analysis spectrum differential value of 0.035 μV/sec or less, which suppresses the dissipation of active components and maintains catalytic activity.
Environmental Impact of HCl Catalysis
The environmental impact of hydrochloric acid (HCl) catalysis is a critical consideration in the advancement of this technology. While HCl-based catalysis offers significant benefits in various industrial processes, it also poses potential risks to the environment that must be carefully managed.
One of the primary environmental concerns associated with HCl catalysis is the potential for acid emissions. When not properly controlled, HCl can be released into the atmosphere, contributing to air pollution and acid rain. These emissions can have detrimental effects on ecosystems, including damage to vegetation, acidification of water bodies, and harm to wildlife. To mitigate these risks, industries employing HCl catalysis have implemented advanced scrubbing systems and emission control technologies to capture and neutralize acid vapors before they are released into the environment.
Water pollution is another significant environmental challenge related to HCl catalysis. Wastewater from industrial processes using HCl catalysts can be highly acidic and may contain dissolved metal ions and other contaminants. If not properly treated, this wastewater can harm aquatic ecosystems and contaminate groundwater resources. As a result, stringent wastewater treatment protocols and recycling systems have been developed to minimize the environmental impact of HCl-containing effluents.
The production and transportation of HCl for catalytic processes also present environmental risks. Accidental spills or leaks during handling and transport can lead to localized environmental damage, including soil contamination and harm to flora and fauna. To address these concerns, industries have implemented rigorous safety protocols and containment measures to prevent and respond to potential incidents.
Despite these challenges, recent advances in HCl catalysis have focused on improving environmental performance. Green chemistry principles are being applied to develop more environmentally friendly catalytic processes that reduce the overall use of HCl or employ it more efficiently. This includes the development of recyclable catalyst systems that minimize waste generation and the exploration of alternative, less corrosive acid catalysts.
Furthermore, life cycle assessments of HCl-based catalytic processes are increasingly being conducted to evaluate their overall environmental impact. These assessments consider factors such as energy consumption, resource depletion, and greenhouse gas emissions associated with the entire catalytic process, from raw material extraction to product disposal. This holistic approach helps identify areas for improvement and guides the development of more sustainable catalytic technologies.
In conclusion, while HCl catalysis presents significant environmental challenges, ongoing research and technological advancements are continually improving its environmental profile. The industry's commitment to sustainable practices and regulatory compliance is driving innovation in emission control, waste management, and process efficiency, ultimately working towards minimizing the environmental footprint of HCl-based catalytic processes.
One of the primary environmental concerns associated with HCl catalysis is the potential for acid emissions. When not properly controlled, HCl can be released into the atmosphere, contributing to air pollution and acid rain. These emissions can have detrimental effects on ecosystems, including damage to vegetation, acidification of water bodies, and harm to wildlife. To mitigate these risks, industries employing HCl catalysis have implemented advanced scrubbing systems and emission control technologies to capture and neutralize acid vapors before they are released into the environment.
Water pollution is another significant environmental challenge related to HCl catalysis. Wastewater from industrial processes using HCl catalysts can be highly acidic and may contain dissolved metal ions and other contaminants. If not properly treated, this wastewater can harm aquatic ecosystems and contaminate groundwater resources. As a result, stringent wastewater treatment protocols and recycling systems have been developed to minimize the environmental impact of HCl-containing effluents.
The production and transportation of HCl for catalytic processes also present environmental risks. Accidental spills or leaks during handling and transport can lead to localized environmental damage, including soil contamination and harm to flora and fauna. To address these concerns, industries have implemented rigorous safety protocols and containment measures to prevent and respond to potential incidents.
Despite these challenges, recent advances in HCl catalysis have focused on improving environmental performance. Green chemistry principles are being applied to develop more environmentally friendly catalytic processes that reduce the overall use of HCl or employ it more efficiently. This includes the development of recyclable catalyst systems that minimize waste generation and the exploration of alternative, less corrosive acid catalysts.
Furthermore, life cycle assessments of HCl-based catalytic processes are increasingly being conducted to evaluate their overall environmental impact. These assessments consider factors such as energy consumption, resource depletion, and greenhouse gas emissions associated with the entire catalytic process, from raw material extraction to product disposal. This holistic approach helps identify areas for improvement and guides the development of more sustainable catalytic technologies.
In conclusion, while HCl catalysis presents significant environmental challenges, ongoing research and technological advancements are continually improving its environmental profile. The industry's commitment to sustainable practices and regulatory compliance is driving innovation in emission control, waste management, and process efficiency, ultimately working towards minimizing the environmental footprint of HCl-based catalytic processes.
Safety Protocols in HCl Catalyst Handling
Safety protocols in handling hydrochloric acid (HCl) catalysts are paramount in the field of HCl-based catalysis. These protocols encompass a comprehensive set of guidelines and procedures designed to protect personnel, equipment, and the environment from the potential hazards associated with HCl catalysts.
Personal protective equipment (PPE) forms the first line of defense in HCl catalyst handling. Workers must wear appropriate acid-resistant gloves, protective eyewear, face shields, and chemical-resistant clothing. Respiratory protection, such as acid gas respirators or self-contained breathing apparatus (SCBA), may be required depending on the concentration and potential for vapor exposure.
Proper storage and containment of HCl catalysts are crucial. These materials should be stored in corrosion-resistant containers in well-ventilated areas, away from incompatible substances. Secondary containment systems are essential to prevent spills and leaks from spreading. Regular inspections of storage areas and containment systems help ensure their integrity and effectiveness.
Handling procedures for HCl catalysts must be meticulously designed and followed. This includes using appropriate transfer equipment, such as corrosion-resistant pumps and piping systems. Closed-loop transfer systems are preferred to minimize exposure risks. Proper labeling of containers and process equipment is essential for clear identification and communication of hazards.
Emergency response planning is a critical component of safety protocols. This includes the development of spill response procedures, installation of emergency showers and eyewash stations, and training personnel in their proper use. Neutralization agents, such as sodium bicarbonate or calcium carbonate, should be readily available for spill containment and cleanup.
Ventilation systems play a crucial role in maintaining a safe working environment. Local exhaust ventilation should be employed in areas where HCl catalysts are handled to remove acid vapors and maintain air quality. Regular monitoring of air quality and the effectiveness of ventilation systems is essential.
Training and education form the backbone of effective safety protocols. All personnel involved in handling HCl catalysts must receive comprehensive training on the hazards, proper handling techniques, emergency procedures, and the use of PPE. Regular refresher courses and safety drills help maintain a high level of preparedness and awareness.
Waste management and disposal procedures for HCl catalysts and related materials must be carefully designed and implemented. This includes proper neutralization techniques, segregation of waste streams, and compliance with local and national regulations governing hazardous waste disposal.
Regular safety audits and risk assessments should be conducted to identify potential hazards and areas for improvement in safety protocols. This proactive approach helps in continuously refining and updating safety measures to address evolving challenges and incorporate best practices in HCl catalyst handling.
Personal protective equipment (PPE) forms the first line of defense in HCl catalyst handling. Workers must wear appropriate acid-resistant gloves, protective eyewear, face shields, and chemical-resistant clothing. Respiratory protection, such as acid gas respirators or self-contained breathing apparatus (SCBA), may be required depending on the concentration and potential for vapor exposure.
Proper storage and containment of HCl catalysts are crucial. These materials should be stored in corrosion-resistant containers in well-ventilated areas, away from incompatible substances. Secondary containment systems are essential to prevent spills and leaks from spreading. Regular inspections of storage areas and containment systems help ensure their integrity and effectiveness.
Handling procedures for HCl catalysts must be meticulously designed and followed. This includes using appropriate transfer equipment, such as corrosion-resistant pumps and piping systems. Closed-loop transfer systems are preferred to minimize exposure risks. Proper labeling of containers and process equipment is essential for clear identification and communication of hazards.
Emergency response planning is a critical component of safety protocols. This includes the development of spill response procedures, installation of emergency showers and eyewash stations, and training personnel in their proper use. Neutralization agents, such as sodium bicarbonate or calcium carbonate, should be readily available for spill containment and cleanup.
Ventilation systems play a crucial role in maintaining a safe working environment. Local exhaust ventilation should be employed in areas where HCl catalysts are handled to remove acid vapors and maintain air quality. Regular monitoring of air quality and the effectiveness of ventilation systems is essential.
Training and education form the backbone of effective safety protocols. All personnel involved in handling HCl catalysts must receive comprehensive training on the hazards, proper handling techniques, emergency procedures, and the use of PPE. Regular refresher courses and safety drills help maintain a high level of preparedness and awareness.
Waste management and disposal procedures for HCl catalysts and related materials must be carefully designed and implemented. This includes proper neutralization techniques, segregation of waste streams, and compliance with local and national regulations governing hazardous waste disposal.
Regular safety audits and risk assessments should be conducted to identify potential hazards and areas for improvement in safety protocols. This proactive approach helps in continuously refining and updating safety measures to address evolving challenges and incorporate best practices in HCl catalyst handling.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!