Biocompatibility Standards Mapping For Transient And Biodegradable Electronics
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Biodegradable Electronics Evolution and Objectives
Biodegradable electronics represent a revolutionary paradigm shift in the field of electronic devices, evolving from conventional permanent electronics to systems designed to degrade safely after fulfilling their intended functions. The evolution of this technology can be traced back to the early 2000s when researchers began exploring materials that could combine electronic functionality with biodegradability. Initial efforts focused primarily on organic semiconductors and conductive polymers that could degrade under specific environmental conditions.
The field gained significant momentum around 2010 when pioneering work by Rogers et al. demonstrated the first fully transient electronic systems capable of dissolving completely in water. This breakthrough catalyzed rapid advancement in the development of biodegradable substrates, conductors, semiconductors, and dielectrics that could function effectively before undergoing controlled degradation.
By 2015, researchers had expanded the material palette to include silk fibroin, poly(lactic-co-glycolic acid) (PLGA), polycaprolactone (PCL), and magnesium-based conductors, establishing the fundamental building blocks for biodegradable electronic systems. The evolution continued with the integration of more complex functionalities, including sensors, actuators, and energy storage components that could maintain performance during their operational lifetime before degrading.
Recent developments have focused on enhancing the controllability of degradation rates, improving electronic performance during the functional period, and expanding the range of biocompatible materials suitable for various physiological environments. The field has progressed from simple circuits to sophisticated systems capable of wireless communication, energy harvesting, and therapeutic delivery.
The primary objectives of biodegradable electronics research center on several key areas. First, developing comprehensive biocompatibility standards that address the unique challenges posed by materials that change their chemical composition during degradation. Second, establishing reliable testing protocols that can predict both short-term biocompatibility and long-term degradation products' safety profiles.
Additional objectives include creating standardized assessment methodologies for degradation kinetics in various biological environments, ensuring consistent performance during the intended functional lifetime, and minimizing potential toxicity of degradation byproducts. The field also aims to bridge the gap between laboratory demonstrations and clinical applications by addressing scalability, reliability, and regulatory compliance challenges.
The ultimate goal is to enable a new generation of implantable medical devices, environmental sensors, and consumer electronics that can perform their functions effectively before harmlessly disappearing, thereby eliminating the need for retrieval surgeries, reducing electronic waste, and opening new application domains previously constrained by the permanence of conventional electronics.
The field gained significant momentum around 2010 when pioneering work by Rogers et al. demonstrated the first fully transient electronic systems capable of dissolving completely in water. This breakthrough catalyzed rapid advancement in the development of biodegradable substrates, conductors, semiconductors, and dielectrics that could function effectively before undergoing controlled degradation.
By 2015, researchers had expanded the material palette to include silk fibroin, poly(lactic-co-glycolic acid) (PLGA), polycaprolactone (PCL), and magnesium-based conductors, establishing the fundamental building blocks for biodegradable electronic systems. The evolution continued with the integration of more complex functionalities, including sensors, actuators, and energy storage components that could maintain performance during their operational lifetime before degrading.
Recent developments have focused on enhancing the controllability of degradation rates, improving electronic performance during the functional period, and expanding the range of biocompatible materials suitable for various physiological environments. The field has progressed from simple circuits to sophisticated systems capable of wireless communication, energy harvesting, and therapeutic delivery.
The primary objectives of biodegradable electronics research center on several key areas. First, developing comprehensive biocompatibility standards that address the unique challenges posed by materials that change their chemical composition during degradation. Second, establishing reliable testing protocols that can predict both short-term biocompatibility and long-term degradation products' safety profiles.
Additional objectives include creating standardized assessment methodologies for degradation kinetics in various biological environments, ensuring consistent performance during the intended functional lifetime, and minimizing potential toxicity of degradation byproducts. The field also aims to bridge the gap between laboratory demonstrations and clinical applications by addressing scalability, reliability, and regulatory compliance challenges.
The ultimate goal is to enable a new generation of implantable medical devices, environmental sensors, and consumer electronics that can perform their functions effectively before harmlessly disappearing, thereby eliminating the need for retrieval surgeries, reducing electronic waste, and opening new application domains previously constrained by the permanence of conventional electronics.
Market Analysis for Transient Electronic Devices
The transient and biodegradable electronics market is experiencing significant growth, driven by increasing applications in healthcare, environmental monitoring, and consumer electronics sectors. Current market estimates value this emerging segment at approximately 2.3 billion USD in 2023, with projections indicating a compound annual growth rate (CAGR) of 18.7% through 2030, potentially reaching 7.5 billion USD by the end of the decade.
Healthcare applications represent the largest market share, accounting for nearly 45% of current demand. Within this sector, implantable medical devices such as temporary sensors, drug delivery systems, and tissue stimulators demonstrate the strongest growth trajectory. The ability of these devices to naturally degrade after fulfilling their function eliminates the need for secondary removal surgeries, reducing patient risk and healthcare costs.
Environmental monitoring applications constitute the second-largest market segment at 28%, with biodegradable sensors being deployed for soil analysis, water quality assessment, and ecological studies. These devices address growing concerns about electronic waste by offering environmentally responsible alternatives to conventional persistent electronics.
Consumer electronics represents a smaller but rapidly expanding segment at 15%, with transient electronic components being incorporated into temporary use products such as event-specific wearables and limited-use devices. This segment is expected to grow at the fastest rate (22.3% CAGR) as manufacturing costs decrease and consumer awareness of electronic waste issues increases.
Geographically, North America leads the market with 38% share, followed by Europe (31%) and Asia-Pacific (24%). However, the Asia-Pacific region is expected to demonstrate the highest growth rate over the next five years due to increasing healthcare expenditure and manufacturing capabilities in countries like China, Japan, and South Korea.
Key market drivers include stringent environmental regulations regarding electronic waste management, growing healthcare focus on patient comfort and safety, and increasing research funding for biodegradable materials. The primary market restraints remain high development costs, technical limitations in device performance compared to conventional electronics, and challenges in achieving consistent degradation rates across different physiological environments.
Customer demand patterns indicate strong interest from medical device manufacturers seeking FDA-approved biocompatible materials, environmental agencies requiring field-deployable degradable sensors, and consumer electronics companies exploring sustainable product alternatives. The willingness to pay premium prices varies significantly across these segments, with healthcare applications demonstrating the highest price tolerance due to clear clinical benefits and cost-offset potential.
Healthcare applications represent the largest market share, accounting for nearly 45% of current demand. Within this sector, implantable medical devices such as temporary sensors, drug delivery systems, and tissue stimulators demonstrate the strongest growth trajectory. The ability of these devices to naturally degrade after fulfilling their function eliminates the need for secondary removal surgeries, reducing patient risk and healthcare costs.
Environmental monitoring applications constitute the second-largest market segment at 28%, with biodegradable sensors being deployed for soil analysis, water quality assessment, and ecological studies. These devices address growing concerns about electronic waste by offering environmentally responsible alternatives to conventional persistent electronics.
Consumer electronics represents a smaller but rapidly expanding segment at 15%, with transient electronic components being incorporated into temporary use products such as event-specific wearables and limited-use devices. This segment is expected to grow at the fastest rate (22.3% CAGR) as manufacturing costs decrease and consumer awareness of electronic waste issues increases.
Geographically, North America leads the market with 38% share, followed by Europe (31%) and Asia-Pacific (24%). However, the Asia-Pacific region is expected to demonstrate the highest growth rate over the next five years due to increasing healthcare expenditure and manufacturing capabilities in countries like China, Japan, and South Korea.
Key market drivers include stringent environmental regulations regarding electronic waste management, growing healthcare focus on patient comfort and safety, and increasing research funding for biodegradable materials. The primary market restraints remain high development costs, technical limitations in device performance compared to conventional electronics, and challenges in achieving consistent degradation rates across different physiological environments.
Customer demand patterns indicate strong interest from medical device manufacturers seeking FDA-approved biocompatible materials, environmental agencies requiring field-deployable degradable sensors, and consumer electronics companies exploring sustainable product alternatives. The willingness to pay premium prices varies significantly across these segments, with healthcare applications demonstrating the highest price tolerance due to clear clinical benefits and cost-offset potential.
Current Biocompatibility Standards and Challenges
The biocompatibility landscape for transient and biodegradable electronics currently lacks comprehensive standardization specifically tailored to these emerging technologies. Existing standards primarily revolve around ISO 10993 series, which provides a framework for biological evaluation of medical devices but was not designed with consideration for materials that intentionally degrade within the body. This creates significant regulatory challenges as manufacturers must navigate standards developed for permanent implants while designing devices intended for controlled dissolution.
FDA guidance documents offer some direction through the 510(k) and Premarket Approval (PMA) pathways, but these frameworks require substantial adaptation when applied to transient electronics. The fundamental assumption in traditional biocompatibility testing—that device materials remain stable over time—is deliberately contradicted in biodegradable electronics, necessitating new testing paradigms that account for degradation products and their potential biological interactions.
European regulations under the Medical Device Regulation (MDR) similarly lack specific provisions for transient electronics, creating regulatory uncertainty in this important market. The gap between existing standards and the unique characteristics of biodegradable electronics has led to inconsistent testing approaches across the industry, hampering innovation and market entry.
A critical challenge lies in the temporal dimension of biocompatibility assessment. Traditional standards evaluate initial tissue response and long-term effects but provide limited guidance on evaluating changing material compositions during controlled degradation. This necessitates custom testing protocols that often lack regulatory precedent, creating uncertainty in approval processes.
Material characterization presents another significant hurdle, as degradable electronics incorporate novel biomaterials whose degradation pathways and byproducts require thorough investigation. Current standards inadequately address the complex interactions between degrading electronic components and surrounding tissues, particularly regarding potential local pH changes, inflammatory responses, and the fate of dissolved electronic materials.
The absence of standardized degradation testing methodologies further complicates assessment, as in vitro models often poorly correlate with in vivo degradation rates and biological responses. This creates challenges in predicting device performance and safety profiles across patient populations.
Internationally, efforts to address these gaps remain fragmented, with organizations like ASTM International and IEEE working on relevant standards but lacking cohesive frameworks specifically for transient electronics. This regulatory uncertainty represents a significant barrier to commercialization despite the tremendous potential these technologies hold for medical applications.
FDA guidance documents offer some direction through the 510(k) and Premarket Approval (PMA) pathways, but these frameworks require substantial adaptation when applied to transient electronics. The fundamental assumption in traditional biocompatibility testing—that device materials remain stable over time—is deliberately contradicted in biodegradable electronics, necessitating new testing paradigms that account for degradation products and their potential biological interactions.
European regulations under the Medical Device Regulation (MDR) similarly lack specific provisions for transient electronics, creating regulatory uncertainty in this important market. The gap between existing standards and the unique characteristics of biodegradable electronics has led to inconsistent testing approaches across the industry, hampering innovation and market entry.
A critical challenge lies in the temporal dimension of biocompatibility assessment. Traditional standards evaluate initial tissue response and long-term effects but provide limited guidance on evaluating changing material compositions during controlled degradation. This necessitates custom testing protocols that often lack regulatory precedent, creating uncertainty in approval processes.
Material characterization presents another significant hurdle, as degradable electronics incorporate novel biomaterials whose degradation pathways and byproducts require thorough investigation. Current standards inadequately address the complex interactions between degrading electronic components and surrounding tissues, particularly regarding potential local pH changes, inflammatory responses, and the fate of dissolved electronic materials.
The absence of standardized degradation testing methodologies further complicates assessment, as in vitro models often poorly correlate with in vivo degradation rates and biological responses. This creates challenges in predicting device performance and safety profiles across patient populations.
Internationally, efforts to address these gaps remain fragmented, with organizations like ASTM International and IEEE working on relevant standards but lacking cohesive frameworks specifically for transient electronics. This regulatory uncertainty represents a significant barrier to commercialization despite the tremendous potential these technologies hold for medical applications.
Existing Biocompatibility Assessment Methodologies
01 Biodegradable materials for transient electronics
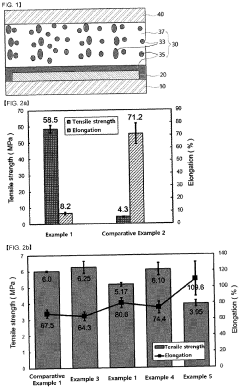
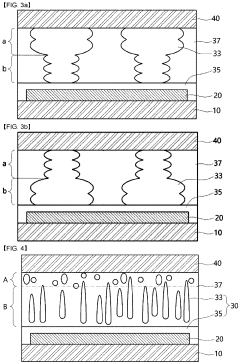
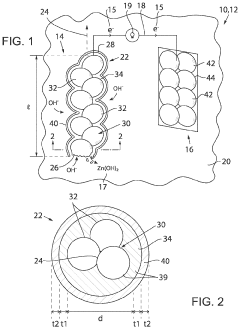
Various biodegradable materials can be used to create transient electronic devices that safely degrade in biological environments. These materials include biodegradable polymers, silk fibroin, magnesium alloys, and other biocompatible substrates that can dissolve or degrade under physiological conditions. The selection of these materials is crucial for ensuring both functionality during the operational period and safe degradation afterward without causing adverse biological reactions.- Biodegradable materials for transient electronics: Various biodegradable materials can be used in transient electronics to ensure biocompatibility. These materials include natural polymers, silk fibroin, magnesium alloys, and zinc oxide that can safely degrade in physiological environments. The degradation products are non-toxic and can be metabolized or excreted by the body, making them suitable for implantable medical devices that don't require surgical removal after their functional lifetime.
- Controlled degradation mechanisms: Transient electronics can be designed with controlled degradation mechanisms that allow for predictable dissolution rates in biological environments. These mechanisms include water-triggered dissolution, enzymatic degradation, and pH-responsive breakdown. By engineering specific degradation pathways, the lifetime of the device can be tailored to match the required therapeutic window, ensuring that the device remains functional for the necessary period before safely degrading.
- Biocompatible encapsulation techniques: Encapsulation techniques play a crucial role in ensuring the biocompatibility of transient electronics. Various biocompatible polymers and hydrogels can be used to encapsulate electronic components, providing a barrier between the electronics and biological tissues. These encapsulation materials protect the electronics from premature degradation while preventing potential inflammatory responses from the host tissue, thus enhancing overall biocompatibility.
- Integration with biological systems: Transient electronics can be designed to integrate seamlessly with biological systems through flexible substrates, conformal contact with tissues, and biomimetic interfaces. These design considerations minimize mechanical mismatch between rigid electronics and soft tissues, reducing inflammation and foreign body responses. Additionally, incorporating bioactive molecules or surface modifications can promote positive biological interactions, such as enhanced cell adhesion or reduced protein adsorption.
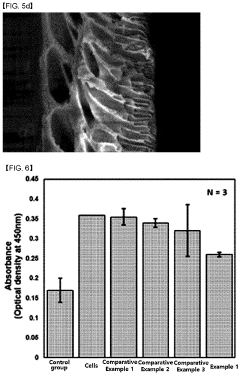
- Biocompatibility testing and assessment: Comprehensive biocompatibility testing protocols are essential for evaluating transient and biodegradable electronics. These assessments include cytotoxicity testing, inflammatory response evaluation, genotoxicity studies, and long-term implantation trials. Advanced imaging techniques and biomarkers can be used to monitor the degradation process in vivo and assess tissue responses over time, ensuring that the materials and their degradation products do not cause adverse biological reactions.
02 Biocompatibility testing and assessment methods
Specific methods and protocols are employed to evaluate the biocompatibility of transient electronic devices. These include in vitro cytotoxicity tests, inflammatory response assessments, implantation studies, and degradation product analysis. These testing procedures ensure that the materials and their degradation byproducts do not cause adverse tissue reactions, immune responses, or toxicity when used in or on the body, which is essential for medical applications of transient electronics.Expand Specific Solutions03 Controlled degradation mechanisms
Transient electronic systems incorporate controlled degradation mechanisms that allow the devices to function for a predetermined period before breaking down. These mechanisms include water-triggered dissolution, enzymatic degradation, pH-responsive materials, and programmable disintegration pathways. By carefully engineering these mechanisms, researchers can create electronics that maintain functionality for the required therapeutic duration before safely degrading, eliminating the need for removal procedures.Expand Specific Solutions04 Encapsulation strategies for biocompatible interfaces
Protective encapsulation techniques are essential for creating biocompatible interfaces between transient electronic components and biological tissues. These strategies include using biocompatible barrier layers, hydrogel coatings, and specialized surface treatments that prevent premature degradation while maintaining biocompatibility. The encapsulation materials must themselves be biocompatible and eventually degrade without releasing harmful substances, while also providing sufficient protection to the functional electronic components during their operational lifetime.Expand Specific Solutions05 Applications in implantable medical devices
Transient and biodegradable electronics are particularly valuable for implantable medical devices that only need to function temporarily. These include biodegradable sensors for post-surgical monitoring, temporary cardiac pacemakers, drug delivery systems, neural interfaces, and wound healing monitors. The biocompatibility of these devices allows them to perform their therapeutic or diagnostic functions and then safely disappear through natural biological processes, eliminating the risks associated with device retrieval surgeries or long-term foreign body responses.Expand Specific Solutions
Leading Organizations in Biocompatible Electronics Development
The biodegradable electronics market is currently in its early growth phase, characterized by rapid technological advancement and expanding applications in medical implants and environmental monitoring. Market size is projected to reach significant scale as these technologies address growing concerns about electronic waste and medical device biocompatibility. Technical maturity varies considerably among key players, with academic institutions like University of Illinois, Northwestern University, and EPFL leading fundamental research, while established corporations such as Philips, Infineon Technologies, and Medtronic (via Covidien) focus on commercialization pathways. Research foundations including Wisconsin Alumni Research Foundation and Purdue Research Foundation are bridging the gap between academic innovation and industry implementation. The regulatory landscape remains complex, with biocompatibility standards still evolving to address the unique challenges of transient electronics that safely degrade within biological systems.
The Board of Trustees of the University of Illinois
Technical Solution: The University of Illinois has pioneered transient electronics with their "born to die" approach, developing silicon-based biodegradable systems that can dissolve completely in biofluids. Their technology platform incorporates water-soluble electronic materials, including silicon nanomembranes, magnesium conductors, and silk or PLGA (poly(lactic-co-glycolic acid)) substrates. They've established comprehensive biocompatibility testing protocols that evaluate both the initial implantation response and the degradation byproducts over time. Their research has demonstrated successful in vivo testing of transient electronic systems that can maintain functionality for predetermined periods before harmlessly dissolving[1][3]. The university has developed standardized dissolution rate models that account for various physiological environments and has created mapping frameworks that correlate material composition with biocompatibility outcomes across different tissue interfaces.
Strengths: Leading expertise in silicon-based transient electronics with proven in vivo performance; established comprehensive testing protocols specific to biodegradable electronics; strong academic-industry partnerships for translation. Weaknesses: Dissolution kinetics can be unpredictable in varied physiological environments; challenges in achieving consistent degradation rates across different batches of materials.
Wisconsin Alumni Research Foundation
Technical Solution: Wisconsin Alumni Research Foundation has developed a biocompatibility mapping framework for transient electronics based on their "controlled degradation" technology platform. Their approach utilizes biodegradable polymers like polyanhydrides and polyesters with tunable degradation rates as substrates for electronic components. They've established a systematic biocompatibility assessment protocol that evaluates both the initial materials and their degradation products across multiple tissue interfaces. Their technology incorporates magnesium-based conductors and semiconductors that can dissolve at predictable rates in physiological environments, with demonstrated applications in implantable sensors and drug delivery systems[5][6]. WARF has developed standardized testing methodologies that correlate material composition and processing parameters with biocompatibility outcomes, creating predictive models for foreign body responses. Their research has established quantitative metrics for evaluating tissue responses to transient electronics, including standardized histological analyses and molecular biomarkers of inflammation.
Strengths: Expertise in polymer-based biodegradable substrates with highly tunable degradation profiles; established predictive models correlating material properties with biocompatibility outcomes; strong intellectual property portfolio. Weaknesses: Challenges in achieving sufficient electronic performance with biodegradable polymers; integration of multiple materials with different degradation rates remains complex.
Key Patents and Research in Transient Electronics
Composite for controlling degradation of transient electronics
PatentPendingEP4316538A1
Innovation
- A composite with a support and a porous polymer layer containing biocompatible oil is used to control the degradation of transient electronics, allowing for controlled release and biodegradation, ensuring flexibility and biocompatibility to prevent organ damage and immune responses.
Biodegradable transient battery built on core-double-shell zinc microparticle networks
PatentActiveUS11791519B2
Innovation
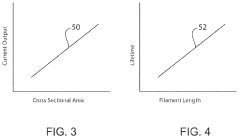
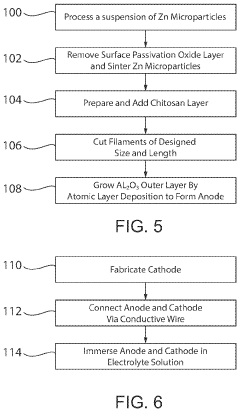
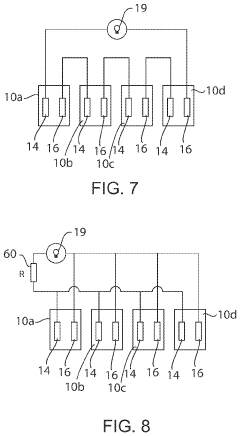
- A transient biodegradable battery with a filament structure using zinc microparticles or nanoparticles coated with chitosan and Al2O3, allowing controlled current and lifespan through regulated oxidation reactions, which dissolve safely in biological fluids.
Regulatory Framework for Implantable Electronic Devices
The regulatory landscape for implantable electronic devices presents a complex framework that manufacturers of transient and biodegradable electronics must navigate. The FDA in the United States classifies most implantable electronic devices as Class III medical devices, requiring the most stringent regulatory controls due to their high-risk nature. These devices typically require Premarket Approval (PMA), which involves comprehensive clinical trials demonstrating both safety and efficacy.
For biodegradable electronics specifically, the FDA has begun developing specialized guidance documents that address their unique characteristics, particularly focusing on degradation profiles and potential metabolite toxicity. The ISO 10993 series remains the cornerstone for biocompatibility testing, with particular emphasis on ISO 10993-1 for evaluation and testing within a risk management process.
The European Union's regulatory approach centers on the Medical Device Regulation (MDR 2017/745), which replaced the previous Medical Device Directive in 2021. The MDR introduces more rigorous requirements for clinical evidence and post-market surveillance, with specific provisions for implantable devices including the requirement for an implant card containing essential information about the device.
In Japan, the Pharmaceuticals and Medical Devices Agency (PMDA) oversees approval through a process that emphasizes quality management systems and often requires local clinical data. China's National Medical Products Administration (NMPA) has similarly strengthened its regulatory framework, implementing a classification system comparable to the FDA's but with unique technical requirements.
Harmonization efforts through the International Medical Device Regulators Forum (IMDRF) have established common principles for medical device regulation globally, though significant regional variations persist. For transient electronics, these variations often center around degradation testing protocols and acceptable residual material thresholds.
A critical regulatory consideration for biodegradable electronics involves the classification of degradation byproducts. Depending on the jurisdiction, these may be regulated as drug components if they exhibit pharmacological activity, potentially triggering additional regulatory requirements under combined product frameworks.
Post-market surveillance requirements are particularly stringent for implantable devices, with manufacturers required to implement comprehensive systems for tracking device performance and adverse events. For biodegradable electronics, these systems must account for the changing risk profile as the device degrades over time.
For biodegradable electronics specifically, the FDA has begun developing specialized guidance documents that address their unique characteristics, particularly focusing on degradation profiles and potential metabolite toxicity. The ISO 10993 series remains the cornerstone for biocompatibility testing, with particular emphasis on ISO 10993-1 for evaluation and testing within a risk management process.
The European Union's regulatory approach centers on the Medical Device Regulation (MDR 2017/745), which replaced the previous Medical Device Directive in 2021. The MDR introduces more rigorous requirements for clinical evidence and post-market surveillance, with specific provisions for implantable devices including the requirement for an implant card containing essential information about the device.
In Japan, the Pharmaceuticals and Medical Devices Agency (PMDA) oversees approval through a process that emphasizes quality management systems and often requires local clinical data. China's National Medical Products Administration (NMPA) has similarly strengthened its regulatory framework, implementing a classification system comparable to the FDA's but with unique technical requirements.
Harmonization efforts through the International Medical Device Regulators Forum (IMDRF) have established common principles for medical device regulation globally, though significant regional variations persist. For transient electronics, these variations often center around degradation testing protocols and acceptable residual material thresholds.
A critical regulatory consideration for biodegradable electronics involves the classification of degradation byproducts. Depending on the jurisdiction, these may be regulated as drug components if they exhibit pharmacological activity, potentially triggering additional regulatory requirements under combined product frameworks.
Post-market surveillance requirements are particularly stringent for implantable devices, with manufacturers required to implement comprehensive systems for tracking device performance and adverse events. For biodegradable electronics, these systems must account for the changing risk profile as the device degrades over time.
Environmental Impact Assessment of Transient Electronics
The environmental impact of transient electronics represents a critical dimension in evaluating their overall sustainability and ecological footprint. Unlike conventional electronic devices that persist in the environment for decades or centuries, transient and biodegradable electronics are designed to decompose naturally after fulfilling their intended functions, potentially offering significant environmental advantages.
Assessment of these technologies reveals several positive environmental implications. Foremost is the reduction in electronic waste (e-waste), which currently constitutes one of the fastest-growing waste streams globally. Conventional electronics contain numerous hazardous materials including heavy metals and flame retardants that can leach into soil and water systems. Transient electronics, by contrast, are engineered to disintegrate into non-toxic components, substantially mitigating long-term contamination risks.
Material selection plays a pivotal role in determining environmental outcomes. Many transient electronic platforms utilize naturally derived polymers such as cellulose, silk fibroin, or poly(lactic-co-glycolic acid) (PLGA) as substrates. These materials generally demonstrate favorable biodegradation profiles with minimal ecological disruption. However, comprehensive life cycle assessments (LCAs) indicate that the environmental benefits vary significantly depending on specific material compositions and manufacturing processes.
Water systems particularly stand to benefit from widespread adoption of transient electronics. Studies examining the dissolution behavior of magnesium, zinc, and silicon-based components in aquatic environments show significantly reduced persistence compared to conventional electronic materials. Nevertheless, potential short-term increases in local ion concentrations during degradation require careful monitoring, especially in sensitive aquatic ecosystems.
Energy consumption during manufacturing remains a challenge for the environmental profile of transient electronics. Current fabrication techniques often require specialized conditions and precision processing that can be energy-intensive. Research indicates that the environmental advantages of biodegradability may be partially offset by higher production-phase energy requirements compared to conventional electronics manufacturing.
Regulatory frameworks for assessing the environmental impact of transient electronics are still evolving. The European Union's Restriction of Hazardous Substances (RoHS) directive and Waste Electrical and Electronic Equipment (WEEE) regulations provide partial guidance, but specialized protocols for evaluating the unique environmental interactions of transient electronics are needed. Several research institutions are developing standardized methodologies for quantifying degradation rates and ecological impacts across diverse environmental conditions.
Assessment of these technologies reveals several positive environmental implications. Foremost is the reduction in electronic waste (e-waste), which currently constitutes one of the fastest-growing waste streams globally. Conventional electronics contain numerous hazardous materials including heavy metals and flame retardants that can leach into soil and water systems. Transient electronics, by contrast, are engineered to disintegrate into non-toxic components, substantially mitigating long-term contamination risks.
Material selection plays a pivotal role in determining environmental outcomes. Many transient electronic platforms utilize naturally derived polymers such as cellulose, silk fibroin, or poly(lactic-co-glycolic acid) (PLGA) as substrates. These materials generally demonstrate favorable biodegradation profiles with minimal ecological disruption. However, comprehensive life cycle assessments (LCAs) indicate that the environmental benefits vary significantly depending on specific material compositions and manufacturing processes.
Water systems particularly stand to benefit from widespread adoption of transient electronics. Studies examining the dissolution behavior of magnesium, zinc, and silicon-based components in aquatic environments show significantly reduced persistence compared to conventional electronic materials. Nevertheless, potential short-term increases in local ion concentrations during degradation require careful monitoring, especially in sensitive aquatic ecosystems.
Energy consumption during manufacturing remains a challenge for the environmental profile of transient electronics. Current fabrication techniques often require specialized conditions and precision processing that can be energy-intensive. Research indicates that the environmental advantages of biodegradability may be partially offset by higher production-phase energy requirements compared to conventional electronics manufacturing.
Regulatory frameworks for assessing the environmental impact of transient electronics are still evolving. The European Union's Restriction of Hazardous Substances (RoHS) directive and Waste Electrical and Electronic Equipment (WEEE) regulations provide partial guidance, but specialized protocols for evaluating the unique environmental interactions of transient electronics are needed. Several research institutions are developing standardized methodologies for quantifying degradation rates and ecological impacts across diverse environmental conditions.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!