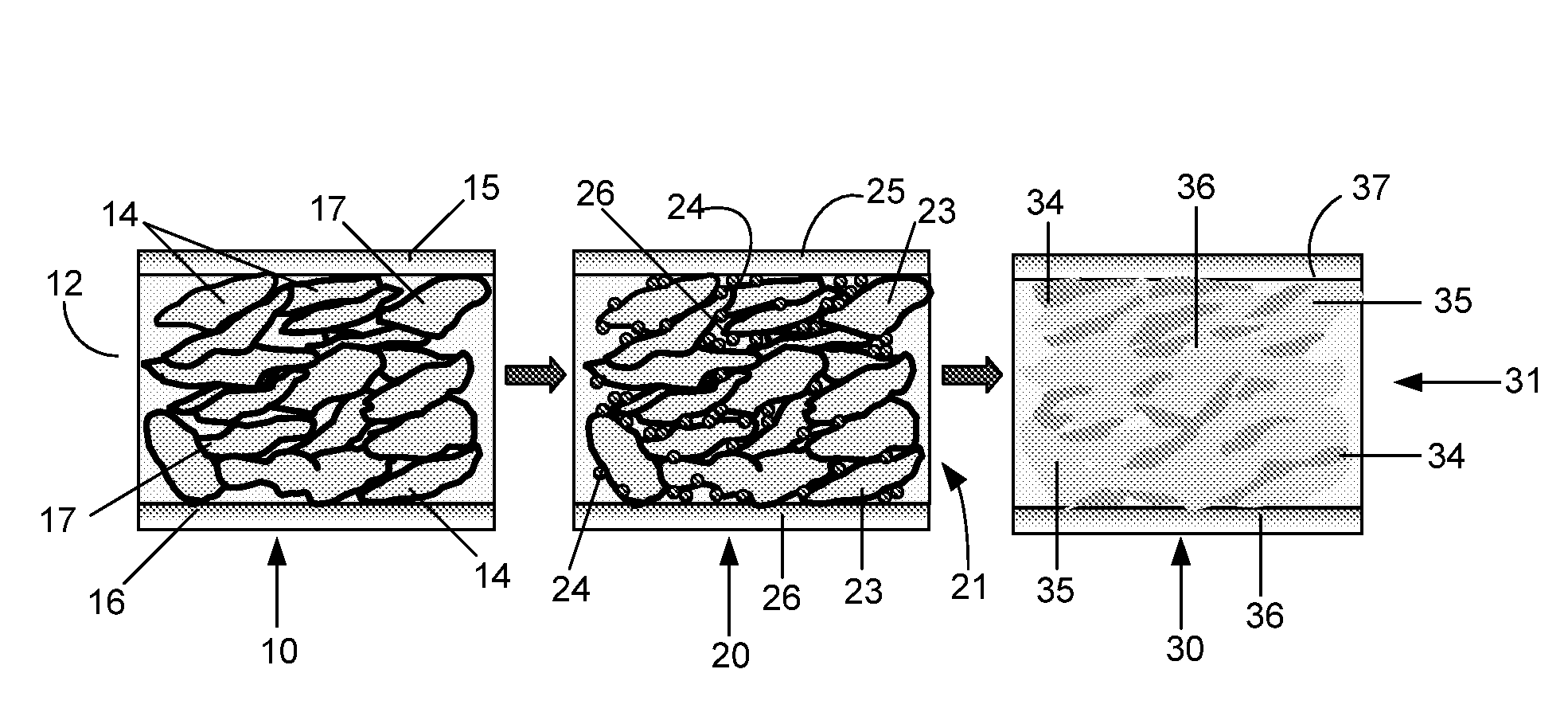
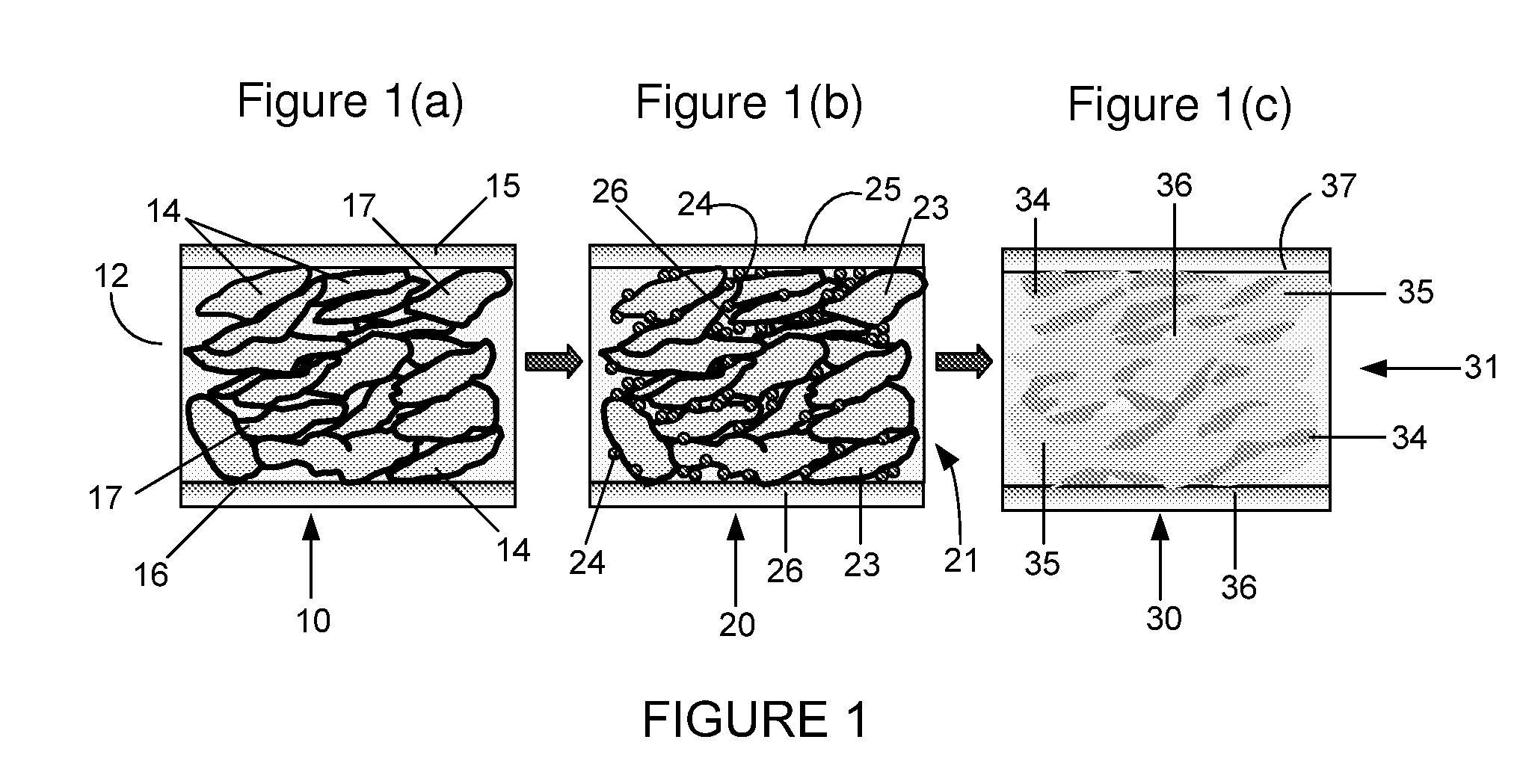
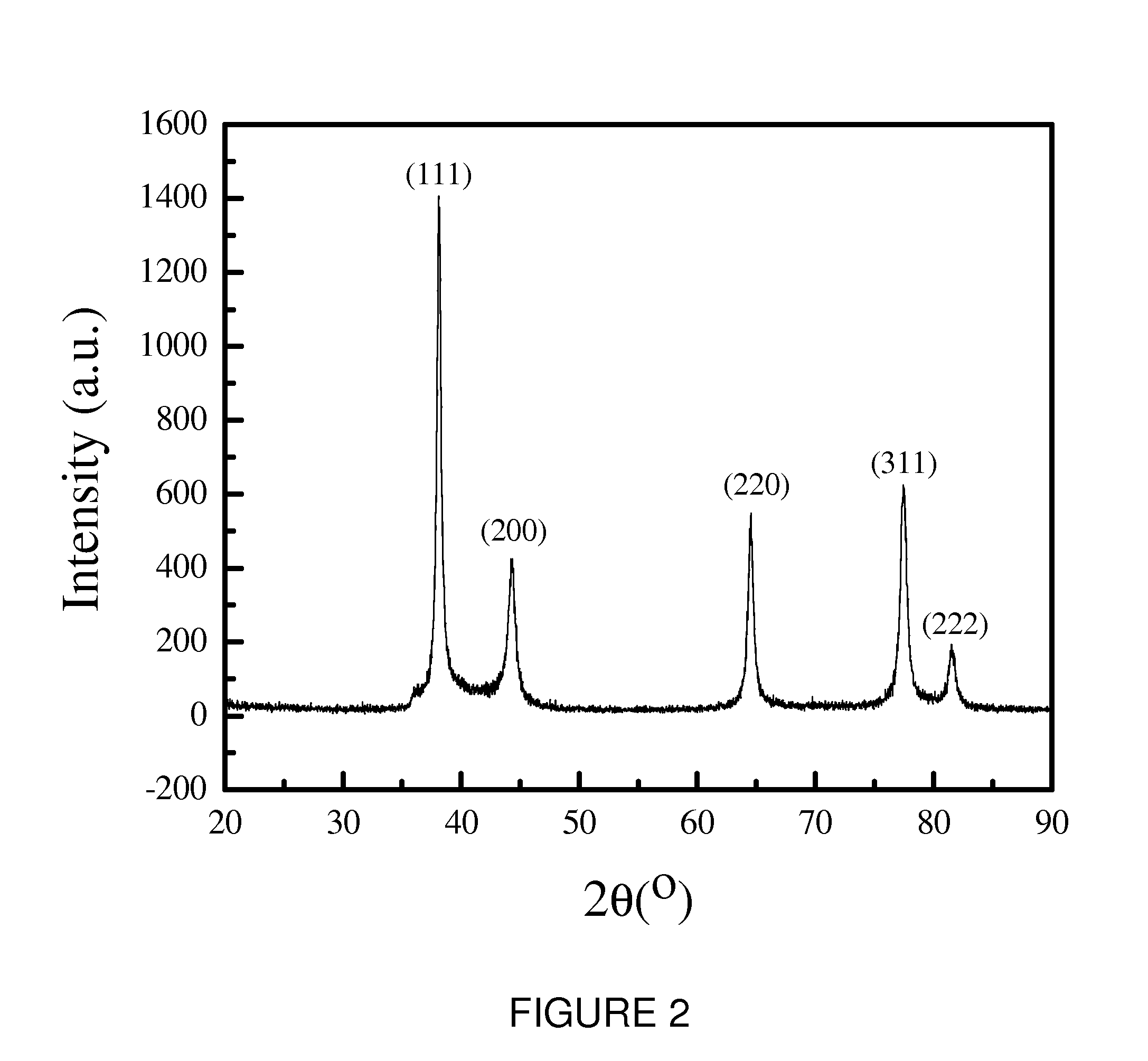
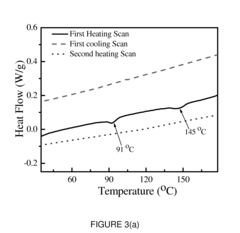
Conductive Polymer Composites: A Comparative Study on Various Catalysts
OCT 23, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Conductive Polymer Evolution and Research Objectives
Conductive polymers have undergone significant evolution since their initial discovery in the 1970s when Alan Heeger, Alan MacDiarmid, and Hideki Shirakawa demonstrated that polyacetylene could conduct electricity when doped with iodine. This groundbreaking work, which earned them the Nobel Prize in Chemistry in 2000, opened a new frontier in materials science by bridging the gap between polymers and conductors.
The 1980s witnessed the development of more stable conductive polymers such as polypyrrole, polyaniline, and polythiophene, which addressed the oxidative instability issues of polyacetylene. During the 1990s, research shifted toward enhancing processability and solubility, leading to the creation of regioregular poly(3-alkylthiophenes) and water-dispersible polyaniline derivatives.
The early 2000s marked the emergence of conductive polymer composites (CPCs), combining conductive polymers with various fillers to achieve synergistic properties. This period saw significant advancements in understanding percolation thresholds and interfacial interactions between polymer matrices and conductive fillers.
In the last decade, research has increasingly focused on the role of catalysts in CPC synthesis, as they critically influence polymer chain length, defect density, and ultimately, electrical conductivity. Traditional metal catalysts like palladium and nickel have been extensively studied, while more recent innovations include organocatalysts and biocatalytic systems that offer more sustainable synthesis routes.
The current technological trajectory is moving toward nanoscale control of polymer morphology and the development of stimuli-responsive CPCs that can change their conductivity in response to external triggers. Additionally, there is growing interest in self-healing CPCs and those with hierarchical structures for enhanced performance.
This technical research aims to comprehensively compare various catalytic systems used in CPC synthesis, evaluating their impact on electrical conductivity, mechanical properties, and processing characteristics. The study will examine traditional metal catalysts, emerging organocatalysts, and novel hybrid systems to establish correlations between catalyst properties and resulting CPC performance.
The research objectives include: quantifying the influence of different catalyst types on conductivity enhancement; determining optimal catalyst concentrations for various polymer matrices; investigating the relationship between catalyst structure and polymer morphology; assessing the environmental and economic sustainability of different catalytic approaches; and developing predictive models for rational catalyst selection in CPC design.
The 1980s witnessed the development of more stable conductive polymers such as polypyrrole, polyaniline, and polythiophene, which addressed the oxidative instability issues of polyacetylene. During the 1990s, research shifted toward enhancing processability and solubility, leading to the creation of regioregular poly(3-alkylthiophenes) and water-dispersible polyaniline derivatives.
The early 2000s marked the emergence of conductive polymer composites (CPCs), combining conductive polymers with various fillers to achieve synergistic properties. This period saw significant advancements in understanding percolation thresholds and interfacial interactions between polymer matrices and conductive fillers.
In the last decade, research has increasingly focused on the role of catalysts in CPC synthesis, as they critically influence polymer chain length, defect density, and ultimately, electrical conductivity. Traditional metal catalysts like palladium and nickel have been extensively studied, while more recent innovations include organocatalysts and biocatalytic systems that offer more sustainable synthesis routes.
The current technological trajectory is moving toward nanoscale control of polymer morphology and the development of stimuli-responsive CPCs that can change their conductivity in response to external triggers. Additionally, there is growing interest in self-healing CPCs and those with hierarchical structures for enhanced performance.
This technical research aims to comprehensively compare various catalytic systems used in CPC synthesis, evaluating their impact on electrical conductivity, mechanical properties, and processing characteristics. The study will examine traditional metal catalysts, emerging organocatalysts, and novel hybrid systems to establish correlations between catalyst properties and resulting CPC performance.
The research objectives include: quantifying the influence of different catalyst types on conductivity enhancement; determining optimal catalyst concentrations for various polymer matrices; investigating the relationship between catalyst structure and polymer morphology; assessing the environmental and economic sustainability of different catalytic approaches; and developing predictive models for rational catalyst selection in CPC design.
Market Applications and Demand Analysis for Conductive Polymers
The global market for conductive polymer composites has witnessed substantial growth over the past decade, driven primarily by increasing demand in electronics, automotive, and healthcare sectors. Current market valuation stands at approximately 3.5 billion USD with a compound annual growth rate of 8.2% projected through 2028, according to industry analyses. This growth trajectory is particularly pronounced in regions with advanced manufacturing capabilities such as North America, Europe, and East Asia.
Consumer electronics represents the largest application segment, accounting for nearly 40% of the total market share. The miniaturization trend in electronic devices has intensified the need for conductive polymers that can replace traditional metallic components while maintaining electrical performance in smaller form factors. Smartphone manufacturers, in particular, have shown increasing interest in conductive polymer composites for flexible displays, touch sensors, and internal components.
The automotive industry presents another significant market opportunity, especially with the accelerating transition toward electric vehicles (EVs). Conductive polymer composites are increasingly utilized in battery systems, sensors, and lightweight components that contribute to improved energy efficiency. The EV market's projected growth rate of 25% annually through 2030 will likely create substantial demand for advanced conductive materials.
Healthcare applications represent an emerging but rapidly growing segment. Medical device manufacturers are incorporating conductive polymers into wearable health monitors, implantable devices, and diagnostic equipment. The biocompatibility of certain conductive polymer formulations makes them particularly valuable in this sector, with market analysts predicting a 12% annual growth rate for medical applications.
Regional market analysis reveals that Asia-Pacific currently dominates consumption, accounting for approximately 45% of global demand, followed by North America (28%) and Europe (22%). China, in particular, has emerged as both a major producer and consumer, driven by its expansive electronics manufacturing ecosystem and government initiatives promoting advanced materials development.
Customer requirements across these markets consistently emphasize several key performance attributes: consistent conductivity across varying environmental conditions, mechanical flexibility, durability, ease of processing, and increasingly, sustainability credentials. The catalyst systems used in conductive polymer synthesis significantly impact these properties, creating market differentiation opportunities for manufacturers who can optimize catalyst performance for specific applications.
Price sensitivity varies considerably across application segments, with consumer electronics manufacturers typically prioritizing cost-effectiveness, while medical and aerospace applications place greater emphasis on performance reliability and consistency. This market segmentation creates opportunities for tiered product offerings based on different catalyst technologies and their resulting performance profiles.
Consumer electronics represents the largest application segment, accounting for nearly 40% of the total market share. The miniaturization trend in electronic devices has intensified the need for conductive polymers that can replace traditional metallic components while maintaining electrical performance in smaller form factors. Smartphone manufacturers, in particular, have shown increasing interest in conductive polymer composites for flexible displays, touch sensors, and internal components.
The automotive industry presents another significant market opportunity, especially with the accelerating transition toward electric vehicles (EVs). Conductive polymer composites are increasingly utilized in battery systems, sensors, and lightweight components that contribute to improved energy efficiency. The EV market's projected growth rate of 25% annually through 2030 will likely create substantial demand for advanced conductive materials.
Healthcare applications represent an emerging but rapidly growing segment. Medical device manufacturers are incorporating conductive polymers into wearable health monitors, implantable devices, and diagnostic equipment. The biocompatibility of certain conductive polymer formulations makes them particularly valuable in this sector, with market analysts predicting a 12% annual growth rate for medical applications.
Regional market analysis reveals that Asia-Pacific currently dominates consumption, accounting for approximately 45% of global demand, followed by North America (28%) and Europe (22%). China, in particular, has emerged as both a major producer and consumer, driven by its expansive electronics manufacturing ecosystem and government initiatives promoting advanced materials development.
Customer requirements across these markets consistently emphasize several key performance attributes: consistent conductivity across varying environmental conditions, mechanical flexibility, durability, ease of processing, and increasingly, sustainability credentials. The catalyst systems used in conductive polymer synthesis significantly impact these properties, creating market differentiation opportunities for manufacturers who can optimize catalyst performance for specific applications.
Price sensitivity varies considerably across application segments, with consumer electronics manufacturers typically prioritizing cost-effectiveness, while medical and aerospace applications place greater emphasis on performance reliability and consistency. This market segmentation creates opportunities for tiered product offerings based on different catalyst technologies and their resulting performance profiles.
Current Catalyst Technologies and Development Challenges
The catalyst landscape for conductive polymer composites (CPCs) has evolved significantly over the past decade, with several distinct technologies emerging as frontrunners. Metal-based catalysts, particularly transition metals such as palladium, platinum, and nickel, continue to dominate commercial applications due to their high activity and selectivity. These catalysts facilitate the polymerization process by lowering activation energy barriers and promoting controlled chain growth, resulting in CPCs with predictable electrical conductivity profiles.
Organometallic catalysts represent another significant category, offering advantages in terms of solubility in organic media and tunable electronic properties. Metallocene catalysts, for instance, have demonstrated exceptional capability in producing CPCs with uniform dispersion of conductive fillers, addressing one of the persistent challenges in composite manufacturing.
Enzyme-based biocatalysts have emerged as an environmentally friendly alternative, though their application remains limited to specific polymer systems. Recent advances in enzyme engineering have improved their stability under industrial processing conditions, making them increasingly viable for specialized CPC applications where biocompatibility is paramount.
Despite these technological advances, several critical challenges persist in catalyst development for conductive polymer composites. Catalyst deactivation remains a significant issue, particularly in systems containing sulfur or nitrogen-rich monomers that can poison catalytic sites. This necessitates higher catalyst loadings, increasing production costs and potentially introducing impurities that compromise electrical performance.
Selectivity challenges also plague current catalyst technologies, with side reactions often leading to branching or crosslinking that can disrupt the conductive pathways within the polymer matrix. This is particularly problematic when incorporating carbon-based conductive fillers such as graphene or carbon nanotubes, which may interact unpredictably with catalytic species.
Scale-up difficulties represent another major hurdle, as catalysts that perform admirably in laboratory settings often exhibit diminished efficiency in industrial-scale reactors. Heat and mass transfer limitations become pronounced at larger scales, affecting reaction kinetics and ultimately the electrical properties of the resulting composites.
Sustainability concerns are increasingly driving research toward catalyst systems with reduced environmental footprints. Current metal-based catalysts often require energy-intensive synthesis procedures and generate hazardous waste streams. Additionally, many high-performance catalysts rely on precious metals with limited global reserves, raising questions about long-term supply security for mass production of conductive polymer composites.
Organometallic catalysts represent another significant category, offering advantages in terms of solubility in organic media and tunable electronic properties. Metallocene catalysts, for instance, have demonstrated exceptional capability in producing CPCs with uniform dispersion of conductive fillers, addressing one of the persistent challenges in composite manufacturing.
Enzyme-based biocatalysts have emerged as an environmentally friendly alternative, though their application remains limited to specific polymer systems. Recent advances in enzyme engineering have improved their stability under industrial processing conditions, making them increasingly viable for specialized CPC applications where biocompatibility is paramount.
Despite these technological advances, several critical challenges persist in catalyst development for conductive polymer composites. Catalyst deactivation remains a significant issue, particularly in systems containing sulfur or nitrogen-rich monomers that can poison catalytic sites. This necessitates higher catalyst loadings, increasing production costs and potentially introducing impurities that compromise electrical performance.
Selectivity challenges also plague current catalyst technologies, with side reactions often leading to branching or crosslinking that can disrupt the conductive pathways within the polymer matrix. This is particularly problematic when incorporating carbon-based conductive fillers such as graphene or carbon nanotubes, which may interact unpredictably with catalytic species.
Scale-up difficulties represent another major hurdle, as catalysts that perform admirably in laboratory settings often exhibit diminished efficiency in industrial-scale reactors. Heat and mass transfer limitations become pronounced at larger scales, affecting reaction kinetics and ultimately the electrical properties of the resulting composites.
Sustainability concerns are increasingly driving research toward catalyst systems with reduced environmental footprints. Current metal-based catalysts often require energy-intensive synthesis procedures and generate hazardous waste streams. Additionally, many high-performance catalysts rely on precious metals with limited global reserves, raising questions about long-term supply security for mass production of conductive polymer composites.
Comparative Analysis of Catalyst Performance in Polymer Composites
01 Carbon-based conductive polymer composites
Carbon-based materials such as carbon nanotubes, graphene, and carbon black can be incorporated into polymer matrices to create conductive composites. These materials provide excellent electrical conductivity while maintaining the mechanical properties of the polymer. The resulting composites can be used in various applications including electromagnetic shielding, antistatic materials, and flexible electronics. The dispersion method and concentration of carbon materials significantly affect the conductivity and performance of the final composite.- Conductive polymer composites with carbon-based fillers: Carbon-based materials such as carbon nanotubes, graphene, and carbon black are commonly used as conductive fillers in polymer composites. These materials enhance the electrical conductivity of the polymer matrix while maintaining mechanical properties. The incorporation of these carbon-based fillers creates a conductive network within the polymer, allowing for efficient electron transport. These composites find applications in electromagnetic shielding, antistatic materials, and flexible electronics.
- Metal-polymer conductive composites: Metal particles or nanostructures can be incorporated into polymer matrices to create conductive composites. Materials such as silver, copper, and nickel are commonly used due to their high electrical conductivity. These metal-polymer composites offer advantages including tunable conductivity, processability, and corrosion resistance compared to pure metals. Applications include printed electronics, conductive adhesives, and sensors where both electrical conductivity and polymer properties are desired.
- Self-healing conductive polymer composites: Self-healing conductive polymer composites incorporate mechanisms that allow the material to restore electrical conductivity after mechanical damage. These composites typically contain microcapsules with healing agents or utilize reversible chemical bonds that can reform after being broken. The self-healing capability extends the lifespan of electronic devices and reduces maintenance costs. These materials are particularly valuable in wearable electronics, flexible displays, and applications subject to mechanical stress or strain.
- Thermally conductive polymer composites: Polymer composites can be engineered to exhibit enhanced thermal conductivity while maintaining electrical insulation properties. These materials incorporate fillers such as boron nitride, aluminum oxide, or specialized carbon structures that facilitate heat transfer without creating electrical pathways. Thermally conductive polymer composites are essential in electronic packaging, heat sinks, and thermal management systems where heat dissipation is critical but electrical insulation must be maintained.
- Stimuli-responsive conductive polymer composites: These advanced composites change their electrical, mechanical, or optical properties in response to external stimuli such as temperature, pH, light, or mechanical force. The responsive behavior is achieved through the incorporation of specialized polymers or additives that undergo reversible structural changes. Applications include smart sensors, actuators, and adaptive electronic systems that can respond to environmental changes. These materials enable the development of self-regulating systems and interactive devices with programmable properties.
02 Metal-polymer conductive composites
Metal particles or nanowires, particularly silver, copper, and gold, can be dispersed within polymer matrices to create conductive composites. These metal-polymer composites offer high electrical conductivity and can be formulated to maintain flexibility. The size, shape, and distribution of metal particles significantly impact the conductivity threshold and mechanical properties. These composites are widely used in printed electronics, conductive adhesives, and electromagnetic interference shielding applications.Expand Specific Solutions03 Intrinsically conductive polymers in composites
Intrinsically conductive polymers such as polyaniline, polypyrrole, and PEDOT:PSS can be blended with conventional polymers to create conductive composites. These materials offer conductivity without requiring additional conductive fillers. The conductivity can be tuned by controlling the doping level, processing conditions, and polymer chain alignment. These composites are particularly valuable in applications requiring biocompatibility, such as biosensors, or in applications where transparency and flexibility are needed.Expand Specific Solutions04 Thermal management in conductive polymer composites
Conductive polymer composites can be engineered for enhanced thermal conductivity alongside electrical conductivity. By incorporating materials such as boron nitride, aluminum oxide, or specific carbon structures, these composites can efficiently dissipate heat while maintaining electrical properties. This dual functionality makes them valuable in electronic packaging, battery components, and thermal interface materials. The orientation and distribution of thermally conductive fillers are critical for optimizing heat transfer pathways within the composite.Expand Specific Solutions05 Processing techniques for conductive polymer composites
Various processing techniques can be employed to enhance the conductivity and performance of polymer composites. These include solution blending, melt mixing, in-situ polymerization, and layer-by-layer assembly. Surface functionalization of conductive fillers can improve their dispersion and interfacial bonding with the polymer matrix. Additionally, techniques such as alignment of conductive fillers through electric or magnetic fields during processing can create anisotropic conductivity. The choice of processing method significantly impacts the final electrical, mechanical, and thermal properties of the composite.Expand Specific Solutions
Leading Manufacturers and Research Institutions in Polymer Catalysis
The conductive polymer composites market is currently in a growth phase, characterized by increasing applications in electronics, automotive, and energy sectors. The market size is projected to expand significantly due to rising demand for lightweight, flexible, and cost-effective conductive materials. Technologically, the field shows varying maturity levels across different catalyst systems. Industry leaders like Dow Global Technologies, BASF, and ExxonMobil are advancing commercial applications, while research institutions such as Zhejiang University and North Carolina State University are pioneering fundamental innovations. Companies including Toyota, Honda, and LG Chem are integrating these materials into next-generation products, particularly for energy storage and automotive applications. The competitive landscape features both established petrochemical giants and specialized materials science companies like Rieke Metals, creating a dynamic environment for technological advancement.
Dow Global Technologies LLC
Technical Solution: Dow has pioneered advanced catalyst technologies for conductive polymer composites focusing on polyolefin-based systems with controlled dispersion of conductive fillers. Their proprietary "Catalyst-Assisted Dispersion Technology" (CADT) uses functionalized metallocene catalysts that simultaneously polymerize the matrix and create chemical affinity sites for conductive fillers. This approach enables uniform dispersion of carbon nanotubes and graphene at lower loading levels (typically 2-5 wt% compared to conventional 8-15 wt%). Dow's catalyst systems feature specially designed single-site catalysts with pendant functional groups that interact with conductive fillers during polymerization, preventing agglomeration. The technology creates hierarchical conductive networks with percolation thresholds as low as 0.5 vol%, resulting in composites with conductivity values of 10^-2 to 10^0 S/cm while maintaining excellent mechanical properties and processability.
Strengths: Significantly lower percolation thresholds reducing the amount of expensive conductive fillers needed; excellent mechanical properties retention; compatibility with existing polymer processing equipment. Weaknesses: Complex catalyst synthesis increases production costs; technology primarily optimized for polyolefin matrices limiting application range; potential catalyst deactivation issues when scaling up production.
Rieke Metals LLC
Technical Solution: Rieke Metals has developed specialized organometallic catalyst systems specifically designed for conductive polymer synthesis. Their technology centers on highly reactive "Rieke metals" (particularly zinc, copper, and nickel) prepared through reduction of metal salts with alkali metals in aprotic solvents. These catalysts enable controlled polymerization of thiophene-based monomers to create regioregular poly(3-alkylthiophenes) with conductivities reaching 10^2 S/cm. The company's proprietary "RCatalyst" series features tailored ligand structures that control polymer molecular weight and regioregularity exceeding 98%. Their latest innovation involves bifunctional catalysts that simultaneously facilitate polymerization and doping, creating self-doped conductive polymers with enhanced environmental stability. Rieke's catalyst systems allow precise control over polymer architecture, enabling the creation of block copolymers with distinct conductive and non-conductive segments for specialized applications in flexible electronics and sensors.
Strengths: Exceptional control over polymer regioregularity leading to superior charge transport properties; ability to create specialized polymer architectures; catalysts that enable room-temperature polymerization reducing energy costs. Weaknesses: Air-sensitive catalyst systems requiring specialized handling; higher cost compared to conventional polymerization methods; limited scalability for some of their more advanced catalyst formulations.
Breakthrough Catalyst Technologies and Patent Landscape
Conductive polymer composites
PatentInactiveUS20080272344A1
Innovation
- A polymer composite is formed by mixing conductive metal flakes and surface-functionalized silver nanoparticles with a polymer precursor, where the nanoparticles are sintered to create a network with reduced contact points, enhancing electrical conductivity without using lead.
Electro-catalyst composition, fuel cell electrode, and membrane-electrode assembly
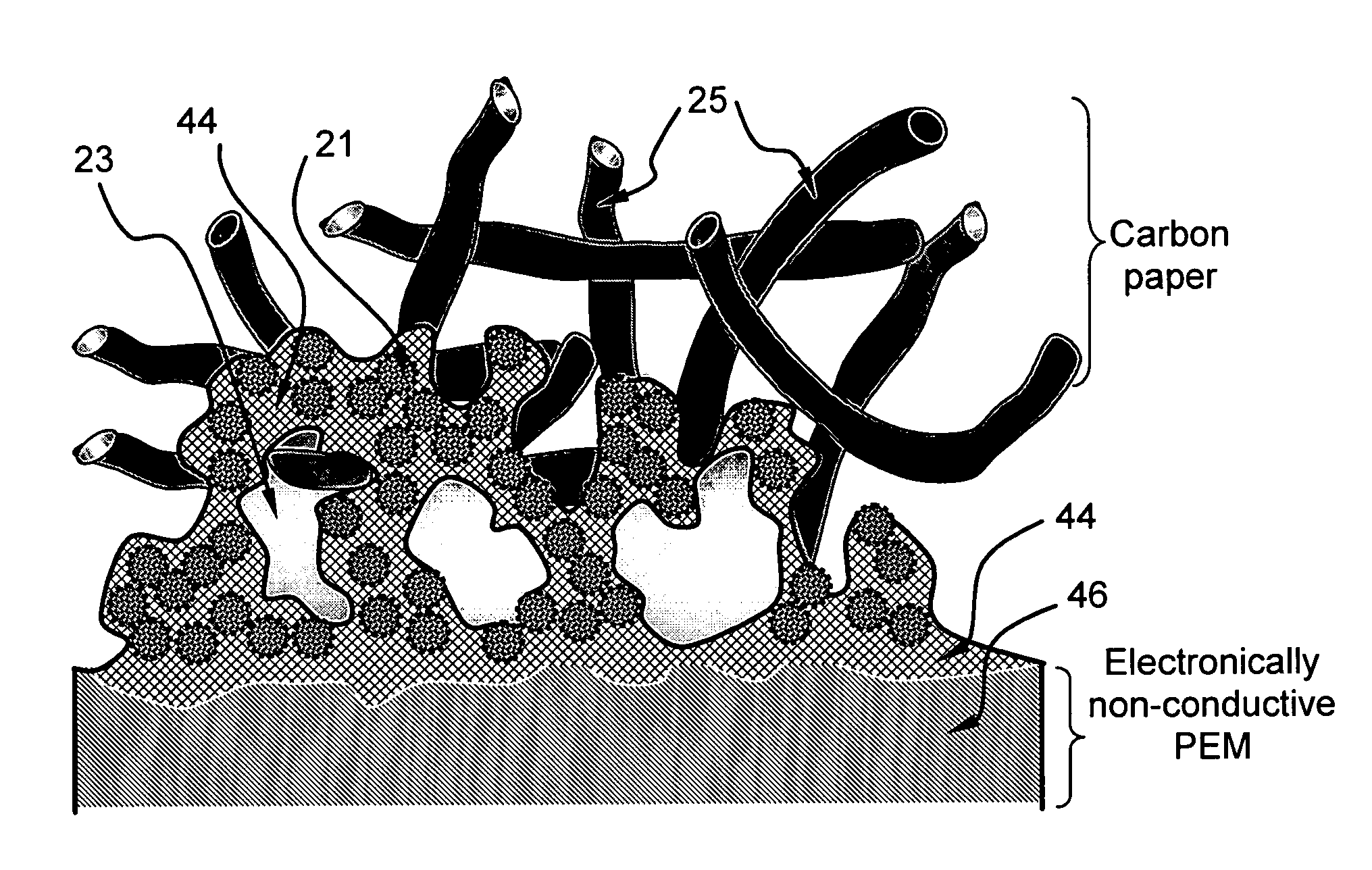
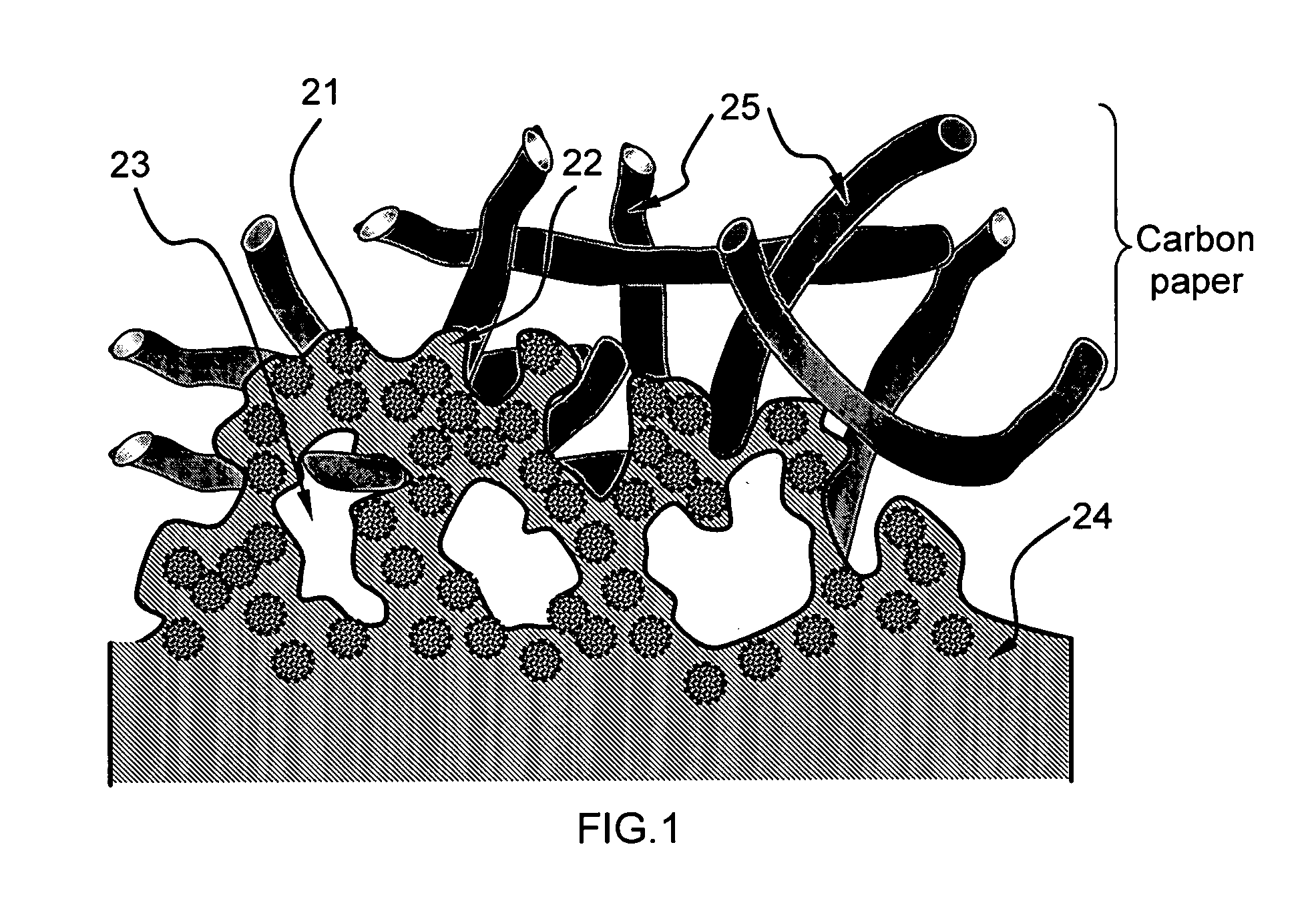
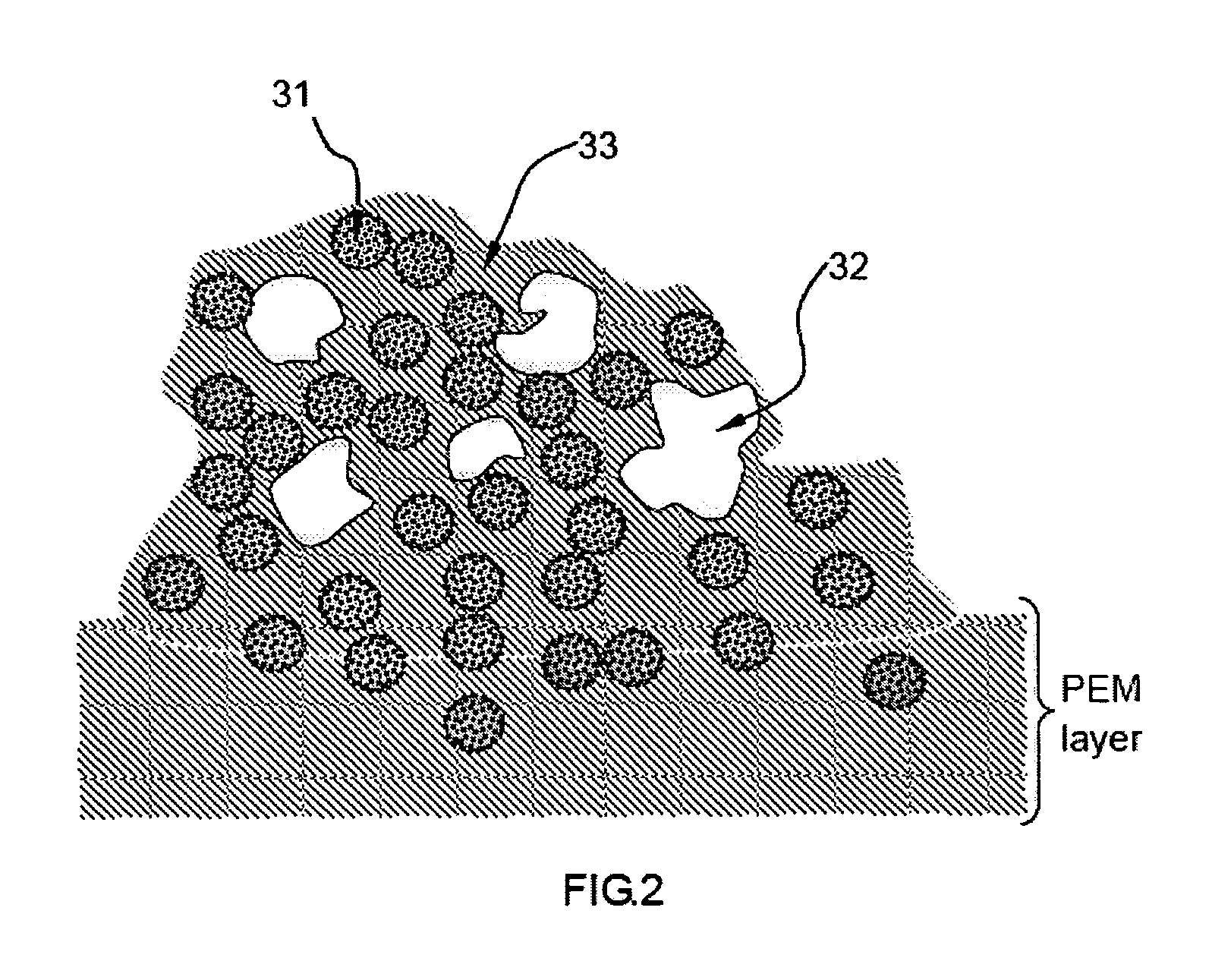
PatentInactiveUS20080063922A1
Innovation
- An electro-catalyst composition comprising a catalyst supported on an electronically conducting carrier, coated with an ion- and electron-conductive polymer that provides both high electronic and ion conductivity, allowing for efficient electron and proton transfer, thereby enhancing catalyst utilization and fuel cell performance.
Environmental Impact and Sustainability of Catalyst Systems
The environmental impact of catalyst systems used in conductive polymer composites (CPCs) represents a critical consideration in their industrial application and future development. Traditional metal-based catalysts, particularly those containing platinum, palladium, and ruthenium, pose significant environmental concerns due to their extraction processes, which often involve extensive mining operations resulting in habitat destruction, soil erosion, and water pollution. Furthermore, the refining of these precious metals generates substantial carbon emissions and requires significant energy inputs, contributing to their overall environmental footprint.
In contrast, newer catalyst systems based on transition metals such as iron, nickel, and copper demonstrate improved environmental profiles. These materials are more abundant, require less energy-intensive extraction methods, and generate fewer toxic byproducts during processing. Recent life cycle assessments indicate that iron-based catalysts can reduce the environmental impact by up to 60% compared to platinum-group metals when used in CPC production processes.
The sustainability of catalyst systems also extends to their recyclability and end-of-life management. Noble metal catalysts offer excellent recovery potential due to their high value, with recovery rates exceeding 95% in specialized facilities. However, this advantage is offset by the energy-intensive nature of the recovery processes. Emerging technologies utilizing biocatalysts and organocatalysts present promising alternatives with inherently lower environmental impacts, as they can be derived from renewable resources and often operate under milder conditions.
Water consumption represents another critical environmental factor in catalyst production and application. Traditional catalyst synthesis methods may require significant quantities of water for washing and purification steps. Recent innovations in green chemistry approaches have reduced water requirements by implementing solvent-free synthesis routes and closed-loop water recycling systems, decreasing water consumption by up to 70% in some manufacturing processes.
The toxicity profiles of different catalyst systems vary considerably, with implications for both environmental and human health. While platinum-group metals exhibit relatively low acute toxicity, their bioaccumulation potential raises long-term ecological concerns. Newer organic catalysts generally demonstrate lower ecotoxicity but may present challenges related to biodegradability and persistence in the environment. Recent regulatory frameworks, including REACH in Europe and similar initiatives globally, have begun addressing these concerns by requiring comprehensive toxicological assessments for catalyst materials.
Looking forward, the development of sustainable catalyst systems for conductive polymer composites will likely focus on earth-abundant elements, biomimetic approaches, and catalyst immobilization techniques that minimize leaching and maximize reusability. These advancements, coupled with green chemistry principles and circular economy approaches, will be essential for reducing the environmental footprint of CPC production while maintaining or enhancing their functional properties.
In contrast, newer catalyst systems based on transition metals such as iron, nickel, and copper demonstrate improved environmental profiles. These materials are more abundant, require less energy-intensive extraction methods, and generate fewer toxic byproducts during processing. Recent life cycle assessments indicate that iron-based catalysts can reduce the environmental impact by up to 60% compared to platinum-group metals when used in CPC production processes.
The sustainability of catalyst systems also extends to their recyclability and end-of-life management. Noble metal catalysts offer excellent recovery potential due to their high value, with recovery rates exceeding 95% in specialized facilities. However, this advantage is offset by the energy-intensive nature of the recovery processes. Emerging technologies utilizing biocatalysts and organocatalysts present promising alternatives with inherently lower environmental impacts, as they can be derived from renewable resources and often operate under milder conditions.
Water consumption represents another critical environmental factor in catalyst production and application. Traditional catalyst synthesis methods may require significant quantities of water for washing and purification steps. Recent innovations in green chemistry approaches have reduced water requirements by implementing solvent-free synthesis routes and closed-loop water recycling systems, decreasing water consumption by up to 70% in some manufacturing processes.
The toxicity profiles of different catalyst systems vary considerably, with implications for both environmental and human health. While platinum-group metals exhibit relatively low acute toxicity, their bioaccumulation potential raises long-term ecological concerns. Newer organic catalysts generally demonstrate lower ecotoxicity but may present challenges related to biodegradability and persistence in the environment. Recent regulatory frameworks, including REACH in Europe and similar initiatives globally, have begun addressing these concerns by requiring comprehensive toxicological assessments for catalyst materials.
Looking forward, the development of sustainable catalyst systems for conductive polymer composites will likely focus on earth-abundant elements, biomimetic approaches, and catalyst immobilization techniques that minimize leaching and maximize reusability. These advancements, coupled with green chemistry principles and circular economy approaches, will be essential for reducing the environmental footprint of CPC production while maintaining or enhancing their functional properties.
Cost-Benefit Analysis of Various Catalyst Technologies
The economic viability of catalyst technologies in conductive polymer composites (CPCs) represents a critical factor in their industrial adoption. When comparing various catalysts used in CPC production, cost considerations must be balanced against performance benefits to determine optimal solutions for different applications.
Metal-based catalysts, particularly those utilizing platinum and palladium, demonstrate superior catalytic efficiency but come with significant cost implications. The initial investment for platinum-based catalysts can be 5-7 times higher than alternatives, though their longevity and reduced replacement frequency may offset this expense over extended operational periods. Palladium catalysts offer a middle-ground solution, providing approximately 85% of platinum's efficiency at roughly 60% of the cost.
Transition metal catalysts, including nickel, copper, and iron-based systems, present a more economical alternative with acquisition costs typically 30-50% lower than noble metal options. However, these savings must be weighed against reduced catalytic activity, which often necessitates higher loading percentages and more frequent replacement cycles. The total cost of ownership analysis reveals that despite lower initial investment, maintenance expenses can accumulate significantly over time.
Non-metal catalysts, particularly carbon-based and organic catalysts, offer the most attractive initial pricing structure, with costs potentially 70-80% lower than platinum-group metals. Their performance limitations in certain high-demand applications, however, may result in efficiency losses that translate to higher energy consumption and operational costs. Recent advancements in nitrogen-doped carbon catalysts show promising cost-performance ratios, achieving 75% of noble metal catalyst efficiency at approximately 25% of the cost.
Scalability considerations further complicate the cost-benefit equation. Noble metal catalysts face supply chain vulnerabilities and price volatility due to limited global reserves, while transition metal and non-metal alternatives generally offer more stable pricing structures and greater supply security. This factor becomes increasingly significant for large-scale industrial applications where material availability directly impacts production continuity.
Lifecycle analysis reveals that while noble metal catalysts carry higher initial costs, their recyclability presents a significant economic advantage. Recovery rates of 85-95% for platinum-group metals substantially improve their long-term economic profile, whereas most non-metal catalysts offer limited recovery potential, effectively functioning as consumable materials.
Emerging hybrid catalyst systems that combine reduced quantities of noble metals with enhanced support structures represent a promising direction for optimizing the cost-benefit ratio, potentially delivering 90% of pure noble metal performance at approximately 40-50% of the cost.
Metal-based catalysts, particularly those utilizing platinum and palladium, demonstrate superior catalytic efficiency but come with significant cost implications. The initial investment for platinum-based catalysts can be 5-7 times higher than alternatives, though their longevity and reduced replacement frequency may offset this expense over extended operational periods. Palladium catalysts offer a middle-ground solution, providing approximately 85% of platinum's efficiency at roughly 60% of the cost.
Transition metal catalysts, including nickel, copper, and iron-based systems, present a more economical alternative with acquisition costs typically 30-50% lower than noble metal options. However, these savings must be weighed against reduced catalytic activity, which often necessitates higher loading percentages and more frequent replacement cycles. The total cost of ownership analysis reveals that despite lower initial investment, maintenance expenses can accumulate significantly over time.
Non-metal catalysts, particularly carbon-based and organic catalysts, offer the most attractive initial pricing structure, with costs potentially 70-80% lower than platinum-group metals. Their performance limitations in certain high-demand applications, however, may result in efficiency losses that translate to higher energy consumption and operational costs. Recent advancements in nitrogen-doped carbon catalysts show promising cost-performance ratios, achieving 75% of noble metal catalyst efficiency at approximately 25% of the cost.
Scalability considerations further complicate the cost-benefit equation. Noble metal catalysts face supply chain vulnerabilities and price volatility due to limited global reserves, while transition metal and non-metal alternatives generally offer more stable pricing structures and greater supply security. This factor becomes increasingly significant for large-scale industrial applications where material availability directly impacts production continuity.
Lifecycle analysis reveals that while noble metal catalysts carry higher initial costs, their recyclability presents a significant economic advantage. Recovery rates of 85-95% for platinum-group metals substantially improve their long-term economic profile, whereas most non-metal catalysts offer limited recovery potential, effectively functioning as consumable materials.
Emerging hybrid catalyst systems that combine reduced quantities of noble metals with enhanced support structures represent a promising direction for optimizing the cost-benefit ratio, potentially delivering 90% of pure noble metal performance at approximately 40-50% of the cost.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!