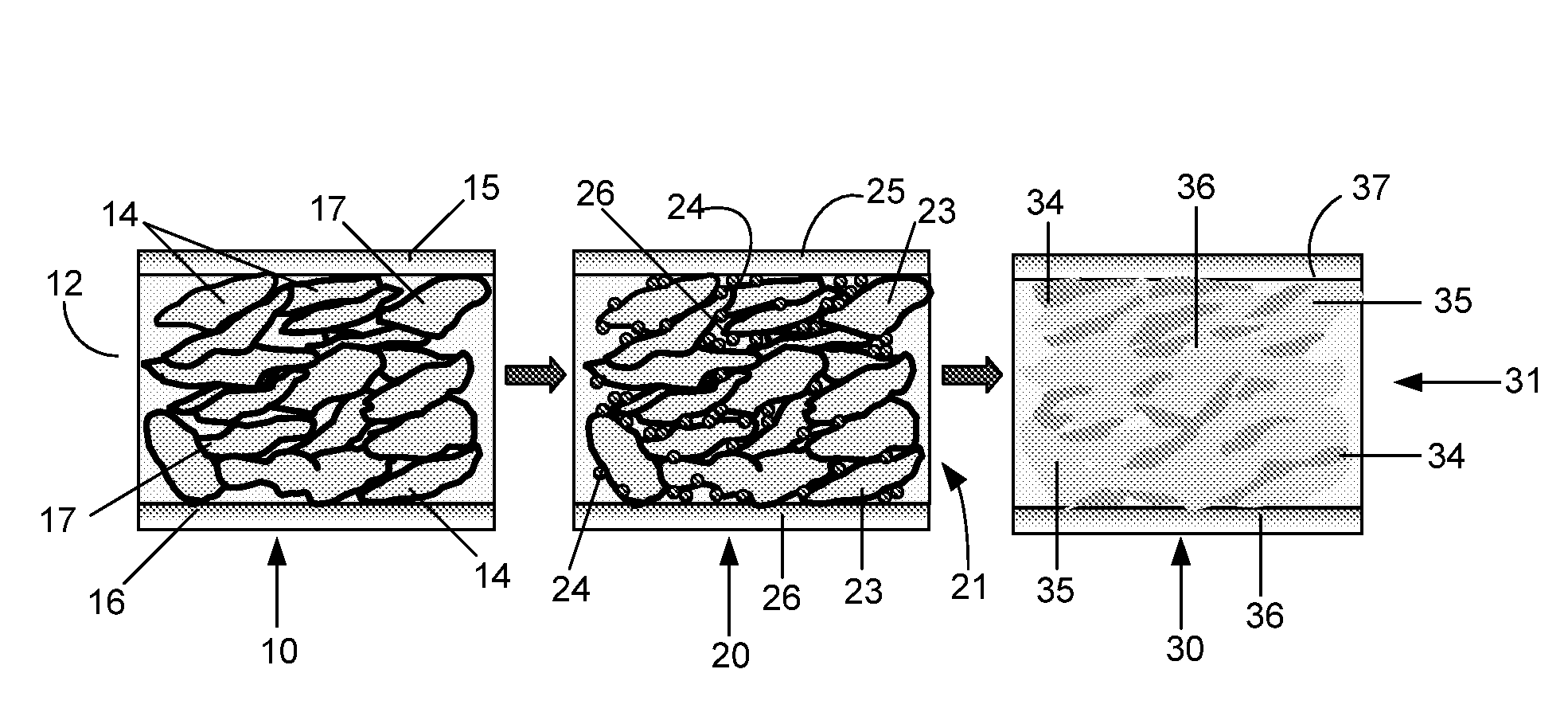
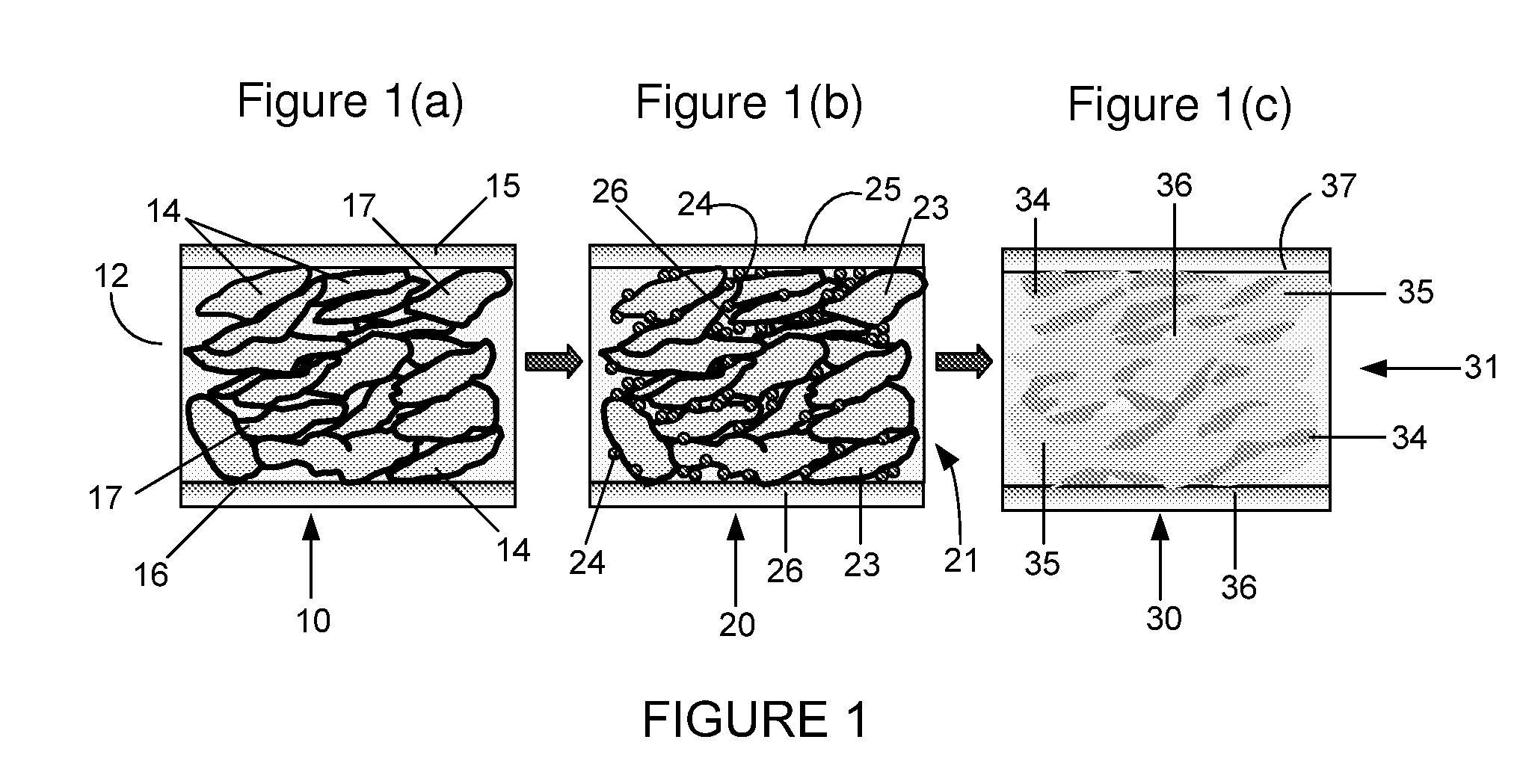
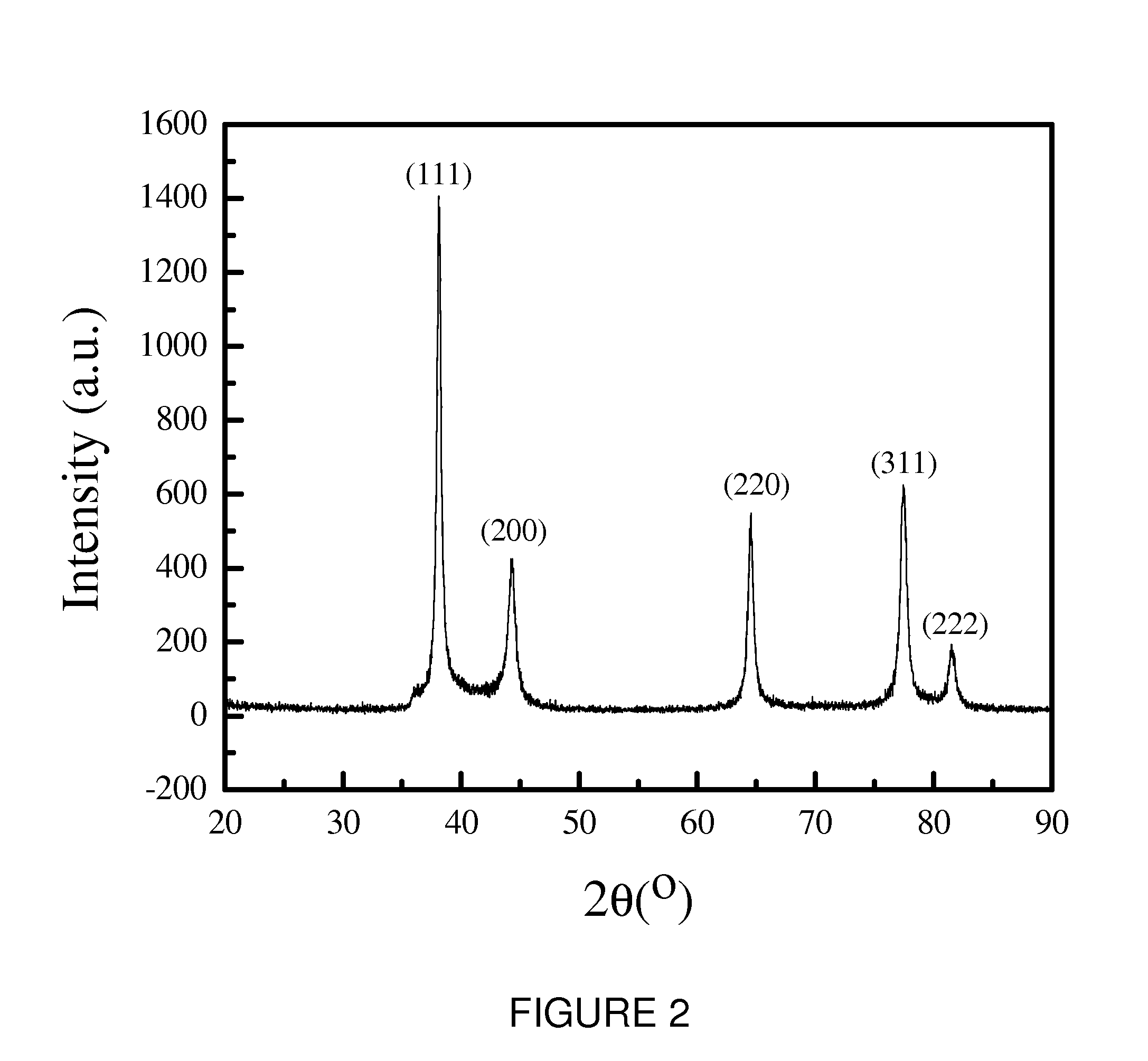
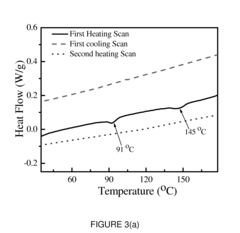
The Interface Chemistry in Conductive Polymer Composites: A Study
OCT 23, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Conductive Polymer Composites Background and Objectives
Conductive polymer composites (CPCs) represent a significant advancement in materials science, combining the electrical conductivity of metals with the processability and lightweight properties of polymers. The evolution of these materials can be traced back to the 1970s with the discovery of conductive polymers by Alan Heeger, Alan MacDiarmid, and Hideki Shirakawa, who were later awarded the Nobel Prize in Chemistry in 2000 for their groundbreaking work.
Over the past five decades, CPCs have evolved from laboratory curiosities to commercially viable materials with applications spanning multiple industries. The fundamental principle behind CPCs involves incorporating conductive fillers such as carbon black, carbon nanotubes, graphene, or metallic particles into polymer matrices to create materials with tailored electrical properties while maintaining the mechanical characteristics of polymers.
The technological trajectory of CPCs has been characterized by continuous improvements in filler dispersion techniques, interface engineering, and polymer matrix selection. Recent advancements have focused on achieving high conductivity at lower filler loadings, enhancing the mechanical-electrical property balance, and developing stimuli-responsive composites that can change their conductivity in response to external triggers.
Current research trends indicate a growing interest in understanding and controlling the interface chemistry between the conductive fillers and polymer matrices, as this interface plays a crucial role in determining the overall performance of the composite. The percolation threshold—the minimum filler concentration required to form a conductive network—remains a central concept in CPC development, with significant efforts directed toward lowering this threshold through innovative processing techniques and filler modifications.
The primary technical objectives in this field include developing CPCs with enhanced electrical conductivity while maintaining mechanical flexibility, improving the stability of electrical properties under mechanical deformation, and achieving uniform dispersion of conductive fillers within polymer matrices. Additionally, there is a growing emphasis on sustainability, with research focusing on bio-based polymers and environmentally friendly processing methods.
Emerging applications in flexible electronics, wearable technology, electromagnetic interference shielding, and sensors are driving the demand for CPCs with increasingly sophisticated property profiles. The interface chemistry between fillers and matrices represents a critical frontier in this field, as it directly influences charge transfer mechanisms, filler dispersion, and composite durability.
This technical research aims to comprehensively investigate the interface chemistry in conductive polymer composites, exploring how molecular interactions at the filler-polymer interface affect macroscopic electrical and mechanical properties, and identifying strategies to optimize these interfaces for next-generation CPC applications.
Over the past five decades, CPCs have evolved from laboratory curiosities to commercially viable materials with applications spanning multiple industries. The fundamental principle behind CPCs involves incorporating conductive fillers such as carbon black, carbon nanotubes, graphene, or metallic particles into polymer matrices to create materials with tailored electrical properties while maintaining the mechanical characteristics of polymers.
The technological trajectory of CPCs has been characterized by continuous improvements in filler dispersion techniques, interface engineering, and polymer matrix selection. Recent advancements have focused on achieving high conductivity at lower filler loadings, enhancing the mechanical-electrical property balance, and developing stimuli-responsive composites that can change their conductivity in response to external triggers.
Current research trends indicate a growing interest in understanding and controlling the interface chemistry between the conductive fillers and polymer matrices, as this interface plays a crucial role in determining the overall performance of the composite. The percolation threshold—the minimum filler concentration required to form a conductive network—remains a central concept in CPC development, with significant efforts directed toward lowering this threshold through innovative processing techniques and filler modifications.
The primary technical objectives in this field include developing CPCs with enhanced electrical conductivity while maintaining mechanical flexibility, improving the stability of electrical properties under mechanical deformation, and achieving uniform dispersion of conductive fillers within polymer matrices. Additionally, there is a growing emphasis on sustainability, with research focusing on bio-based polymers and environmentally friendly processing methods.
Emerging applications in flexible electronics, wearable technology, electromagnetic interference shielding, and sensors are driving the demand for CPCs with increasingly sophisticated property profiles. The interface chemistry between fillers and matrices represents a critical frontier in this field, as it directly influences charge transfer mechanisms, filler dispersion, and composite durability.
This technical research aims to comprehensively investigate the interface chemistry in conductive polymer composites, exploring how molecular interactions at the filler-polymer interface affect macroscopic electrical and mechanical properties, and identifying strategies to optimize these interfaces for next-generation CPC applications.
Market Analysis for Conductive Polymer Applications
The conductive polymer composites (CPCs) market has witnessed substantial growth in recent years, driven by increasing demand for lightweight, flexible, and cost-effective alternatives to traditional metal conductors. The global market for conductive polymers was valued at approximately 3.9 billion USD in 2022 and is projected to reach 7.6 billion USD by 2028, representing a compound annual growth rate of 11.8% during the forecast period.
The automotive sector constitutes the largest application segment for conductive polymer composites, accounting for roughly 32% of the total market share. This dominance is attributed to the growing adoption of electric vehicles and the increasing integration of electronic components in modern automobiles. CPCs are extensively used in electromagnetic interference (EMI) shielding, antistatic applications, and lightweight components that contribute to fuel efficiency.
Electronics and semiconductor industries represent the second-largest market segment, with approximately 28% market share. The miniaturization trend in consumer electronics and the growing demand for flexible displays and wearable technology have significantly boosted the adoption of conductive polymer composites. Interface chemistry innovations have enabled better integration of these materials into complex electronic systems.
Healthcare applications are emerging as the fastest-growing segment, with a projected growth rate of 14.2% through 2028. Medical devices, biosensors, and drug delivery systems increasingly utilize conductive polymers due to their biocompatibility and controllable electrical properties. The interface chemistry between conductive polymers and biological tissues presents unique opportunities for advanced medical applications.
Regionally, Asia-Pacific dominates the market with approximately 42% share, led by China, Japan, and South Korea. This dominance is attributed to the region's robust electronics manufacturing ecosystem and increasing automotive production. North America and Europe follow with 27% and 23% market shares respectively, with significant research activities focused on interface chemistry improvements.
Key market challenges include performance limitations at extreme temperatures, long-term stability issues, and batch-to-batch consistency in production. The interface chemistry between polymer matrices and conductive fillers remains a critical factor affecting these limitations. Recent advancements in surface functionalization techniques have shown promise in addressing these challenges.
Market analysts predict that innovations in interface chemistry will be a primary differentiator for companies in this space, potentially unlocking new application areas such as advanced energy storage, smart textiles, and 3D-printable electronics. Companies investing in research focused on interface optimization are likely to gain significant competitive advantages in the rapidly evolving CPC market landscape.
The automotive sector constitutes the largest application segment for conductive polymer composites, accounting for roughly 32% of the total market share. This dominance is attributed to the growing adoption of electric vehicles and the increasing integration of electronic components in modern automobiles. CPCs are extensively used in electromagnetic interference (EMI) shielding, antistatic applications, and lightweight components that contribute to fuel efficiency.
Electronics and semiconductor industries represent the second-largest market segment, with approximately 28% market share. The miniaturization trend in consumer electronics and the growing demand for flexible displays and wearable technology have significantly boosted the adoption of conductive polymer composites. Interface chemistry innovations have enabled better integration of these materials into complex electronic systems.
Healthcare applications are emerging as the fastest-growing segment, with a projected growth rate of 14.2% through 2028. Medical devices, biosensors, and drug delivery systems increasingly utilize conductive polymers due to their biocompatibility and controllable electrical properties. The interface chemistry between conductive polymers and biological tissues presents unique opportunities for advanced medical applications.
Regionally, Asia-Pacific dominates the market with approximately 42% share, led by China, Japan, and South Korea. This dominance is attributed to the region's robust electronics manufacturing ecosystem and increasing automotive production. North America and Europe follow with 27% and 23% market shares respectively, with significant research activities focused on interface chemistry improvements.
Key market challenges include performance limitations at extreme temperatures, long-term stability issues, and batch-to-batch consistency in production. The interface chemistry between polymer matrices and conductive fillers remains a critical factor affecting these limitations. Recent advancements in surface functionalization techniques have shown promise in addressing these challenges.
Market analysts predict that innovations in interface chemistry will be a primary differentiator for companies in this space, potentially unlocking new application areas such as advanced energy storage, smart textiles, and 3D-printable electronics. Companies investing in research focused on interface optimization are likely to gain significant competitive advantages in the rapidly evolving CPC market landscape.
Interface Chemistry Challenges and Current Status
The interface chemistry in conductive polymer composites (CPCs) represents one of the most critical aspects determining their electrical, mechanical, and thermal properties. Currently, the field faces several significant challenges that limit the widespread application and performance optimization of these materials. The primary challenge lies in achieving consistent and predictable interfacial interactions between polymer matrices and conductive fillers, which directly impacts electron transfer efficiency across phase boundaries.
Research indicates that surface chemistry incompatibility between hydrophobic polymer matrices and often hydrophilic fillers (particularly carbon-based nanomaterials) creates interfacial resistance that significantly diminishes conductivity. This mismatch leads to poor dispersion, agglomeration of conductive particles, and ultimately inconsistent electrical properties throughout the composite material.
Another substantial challenge is the lack of standardized characterization methods for quantitatively assessing interfacial properties at the nanoscale. Current analytical techniques provide limited insight into the dynamic nature of these interfaces, especially under varying environmental conditions such as temperature fluctuations, mechanical stress, or chemical exposure.
The stability of interfacial chemistry over time presents an additional hurdle. Many CPCs exhibit performance degradation due to interfacial changes caused by oxidation, moisture absorption, or polymer chain rearrangement. This aging effect significantly impacts the long-term reliability of devices incorporating these materials, particularly in harsh operating environments.
Recent advances in surface functionalization have shown promise in addressing some of these challenges. Techniques such as plasma treatment, chemical grafting, and nanoparticle surface modification have demonstrated improved compatibility between components. However, these approaches often introduce complexity in manufacturing processes and may alter other desirable properties of the composite.
The current status of interface chemistry research in CPCs is characterized by a shift toward multifunctional interfaces that not only enhance electrical conductivity but also improve mechanical properties and environmental stability. Self-healing interfaces and stimuli-responsive designs represent emerging frontiers in this field, though they remain largely in experimental stages.
Computational modeling of interfacial phenomena has gained significant traction, with molecular dynamics simulations and density functional theory calculations providing insights into atomic-level interactions that experimental methods cannot easily capture. These theoretical approaches are increasingly being coupled with experimental validation to accelerate the development of next-generation interface engineering strategies.
Research indicates that surface chemistry incompatibility between hydrophobic polymer matrices and often hydrophilic fillers (particularly carbon-based nanomaterials) creates interfacial resistance that significantly diminishes conductivity. This mismatch leads to poor dispersion, agglomeration of conductive particles, and ultimately inconsistent electrical properties throughout the composite material.
Another substantial challenge is the lack of standardized characterization methods for quantitatively assessing interfacial properties at the nanoscale. Current analytical techniques provide limited insight into the dynamic nature of these interfaces, especially under varying environmental conditions such as temperature fluctuations, mechanical stress, or chemical exposure.
The stability of interfacial chemistry over time presents an additional hurdle. Many CPCs exhibit performance degradation due to interfacial changes caused by oxidation, moisture absorption, or polymer chain rearrangement. This aging effect significantly impacts the long-term reliability of devices incorporating these materials, particularly in harsh operating environments.
Recent advances in surface functionalization have shown promise in addressing some of these challenges. Techniques such as plasma treatment, chemical grafting, and nanoparticle surface modification have demonstrated improved compatibility between components. However, these approaches often introduce complexity in manufacturing processes and may alter other desirable properties of the composite.
The current status of interface chemistry research in CPCs is characterized by a shift toward multifunctional interfaces that not only enhance electrical conductivity but also improve mechanical properties and environmental stability. Self-healing interfaces and stimuli-responsive designs represent emerging frontiers in this field, though they remain largely in experimental stages.
Computational modeling of interfacial phenomena has gained significant traction, with molecular dynamics simulations and density functional theory calculations providing insights into atomic-level interactions that experimental methods cannot easily capture. These theoretical approaches are increasingly being coupled with experimental validation to accelerate the development of next-generation interface engineering strategies.
Current Interface Chemistry Solutions and Methodologies
01 Interface modification techniques for conductive polymer composites
Various interface modification techniques can be employed to enhance the compatibility and adhesion between conductive polymers and other materials in composites. These techniques include surface functionalization, grafting, and the use of coupling agents to improve the interfacial bonding. By modifying the interface chemistry, the electrical conductivity, mechanical properties, and overall performance of the composite can be significantly improved.- Interface modification techniques for conductive polymer composites: Various interface modification techniques can be employed to enhance the compatibility and interaction between conductive polymers and fillers in composite materials. These techniques include surface functionalization, grafting, and the use of coupling agents to improve the interfacial adhesion. By modifying the interface chemistry, the electrical conductivity, mechanical properties, and overall performance of the composite can be significantly improved.
- Nanoparticle incorporation in conductive polymer matrices: The incorporation of nanoparticles such as carbon nanotubes, graphene, and metal nanoparticles into polymer matrices can create highly conductive composites with enhanced properties. The interface chemistry between these nanoparticles and the polymer matrix plays a crucial role in determining the dispersion quality, electrical percolation threshold, and overall conductivity of the composite material.
- Self-assembly and hierarchical structures in conductive composites: Self-assembly processes and the formation of hierarchical structures at the interface between conductive polymers and fillers can lead to improved electrical pathways and enhanced conductivity. These structures can be controlled through specific interface chemistry approaches, including the use of block copolymers, template-directed assembly, and controlled phase separation techniques.
- Stimuli-responsive interface chemistry for smart conductive composites: Stimuli-responsive interface chemistry enables the development of smart conductive polymer composites that can change their electrical properties in response to external stimuli such as temperature, pH, light, or mechanical stress. This is achieved through the incorporation of dynamic bonds, switchable functional groups, or responsive polymers at the interface between the conductive components.
- Interfacial polymerization and in-situ synthesis methods: Interfacial polymerization and in-situ synthesis methods provide effective approaches for creating well-defined interfaces in conductive polymer composites. These techniques involve the formation of conductive polymers directly at the interface with fillers or substrates, leading to improved interfacial adhesion, reduced contact resistance, and enhanced charge transfer across the composite material.
02 Nanoparticle incorporation in conductive polymer matrices
The incorporation of nanoparticles such as carbon nanotubes, graphene, and metal nanoparticles into polymer matrices can enhance the electrical conductivity and mechanical properties of the resulting composites. The interface chemistry between the nanoparticles and the polymer matrix plays a crucial role in determining the dispersion quality and the formation of conductive networks within the composite structure.Expand Specific Solutions03 Self-assembly and molecular interactions at polymer interfaces
Self-assembly processes and molecular interactions at polymer interfaces can be leveraged to create well-organized conductive polymer composite structures. These interactions, including hydrogen bonding, π-π stacking, and ionic interactions, can be controlled to achieve desired electrical properties and structural arrangements. Understanding and manipulating these molecular-level interactions is essential for designing advanced conductive polymer composites with tailored properties.Expand Specific Solutions04 Surface treatment and functionalization for improved conductivity
Surface treatment and functionalization of components in conductive polymer composites can significantly enhance electrical conductivity by improving charge transfer across interfaces. Techniques such as plasma treatment, chemical oxidation, and the introduction of functional groups can modify the surface chemistry of fillers or polymer matrices, leading to better dispersion and stronger interfacial interactions. These modifications can reduce contact resistance and create more efficient pathways for electron transport.Expand Specific Solutions05 Polymer blending and copolymerization strategies
Polymer blending and copolymerization strategies can be employed to create conductive polymer composites with enhanced interface properties. By combining different polymers or synthesizing copolymers with specific functional groups, the compatibility between components can be improved, leading to better dispersion of conductive fillers and enhanced electrical properties. These approaches allow for the fine-tuning of interface chemistry to achieve desired conductivity while maintaining processability and mechanical properties.Expand Specific Solutions
Leading Research Groups and Industrial Players
The interface chemistry in conductive polymer composites is currently in a growth phase, with the market expected to reach significant expansion due to increasing applications in electronics, automotive, and energy sectors. Research institutions like University of Alabama, Centre National de la Recherche Scientifique, and California Institute of Technology are driving fundamental research, while commercial players such as DuPont, LG Chem, and Samsung Electro Mechanics are advancing practical applications. The technology shows varying maturity levels across different application domains, with established players like Honeywell and Boeing focusing on specialized industrial applications, while academic-industrial partnerships (exemplified by Georgia Tech Research Corp and Korea University Research & Business Foundation) are accelerating innovation in emerging areas like flexible electronics and energy storage solutions.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed advanced conductive polymer composites (CPCs) utilizing their proprietary interface chemistry approach. Their technology focuses on creating controlled interfaces between polymer matrices and conductive fillers through surface functionalization techniques. DuPont's research has demonstrated that by modifying the surface chemistry of carbon-based fillers (carbon nanotubes, graphene) with specific functional groups, they can achieve enhanced dispersion and stronger interfacial bonding with various polymer matrices. Their patented process involves grafting polymer chains directly onto conductive fillers, creating "bridge molecules" that improve compatibility between components. This approach has enabled them to develop CPCs with electrical conductivity at significantly lower filler loadings (typically 2-5 wt% compared to conventional 15-20 wt%), maintaining mechanical properties while achieving percolation thresholds below industry standards.
Strengths: Superior dispersion of conductive fillers at lower loading levels, maintaining mechanical integrity while achieving high conductivity. Proprietary surface modification techniques create stronger polymer-filler interfaces. Weaknesses: Complex manufacturing processes increase production costs; specialized surface treatments may limit scalability for mass production applications.
Centre National de la Recherche Scientifique
Technical Solution: CNRS has pioneered innovative approaches to interface chemistry in conductive polymer composites through their comprehensive research program. Their scientists have developed novel in-situ polymerization techniques where conductive polymers like polyaniline (PANI) and polypyrrole (PPy) are synthesized directly on the surface of nanofillers. This creates intimate molecular-level interfaces between components. Their research has demonstrated that controlling the oxidation state of conductive polymers at these interfaces significantly impacts charge transfer mechanisms. CNRS has also developed advanced characterization methodologies combining spectroscopic techniques (XPS, FTIR) with electrical measurements to correlate interface chemistry with macroscopic electrical properties. Their recent work has focused on creating hierarchical structures within CPCs, where primary and secondary conductive networks form at different length scales, creating multiple pathways for electrical conduction while maintaining structural integrity of the composite.
Strengths: Exceptional fundamental understanding of interface phenomena at molecular level; sophisticated characterization techniques provide deep insights into structure-property relationships. Weaknesses: Research primarily focused on fundamental aspects rather than commercial applications; some techniques require specialized equipment limiting industrial implementation.
Key Patents and Scientific Breakthroughs in Interface Chemistry
Conductive polymer composites
PatentInactiveUS20080272344A1
Innovation
- A polymer composite is formed by mixing conductive metal flakes and surface-functionalized silver nanoparticles with a polymer precursor, where the nanoparticles are sintered to create a network with reduced contact points, enhancing electrical conductivity without using lead.
Highly conductive electrically conductive adhesives
PatentInactiveUS20130056689A1
Innovation
- Incorporating a reducing agent, such as ethylene glycol or diglycidyl ether of polyethylene glycol, to reduce silver carboxylate on silver flakes, forming nano/submicron-sized particles that sinter at low temperatures, creating metallurgical joints and reducing contact resistance, thereby enhancing electrical conductivity.
Sustainability and Environmental Impact Assessment
The environmental implications of conductive polymer composites (CPCs) represent a critical dimension in their development and application trajectory. These materials, while offering significant technological advantages, present complex sustainability challenges that must be systematically addressed. The production processes for CPCs often involve energy-intensive methods and potentially hazardous chemicals, particularly in the synthesis of conductive polymers and the functionalization of fillers to enhance interface chemistry.
When examining the life cycle assessment (LCA) of CPCs, several environmental hotspots emerge. The extraction and processing of raw materials for conductive fillers, especially carbon-based materials like graphene and carbon nanotubes, generate substantial carbon footprints. Additionally, the chemical treatments employed to modify interface properties frequently utilize solvents and reagents with significant environmental toxicity profiles. These processes contribute to various impact categories including global warming potential, acidification, and resource depletion.
Encouragingly, recent research has demonstrated promising approaches to mitigate these environmental concerns. Bio-based polymers derived from renewable resources are increasingly being incorporated into CPC formulations, reducing dependence on petroleum-based polymers. Furthermore, green chemistry principles are being applied to interface modification techniques, with water-based processing and solvent-free methods gaining traction. These innovations substantially reduce volatile organic compound (VOC) emissions and hazardous waste generation.
End-of-life considerations for CPCs present both challenges and opportunities. The intimate mixing of different materials at the interface level complicates recycling efforts, as separation of components becomes technically difficult and economically prohibitive. However, emerging technologies for selective dissolution and chemical recycling show potential for recovering valuable conductive fillers while minimizing environmental impact. Some researchers have also explored the biodegradability of certain CPC formulations, particularly those incorporating natural polymers with engineered degradation pathways.
Regulatory frameworks worldwide are increasingly addressing the environmental aspects of nanomaterials used in CPCs. The European Union's REACH regulation and similar initiatives globally are establishing guidelines for risk assessment and safe handling of nanomaterials throughout their lifecycle. Manufacturers are responding by developing greener interface chemistry approaches that maintain performance while reducing environmental footprint, including solvent-free processing and the elimination of persistent toxic substances from formulations.
The transition toward more sustainable CPCs necessitates a holistic approach that considers interface chemistry optimization not only for performance but also for environmental compatibility. This includes designing interfaces that maintain stability during use but allow for controlled disassembly at end-of-life, facilitating material recovery and recycling. Such design strategies represent a promising frontier in the development of next-generation conductive polymer composites that balance technological advancement with environmental stewardship.
When examining the life cycle assessment (LCA) of CPCs, several environmental hotspots emerge. The extraction and processing of raw materials for conductive fillers, especially carbon-based materials like graphene and carbon nanotubes, generate substantial carbon footprints. Additionally, the chemical treatments employed to modify interface properties frequently utilize solvents and reagents with significant environmental toxicity profiles. These processes contribute to various impact categories including global warming potential, acidification, and resource depletion.
Encouragingly, recent research has demonstrated promising approaches to mitigate these environmental concerns. Bio-based polymers derived from renewable resources are increasingly being incorporated into CPC formulations, reducing dependence on petroleum-based polymers. Furthermore, green chemistry principles are being applied to interface modification techniques, with water-based processing and solvent-free methods gaining traction. These innovations substantially reduce volatile organic compound (VOC) emissions and hazardous waste generation.
End-of-life considerations for CPCs present both challenges and opportunities. The intimate mixing of different materials at the interface level complicates recycling efforts, as separation of components becomes technically difficult and economically prohibitive. However, emerging technologies for selective dissolution and chemical recycling show potential for recovering valuable conductive fillers while minimizing environmental impact. Some researchers have also explored the biodegradability of certain CPC formulations, particularly those incorporating natural polymers with engineered degradation pathways.
Regulatory frameworks worldwide are increasingly addressing the environmental aspects of nanomaterials used in CPCs. The European Union's REACH regulation and similar initiatives globally are establishing guidelines for risk assessment and safe handling of nanomaterials throughout their lifecycle. Manufacturers are responding by developing greener interface chemistry approaches that maintain performance while reducing environmental footprint, including solvent-free processing and the elimination of persistent toxic substances from formulations.
The transition toward more sustainable CPCs necessitates a holistic approach that considers interface chemistry optimization not only for performance but also for environmental compatibility. This includes designing interfaces that maintain stability during use but allow for controlled disassembly at end-of-life, facilitating material recovery and recycling. Such design strategies represent a promising frontier in the development of next-generation conductive polymer composites that balance technological advancement with environmental stewardship.
Standardization and Quality Control Frameworks
Standardization and quality control frameworks are essential for ensuring consistent performance and reliability in conductive polymer composites (CPCs). The interface chemistry between polymer matrices and conductive fillers represents a critical aspect requiring standardized approaches for characterization, measurement, and production.
Currently, several international organizations including ASTM International, ISO, and IEC have developed standards applicable to polymer composites, though specific frameworks for interface chemistry in CPCs remain limited. ASTM D5739 addresses electrical conductivity measurements in conductive composites, while ISO 22007 series covers thermal conductivity testing methods that indirectly reflect interfacial properties.
Quality control protocols for interface chemistry typically involve multi-parameter assessment frameworks. These include contact angle measurements to quantify wetting behaviors, surface energy determinations, and interfacial adhesion strength tests. Standardized procedures for surface functionalization of fillers, which directly impact interface chemistry, have been established by organizations like SEMI for semiconductor applications but require adaptation for broader CPC applications.
Statistical process control (SPC) methodologies have been implemented by industry leaders to monitor interface chemistry parameters during production. These frameworks typically establish control limits for key indicators such as dispersion indices, interfacial shear strength, and electrical percolation thresholds. Companies like DuPont and BASF have developed proprietary quality control systems specifically targeting interface chemistry in their conductive composite product lines.
Emerging quality control approaches incorporate real-time monitoring technologies. Raman spectroscopy and infrared imaging systems can now provide in-line assessment of interfacial bonding during composite manufacturing. These advanced techniques are gradually being incorporated into updated standards, though industry-wide adoption remains in progress.
Certification frameworks for CPC materials increasingly require documentation of interface chemistry parameters. The UL certification process for conductive materials used in electronics now includes specific requirements for interfacial stability under environmental stress conditions. Similarly, automotive industry standards like IATF 16949 have expanded to address interface chemistry quality in conductive composites used in vehicle electronics.
Challenges in standardization persist due to the complex nature of polymer-filler interfaces. Variations in testing conditions, sample preparation methods, and measurement techniques contribute to inconsistencies across laboratories. International efforts to harmonize these approaches are underway through joint working groups between IEEE, IEC, and ISO, focusing specifically on nanomaterial-polymer interfaces in conductive applications.
Currently, several international organizations including ASTM International, ISO, and IEC have developed standards applicable to polymer composites, though specific frameworks for interface chemistry in CPCs remain limited. ASTM D5739 addresses electrical conductivity measurements in conductive composites, while ISO 22007 series covers thermal conductivity testing methods that indirectly reflect interfacial properties.
Quality control protocols for interface chemistry typically involve multi-parameter assessment frameworks. These include contact angle measurements to quantify wetting behaviors, surface energy determinations, and interfacial adhesion strength tests. Standardized procedures for surface functionalization of fillers, which directly impact interface chemistry, have been established by organizations like SEMI for semiconductor applications but require adaptation for broader CPC applications.
Statistical process control (SPC) methodologies have been implemented by industry leaders to monitor interface chemistry parameters during production. These frameworks typically establish control limits for key indicators such as dispersion indices, interfacial shear strength, and electrical percolation thresholds. Companies like DuPont and BASF have developed proprietary quality control systems specifically targeting interface chemistry in their conductive composite product lines.
Emerging quality control approaches incorporate real-time monitoring technologies. Raman spectroscopy and infrared imaging systems can now provide in-line assessment of interfacial bonding during composite manufacturing. These advanced techniques are gradually being incorporated into updated standards, though industry-wide adoption remains in progress.
Certification frameworks for CPC materials increasingly require documentation of interface chemistry parameters. The UL certification process for conductive materials used in electronics now includes specific requirements for interfacial stability under environmental stress conditions. Similarly, automotive industry standards like IATF 16949 have expanded to address interface chemistry quality in conductive composites used in vehicle electronics.
Challenges in standardization persist due to the complex nature of polymer-filler interfaces. Variations in testing conditions, sample preparation methods, and measurement techniques contribute to inconsistencies across laboratories. International efforts to harmonize these approaches are underway through joint working groups between IEEE, IEC, and ISO, focusing specifically on nanomaterial-polymer interfaces in conductive applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!