Electrocatalyst Layer Engineering For Single-Atom Sites
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Electrocatalyst SAC Development Background and Objectives
Single-atom catalysts (SACs) have emerged as a frontier in heterogeneous catalysis research over the past decade, representing a paradigm shift in catalyst design. The evolution of electrocatalyst technology has progressed from traditional bulk materials to nanoparticles, and now to atomically dispersed active sites, marking a significant advancement in maximizing atomic efficiency and selectivity. This technological progression aims to address critical challenges in energy conversion and environmental remediation through more efficient catalytic processes.
The development of single-atom catalysts originated from fundamental studies in the early 2000s, with pioneering work demonstrating the feasibility of stabilizing isolated metal atoms on various supports. However, it wasn't until 2011 that the term "single-atom catalyst" was formally introduced, catalyzing rapid growth in this field. Since then, research has expanded exponentially, with particular focus on electrocatalytic applications for reactions such as hydrogen evolution, oxygen reduction, and CO2 conversion.
The primary objective of single-atom catalyst layer engineering is to develop highly efficient, stable, and scalable electrocatalytic systems that can operate under practical conditions. This involves precise control over the atomic architecture of catalytic sites, optimization of the surrounding coordination environment, and enhancement of electron transfer pathways within the catalyst layer. These improvements aim to overcome the limitations of conventional catalysts, including low active site density, poor stability, and inefficient mass transport.
Current research trends focus on several key aspects: developing synthetic methods for high-density SAC production, understanding structure-performance relationships at the atomic level, designing multi-functional supports that enhance catalytic activity, and creating hierarchical structures that facilitate mass transport. Additionally, in situ characterization techniques are being advanced to provide deeper insights into the dynamic behavior of single-atom sites during catalytic processes.
The technological objectives extend beyond laboratory demonstrations to practical implementation in energy devices such as fuel cells, electrolyzers, and metal-air batteries. This transition requires addressing challenges related to scalable synthesis, long-term stability under operating conditions, and integration with existing device architectures. Furthermore, there is growing interest in developing computational models that can accurately predict the behavior of single-atom catalysts, enabling more rational design approaches.
Industry adoption represents another critical objective, necessitating cost-effective production methods and demonstration of performance advantages over incumbent technologies. This includes reducing dependence on precious metals through the development of earth-abundant alternatives without compromising activity or durability.
The development of single-atom catalysts originated from fundamental studies in the early 2000s, with pioneering work demonstrating the feasibility of stabilizing isolated metal atoms on various supports. However, it wasn't until 2011 that the term "single-atom catalyst" was formally introduced, catalyzing rapid growth in this field. Since then, research has expanded exponentially, with particular focus on electrocatalytic applications for reactions such as hydrogen evolution, oxygen reduction, and CO2 conversion.
The primary objective of single-atom catalyst layer engineering is to develop highly efficient, stable, and scalable electrocatalytic systems that can operate under practical conditions. This involves precise control over the atomic architecture of catalytic sites, optimization of the surrounding coordination environment, and enhancement of electron transfer pathways within the catalyst layer. These improvements aim to overcome the limitations of conventional catalysts, including low active site density, poor stability, and inefficient mass transport.
Current research trends focus on several key aspects: developing synthetic methods for high-density SAC production, understanding structure-performance relationships at the atomic level, designing multi-functional supports that enhance catalytic activity, and creating hierarchical structures that facilitate mass transport. Additionally, in situ characterization techniques are being advanced to provide deeper insights into the dynamic behavior of single-atom sites during catalytic processes.
The technological objectives extend beyond laboratory demonstrations to practical implementation in energy devices such as fuel cells, electrolyzers, and metal-air batteries. This transition requires addressing challenges related to scalable synthesis, long-term stability under operating conditions, and integration with existing device architectures. Furthermore, there is growing interest in developing computational models that can accurately predict the behavior of single-atom catalysts, enabling more rational design approaches.
Industry adoption represents another critical objective, necessitating cost-effective production methods and demonstration of performance advantages over incumbent technologies. This includes reducing dependence on precious metals through the development of earth-abundant alternatives without compromising activity or durability.
Market Analysis for Single-Atom Catalysts
The single-atom catalyst (SAC) market is experiencing rapid growth, driven by increasing demand for sustainable and efficient catalytic solutions across multiple industries. Current market valuations indicate that the global catalyst market exceeds $33 billion, with single-atom catalysts representing an emerging segment projected to grow at a compound annual growth rate of 6.8% through 2030. This growth trajectory is particularly pronounced in regions with strong industrial bases and environmental regulations, including North America, Europe, and East Asia.
The primary market drivers for single-atom catalysts include stringent environmental regulations, growing industrial demand for energy-efficient processes, and the push toward green hydrogen production. The automotive sector represents a significant market, with single-atom catalysts offering superior performance in emission control systems at lower precious metal loadings than conventional catalysts. This provides both environmental and economic benefits that align with increasingly strict vehicle emission standards worldwide.
Chemical manufacturing constitutes another substantial market segment, where single-atom catalysts demonstrate enhanced selectivity and activity for various reactions, potentially reducing energy consumption and waste generation. The pharmaceutical industry is also showing increased interest, particularly for complex synthesis reactions where catalyst selectivity is paramount.
Energy applications represent perhaps the most promising growth sector, with single-atom catalysts showing exceptional performance in fuel cells, water splitting for hydrogen production, and CO2 reduction technologies. The market potential in renewable energy applications alone is estimated to reach $5 billion by 2028, with electrocatalysis applications leading this growth.
Market barriers include high production costs, scalability challenges, and stability issues in industrial conditions. The cost-performance ratio remains a critical factor for widespread commercial adoption, with current production methods often requiring sophisticated equipment and precise control that limit mass production capabilities.
Customer segments can be categorized into three tiers: early adopters (primarily R&D-focused organizations and specialty chemical producers), mainstream industrial users (automotive catalyst manufacturers, chemical companies), and emerging applications (renewable energy developers, carbon capture technology providers).
The competitive landscape features both established catalyst manufacturers investing in SAC technology and specialized startups focused exclusively on single-atom catalyst innovations. Strategic partnerships between academic institutions, technology startups, and industrial end-users are becoming increasingly common, accelerating the commercialization timeline for novel electrocatalyst layer engineering approaches.
The primary market drivers for single-atom catalysts include stringent environmental regulations, growing industrial demand for energy-efficient processes, and the push toward green hydrogen production. The automotive sector represents a significant market, with single-atom catalysts offering superior performance in emission control systems at lower precious metal loadings than conventional catalysts. This provides both environmental and economic benefits that align with increasingly strict vehicle emission standards worldwide.
Chemical manufacturing constitutes another substantial market segment, where single-atom catalysts demonstrate enhanced selectivity and activity for various reactions, potentially reducing energy consumption and waste generation. The pharmaceutical industry is also showing increased interest, particularly for complex synthesis reactions where catalyst selectivity is paramount.
Energy applications represent perhaps the most promising growth sector, with single-atom catalysts showing exceptional performance in fuel cells, water splitting for hydrogen production, and CO2 reduction technologies. The market potential in renewable energy applications alone is estimated to reach $5 billion by 2028, with electrocatalysis applications leading this growth.
Market barriers include high production costs, scalability challenges, and stability issues in industrial conditions. The cost-performance ratio remains a critical factor for widespread commercial adoption, with current production methods often requiring sophisticated equipment and precise control that limit mass production capabilities.
Customer segments can be categorized into three tiers: early adopters (primarily R&D-focused organizations and specialty chemical producers), mainstream industrial users (automotive catalyst manufacturers, chemical companies), and emerging applications (renewable energy developers, carbon capture technology providers).
The competitive landscape features both established catalyst manufacturers investing in SAC technology and specialized startups focused exclusively on single-atom catalyst innovations. Strategic partnerships between academic institutions, technology startups, and industrial end-users are becoming increasingly common, accelerating the commercialization timeline for novel electrocatalyst layer engineering approaches.
Current Status and Challenges in SAC Layer Engineering
Single-atom catalysts (SACs) have emerged as a frontier in heterogeneous catalysis research, offering maximum atom efficiency and unique catalytic properties. The current landscape of SAC layer engineering reveals significant progress alongside persistent challenges that require innovative solutions.
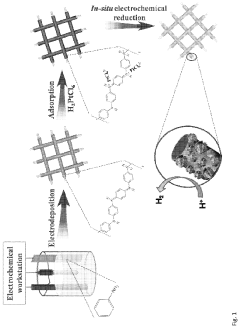
Global research efforts have established several successful approaches for SAC layer engineering. Wet chemistry methods, including impregnation and co-precipitation, remain widely adopted due to their simplicity and scalability. Advanced techniques such as atomic layer deposition (ALD) and chemical vapor deposition (CVD) have demonstrated superior control over atomic dispersion but face limitations in large-scale implementation. Electrochemical deposition has gained traction for in-situ formation of single-atom sites directly on electrode surfaces.
Despite these advances, maintaining the stability of single-atom sites during catalytic reactions presents a formidable challenge. Under reaction conditions, single atoms tend to migrate and aggregate, diminishing catalytic performance. This instability is particularly pronounced in harsh electrochemical environments involving extreme pH values or high current densities.
Another critical challenge lies in achieving precise control over the coordination environment of single atoms. The catalytic performance of SACs depends heavily on the local electronic structure and coordination geometry around the metal center. Current synthetic methods often yield heterogeneous distributions of coordination environments, complicating structure-property relationships and hindering rational design.
The loading density of single atoms represents another significant limitation. Most SAC preparation methods achieve metal loadings below 2 wt%, restricting mass activity and practical applications. Increasing loading while maintaining atomic dispersion remains a delicate balance that few techniques have successfully navigated.
Characterization challenges further complicate progress in this field. Conventional techniques struggle to provide comprehensive information about the exact location, coordination environment, and electronic state of single atoms. Advanced methods like aberration-corrected electron microscopy and X-ray absorption spectroscopy offer valuable insights but require specialized equipment and expertise.
Scalable production represents perhaps the most significant barrier to commercial implementation. Laboratory-scale synthesis methods often involve complex procedures, expensive precursors, or specialized equipment that pose challenges for industrial-scale manufacturing. The development of cost-effective, scalable synthesis routes remains a priority for transitioning SACs from academic research to practical applications.
Global research efforts have established several successful approaches for SAC layer engineering. Wet chemistry methods, including impregnation and co-precipitation, remain widely adopted due to their simplicity and scalability. Advanced techniques such as atomic layer deposition (ALD) and chemical vapor deposition (CVD) have demonstrated superior control over atomic dispersion but face limitations in large-scale implementation. Electrochemical deposition has gained traction for in-situ formation of single-atom sites directly on electrode surfaces.
Despite these advances, maintaining the stability of single-atom sites during catalytic reactions presents a formidable challenge. Under reaction conditions, single atoms tend to migrate and aggregate, diminishing catalytic performance. This instability is particularly pronounced in harsh electrochemical environments involving extreme pH values or high current densities.
Another critical challenge lies in achieving precise control over the coordination environment of single atoms. The catalytic performance of SACs depends heavily on the local electronic structure and coordination geometry around the metal center. Current synthetic methods often yield heterogeneous distributions of coordination environments, complicating structure-property relationships and hindering rational design.
The loading density of single atoms represents another significant limitation. Most SAC preparation methods achieve metal loadings below 2 wt%, restricting mass activity and practical applications. Increasing loading while maintaining atomic dispersion remains a delicate balance that few techniques have successfully navigated.
Characterization challenges further complicate progress in this field. Conventional techniques struggle to provide comprehensive information about the exact location, coordination environment, and electronic state of single atoms. Advanced methods like aberration-corrected electron microscopy and X-ray absorption spectroscopy offer valuable insights but require specialized equipment and expertise.
Scalable production represents perhaps the most significant barrier to commercial implementation. Laboratory-scale synthesis methods often involve complex procedures, expensive precursors, or specialized equipment that pose challenges for industrial-scale manufacturing. The development of cost-effective, scalable synthesis routes remains a priority for transitioning SACs from academic research to practical applications.
Current Engineering Approaches for Single-Atom Sites
01 Single-atom catalyst synthesis methods
Various methods can be employed to synthesize single-atom catalysts for electrocatalyst layers. These include atomic layer deposition, wet chemical synthesis, and high-temperature pyrolysis techniques. The precise control of synthesis conditions allows for the uniform dispersion of metal atoms on support materials, preventing aggregation and maintaining the single-atom state. These methods enable the creation of catalysts with maximized atom utilization efficiency and enhanced catalytic performance for various electrochemical reactions.- Single-atom catalyst synthesis methods: Various methods can be employed to synthesize single-atom catalysts for electrocatalyst layers. These include atomic layer deposition, wet chemical synthesis, and high-temperature pyrolysis techniques. The synthesis approach significantly influences the distribution, stability, and activity of single-atom sites within the electrocatalyst layer. Precise control over synthesis parameters enables the creation of well-dispersed single-atom active sites with optimized coordination environments for specific electrochemical reactions.
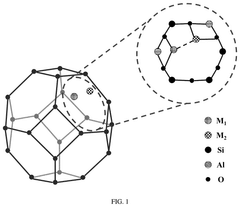
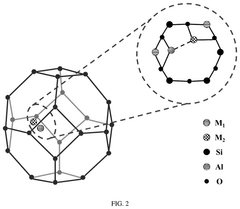
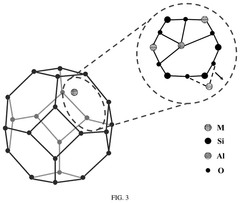
- Support material selection for single-atom sites: The choice of support material plays a crucial role in anchoring single-atom catalysts and maintaining their stability during electrochemical reactions. Carbon-based materials (graphene, carbon nanotubes), metal oxides, and nitrogen-doped carbon frameworks serve as effective supports for single-atom sites. The support material not only prevents aggregation of metal atoms but also can modify the electronic structure of the single atoms through strong metal-support interactions, enhancing catalytic performance and selectivity.
- Coordination environment engineering: Engineering the coordination environment around single-atom sites is essential for optimizing their catalytic performance. By controlling the type and number of coordinating atoms (such as N, O, S, or P), the electronic structure and binding properties of the metal center can be precisely tuned. Different coordination environments can be created through the introduction of specific heteroatoms or functional groups into the support material, allowing for customized catalytic properties tailored to specific electrochemical reactions.
- Interface engineering for enhanced activity: Interface engineering between single-atom sites and the surrounding environment significantly impacts catalytic performance. Creating specific interfaces, such as metal-support interfaces or heterojunctions, can enhance electron transfer, modify adsorption energies of reactants, and improve reaction kinetics. Advanced interface engineering strategies include creating dual-atom sites, constructing single-atom/nanoparticle hybrid structures, and developing multi-metal single-atom systems to achieve synergistic effects and improved catalytic efficiency.
- Stability enhancement strategies: Enhancing the stability of single-atom sites in electrocatalyst layers is crucial for practical applications. Various strategies have been developed, including strong covalent anchoring to support materials, encapsulation within protective structures, and the creation of confined spaces to prevent migration and aggregation. Additionally, the incorporation of secondary elements or the formation of specific coordination structures can significantly improve the durability of single-atom catalysts under harsh electrochemical conditions, maintaining high activity during long-term operation.
02 Support material selection for single-atom sites
The choice of support material significantly influences the stability and activity of single-atom catalysts. Carbon-based materials (graphene, carbon nanotubes), metal oxides, and nitrogen-doped carbon frameworks serve as effective supports by providing strong anchoring sites for metal atoms. The support material's electronic structure can be engineered to optimize the interaction with single metal atoms, preventing migration and aggregation while enhancing electron transfer properties during catalytic reactions.Expand Specific Solutions03 Metal-nitrogen-carbon (M-N-C) coordination structures
Metal-nitrogen-carbon (M-N-C) coordination structures represent a significant advancement in single-atom catalyst design. These structures feature metal atoms coordinated with nitrogen atoms embedded in carbon matrices, creating active sites with unique electronic properties. The coordination environment can be precisely tuned by adjusting nitrogen content and configuration, which directly influences the catalytic performance. M-N-C structures have demonstrated exceptional activity for oxygen reduction, hydrogen evolution, and CO2 reduction reactions.Expand Specific Solutions04 Interface engineering for enhanced catalytic activity
Interface engineering involves optimizing the boundary between single-atom sites and surrounding materials to enhance catalytic performance. This includes creating defects, vacancies, or heterostructures that modify the electronic environment around the active sites. Strategic placement of single atoms at interfaces between different materials can create synergistic effects that lower activation barriers for electrochemical reactions. Advanced characterization techniques are employed to understand and optimize these interfacial structures at the atomic level.Expand Specific Solutions05 Dual-atom and cluster site engineering
Beyond single-atom sites, dual-atom and small cluster configurations offer unique catalytic properties for electrocatalyst layers. These structures feature two or more metal atoms in close proximity, creating distinctive electronic structures and reaction pathways not available to isolated atoms. The controlled synthesis of these multi-atomic sites involves precise tuning of metal loading, thermal treatment conditions, and stabilizing ligands. These advanced structures can provide superior performance for complex multi-electron transfer reactions while maintaining high atom utilization efficiency.Expand Specific Solutions
Leading Research Groups and Companies in SAC Field
The electrocatalyst layer engineering for single-atom sites market is in an early growth phase, characterized by intensive research and development activities. The global market size is estimated to be relatively modest but growing rapidly, driven by increasing demand for efficient energy conversion and storage solutions. Academic institutions like Dalian University of Technology, University of Florida, and Korea Advanced Institute of Science & Technology are leading fundamental research, while companies such as Toshiba, Lam Research, and Applied Materials are developing commercial applications. The technology is approaching early commercial maturity, with key players like PolyPlus Battery and Ballard Power Systems focusing on practical implementations in battery and fuel cell technologies. Chinese research institutions demonstrate particular strength in this field, suggesting regional competitive advantages in future commercialization.
Dalian University of Technology
Technical Solution: Dalian University of Technology has pioneered advanced atomic layer deposition (ALD) techniques for single-atom catalyst engineering, focusing on precise control of metal-nitrogen-carbon (M-N-C) structures. Their approach involves coordinating transition metal atoms with nitrogen-doped carbon supports to create isolated single-atom active sites with maximized atom utilization efficiency. The university has developed innovative "host-guest" strategies where the carbon substrate acts as a host for anchoring single metal atoms through strong metal-N coordination bonds. Their recent breakthroughs include developing a two-step pyrolysis method that prevents metal aggregation during high-temperature treatment, maintaining single-atom dispersion while optimizing the surrounding electronic environment. This method has achieved remarkable oxygen reduction reaction (ORR) performance comparable to commercial Pt/C catalysts but with significantly higher durability in acidic conditions.
Strengths: Superior atomic-level precision in catalyst synthesis, excellent stability of single-atom sites, and cost-effective alternatives to precious metal catalysts. Weaknesses: Laboratory-scale production methods may face challenges in industrial scaling, and the complex synthesis procedures require specialized equipment and expertise.
Institute of Process Engineering, Chinese Academy of Sciences
Technical Solution: The Institute of Process Engineering at CAS has developed a groundbreaking "coordination-confinement strategy" for single-atom catalyst engineering. Their approach focuses on creating atomically dispersed metal sites embedded in nitrogen-doped carbon matrices through precise control of coordination chemistry. The institute has pioneered the use of metal-organic frameworks (MOFs) as precursors, which are transformed through controlled pyrolysis to yield single-atom catalysts with exceptional stability. Their innovative "dual-confinement" technique employs both spatial and electronic confinement mechanisms to prevent metal atom aggregation during high-temperature treatments. This method has enabled the creation of M-N4 (where M = Fe, Co, Ni) active sites with optimized electronic structures for various electrocatalytic reactions. Recent advances include the development of bimetallic single-atom catalysts that demonstrate synergistic effects, significantly enhancing catalytic performance for hydrogen evolution reaction (HER) and oxygen evolution reaction (OER).
Strengths: Exceptional control over atomic coordination environments, innovative precursor design strategies, and demonstrated scalability of certain synthesis routes. Weaknesses: Some synthesis methods require expensive precursors, and performance in industrial operating conditions still needs further validation.
Key Innovations in Electrocatalyst Layer Architecture
Single-atom catalysts and method of manufacture thereof
PatentPendingUS20230366111A1
Innovation
- The synthesis of single-atom catalysts (SACs) comprising nanofibers with uniformly dispersed single-atom metal sites, particularly Pt, Ru, and Pd, anchored on conductive polymers like polyaniline (PANI), which are produced through an electrochemical method that avoids the formation of metal clusters and nanoparticles, thereby maximizing active site exposure and catalytic efficiency.
Single-atom catalyst with molecular sieve-confined domains, preparation method and application thereof
PatentPendingUS20240399346A1
Innovation
- A single-atom catalyst with molecular sieve-confined domains is developed, where bimetallic ions are uniformly dispersed within the molecular sieve using a post-processing or in-situ synthesis method, leveraging oxygen vacancies and aluminum-rich sites for enhanced NO adsorption and dissociation, improving catalytic activity and stability.
Sustainability Aspects of Single-Atom Catalysts
The sustainability of single-atom catalysts (SACs) represents a critical dimension in the advancement of electrocatalyst layer engineering. SACs offer remarkable advantages in terms of atom economy, utilizing nearly 100% of the metal atoms as catalytically active sites, which significantly reduces the consumption of precious metals compared to traditional nanoparticle catalysts. This efficiency translates directly into resource conservation, particularly for scarce platinum group metals that face supply constraints and geopolitical challenges.
Environmental impact assessments of SAC production processes reveal substantially lower carbon footprints compared to conventional catalyst manufacturing. The synthesis methods for single-atom sites often require lower temperatures and pressures, resulting in reduced energy consumption. Additionally, the precision placement of atoms eliminates wasteful byproducts associated with traditional catalyst preparation methods, further enhancing their environmental credentials.
Life cycle analyses of SAC-based electrocatalytic systems demonstrate extended operational lifespans due to their resistance to degradation mechanisms like sintering and Ostwald ripening. This longevity reduces the frequency of catalyst replacement, minimizing waste generation and resource consumption throughout the system's operational life. Furthermore, the enhanced catalytic activity of SACs enables lower operating temperatures and pressures in many applications, contributing to overall energy efficiency improvements in industrial processes.
The recyclability of SAC substrates presents another sustainability advantage. Research indicates that carbon-based supports used in many SAC configurations can be regenerated through controlled oxidation processes, allowing for metal atom recovery and support material reuse. This circular approach significantly reduces the end-of-life environmental impact compared to conventional catalysts that often become hazardous waste.
Economic sustainability analyses suggest that despite higher initial development costs, the total cost of ownership for SAC-based systems is frequently lower over their operational lifetime. The reduced precious metal loading, extended service intervals, and improved energy efficiency create compelling economic incentives that align with environmental benefits, driving market adoption beyond regulatory compliance motivations.
Future sustainability improvements focus on developing earth-abundant metal SACs to replace precious metal variants, with promising results emerging for iron, cobalt, and nickel-based single-atom catalysts in certain applications. Additionally, research into biologically-derived support materials offers pathways to fully renewable catalyst systems that maintain the performance advantages of traditional SACs while further reducing environmental impacts.
Environmental impact assessments of SAC production processes reveal substantially lower carbon footprints compared to conventional catalyst manufacturing. The synthesis methods for single-atom sites often require lower temperatures and pressures, resulting in reduced energy consumption. Additionally, the precision placement of atoms eliminates wasteful byproducts associated with traditional catalyst preparation methods, further enhancing their environmental credentials.
Life cycle analyses of SAC-based electrocatalytic systems demonstrate extended operational lifespans due to their resistance to degradation mechanisms like sintering and Ostwald ripening. This longevity reduces the frequency of catalyst replacement, minimizing waste generation and resource consumption throughout the system's operational life. Furthermore, the enhanced catalytic activity of SACs enables lower operating temperatures and pressures in many applications, contributing to overall energy efficiency improvements in industrial processes.
The recyclability of SAC substrates presents another sustainability advantage. Research indicates that carbon-based supports used in many SAC configurations can be regenerated through controlled oxidation processes, allowing for metal atom recovery and support material reuse. This circular approach significantly reduces the end-of-life environmental impact compared to conventional catalysts that often become hazardous waste.
Economic sustainability analyses suggest that despite higher initial development costs, the total cost of ownership for SAC-based systems is frequently lower over their operational lifetime. The reduced precious metal loading, extended service intervals, and improved energy efficiency create compelling economic incentives that align with environmental benefits, driving market adoption beyond regulatory compliance motivations.
Future sustainability improvements focus on developing earth-abundant metal SACs to replace precious metal variants, with promising results emerging for iron, cobalt, and nickel-based single-atom catalysts in certain applications. Additionally, research into biologically-derived support materials offers pathways to fully renewable catalyst systems that maintain the performance advantages of traditional SACs while further reducing environmental impacts.
Scalability and Industrial Implementation Challenges
The transition from laboratory-scale single-atom catalyst (SAC) synthesis to industrial production presents significant challenges that must be addressed for commercial viability. Current laboratory methods typically produce SACs in milligram quantities under highly controlled conditions, whereas industrial applications require kilogram to ton-scale production with consistent quality. The scale-up process faces several critical bottlenecks, including maintaining atomic dispersion during mass production, preventing aggregation of metal atoms into clusters or nanoparticles, and ensuring uniform distribution across support materials.
Production cost remains a major barrier to industrial implementation. The synthesis of SACs often involves expensive precursors, specialized equipment, and energy-intensive processes. Additionally, many current methods utilize precious metals like platinum, palladium, and iridium, which are cost-prohibitive for large-scale applications. Developing economically viable production routes using earth-abundant metals while maintaining catalytic performance represents a crucial research direction.
Quality control and characterization pose another significant challenge. At industrial scales, it becomes increasingly difficult to verify the atomic dispersion and coordination environment of metal sites. Conventional characterization techniques such as X-ray absorption spectroscopy (XAS) and aberration-corrected transmission electron microscopy (AC-TEM) are time-consuming and not suitable for in-line quality control during mass production. New rapid analytical methods must be developed to ensure consistent product quality.
Stability under industrial operating conditions presents further complications. While laboratory tests often demonstrate excellent performance in controlled environments, industrial applications subject catalysts to harsh conditions including high temperatures, pressure fluctuations, and contaminants. Engineering SACs with enhanced durability against sintering, leaching, and poisoning is essential for practical implementation.
Integration with existing manufacturing infrastructure represents another hurdle. Many industries have established production lines optimized for conventional catalysts. Retrofitting these systems for SAC implementation requires significant capital investment and process redesign. Developing drop-in replacement SACs that can utilize existing infrastructure would significantly accelerate industrial adoption.
Regulatory and safety considerations must also be addressed, particularly for novel SAC formulations. New materials require extensive testing and certification before industrial deployment, especially in sensitive applications like pharmaceutical production or food processing. Establishing standardized testing protocols and safety guidelines specific to SACs will facilitate their regulatory approval and market acceptance.
Production cost remains a major barrier to industrial implementation. The synthesis of SACs often involves expensive precursors, specialized equipment, and energy-intensive processes. Additionally, many current methods utilize precious metals like platinum, palladium, and iridium, which are cost-prohibitive for large-scale applications. Developing economically viable production routes using earth-abundant metals while maintaining catalytic performance represents a crucial research direction.
Quality control and characterization pose another significant challenge. At industrial scales, it becomes increasingly difficult to verify the atomic dispersion and coordination environment of metal sites. Conventional characterization techniques such as X-ray absorption spectroscopy (XAS) and aberration-corrected transmission electron microscopy (AC-TEM) are time-consuming and not suitable for in-line quality control during mass production. New rapid analytical methods must be developed to ensure consistent product quality.
Stability under industrial operating conditions presents further complications. While laboratory tests often demonstrate excellent performance in controlled environments, industrial applications subject catalysts to harsh conditions including high temperatures, pressure fluctuations, and contaminants. Engineering SACs with enhanced durability against sintering, leaching, and poisoning is essential for practical implementation.
Integration with existing manufacturing infrastructure represents another hurdle. Many industries have established production lines optimized for conventional catalysts. Retrofitting these systems for SAC implementation requires significant capital investment and process redesign. Developing drop-in replacement SACs that can utilize existing infrastructure would significantly accelerate industrial adoption.
Regulatory and safety considerations must also be addressed, particularly for novel SAC formulations. New materials require extensive testing and certification before industrial deployment, especially in sensitive applications like pharmaceutical production or food processing. Establishing standardized testing protocols and safety guidelines specific to SACs will facilitate their regulatory approval and market acceptance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!