High-bandwidth intracortical Brain-Computer Interfaces for fine motor tasks
SEP 2, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
BCI Evolution and Neural Interface Goals
Brain-Computer Interfaces (BCIs) have evolved significantly since their inception in the 1970s, transitioning from rudimentary systems capable of basic binary commands to sophisticated neural interfaces that can interpret complex neural patterns for fine motor control. The initial BCI systems developed by researchers like Jacques Vidal at UCLA demonstrated the possibility of using brain signals for external device control, albeit with limited bandwidth and functionality.
The 1990s marked a pivotal shift with the development of the first intracortical BCIs, which involved implanting electrodes directly into the brain tissue to record neural activity with higher fidelity. This approach significantly improved signal quality compared to non-invasive methods like EEG, enabling more precise control capabilities. By the early 2000s, researchers at Brown University and the University of Pittsburgh had successfully demonstrated that humans with paralysis could control computer cursors and robotic arms using intracortical BCIs.
The evolution of BCI technology has been driven by advances in multiple disciplines, including neuroscience, materials science, signal processing, and machine learning. Miniaturization of electronics and development of biocompatible materials have enabled the creation of increasingly sophisticated neural implants with higher electrode densities and improved longevity. Concurrently, advances in signal processing algorithms and machine learning techniques have enhanced the ability to decode neural signals accurately in real-time.
Current high-bandwidth intracortical BCIs aim to restore natural motor function for individuals with paralysis or limb loss. These systems typically employ arrays of microelectrodes implanted in motor cortex regions to record from hundreds of individual neurons simultaneously. The primary goal is to achieve control capabilities that closely mimic natural movement, including multi-dimensional control, fine motor precision, and intuitive operation without extensive training requirements.
Looking forward, the field is moving toward fully implantable wireless systems that eliminate infection risks associated with transcutaneous connections. Researchers are also focusing on increasing electrode density to record from thousands or tens of thousands of neurons simultaneously, which would theoretically enable more naturalistic control of complex movements. Another critical goal is developing systems with bidirectional capabilities that not only record neural activity but also provide sensory feedback through direct neural stimulation.
The ultimate vision for high-bandwidth intracortical BCIs is to create seamless neural interfaces that restore or augment human capabilities with minimal cognitive burden. This includes enabling users to perform delicate tasks requiring fine motor control, such as typing, drawing, or manipulating small objects with dexterity comparable to natural limbs. Achieving this vision requires overcoming significant challenges in electrode longevity, signal stability, and the development of algorithms capable of adapting to changes in neural activity over time.
The 1990s marked a pivotal shift with the development of the first intracortical BCIs, which involved implanting electrodes directly into the brain tissue to record neural activity with higher fidelity. This approach significantly improved signal quality compared to non-invasive methods like EEG, enabling more precise control capabilities. By the early 2000s, researchers at Brown University and the University of Pittsburgh had successfully demonstrated that humans with paralysis could control computer cursors and robotic arms using intracortical BCIs.
The evolution of BCI technology has been driven by advances in multiple disciplines, including neuroscience, materials science, signal processing, and machine learning. Miniaturization of electronics and development of biocompatible materials have enabled the creation of increasingly sophisticated neural implants with higher electrode densities and improved longevity. Concurrently, advances in signal processing algorithms and machine learning techniques have enhanced the ability to decode neural signals accurately in real-time.
Current high-bandwidth intracortical BCIs aim to restore natural motor function for individuals with paralysis or limb loss. These systems typically employ arrays of microelectrodes implanted in motor cortex regions to record from hundreds of individual neurons simultaneously. The primary goal is to achieve control capabilities that closely mimic natural movement, including multi-dimensional control, fine motor precision, and intuitive operation without extensive training requirements.
Looking forward, the field is moving toward fully implantable wireless systems that eliminate infection risks associated with transcutaneous connections. Researchers are also focusing on increasing electrode density to record from thousands or tens of thousands of neurons simultaneously, which would theoretically enable more naturalistic control of complex movements. Another critical goal is developing systems with bidirectional capabilities that not only record neural activity but also provide sensory feedback through direct neural stimulation.
The ultimate vision for high-bandwidth intracortical BCIs is to create seamless neural interfaces that restore or augment human capabilities with minimal cognitive burden. This includes enabling users to perform delicate tasks requiring fine motor control, such as typing, drawing, or manipulating small objects with dexterity comparable to natural limbs. Achieving this vision requires overcoming significant challenges in electrode longevity, signal stability, and the development of algorithms capable of adapting to changes in neural activity over time.
Market Analysis for High-Bandwidth Neural Interfaces
The high-bandwidth intracortical Brain-Computer Interface (BCI) market is experiencing significant growth, driven by advancements in neural recording technology and increasing applications in medical and consumer sectors. Current market estimates value the neural interface industry at approximately $2.4 billion globally, with projections suggesting expansion to reach $8.5 billion by 2030, representing a compound annual growth rate of 15.1%.
The market segmentation reveals distinct application areas with varying growth trajectories. Medical applications currently dominate, accounting for roughly 70% of market share, with rehabilitation and assistive technologies for paralysis and motor impairments leading demand. The fine motor control capabilities of high-bandwidth BCIs make them particularly valuable for restoring function in patients with spinal cord injuries, stroke, and neurodegenerative conditions.
Consumer applications represent a smaller but rapidly expanding segment, growing at nearly 22% annually. This includes gaming, virtual reality interaction, and productivity enhancement tools. Military and defense applications constitute approximately 8% of the market, focusing on enhanced soldier capabilities and remote operation of complex systems.
Geographically, North America leads with 45% market share, followed by Europe (28%) and Asia-Pacific (20%). China and South Korea are demonstrating the fastest growth rates in the Asia-Pacific region, investing heavily in neural interface research and development.
Key market drivers include decreasing costs of high-resolution neural recording equipment, miniaturization of implantable devices, and improvements in wireless data transmission capabilities. The development of more biocompatible materials has extended device longevity, improving the cost-benefit ratio for medical applications.
Regulatory environments significantly impact market dynamics, with medical applications facing stringent approval processes. The FDA's recent introduction of the Innovation Pathway for neurological devices has accelerated commercialization timelines in the United States, while the European Medical Device Regulation provides a structured framework for CE marking.
Market barriers include high initial costs, with current high-bandwidth systems ranging from $50,000 to $200,000, limiting widespread adoption. Public perception and ethical concerns regarding neural data privacy represent significant challenges, with surveys indicating 65% of consumers express concerns about neural data security.
Reimbursement structures in healthcare systems are evolving to accommodate BCI technologies, with several major insurers now covering implantable neural interfaces for specific medical conditions, substantially expanding the addressable market for therapeutic applications focused on fine motor control restoration.
The market segmentation reveals distinct application areas with varying growth trajectories. Medical applications currently dominate, accounting for roughly 70% of market share, with rehabilitation and assistive technologies for paralysis and motor impairments leading demand. The fine motor control capabilities of high-bandwidth BCIs make them particularly valuable for restoring function in patients with spinal cord injuries, stroke, and neurodegenerative conditions.
Consumer applications represent a smaller but rapidly expanding segment, growing at nearly 22% annually. This includes gaming, virtual reality interaction, and productivity enhancement tools. Military and defense applications constitute approximately 8% of the market, focusing on enhanced soldier capabilities and remote operation of complex systems.
Geographically, North America leads with 45% market share, followed by Europe (28%) and Asia-Pacific (20%). China and South Korea are demonstrating the fastest growth rates in the Asia-Pacific region, investing heavily in neural interface research and development.
Key market drivers include decreasing costs of high-resolution neural recording equipment, miniaturization of implantable devices, and improvements in wireless data transmission capabilities. The development of more biocompatible materials has extended device longevity, improving the cost-benefit ratio for medical applications.
Regulatory environments significantly impact market dynamics, with medical applications facing stringent approval processes. The FDA's recent introduction of the Innovation Pathway for neurological devices has accelerated commercialization timelines in the United States, while the European Medical Device Regulation provides a structured framework for CE marking.
Market barriers include high initial costs, with current high-bandwidth systems ranging from $50,000 to $200,000, limiting widespread adoption. Public perception and ethical concerns regarding neural data privacy represent significant challenges, with surveys indicating 65% of consumers express concerns about neural data security.
Reimbursement structures in healthcare systems are evolving to accommodate BCI technologies, with several major insurers now covering implantable neural interfaces for specific medical conditions, substantially expanding the addressable market for therapeutic applications focused on fine motor control restoration.
Current Intracortical BCI Technology Landscape
The intracortical Brain-Computer Interface (BCI) landscape has evolved significantly over the past decade, with several key technologies emerging as frontrunners in enabling high-bandwidth neural interfaces for fine motor tasks. Currently, the field is dominated by microelectrode array technologies, with Utah arrays and Michigan probes representing the most clinically validated approaches. These systems typically record from 100-1000 neurons simultaneously, providing sufficient neural data to decode complex motor intentions.
Utah arrays, developed at the University of Utah, feature a grid of silicon microelectrodes that penetrate the cortex to record from individual neurons. These arrays have been the workhorse of clinical BCI research, most notably in the BrainGate trials, where they have enabled tetraplegic patients to control robotic arms and computer cursors with remarkable precision. The current generation of Utah arrays can record from approximately 100 channels, though newer iterations are pushing toward higher channel counts.
Michigan probes, developed at the University of Michigan, offer a different form factor with electrodes arranged along a shank, allowing for recording across different cortical layers. These probes have been further refined through the Neural Engineering System Design (NESD) program, resulting in higher-density recording capabilities that can capture neural activity across broader cortical regions.
More recently, Neuralink has introduced a novel "neural lace" technology featuring flexible polymer threads with thousands of electrodes, potentially offering dramatically increased channel counts compared to traditional arrays. Their robotic insertion system aims to minimize tissue damage during implantation, addressing a key limitation of rigid electrode arrays. While promising, this technology remains in pre-clinical testing phases.
Paradromics and Blackrock Neurotech represent other significant players, with the former developing microwire arrays capable of recording from up to 65,000 channels, and the latter continuing to refine the Utah array technology for commercial and clinical applications. These high-density recording systems are critical for capturing the neural activity patterns necessary for fine motor control tasks.
Signal processing technologies have also advanced considerably, with real-time spike sorting algorithms and neural decoders becoming increasingly sophisticated. Modern systems employ machine learning approaches, particularly recurrent neural networks and deep learning architectures, to translate neural activity patterns into control signals for external devices with minimal latency.
Wireless transmission capabilities have improved as well, with several groups demonstrating fully implantable systems that can transmit neural data without transcutaneous connections, reducing infection risk and improving usability. However, power consumption and heat generation remain significant challenges for these wireless systems, particularly as channel counts increase.
Utah arrays, developed at the University of Utah, feature a grid of silicon microelectrodes that penetrate the cortex to record from individual neurons. These arrays have been the workhorse of clinical BCI research, most notably in the BrainGate trials, where they have enabled tetraplegic patients to control robotic arms and computer cursors with remarkable precision. The current generation of Utah arrays can record from approximately 100 channels, though newer iterations are pushing toward higher channel counts.
Michigan probes, developed at the University of Michigan, offer a different form factor with electrodes arranged along a shank, allowing for recording across different cortical layers. These probes have been further refined through the Neural Engineering System Design (NESD) program, resulting in higher-density recording capabilities that can capture neural activity across broader cortical regions.
More recently, Neuralink has introduced a novel "neural lace" technology featuring flexible polymer threads with thousands of electrodes, potentially offering dramatically increased channel counts compared to traditional arrays. Their robotic insertion system aims to minimize tissue damage during implantation, addressing a key limitation of rigid electrode arrays. While promising, this technology remains in pre-clinical testing phases.
Paradromics and Blackrock Neurotech represent other significant players, with the former developing microwire arrays capable of recording from up to 65,000 channels, and the latter continuing to refine the Utah array technology for commercial and clinical applications. These high-density recording systems are critical for capturing the neural activity patterns necessary for fine motor control tasks.
Signal processing technologies have also advanced considerably, with real-time spike sorting algorithms and neural decoders becoming increasingly sophisticated. Modern systems employ machine learning approaches, particularly recurrent neural networks and deep learning architectures, to translate neural activity patterns into control signals for external devices with minimal latency.
Wireless transmission capabilities have improved as well, with several groups demonstrating fully implantable systems that can transmit neural data without transcutaneous connections, reducing infection risk and improving usability. However, power consumption and heat generation remain significant challenges for these wireless systems, particularly as channel counts increase.
Existing High-Bandwidth Intracortical Solutions
01 High-bandwidth neural signal acquisition and processing
Advanced technologies for acquiring and processing neural signals at high bandwidth from intracortical interfaces. These systems enable the capture of large volumes of neural data with improved temporal and spatial resolution, allowing for more precise brain activity monitoring and interpretation. The technologies include specialized microelectrode arrays, signal amplification circuits, and digital processing algorithms that can handle the high data throughput required for advanced brain-computer interface applications.- High-bandwidth neural signal acquisition systems: Advanced neural signal acquisition systems are designed to capture high-bandwidth intracortical signals with improved resolution and fidelity. These systems incorporate specialized electrodes and signal processing techniques to maximize the information transfer rate from the brain to external devices. The high-bandwidth acquisition enables more precise detection of neural activity patterns, which is crucial for complex BCI applications requiring detailed neural information.
- Wireless data transmission for BCIs: Wireless data transmission technologies are being developed to eliminate the need for transcutaneous connections in intracortical BCIs. These systems utilize advanced communication protocols and compression algorithms to transmit large volumes of neural data wirelessly while maintaining signal integrity. Wireless transmission enhances user mobility and reduces infection risks associated with wired connections, while addressing the challenge of transmitting high-bandwidth neural data with minimal latency and power consumption.
- Signal processing and bandwidth optimization techniques: Novel signal processing algorithms and bandwidth optimization techniques are being implemented to enhance the efficiency of data transmission in intracortical BCIs. These approaches include adaptive filtering, dimensionality reduction, and neural compression methods that preserve essential information while reducing data volume. By optimizing bandwidth usage, these techniques enable more efficient neural decoding and improve the overall performance of brain-computer interfaces in real-time applications.
- Multi-channel electrode arrays for increased bandwidth: Advanced multi-channel electrode arrays are being developed to increase the bandwidth of intracortical BCIs by sampling from larger populations of neurons simultaneously. These high-density arrays feature numerous recording sites with optimized spatial distribution to capture diverse neural signals across different brain regions. The increased channel count enables parallel processing of neural information, significantly enhancing the information transfer rate and allowing for more complex control signals to be extracted from brain activity.
- AI-enhanced neural decoding for bandwidth efficiency: Artificial intelligence and machine learning algorithms are being integrated into intracortical BCI systems to improve neural decoding efficiency and bandwidth utilization. These AI-based approaches can extract meaningful information from complex neural patterns with fewer data points, effectively increasing the functional bandwidth of the interface. By learning and adapting to individual neural signatures, these systems can achieve higher performance with lower raw bandwidth requirements, enabling more natural and intuitive control of external devices.
02 Wireless data transmission for brain-computer interfaces
Wireless communication systems designed specifically for transmitting neural data from implanted intracortical devices to external processors. These technologies address the bandwidth limitations of traditional wireless protocols by implementing specialized compression algorithms, efficient encoding schemes, and optimized radio frequency transmission methods. Wireless solutions eliminate the need for transcranial wires, reducing infection risk while maintaining the high bandwidth necessary for complex neural decoding and control applications.Expand Specific Solutions03 Signal processing algorithms for neural data compression
Computational methods and algorithms specifically designed to compress and efficiently process the large volumes of data generated by intracortical interfaces. These approaches include machine learning techniques, neural network implementations, and specialized signal processing that identify and extract the most relevant neural features while reducing redundant information. By optimizing data representation, these algorithms effectively increase the functional bandwidth of brain-computer interface systems without requiring proportional increases in physical transmission capacity.Expand Specific Solutions04 Implantable neural interface hardware optimization
Hardware designs and materials that optimize the physical components of intracortical interfaces to increase bandwidth capabilities. These innovations include miniaturized integrated circuits, advanced electrode materials with improved biocompatibility, and novel form factors that maximize the number of recording sites while minimizing tissue damage. The hardware optimizations focus on power efficiency, signal-to-noise ratio improvement, and long-term stability to maintain high bandwidth neural recording capabilities over extended periods.Expand Specific Solutions05 Real-time neural decoding for increased effective bandwidth
Systems that implement real-time neural signal decoding directly at or near the recording site to increase the effective bandwidth of brain-computer interfaces. By performing initial processing and feature extraction before data transmission, these approaches reduce the amount of raw data that needs to be transmitted while preserving the information content. These technologies include edge computing implementations, embedded neural processors, and adaptive algorithms that continuously optimize the decoding process based on the specific neural activity patterns of the user.Expand Specific Solutions
Leading BCI Research Organizations and Companies
The high-bandwidth intracortical Brain-Computer Interface (BCI) market for fine motor tasks is in its early growth phase, characterized by rapid technological advancement but limited commercial deployment. The global BCI market is projected to reach $3.7 billion by 2027, with the intracortical segment showing the highest growth potential. Companies like Neuralink, Science Corp., and Precision Neuroscience are leading commercial development with innovative implantable technologies, while academic institutions including Zhejiang University, California Institute of Technology, and University of California contribute significant research advancements. The technology remains in early clinical trials, with challenges in biocompatibility, longevity, and signal processing still being addressed. Recent breakthroughs in graphene-based interfaces by INBRAIN Neuroelectronics and miniaturized systems by Neurable represent promising developments toward practical applications.
Precision Neuroscience Corp.
Technical Solution: Precision Neuroscience has developed the Layer 7 Cortical Interface, an ultra-thin, flexible electrode array designed specifically for high-bandwidth intracortical recording with minimal invasiveness. Their technology uses a film-like array approximately 25 microns thick (about one-third the thickness of a human hair) containing hundreds of microelectrodes that conform to the brain's surface. The system employs a novel insertion technique that allows placement through a cranial slit smaller than 23mm, rather than traditional craniotomy. Their proprietary signal processing architecture enables simultaneous recording from up to 1,600 channels, providing exceptional spatial resolution for decoding fine motor intentions. The interface targets the cortical layer without penetrating deep into brain tissue, balancing signal quality with reduced tissue damage. Their approach combines high-density microelectrode arrays with advanced machine learning algorithms specifically optimized for decoding complex fine motor movements from neural signals.
Strengths: Minimally invasive insertion technique; extremely thin and flexible substrate reducing foreign body response; high channel count enabling detailed motor decoding; potentially easier removal than penetrating electrodes. Weaknesses: Surface positioning may capture fewer action potentials than penetrating electrodes; signal quality might degrade over time due to natural meningeal thickening; limited published human trial data; potential challenges with long-term stability.
Neuralink Corp.
Technical Solution: Neuralink has developed an advanced high-bandwidth BCI system called the N1 Link, which utilizes flexible polymer threads (4-6 μm in width) containing thousands of electrodes. Their system can record from 1,024 electrodes across 64 threads, enabling high-resolution neural recording. Neuralink's proprietary ASIC (application-specific integrated circuit) can process neural signals at 20,000 samples per second per channel with a custom-designed robot for precise thread insertion to minimize tissue damage. Their system incorporates wireless data transmission capabilities with a custom-designed chip that processes, filters, and amplifies signals, allowing for real-time decoding of neural activity for fine motor control applications. Recent demonstrations showed monkeys successfully controlling computer cursors and playing video games through thought alone, with signal quality maintained over extended periods.
Strengths: Highest electrode density among commercial BCIs; custom surgical robot for precise implantation; wireless operation eliminating infection risk from percutaneous connectors; proprietary signal processing algorithms. Weaknesses: Limited human trial data compared to academic systems; concerns about long-term biocompatibility of materials; potential thermal management issues with implanted electronics; regulatory approval challenges.
Key Neural Decoding Algorithms and Electrode Technologies
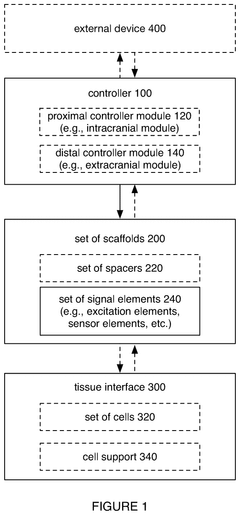
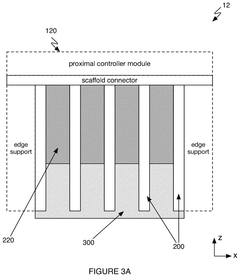
Neural interface device
PatentPendingUS20250181903A1
Innovation
- The system employs a scalable intracortical brain-computer interface with a high-density of engineered cells seeded between stacked scaffolds, which grow projections to interface with native brain tissue, enabling high-bandwidth signal transmission with minimal brain damage.
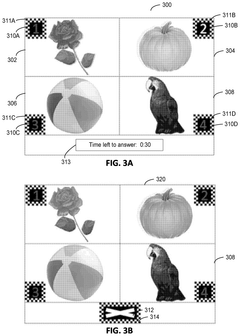
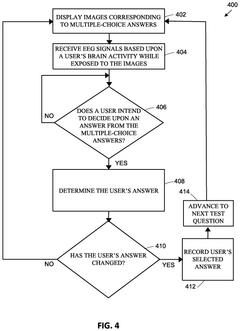
Brain-computer interface for facilitating direct selection of multiple-choice answers and the identification of state changes
PatentPendingUS20250009284A1
Innovation
- A BCI system that uses electroencephalograph (EEG) measurements to directly determine user intentions and selections through a three-step process, allowing users to select answers without motor or oral feedback, maintaining the standardization of cognitive tests and reducing test data skewing.
Clinical Translation and Regulatory Pathways
The clinical translation of high-bandwidth intracortical Brain-Computer Interfaces (BCIs) for fine motor tasks represents a complex journey from laboratory research to patient application. This process requires navigating multiple regulatory frameworks that vary significantly across global jurisdictions, with the FDA in the United States, the EMA in Europe, and the NMPA in China each maintaining distinct requirements for implantable neural devices.
For intracortical BCIs targeting fine motor control, the regulatory pathway typically begins with classification as a Class III medical device due to the high-risk nature of brain implantation. This classification necessitates comprehensive clinical trials demonstrating both safety and efficacy. The FDA's Investigational Device Exemption (IDE) pathway has been the primary route for pioneering BCI systems like those developed by BrainGate and Synchron, requiring rigorous preclinical testing before human trials can commence.
Recent regulatory developments have shown increasing recognition of the unique challenges posed by neural interface technologies. The FDA's Breakthrough Devices Program has accelerated several high-bandwidth BCI systems, acknowledging their potential to address unmet medical needs for patients with severe motor impairments. This program provides prioritized review and interactive communication with regulatory officials during development phases.
Clinical translation further demands addressing biocompatibility concerns, long-term stability of recording capabilities, and surgical implantation protocols. Successful regulatory submissions must include comprehensive data on electrode materials, hermetic packaging, wireless transmission security, and potential tissue reactions over extended periods. The development of consensus standards specifically for neural interfaces remains an evolving area, with organizations like ASTM International and IEEE working to establish testing methodologies appropriate for these novel technologies.
Reimbursement pathways present another critical consideration for clinical translation. Current healthcare systems lack established codes for high-bandwidth BCI technologies, requiring developers to pursue novel reimbursement strategies or demonstrate exceptional cost-effectiveness compared to existing assistive technologies. Several companies have begun engaging with insurance providers and government healthcare systems early in development to address these challenges.
International harmonization efforts, including the Medical Device Single Audit Program (MDSAP) and the International Medical Device Regulators Forum (IMDRF), are gradually reducing regulatory barriers for global deployment of neural interface technologies. However, significant differences remain in how various jurisdictions approach risk assessment and post-market surveillance requirements for implantable brain-computer interfaces.
For intracortical BCIs targeting fine motor control, the regulatory pathway typically begins with classification as a Class III medical device due to the high-risk nature of brain implantation. This classification necessitates comprehensive clinical trials demonstrating both safety and efficacy. The FDA's Investigational Device Exemption (IDE) pathway has been the primary route for pioneering BCI systems like those developed by BrainGate and Synchron, requiring rigorous preclinical testing before human trials can commence.
Recent regulatory developments have shown increasing recognition of the unique challenges posed by neural interface technologies. The FDA's Breakthrough Devices Program has accelerated several high-bandwidth BCI systems, acknowledging their potential to address unmet medical needs for patients with severe motor impairments. This program provides prioritized review and interactive communication with regulatory officials during development phases.
Clinical translation further demands addressing biocompatibility concerns, long-term stability of recording capabilities, and surgical implantation protocols. Successful regulatory submissions must include comprehensive data on electrode materials, hermetic packaging, wireless transmission security, and potential tissue reactions over extended periods. The development of consensus standards specifically for neural interfaces remains an evolving area, with organizations like ASTM International and IEEE working to establish testing methodologies appropriate for these novel technologies.
Reimbursement pathways present another critical consideration for clinical translation. Current healthcare systems lack established codes for high-bandwidth BCI technologies, requiring developers to pursue novel reimbursement strategies or demonstrate exceptional cost-effectiveness compared to existing assistive technologies. Several companies have begun engaging with insurance providers and government healthcare systems early in development to address these challenges.
International harmonization efforts, including the Medical Device Single Audit Program (MDSAP) and the International Medical Device Regulators Forum (IMDRF), are gradually reducing regulatory barriers for global deployment of neural interface technologies. However, significant differences remain in how various jurisdictions approach risk assessment and post-market surveillance requirements for implantable brain-computer interfaces.
Ethical Implications of Invasive Neural Interfaces
The integration of invasive neural interfaces, particularly high-bandwidth intracortical Brain-Computer Interfaces (BCIs) for fine motor tasks, raises profound ethical considerations that must be addressed before widespread implementation. These technologies, which directly interface with neural tissue, present unique ethical challenges beyond those of non-invasive alternatives.
Privacy and data security constitute primary concerns, as intracortical BCIs capture unprecedented volumes of neural data that could reveal sensitive information about cognitive processes, emotional states, and even subconscious thoughts. The potential for unauthorized access to this intimate neural data presents risks far exceeding traditional privacy breaches, potentially exposing the very essence of human cognition and identity.
Informed consent presents another significant ethical challenge, particularly given the experimental nature of many high-bandwidth BCIs. Ensuring that participants fully comprehend the short and long-term implications of having devices implanted in their brains remains problematic, especially when considering vulnerable populations or those with cognitive impairments who might benefit most from these technologies.
Questions of autonomy and agency emerge when neural interfaces enable direct control of devices or potentially influence decision-making processes. As BCIs become more sophisticated in interpreting and potentially modifying neural activity for fine motor control, the boundary between user intention and machine interpretation blurs, raising fundamental questions about human agency.
The risk of creating new forms of social inequality through "neural enhancement" must also be considered. If high-bandwidth BCIs provide significant advantages in motor control, cognitive function, or communication, access disparities could create a "neural divide" between enhanced and non-enhanced individuals, potentially exacerbating existing social inequalities.
Long-term neural adaptation and identity concerns also warrant attention. The brain's neuroplasticity means it will adapt to interface with implanted devices, potentially altering neural pathways and, by extension, aspects of personality or identity. This raises profound questions about personhood and continuity of identity when neural function becomes increasingly integrated with technology.
Regulatory frameworks currently lag behind technological developments in this field. The unique ethical challenges of intracortical BCIs necessitate specialized oversight that balances innovation with protection of fundamental human rights and values, requiring international cooperation to establish consistent ethical standards.
Privacy and data security constitute primary concerns, as intracortical BCIs capture unprecedented volumes of neural data that could reveal sensitive information about cognitive processes, emotional states, and even subconscious thoughts. The potential for unauthorized access to this intimate neural data presents risks far exceeding traditional privacy breaches, potentially exposing the very essence of human cognition and identity.
Informed consent presents another significant ethical challenge, particularly given the experimental nature of many high-bandwidth BCIs. Ensuring that participants fully comprehend the short and long-term implications of having devices implanted in their brains remains problematic, especially when considering vulnerable populations or those with cognitive impairments who might benefit most from these technologies.
Questions of autonomy and agency emerge when neural interfaces enable direct control of devices or potentially influence decision-making processes. As BCIs become more sophisticated in interpreting and potentially modifying neural activity for fine motor control, the boundary between user intention and machine interpretation blurs, raising fundamental questions about human agency.
The risk of creating new forms of social inequality through "neural enhancement" must also be considered. If high-bandwidth BCIs provide significant advantages in motor control, cognitive function, or communication, access disparities could create a "neural divide" between enhanced and non-enhanced individuals, potentially exacerbating existing social inequalities.
Long-term neural adaptation and identity concerns also warrant attention. The brain's neuroplasticity means it will adapt to interface with implanted devices, potentially altering neural pathways and, by extension, aspects of personality or identity. This raises profound questions about personhood and continuity of identity when neural function becomes increasingly integrated with technology.
Regulatory frameworks currently lag behind technological developments in this field. The unique ethical challenges of intracortical BCIs necessitate specialized oversight that balances innovation with protection of fundamental human rights and values, requiring international cooperation to establish consistent ethical standards.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!