How to Develop Customized PEMF Therapy Protocols?
AUG 11, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
PEMF Therapy Evolution
Pulsed Electromagnetic Field (PEMF) therapy has undergone significant evolution since its inception in the mid-20th century. Initially developed for bone healing, PEMF therapy has expanded its applications across various medical fields. The therapy's evolution can be traced through several key phases, each marked by technological advancements and broadening therapeutic applications.
In the 1950s, scientists first observed the piezoelectric effect in bones, leading to the development of early PEMF devices for bone healing. These initial devices were large, stationary, and primarily used in clinical settings. The 1970s saw a surge in research and development, resulting in the FDA approval of PEMF devices for non-union fractures in 1979.
The 1980s and 1990s witnessed a expansion of PEMF applications beyond orthopedics. Researchers began exploring its potential in pain management, wound healing, and neurological disorders. This period also saw the miniaturization of PEMF devices, making them more portable and accessible for home use.
The turn of the millennium brought about a new era in PEMF therapy. Advancements in electronics and materials science led to the development of more sophisticated devices capable of generating precise electromagnetic fields. This precision allowed for the targeting of specific tissues and conditions, paving the way for more personalized treatment protocols.
In recent years, the integration of digital technologies has further revolutionized PEMF therapy. Smart devices now offer programmable settings, allowing for customized treatment plans tailored to individual patient needs. Additionally, the incorporation of biofeedback mechanisms has enabled real-time adjustments to therapy parameters based on physiological responses.
The evolution of PEMF therapy has also been marked by an increasing focus on understanding the underlying mechanisms of action. Research has delved into the effects of electromagnetic fields on cellular processes, including ion transport, gene expression, and protein synthesis. This deeper understanding has led to more targeted and effective treatment protocols.
As PEMF therapy continues to evolve, current trends point towards the development of wearable devices for continuous therapy, integration with other treatment modalities, and the use of artificial intelligence to optimize treatment parameters. These advancements are expected to further enhance the efficacy and accessibility of PEMF therapy, potentially expanding its role in both conventional and complementary medicine.
In the 1950s, scientists first observed the piezoelectric effect in bones, leading to the development of early PEMF devices for bone healing. These initial devices were large, stationary, and primarily used in clinical settings. The 1970s saw a surge in research and development, resulting in the FDA approval of PEMF devices for non-union fractures in 1979.
The 1980s and 1990s witnessed a expansion of PEMF applications beyond orthopedics. Researchers began exploring its potential in pain management, wound healing, and neurological disorders. This period also saw the miniaturization of PEMF devices, making them more portable and accessible for home use.
The turn of the millennium brought about a new era in PEMF therapy. Advancements in electronics and materials science led to the development of more sophisticated devices capable of generating precise electromagnetic fields. This precision allowed for the targeting of specific tissues and conditions, paving the way for more personalized treatment protocols.
In recent years, the integration of digital technologies has further revolutionized PEMF therapy. Smart devices now offer programmable settings, allowing for customized treatment plans tailored to individual patient needs. Additionally, the incorporation of biofeedback mechanisms has enabled real-time adjustments to therapy parameters based on physiological responses.
The evolution of PEMF therapy has also been marked by an increasing focus on understanding the underlying mechanisms of action. Research has delved into the effects of electromagnetic fields on cellular processes, including ion transport, gene expression, and protein synthesis. This deeper understanding has led to more targeted and effective treatment protocols.
As PEMF therapy continues to evolve, current trends point towards the development of wearable devices for continuous therapy, integration with other treatment modalities, and the use of artificial intelligence to optimize treatment parameters. These advancements are expected to further enhance the efficacy and accessibility of PEMF therapy, potentially expanding its role in both conventional and complementary medicine.
Market Demand Analysis
The market demand for customized PEMF (Pulsed Electromagnetic Field) therapy protocols has been steadily increasing in recent years, driven by a growing awareness of the potential benefits of this non-invasive treatment modality. As more healthcare providers and patients seek personalized approaches to managing various health conditions, the need for tailored PEMF protocols has become increasingly apparent.
The global PEMF therapy devices market is experiencing significant growth, with projections indicating continued expansion in the coming years. This growth is fueled by the rising prevalence of chronic diseases, an aging population, and a shift towards non-pharmacological treatment options. The versatility of PEMF therapy in addressing a wide range of conditions, from pain management to bone healing and neurological disorders, has contributed to its increasing adoption across various medical specialties.
One of the key drivers of market demand for customized PEMF protocols is the growing emphasis on precision medicine. Healthcare providers are recognizing the importance of tailoring treatments to individual patient needs, considering factors such as age, medical history, and specific health conditions. This trend has created a demand for PEMF devices and protocols that can be adjusted and optimized for each patient, leading to improved treatment outcomes and patient satisfaction.
The sports medicine and rehabilitation sectors have emerged as significant contributors to the demand for customized PEMF therapy protocols. Professional athletes and sports teams are increasingly incorporating PEMF therapy into their training and recovery regimens, seeking personalized protocols to enhance performance and accelerate injury healing. This has led to a surge in demand for advanced PEMF devices capable of delivering targeted treatments for specific muscle groups and injuries.
In the realm of chronic pain management, there is a growing interest in alternative therapies, including PEMF, as patients and healthcare providers seek non-opioid solutions. The ability to customize PEMF protocols for different types of pain and individual patient responses has made it an attractive option for pain clinics and rehabilitation centers. This trend is expected to continue driving market growth as the healthcare industry focuses on reducing opioid dependence and improving long-term pain management strategies.
The home healthcare market has also shown increasing demand for customizable PEMF devices, as patients seek convenient and cost-effective treatment options for ongoing health management. This has led to the development of user-friendly, portable PEMF devices that allow for personalized therapy sessions in the comfort of one's home, further expanding the market reach of customized PEMF protocols.
As research in PEMF therapy continues to advance, there is a growing body of evidence supporting its efficacy in various medical applications. This has led to increased acceptance among healthcare professionals and insurance providers, potentially expanding market access and reimbursement opportunities for customized PEMF treatments. The ongoing clinical trials and research initiatives focused on optimizing PEMF protocols for specific conditions are expected to further drive market demand and innovation in the coming years.
The global PEMF therapy devices market is experiencing significant growth, with projections indicating continued expansion in the coming years. This growth is fueled by the rising prevalence of chronic diseases, an aging population, and a shift towards non-pharmacological treatment options. The versatility of PEMF therapy in addressing a wide range of conditions, from pain management to bone healing and neurological disorders, has contributed to its increasing adoption across various medical specialties.
One of the key drivers of market demand for customized PEMF protocols is the growing emphasis on precision medicine. Healthcare providers are recognizing the importance of tailoring treatments to individual patient needs, considering factors such as age, medical history, and specific health conditions. This trend has created a demand for PEMF devices and protocols that can be adjusted and optimized for each patient, leading to improved treatment outcomes and patient satisfaction.
The sports medicine and rehabilitation sectors have emerged as significant contributors to the demand for customized PEMF therapy protocols. Professional athletes and sports teams are increasingly incorporating PEMF therapy into their training and recovery regimens, seeking personalized protocols to enhance performance and accelerate injury healing. This has led to a surge in demand for advanced PEMF devices capable of delivering targeted treatments for specific muscle groups and injuries.
In the realm of chronic pain management, there is a growing interest in alternative therapies, including PEMF, as patients and healthcare providers seek non-opioid solutions. The ability to customize PEMF protocols for different types of pain and individual patient responses has made it an attractive option for pain clinics and rehabilitation centers. This trend is expected to continue driving market growth as the healthcare industry focuses on reducing opioid dependence and improving long-term pain management strategies.
The home healthcare market has also shown increasing demand for customizable PEMF devices, as patients seek convenient and cost-effective treatment options for ongoing health management. This has led to the development of user-friendly, portable PEMF devices that allow for personalized therapy sessions in the comfort of one's home, further expanding the market reach of customized PEMF protocols.
As research in PEMF therapy continues to advance, there is a growing body of evidence supporting its efficacy in various medical applications. This has led to increased acceptance among healthcare professionals and insurance providers, potentially expanding market access and reimbursement opportunities for customized PEMF treatments. The ongoing clinical trials and research initiatives focused on optimizing PEMF protocols for specific conditions are expected to further drive market demand and innovation in the coming years.
Technical Challenges
The development of customized PEMF (Pulsed Electromagnetic Field) therapy protocols faces several technical challenges that need to be addressed for effective implementation. One of the primary obstacles is the lack of standardized parameters for PEMF therapy across different medical conditions. This variability in treatment protocols makes it difficult to establish a universal approach, necessitating extensive research and clinical trials for each specific application.
Another significant challenge lies in the precise control and modulation of electromagnetic fields. Generating consistent and reproducible PEMF signals with specific frequencies, intensities, and waveforms requires sophisticated hardware and software integration. The development of compact, portable devices capable of delivering accurate PEMF therapy while maintaining user-friendliness poses a considerable engineering challenge.
The optimization of treatment duration and frequency presents another hurdle. Determining the ideal exposure time and repetition rate for various conditions demands extensive experimentation and data analysis. This process is further complicated by individual patient variability, as factors such as age, health status, and concurrent treatments can influence the efficacy of PEMF therapy.
Ensuring the safety and biocompatibility of PEMF devices is crucial. While PEMF therapy is generally considered safe, long-term effects of repeated exposure to electromagnetic fields need thorough investigation. Developing protocols that maximize therapeutic benefits while minimizing potential risks requires a delicate balance and comprehensive safety studies.
The integration of PEMF therapy with existing medical treatments and technologies presents both opportunities and challenges. Compatibility issues may arise when combining PEMF with other therapeutic modalities or medical devices, necessitating careful consideration of potential interactions and interference.
Another technical challenge is the development of accurate and reliable methods for measuring the biological effects of PEMF therapy. Current limitations in quantifying cellular and tissue responses to electromagnetic fields hinder the precise evaluation of treatment efficacy and the optimization of protocols.
Lastly, the customization of PEMF therapy protocols for individual patients remains a complex task. Creating adaptive systems that can adjust treatment parameters based on real-time physiological feedback requires advanced sensing technologies and sophisticated algorithms. The development of such personalized approaches demands interdisciplinary collaboration between medical professionals, engineers, and data scientists to create truly tailored PEMF therapy solutions.
Another significant challenge lies in the precise control and modulation of electromagnetic fields. Generating consistent and reproducible PEMF signals with specific frequencies, intensities, and waveforms requires sophisticated hardware and software integration. The development of compact, portable devices capable of delivering accurate PEMF therapy while maintaining user-friendliness poses a considerable engineering challenge.
The optimization of treatment duration and frequency presents another hurdle. Determining the ideal exposure time and repetition rate for various conditions demands extensive experimentation and data analysis. This process is further complicated by individual patient variability, as factors such as age, health status, and concurrent treatments can influence the efficacy of PEMF therapy.
Ensuring the safety and biocompatibility of PEMF devices is crucial. While PEMF therapy is generally considered safe, long-term effects of repeated exposure to electromagnetic fields need thorough investigation. Developing protocols that maximize therapeutic benefits while minimizing potential risks requires a delicate balance and comprehensive safety studies.
The integration of PEMF therapy with existing medical treatments and technologies presents both opportunities and challenges. Compatibility issues may arise when combining PEMF with other therapeutic modalities or medical devices, necessitating careful consideration of potential interactions and interference.
Another technical challenge is the development of accurate and reliable methods for measuring the biological effects of PEMF therapy. Current limitations in quantifying cellular and tissue responses to electromagnetic fields hinder the precise evaluation of treatment efficacy and the optimization of protocols.
Lastly, the customization of PEMF therapy protocols for individual patients remains a complex task. Creating adaptive systems that can adjust treatment parameters based on real-time physiological feedback requires advanced sensing technologies and sophisticated algorithms. The development of such personalized approaches demands interdisciplinary collaboration between medical professionals, engineers, and data scientists to create truly tailored PEMF therapy solutions.
Current PEMF Protocols
01 Customizable PEMF therapy protocols
PEMF therapy devices can be designed with customizable protocols, allowing for adjustable parameters such as frequency, intensity, and duration. This customization enables tailored treatments for various conditions and individual patient needs, potentially improving therapeutic outcomes.- Customizable PEMF therapy protocols: PEMF therapy devices can be designed with customizable protocols, allowing for adjustable parameters such as frequency, intensity, and duration. This customization enables tailored treatments for various conditions and individual patient needs, potentially improving therapeutic outcomes.
- Integration of AI and machine learning in PEMF therapy: Advanced PEMF systems incorporate artificial intelligence and machine learning algorithms to analyze patient data and optimize treatment protocols. These technologies can adapt therapy settings based on real-time feedback and historical treatment outcomes, enhancing the effectiveness of personalized PEMF protocols.
- Mobile app-controlled PEMF therapy customization: PEMF devices can be controlled and customized through mobile applications, allowing users to adjust therapy settings, track progress, and receive personalized recommendations. This user-friendly interface enhances accessibility and enables remote monitoring by healthcare professionals.
- Multi-coil PEMF systems for targeted therapy: Advanced PEMF devices utilize multiple coils strategically placed to target specific body areas or conditions. This multi-coil approach allows for more precise and customized treatment protocols, potentially improving therapeutic efficacy for localized issues.
- Integration of biofeedback in PEMF protocol customization: PEMF therapy systems can incorporate biofeedback mechanisms to measure physiological responses during treatment. This real-time data is used to dynamically adjust therapy parameters, ensuring optimal and personalized treatment protocols based on individual patient responses.
02 Integration of AI and machine learning in PEMF therapy
Advanced PEMF systems incorporate artificial intelligence and machine learning algorithms to analyze patient data and optimize treatment protocols. These technologies can adapt therapy settings based on real-time feedback and historical treatment outcomes, enhancing the effectiveness of personalized PEMF therapy.Expand Specific Solutions03 Mobile app-controlled PEMF therapy customization
PEMF devices can be integrated with mobile applications, allowing users to control and customize their therapy protocols remotely. These apps may offer pre-set programs, user-defined settings, and the ability to track treatment progress, making PEMF therapy more accessible and user-friendly.Expand Specific Solutions04 Multi-coil PEMF systems for targeted therapy
Advanced PEMF devices utilize multiple coils or applicators that can be independently controlled and positioned. This design allows for targeted therapy to specific body areas and the creation of complex electromagnetic field patterns, enhancing the customization capabilities of PEMF treatments.Expand Specific Solutions05 Biofeedback-guided PEMF protocol adjustment
PEMF systems can incorporate biofeedback sensors to monitor physiological responses during therapy. This real-time data can be used to automatically adjust treatment parameters, ensuring that the PEMF protocol remains optimized throughout the session and adapts to the patient's immediate needs.Expand Specific Solutions
Key Industry Players
The development of customized PEMF therapy protocols is in a growth phase, with increasing market size and technological advancements. The industry is characterized by a mix of established medical device companies and innovative startups, indicating a maturing but still evolving market. Key players like Galvanize Therapeutics, Regenesis Biomedical, and Venus Concept are driving innovation in this field. Academic institutions such as the National University of Singapore and the Swiss Federal Institute of Technology are contributing to research and development, suggesting a strong focus on scientific advancement. The technology's maturity is progressing, with companies like Medtronic and Zomedica investing in PEMF applications, signaling growing interest from major healthcare players.
Galvanize Therapeutics, Inc.
Technical Solution: Galvanize Therapeutics has developed a proprietary PEMF therapy platform called AlterG. This system utilizes precise electromagnetic field generation to create customized treatment protocols. The AlterG platform allows for the adjustment of frequency, intensity, and duration of PEMF pulses based on specific patient needs and conditions. The system incorporates advanced sensors to monitor tissue response in real-time, enabling dynamic protocol adjustments during treatment sessions[1]. Galvanize's approach also includes a machine learning algorithm that analyzes treatment outcomes to continuously refine and optimize protocols for different pathologies[2].
Strengths: Highly customizable, real-time monitoring capabilities, and AI-driven protocol optimization. Weaknesses: May require specialized training for operators and potentially higher cost compared to simpler PEMF devices.
Regenesis Biomedical, Inc.
Technical Solution: Regenesis Biomedical has developed the Provant Therapy System, a PEMF device that utilizes a specific pulsed electromagnetic field to stimulate cellular repair and regeneration. Their approach to customized protocols involves a proprietary "Time-Varying Electromagnetic Field" (TVEMF) technology, which generates a precise sequence of electromagnetic pulses tailored to different tissue types and healing stages[3]. The Provant system allows for the adjustment of treatment duration and intensity based on the patient's condition and response. Regenesis has also implemented a cloud-based data collection system that enables healthcare providers to track patient progress and adjust protocols remotely, enhancing the personalization of treatments over time[4].
Strengths: Targeted TVEMF technology, remote monitoring and adjustment capabilities. Weaknesses: May be limited to specific frequency ranges, potentially reducing versatility for some applications.
Core PEMF Innovations
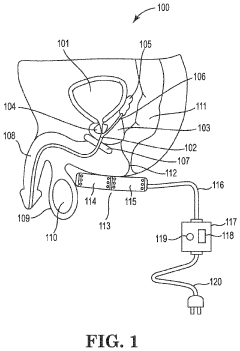
Method and apparatus for treatment of benign prostatic hyperplasia (BPH)
PatentInactiveUS20230398368A1
Innovation
- A non-invasive method utilizing pulsed electromagnetic field (PEMF) stimulation to increase the number of A2a receptors on cell membranes, enhancing the anti-inflammatory effects of adenosine and providing immunosuppressive action to reduce chronic inflammation and tissue damage in the prostate.
A pulsed electromagnetic field apparatus and method for generating frequencies
PatentWO2024127242A1
Innovation
- A PEMF apparatus with a pulse generator and electromagnetic field generation means that uses modified sawtooth waveforms with pre-stress and relaxation periods, and quasi-sine signals with pulse width modulation, along with a feedback circuit for frequency stability and precision, and a bifilar antenna for scalar wave generation.
Regulatory Framework
The regulatory framework for Pulsed Electromagnetic Field (PEMF) therapy protocols is complex and varies across different regions. In the United States, the Food and Drug Administration (FDA) classifies PEMF devices as Class II medical devices, requiring premarket notification (510(k)) clearance before commercialization. The FDA evaluates the safety and effectiveness of these devices based on clinical data and intended use. Manufacturers must demonstrate substantial equivalence to a predicate device or provide sufficient evidence of safety and efficacy through clinical trials.
In the European Union, PEMF devices fall under the Medical Device Regulation (MDR) 2017/745. Manufacturers must obtain CE marking by demonstrating compliance with essential requirements, including clinical evaluation and risk management. The classification of PEMF devices in the EU depends on their intended use and risk level, typically falling into Class IIa or IIb.
Regulatory bodies in other countries, such as Health Canada and Australia's Therapeutic Goods Administration (TGA), have similar requirements for PEMF devices. These agencies often require clinical evidence, risk assessments, and quality management systems for approval.
When developing customized PEMF therapy protocols, it is crucial to consider the regulatory requirements for both the device and the specific protocol. Manufacturers must ensure that their devices and protocols comply with the relevant regulations in each target market. This may involve conducting clinical trials to demonstrate the safety and efficacy of customized protocols for specific indications.
The regulatory landscape also addresses the claims made about PEMF therapy. Manufacturers must ensure that marketing materials and product labeling accurately reflect the approved indications and do not make unsupported claims. Regulatory bodies closely scrutinize promotional materials to prevent misleading or false advertising.
As PEMF technology advances and new applications emerge, regulatory frameworks continue to evolve. Manufacturers and researchers must stay informed about changes in regulations and guidelines to ensure compliance throughout the development and commercialization process. Engaging with regulatory agencies early in the development process can help navigate the complex landscape and streamline the approval process for customized PEMF therapy protocols.
In the European Union, PEMF devices fall under the Medical Device Regulation (MDR) 2017/745. Manufacturers must obtain CE marking by demonstrating compliance with essential requirements, including clinical evaluation and risk management. The classification of PEMF devices in the EU depends on their intended use and risk level, typically falling into Class IIa or IIb.
Regulatory bodies in other countries, such as Health Canada and Australia's Therapeutic Goods Administration (TGA), have similar requirements for PEMF devices. These agencies often require clinical evidence, risk assessments, and quality management systems for approval.
When developing customized PEMF therapy protocols, it is crucial to consider the regulatory requirements for both the device and the specific protocol. Manufacturers must ensure that their devices and protocols comply with the relevant regulations in each target market. This may involve conducting clinical trials to demonstrate the safety and efficacy of customized protocols for specific indications.
The regulatory landscape also addresses the claims made about PEMF therapy. Manufacturers must ensure that marketing materials and product labeling accurately reflect the approved indications and do not make unsupported claims. Regulatory bodies closely scrutinize promotional materials to prevent misleading or false advertising.
As PEMF technology advances and new applications emerge, regulatory frameworks continue to evolve. Manufacturers and researchers must stay informed about changes in regulations and guidelines to ensure compliance throughout the development and commercialization process. Engaging with regulatory agencies early in the development process can help navigate the complex landscape and streamline the approval process for customized PEMF therapy protocols.
Safety Considerations
Safety considerations are paramount when developing customized PEMF therapy protocols. The primary concern is to ensure that the electromagnetic fields generated do not exceed safety thresholds established by regulatory bodies such as the International Commission on Non-Ionizing Radiation Protection (ICNIRP). These guidelines typically set limits on the strength and frequency of electromagnetic fields to prevent adverse biological effects.
One crucial aspect is the careful calibration of PEMF devices to deliver precise field strengths and frequencies. Overexposure to electromagnetic fields can potentially lead to tissue heating or other unintended physiological responses. Therefore, it is essential to implement robust safety mechanisms that prevent accidental overexposure, such as automatic shut-off features and real-time monitoring of field parameters.
The duration and frequency of PEMF therapy sessions must also be carefully considered. Prolonged exposure, even at lower field strengths, may have cumulative effects that are not yet fully understood. Establishing clear treatment protocols with defined session lengths and intervals between treatments is crucial to minimize potential risks.
Patient-specific factors play a significant role in safety considerations. Pre-existing medical conditions, implanted medical devices, and pregnancy are key factors that may contraindicate PEMF therapy or require modified protocols. Comprehensive patient screening and ongoing monitoring are essential to identify and mitigate potential risks.
Electromagnetic interference with other medical devices is another critical safety concern. PEMF devices must be designed and tested to ensure they do not interfere with pacemakers, insulin pumps, or other electronic medical equipment. This may involve incorporating shielding technologies or establishing safe operating distances from sensitive devices.
Long-term safety studies are crucial for understanding the potential cumulative effects of PEMF therapy. While short-term studies have generally shown PEMF to be safe, more research is needed to assess the impact of prolonged or repeated exposure over extended periods. This underscores the importance of ongoing clinical trials and post-market surveillance to identify any emerging safety concerns.
Lastly, patient education is a vital component of safety considerations. Providing clear instructions on proper device use, potential side effects, and when to seek medical attention ensures that patients can safely administer PEMF therapy at home. Regular follow-ups and assessments can help identify any adverse reactions early and allow for timely adjustments to the treatment protocol.
One crucial aspect is the careful calibration of PEMF devices to deliver precise field strengths and frequencies. Overexposure to electromagnetic fields can potentially lead to tissue heating or other unintended physiological responses. Therefore, it is essential to implement robust safety mechanisms that prevent accidental overexposure, such as automatic shut-off features and real-time monitoring of field parameters.
The duration and frequency of PEMF therapy sessions must also be carefully considered. Prolonged exposure, even at lower field strengths, may have cumulative effects that are not yet fully understood. Establishing clear treatment protocols with defined session lengths and intervals between treatments is crucial to minimize potential risks.
Patient-specific factors play a significant role in safety considerations. Pre-existing medical conditions, implanted medical devices, and pregnancy are key factors that may contraindicate PEMF therapy or require modified protocols. Comprehensive patient screening and ongoing monitoring are essential to identify and mitigate potential risks.
Electromagnetic interference with other medical devices is another critical safety concern. PEMF devices must be designed and tested to ensure they do not interfere with pacemakers, insulin pumps, or other electronic medical equipment. This may involve incorporating shielding technologies or establishing safe operating distances from sensitive devices.
Long-term safety studies are crucial for understanding the potential cumulative effects of PEMF therapy. While short-term studies have generally shown PEMF to be safe, more research is needed to assess the impact of prolonged or repeated exposure over extended periods. This underscores the importance of ongoing clinical trials and post-market surveillance to identify any emerging safety concerns.
Lastly, patient education is a vital component of safety considerations. Providing clear instructions on proper device use, potential side effects, and when to seek medical attention ensures that patients can safely administer PEMF therapy at home. Regular follow-ups and assessments can help identify any adverse reactions early and allow for timely adjustments to the treatment protocol.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!