How to Ensure Brain-Computer Interface Equipment Operates within Safe Limits
MAR 5, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
BCI Safety Background and Objectives
Brain-computer interfaces represent a revolutionary convergence of neuroscience, engineering, and computer science that has evolved from experimental laboratory concepts to clinical realities over the past five decades. The journey began in the 1970s with early neural signal recording experiments and has progressed through significant milestones including the development of cochlear implants, deep brain stimulation systems, and modern bidirectional neural interfaces. This technological evolution has consistently been accompanied by growing awareness of safety imperatives, as these systems directly interact with the most critical organ in the human body.
The historical development of BCI technology reveals a pattern where each advancement has necessitated increasingly sophisticated safety protocols. Early invasive BCIs focused primarily on signal acquisition and basic functionality, but contemporary systems must address complex safety challenges including biocompatibility, signal integrity, thermal management, and long-term tissue compatibility. The transition from research prototypes to clinical devices has highlighted the critical importance of establishing comprehensive safety frameworks that can adapt to rapidly evolving technological capabilities.
Current safety objectives in BCI development encompass multiple interconnected domains that reflect the multidisciplinary nature of these systems. Primary safety goals include ensuring biocompatible material selection and device design that minimizes inflammatory responses and tissue damage over extended operational periods. Electrical safety represents another fundamental objective, requiring precise control of current densities, voltage levels, and stimulation parameters to prevent neural tissue damage while maintaining therapeutic or functional efficacy.
Thermal management constitutes a critical safety objective, as neural tissue is particularly sensitive to temperature variations. Modern BCI systems must incorporate sophisticated heat dissipation mechanisms and real-time temperature monitoring to prevent thermal injury during both passive recording and active stimulation operations. Additionally, infection prevention through sterile design principles and antimicrobial surface treatments has become a paramount consideration for implantable systems.
The establishment of fail-safe operational boundaries represents an overarching objective that integrates multiple safety domains. This includes developing robust monitoring systems capable of detecting anomalous conditions, implementing automatic shutdown protocols when safety thresholds are exceeded, and ensuring graceful degradation of system performance rather than catastrophic failure modes. These objectives collectively aim to create BCI systems that not only enhance human capabilities but do so within rigorously defined safety parameters that protect user wellbeing across all operational scenarios.
The historical development of BCI technology reveals a pattern where each advancement has necessitated increasingly sophisticated safety protocols. Early invasive BCIs focused primarily on signal acquisition and basic functionality, but contemporary systems must address complex safety challenges including biocompatibility, signal integrity, thermal management, and long-term tissue compatibility. The transition from research prototypes to clinical devices has highlighted the critical importance of establishing comprehensive safety frameworks that can adapt to rapidly evolving technological capabilities.
Current safety objectives in BCI development encompass multiple interconnected domains that reflect the multidisciplinary nature of these systems. Primary safety goals include ensuring biocompatible material selection and device design that minimizes inflammatory responses and tissue damage over extended operational periods. Electrical safety represents another fundamental objective, requiring precise control of current densities, voltage levels, and stimulation parameters to prevent neural tissue damage while maintaining therapeutic or functional efficacy.
Thermal management constitutes a critical safety objective, as neural tissue is particularly sensitive to temperature variations. Modern BCI systems must incorporate sophisticated heat dissipation mechanisms and real-time temperature monitoring to prevent thermal injury during both passive recording and active stimulation operations. Additionally, infection prevention through sterile design principles and antimicrobial surface treatments has become a paramount consideration for implantable systems.
The establishment of fail-safe operational boundaries represents an overarching objective that integrates multiple safety domains. This includes developing robust monitoring systems capable of detecting anomalous conditions, implementing automatic shutdown protocols when safety thresholds are exceeded, and ensuring graceful degradation of system performance rather than catastrophic failure modes. These objectives collectively aim to create BCI systems that not only enhance human capabilities but do so within rigorously defined safety parameters that protect user wellbeing across all operational scenarios.
Market Demand for Safe BCI Systems
The global brain-computer interface market is experiencing unprecedented growth driven by increasing awareness of neurological disorders and the urgent need for safe, reliable therapeutic solutions. Healthcare institutions worldwide are prioritizing patient safety protocols, creating substantial demand for BCI systems that incorporate comprehensive safety mechanisms and real-time monitoring capabilities.
Medical device manufacturers face mounting pressure from regulatory bodies to demonstrate robust safety frameworks before market approval. This regulatory landscape has intensified demand for BCI equipment featuring advanced fail-safe mechanisms, biocompatibility assurance, and continuous physiological monitoring. Hospitals and research institutions are specifically seeking systems that can prevent neural tissue damage, minimize infection risks, and provide immediate alerts for any operational anomalies.
The aging global population presents a significant market driver, as neurological conditions such as stroke, spinal cord injuries, and neurodegenerative diseases become more prevalent. Healthcare providers require BCI solutions that not only restore functionality but also guarantee long-term safety for vulnerable patient populations. This demographic shift has created substantial market opportunities for companies developing safety-first BCI architectures.
Research institutions and clinical trial organizations represent another critical market segment demanding enhanced safety protocols. These entities require BCI systems capable of supporting extended research periods while maintaining participant safety through sophisticated monitoring algorithms and automated shutdown procedures. The academic sector's emphasis on ethical research practices has further amplified demand for transparent safety reporting and real-time risk assessment capabilities.
Consumer healthcare markets are emerging as potential growth areas, though safety concerns remain paramount. Early adopters in rehabilitation centers and specialized clinics are driving demand for user-friendly BCI systems that maintain clinical-grade safety standards while offering improved accessibility and reduced operational complexity.
The competitive landscape reveals significant investment in safety-oriented BCI development, with market participants recognizing that safety assurance represents a key differentiator. Companies demonstrating superior safety protocols and regulatory compliance are capturing larger market shares, indicating strong commercial incentives for continued safety innovation in BCI technology development.
Medical device manufacturers face mounting pressure from regulatory bodies to demonstrate robust safety frameworks before market approval. This regulatory landscape has intensified demand for BCI equipment featuring advanced fail-safe mechanisms, biocompatibility assurance, and continuous physiological monitoring. Hospitals and research institutions are specifically seeking systems that can prevent neural tissue damage, minimize infection risks, and provide immediate alerts for any operational anomalies.
The aging global population presents a significant market driver, as neurological conditions such as stroke, spinal cord injuries, and neurodegenerative diseases become more prevalent. Healthcare providers require BCI solutions that not only restore functionality but also guarantee long-term safety for vulnerable patient populations. This demographic shift has created substantial market opportunities for companies developing safety-first BCI architectures.
Research institutions and clinical trial organizations represent another critical market segment demanding enhanced safety protocols. These entities require BCI systems capable of supporting extended research periods while maintaining participant safety through sophisticated monitoring algorithms and automated shutdown procedures. The academic sector's emphasis on ethical research practices has further amplified demand for transparent safety reporting and real-time risk assessment capabilities.
Consumer healthcare markets are emerging as potential growth areas, though safety concerns remain paramount. Early adopters in rehabilitation centers and specialized clinics are driving demand for user-friendly BCI systems that maintain clinical-grade safety standards while offering improved accessibility and reduced operational complexity.
The competitive landscape reveals significant investment in safety-oriented BCI development, with market participants recognizing that safety assurance represents a key differentiator. Companies demonstrating superior safety protocols and regulatory compliance are capturing larger market shares, indicating strong commercial incentives for continued safety innovation in BCI technology development.
Current BCI Safety Status and Challenges
Brain-computer interface technology currently operates within a complex safety landscape characterized by significant regulatory gaps and evolving technical standards. The FDA has established preliminary guidelines for invasive BCI devices under Class III medical device regulations, requiring extensive clinical trials and safety demonstrations. However, non-invasive BCI systems face less stringent oversight, creating inconsistencies in safety protocols across different device categories. The European Union's Medical Device Regulation (MDR) provides additional framework, yet harmonization between international standards remains incomplete.
Current safety assessment protocols primarily focus on immediate physical risks such as tissue damage, infection, and electromagnetic interference. Established testing procedures evaluate biocompatibility of implanted materials, signal quality degradation over time, and thermal effects from prolonged device operation. However, these protocols inadequately address long-term neuroplasticity changes, cognitive side effects, and potential psychological dependencies that may emerge from extended BCI usage.
The industry faces substantial challenges in establishing comprehensive safety thresholds due to limited long-term clinical data. Most BCI studies span months rather than years, creating knowledge gaps regarding chronic exposure effects. Variability in individual neural responses complicates the development of universal safety parameters, as optimal stimulation levels and signal processing algorithms may differ significantly between users based on neurological conditions, age, and baseline brain activity patterns.
Technical challenges include real-time monitoring of neural tissue health, detection of device malfunction before adverse events occur, and implementation of fail-safe mechanisms that can gracefully degrade system performance rather than causing abrupt disconnection. Current monitoring systems rely heavily on impedance measurements and signal quality metrics, which may not capture subtle inflammatory responses or gradual tissue encapsulation that could compromise long-term safety.
Cybersecurity vulnerabilities represent an emerging safety concern as BCI systems become more connected and sophisticated. Potential unauthorized access to neural data or malicious manipulation of device parameters poses unprecedented risks that traditional medical device safety frameworks were not designed to address. The integration of artificial intelligence algorithms in BCI systems introduces additional complexity, as machine learning models may exhibit unpredictable behaviors that could compromise user safety under certain conditions.
Current safety assessment protocols primarily focus on immediate physical risks such as tissue damage, infection, and electromagnetic interference. Established testing procedures evaluate biocompatibility of implanted materials, signal quality degradation over time, and thermal effects from prolonged device operation. However, these protocols inadequately address long-term neuroplasticity changes, cognitive side effects, and potential psychological dependencies that may emerge from extended BCI usage.
The industry faces substantial challenges in establishing comprehensive safety thresholds due to limited long-term clinical data. Most BCI studies span months rather than years, creating knowledge gaps regarding chronic exposure effects. Variability in individual neural responses complicates the development of universal safety parameters, as optimal stimulation levels and signal processing algorithms may differ significantly between users based on neurological conditions, age, and baseline brain activity patterns.
Technical challenges include real-time monitoring of neural tissue health, detection of device malfunction before adverse events occur, and implementation of fail-safe mechanisms that can gracefully degrade system performance rather than causing abrupt disconnection. Current monitoring systems rely heavily on impedance measurements and signal quality metrics, which may not capture subtle inflammatory responses or gradual tissue encapsulation that could compromise long-term safety.
Cybersecurity vulnerabilities represent an emerging safety concern as BCI systems become more connected and sophisticated. Potential unauthorized access to neural data or malicious manipulation of device parameters poses unprecedented risks that traditional medical device safety frameworks were not designed to address. The integration of artificial intelligence algorithms in BCI systems introduces additional complexity, as machine learning models may exhibit unpredictable behaviors that could compromise user safety under certain conditions.
Existing BCI Safety Solutions
01 Electrical safety limits and current control in BCI systems
Brain-computer interface equipment must adhere to strict electrical safety standards to prevent harm to users. This includes controlling the maximum current that can flow through electrodes in contact with the scalp or brain tissue, implementing current limiting circuits, and ensuring proper insulation. Safety mechanisms include automatic shutdown when current thresholds are exceeded, voltage regulation systems, and impedance monitoring to detect abnormal conditions that could pose risks to users.- Electrical safety limits and current control in BCI systems: Brain-computer interface equipment must adhere to strict electrical safety standards to prevent harm to users. This includes controlling the maximum current that can flow through electrodes in contact with the scalp or brain tissue, implementing current limiting circuits, and ensuring proper insulation. Safety mechanisms include automatic shutdown when current thresholds are exceeded, voltage regulation systems, and impedance monitoring to detect abnormal conditions that could pose risks to users.
- Signal quality monitoring and safety thresholds: BCI systems incorporate real-time monitoring of signal quality parameters to ensure safe operation. This includes tracking electrode impedance levels, signal-to-noise ratios, and detecting artifacts that may indicate equipment malfunction or improper contact. When signals fall outside predetermined safe ranges, the system can alert users or automatically adjust parameters. These monitoring systems help prevent both physical harm from electrical issues and ensure the device operates within validated performance boundaries.
- Thermal management and temperature limits: Safe operation of brain-computer interface equipment requires careful thermal management to prevent tissue damage from excessive heat. This involves setting maximum temperature limits for components in contact with or near the user's head, implementing heat dissipation structures, and incorporating temperature sensors with automatic power reduction or shutdown capabilities. Design considerations include material selection for thermal conductivity, ventilation systems, and continuous temperature monitoring to maintain operation within safe thermal boundaries.
- Electromagnetic compatibility and exposure limits: BCI equipment must comply with electromagnetic safety standards to limit user exposure to electromagnetic fields and prevent interference with other medical devices. This includes controlling the strength and frequency of electromagnetic signals emitted by the device, implementing shielding to contain electromagnetic radiation, and ensuring compatibility with pacemakers and other implanted devices. Safety protocols define maximum specific absorption rates and field strength limits based on international standards for electromagnetic exposure to protect users from potential biological effects.
- Duration limits and usage protocols for safe operation: Safe use of brain-computer interfaces involves establishing maximum continuous usage durations and mandatory rest periods to prevent fatigue, skin irritation, and potential neurological effects from prolonged stimulation or signal acquisition. Safety protocols define recommended session lengths, break intervals, and cumulative daily usage limits. These guidelines are based on clinical studies and consider factors such as electrode contact pressure, electrical stimulation duration, and cognitive load to ensure long-term user safety and device efficacy.
02 Signal quality monitoring and safety thresholds
BCI systems incorporate real-time monitoring of signal quality parameters to ensure safe operation. This includes tracking electrode impedance levels, signal-to-noise ratios, and detecting artifacts that may indicate equipment malfunction or improper contact. When signals fall outside predetermined safe ranges, the system can alert users or automatically adjust parameters. These monitoring systems help prevent both safety hazards and ensure reliable brain signal acquisition.Expand Specific Solutions03 Thermal management and temperature limits
Safe operation of BCI equipment requires careful thermal management to prevent overheating of components in contact with or near the user's head. This involves setting maximum temperature thresholds for electrodes, amplifiers, and processing units, implementing heat dissipation structures, and incorporating temperature sensors with automatic power reduction or shutdown capabilities. Proper thermal design ensures user comfort and prevents tissue damage from prolonged exposure to elevated temperatures.Expand Specific Solutions04 Electromagnetic compatibility and exposure limits
BCI devices must comply with electromagnetic safety standards to limit user exposure to electromagnetic fields and prevent interference with other medical devices. This includes controlling the strength and frequency of any electromagnetic emissions, implementing shielding to contain fields within safe levels, and ensuring the device can operate safely in the presence of external electromagnetic sources. Compliance with international standards for specific absorption rate and electromagnetic field exposure is essential for user safety.Expand Specific Solutions05 Duration limits and usage time restrictions
Safe BCI operation involves establishing maximum continuous usage times and mandatory rest periods to prevent fatigue, discomfort, or potential long-term effects from prolonged brain signal monitoring. Systems may include automatic timers that alert users or pause operation after specified durations, adaptive algorithms that adjust stimulation parameters based on usage duration, and protocols for gradual intensity reduction. These temporal safety limits help ensure that BCI technology is used within parameters that have been validated for safety.Expand Specific Solutions
Key Players in BCI Safety Industry
The brain-computer interface (BCI) safety landscape represents an emerging yet rapidly evolving sector characterized by significant technological complexity and diverse stakeholder involvement. The industry is in its early commercialization phase, with market potential estimated in billions but still facing substantial regulatory and technical hurdles. Technology maturity varies considerably across players, with established companies like Neuralink Corp. and Precision Neuroscience Corp. leading invasive BCI development, while research institutions including Carnegie Mellon University, California Institute of Technology, and Cornell University drive fundamental safety research. Technology giants such as Intel Corp. and Huawei Technologies provide essential computing infrastructure, while specialized firms like CoMind Technologies focus on non-invasive monitoring solutions. The competitive landscape spans from pure research entities to commercial ventures, indicating a fragmented but rapidly consolidating market where safety protocols remain critical differentiators for successful deployment.
South China Brain Control Guangdong Intelligent Tech Co Ltd.
Technical Solution: South China Brain Control has developed safety protocols specifically tailored for non-invasive brain-computer interface systems, focusing on EEG-based applications with strict electromagnetic field exposure limits. Their safety framework includes continuous monitoring of signal acquisition parameters and automatic adjustment of stimulation intensities to prevent overstimulation. The company has implemented multi-layer safety checks including software-based monitoring, hardware circuit protection, and manual override capabilities. Their systems incorporate real-time analysis of brain state indicators to ensure that the interface operates within physiologically safe parameters and can detect signs of user fatigue or stress.
Strengths: Specialized in non-invasive BCI safety, comprehensive multi-layer protection, real-time physiological monitoring. Weaknesses: Limited to non-invasive applications, smaller scale compared to international competitors.
Precision Neuroscience Corp.
Technical Solution: Precision Neuroscience focuses on ultra-thin, flexible electrode arrays that minimize tissue displacement and reduce inflammatory responses. Their safety approach emphasizes surface-level implantation rather than penetrating electrodes, significantly reducing the risk of brain tissue damage. The company has developed proprietary materials and manufacturing processes that ensure consistent electrode performance while maintaining biocompatibility. Their systems incorporate real-time impedance monitoring and adaptive signal processing to maintain safe operating parameters. The flexible nature of their electrodes allows for natural brain movement without causing mechanical stress or tissue tearing.
Strengths: Minimally invasive design, reduced tissue damage risk, flexible electrode technology. Weaknesses: Limited signal quality compared to penetrating electrodes, newer technology with less clinical validation.
Core BCI Safety Innovations
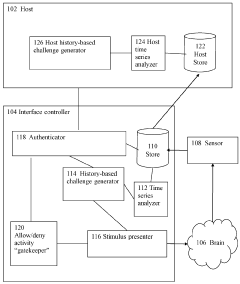
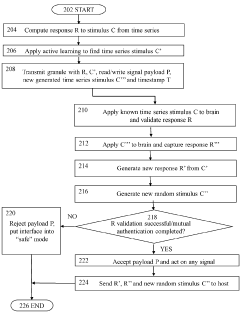
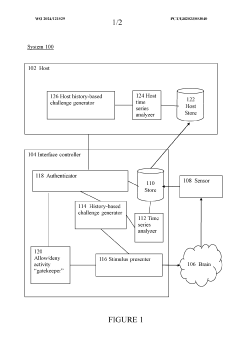
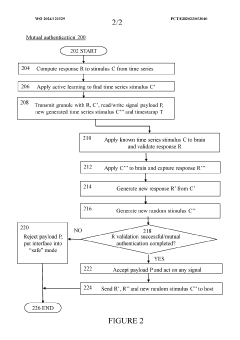
Brain-computer interface device, system and operating method
PatentWO2024121529A1
Innovation
- A time-series authentication system using a long-short-term memory (LSTM) neural network and autoencoders to generate and verify stimulus-response pairs, providing a firewall-like protection between the brain and external entities, ensuring that only valid signals are processed and preventing replay attacks by using temporal authentication and biometric proof of life.
Operation control device, operation control method, and program
PatentWO2023042501A1
Innovation
- An operation control device and method that includes a brain information acquisition unit, a determining unit, and an execution unit, which acquires and processes electroencephalogram signals to determine intended commands and notifies the surroundings of command processing status, ensuring safe execution by informing the environment of the user's intent and the robot's operation state.
BCI Regulatory Framework
The regulatory landscape for brain-computer interface equipment is rapidly evolving as governments and international bodies recognize the critical need for comprehensive safety standards. Currently, BCI devices fall under multiple regulatory jurisdictions depending on their intended use, with medical applications typically governed by medical device regulations such as the FDA's Class II or III classifications in the United States and the Medical Device Regulation in Europe. Non-medical BCI applications face a more fragmented regulatory environment, often subject to general electronic device standards and emerging neurotechnology-specific guidelines.
The FDA has established preliminary frameworks for invasive BCI devices through its breakthrough device designation program, expediting review processes for innovative neural interfaces while maintaining rigorous safety requirements. The agency requires extensive preclinical testing, biocompatibility assessments, and clinical trials demonstrating both efficacy and long-term safety profiles. European regulators have adopted similar approaches through the CE marking process, emphasizing risk management and post-market surveillance.
International standardization efforts are gaining momentum through organizations like the International Electrotechnical Commission and IEEE, which are developing specific standards for BCI safety protocols. These emerging standards address electromagnetic compatibility, signal processing accuracy, cybersecurity requirements, and user protection measures. The IEEE 2857 standard for privacy engineering in BCI systems represents a significant step toward comprehensive regulatory coverage.
Key regulatory challenges include establishing appropriate safety thresholds for neural stimulation, defining acceptable risk-benefit ratios for different user populations, and creating adaptive frameworks that can accommodate rapid technological advancement. Regulators are particularly focused on addressing potential long-term neurological effects, data privacy concerns, and the unique ethical considerations surrounding direct brain interfaces.
The regulatory framework continues to evolve through collaborative efforts between industry stakeholders, academic researchers, and government agencies, with ongoing initiatives aimed at harmonizing international standards and establishing clear pathways for BCI device approval while ensuring robust safety protections for users.
The FDA has established preliminary frameworks for invasive BCI devices through its breakthrough device designation program, expediting review processes for innovative neural interfaces while maintaining rigorous safety requirements. The agency requires extensive preclinical testing, biocompatibility assessments, and clinical trials demonstrating both efficacy and long-term safety profiles. European regulators have adopted similar approaches through the CE marking process, emphasizing risk management and post-market surveillance.
International standardization efforts are gaining momentum through organizations like the International Electrotechnical Commission and IEEE, which are developing specific standards for BCI safety protocols. These emerging standards address electromagnetic compatibility, signal processing accuracy, cybersecurity requirements, and user protection measures. The IEEE 2857 standard for privacy engineering in BCI systems represents a significant step toward comprehensive regulatory coverage.
Key regulatory challenges include establishing appropriate safety thresholds for neural stimulation, defining acceptable risk-benefit ratios for different user populations, and creating adaptive frameworks that can accommodate rapid technological advancement. Regulators are particularly focused on addressing potential long-term neurological effects, data privacy concerns, and the unique ethical considerations surrounding direct brain interfaces.
The regulatory framework continues to evolve through collaborative efforts between industry stakeholders, academic researchers, and government agencies, with ongoing initiatives aimed at harmonizing international standards and establishing clear pathways for BCI device approval while ensuring robust safety protections for users.
Bioethics in BCI Safety
The bioethical landscape surrounding brain-computer interface safety encompasses fundamental principles that must guide the development and deployment of these transformative technologies. At its core, the principle of beneficence demands that BCI systems maximize therapeutic benefits while minimizing potential harm to users. This requires establishing rigorous safety protocols that extend beyond traditional medical device standards to address the unique vulnerabilities associated with direct neural intervention.
Informed consent presents unprecedented challenges in BCI applications, particularly when dealing with patients who have compromised cognitive abilities or communication disorders. The complexity of neural interfaces necessitates developing new frameworks for ensuring that participants truly understand the long-term implications of brain implantation, including potential irreversible changes to neural function and personality. This ethical imperative requires creating accessible communication methods that can convey technical risks to diverse patient populations.
The principle of autonomy becomes particularly complex when considering BCIs designed to restore motor or cognitive function. While these devices aim to enhance user independence, they simultaneously create new forms of technological dependence that raise questions about authentic self-determination. Ethical frameworks must address scenarios where BCI malfunction or removal could significantly impact a user's quality of life or sense of identity.
Privacy and mental integrity represent emerging bioethical frontiers in BCI safety. The unprecedented access to neural data raises fundamental questions about cognitive liberty and the right to mental privacy. Establishing ethical boundaries for neural data collection, storage, and interpretation requires balancing therapeutic benefits with protection of users' innermost thoughts and mental processes.
Justice considerations demand equitable access to safe BCI technologies while preventing the creation of new forms of social stratification based on neural enhancement capabilities. Ethical frameworks must address how safety standards and regulatory oversight can be maintained without creating barriers that limit access for underserved populations who might benefit most from these interventions.
The long-term societal implications of widespread BCI adoption require proactive ethical consideration of how these technologies might reshape human identity, social relationships, and concepts of normalcy. Safety protocols must therefore incorporate not only immediate physical risks but also broader psychological and social consequences that may emerge as BCIs become more prevalent in medical and enhancement applications.
Informed consent presents unprecedented challenges in BCI applications, particularly when dealing with patients who have compromised cognitive abilities or communication disorders. The complexity of neural interfaces necessitates developing new frameworks for ensuring that participants truly understand the long-term implications of brain implantation, including potential irreversible changes to neural function and personality. This ethical imperative requires creating accessible communication methods that can convey technical risks to diverse patient populations.
The principle of autonomy becomes particularly complex when considering BCIs designed to restore motor or cognitive function. While these devices aim to enhance user independence, they simultaneously create new forms of technological dependence that raise questions about authentic self-determination. Ethical frameworks must address scenarios where BCI malfunction or removal could significantly impact a user's quality of life or sense of identity.
Privacy and mental integrity represent emerging bioethical frontiers in BCI safety. The unprecedented access to neural data raises fundamental questions about cognitive liberty and the right to mental privacy. Establishing ethical boundaries for neural data collection, storage, and interpretation requires balancing therapeutic benefits with protection of users' innermost thoughts and mental processes.
Justice considerations demand equitable access to safe BCI technologies while preventing the creation of new forms of social stratification based on neural enhancement capabilities. Ethical frameworks must address how safety standards and regulatory oversight can be maintained without creating barriers that limit access for underserved populations who might benefit most from these interventions.
The long-term societal implications of widespread BCI adoption require proactive ethical consideration of how these technologies might reshape human identity, social relationships, and concepts of normalcy. Safety protocols must therefore incorporate not only immediate physical risks but also broader psychological and social consequences that may emerge as BCIs become more prevalent in medical and enhancement applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!