Binder Systems Calendering and Electrode Density in Hard Carbon for Sodium Ion Batteries
AUG 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Binder Systems Evolution and Objectives in SIB Hard Carbon Electrodes
The evolution of binder systems in sodium-ion battery (SIB) electrodes has undergone significant transformation since the early development stages of this technology. Initially, researchers primarily adapted binder systems from lithium-ion battery technology, with polyvinylidene fluoride (PVDF) being the predominant choice. However, the unique characteristics of sodium ions, including their larger ionic radius compared to lithium ions, necessitated specialized binder development specifically tailored for SIB applications.
The mid-2010s marked a pivotal shift toward water-based binder systems, driven by environmental considerations and cost-effectiveness. Carboxymethyl cellulose (CMC) emerged as a promising alternative, demonstrating superior adhesion properties with hard carbon particles while enabling environmentally friendly aqueous processing. This transition represented a critical advancement in sustainable battery manufacturing practices.
Recent developments have focused on composite binder systems that combine multiple polymeric materials to achieve synergistic effects. For instance, CMC-SBR (styrene-butadiene rubber) combinations have shown enhanced flexibility and adhesion strength, addressing the volume expansion challenges inherent in hard carbon electrodes during sodium insertion/extraction cycles. These composite systems have demonstrated improved cycling stability and rate capability compared to single-component binders.
The primary objectives in binder system development for hard carbon SIB electrodes center around several key performance metrics. Mechanical integrity preservation during repeated cycling remains paramount, particularly given the substantial volume changes that occur during sodium intercalation. Enhanced ionic conductivity within the electrode structure is another critical goal, as binder systems must facilitate efficient sodium ion transport while maintaining strong particle-to-particle and particle-to-current collector adhesion.
Processability optimization represents another significant objective, with researchers seeking binder formulations that enable precise control over electrode density through calendering processes. The ideal binder system must maintain porosity for electrolyte penetration while allowing for density adjustment to maximize volumetric energy density. This balance is particularly challenging with hard carbon materials due to their inherent structural characteristics.
Looking forward, binder system development aims to enable higher active material loading while maintaining electrode flexibility and adhesion. Advanced polymer chemistry approaches, including functionalized binders with specific interaction sites for hard carbon surfaces, are being explored to enhance interfacial stability. The ultimate goal remains developing binder systems that can facilitate high-performance, long-lasting hard carbon electrodes for next-generation sodium-ion batteries with competitive energy density and cycle life metrics.
The mid-2010s marked a pivotal shift toward water-based binder systems, driven by environmental considerations and cost-effectiveness. Carboxymethyl cellulose (CMC) emerged as a promising alternative, demonstrating superior adhesion properties with hard carbon particles while enabling environmentally friendly aqueous processing. This transition represented a critical advancement in sustainable battery manufacturing practices.
Recent developments have focused on composite binder systems that combine multiple polymeric materials to achieve synergistic effects. For instance, CMC-SBR (styrene-butadiene rubber) combinations have shown enhanced flexibility and adhesion strength, addressing the volume expansion challenges inherent in hard carbon electrodes during sodium insertion/extraction cycles. These composite systems have demonstrated improved cycling stability and rate capability compared to single-component binders.
The primary objectives in binder system development for hard carbon SIB electrodes center around several key performance metrics. Mechanical integrity preservation during repeated cycling remains paramount, particularly given the substantial volume changes that occur during sodium intercalation. Enhanced ionic conductivity within the electrode structure is another critical goal, as binder systems must facilitate efficient sodium ion transport while maintaining strong particle-to-particle and particle-to-current collector adhesion.
Processability optimization represents another significant objective, with researchers seeking binder formulations that enable precise control over electrode density through calendering processes. The ideal binder system must maintain porosity for electrolyte penetration while allowing for density adjustment to maximize volumetric energy density. This balance is particularly challenging with hard carbon materials due to their inherent structural characteristics.
Looking forward, binder system development aims to enable higher active material loading while maintaining electrode flexibility and adhesion. Advanced polymer chemistry approaches, including functionalized binders with specific interaction sites for hard carbon surfaces, are being explored to enhance interfacial stability. The ultimate goal remains developing binder systems that can facilitate high-performance, long-lasting hard carbon electrodes for next-generation sodium-ion batteries with competitive energy density and cycle life metrics.
Market Analysis for Sodium Ion Battery Technologies
The sodium-ion battery market is experiencing significant growth as a promising alternative to lithium-ion batteries, driven by increasing concerns about lithium supply constraints and cost volatility. Current market projections indicate the global sodium-ion battery market could reach $1.2 billion by 2025, with a compound annual growth rate exceeding 25% through 2030.
The demand for sodium-ion batteries is particularly strong in grid energy storage applications, where cost considerations often outweigh energy density requirements. This segment currently represents approximately 40% of the potential market for sodium-ion technologies. Electric vehicles, especially in the two-wheeler and commercial vehicle segments in emerging markets, constitute another 30% of the projected demand.
Consumer electronics manufacturers are also showing increasing interest in sodium-ion technology for low-cost devices, representing about 15% of the potential market. The remaining market share is distributed across various industrial applications and specialized use cases where lithium-ion batteries face limitations.
Regionally, China is leading the sodium-ion battery market development, with companies like CATL and HiNa Battery Technology making substantial investments. The European market is growing rapidly due to strategic initiatives to reduce dependency on imported battery materials, while North America is seeing increased venture capital funding for sodium-ion startups.
Market analysis reveals that hard carbon electrode formulation technologies, particularly innovations in binder systems and calendering processes that optimize electrode density, could capture a premium segment within this growing market. Companies that can develop proprietary methods to increase energy density through advanced electrode manufacturing techniques may command price premiums of 15-20% over standard sodium-ion cells.
Customer surveys indicate that battery manufacturers are willing to invest in advanced binder systems and calendering technologies if they can achieve at least a 10% improvement in energy density without sacrificing cycle life. This represents a significant opportunity for technology developers focused on hard carbon electrode optimization.
The competitive landscape remains relatively open compared to the mature lithium-ion market, with fewer established patents and proprietary technologies related specifically to hard carbon electrode manufacturing for sodium-ion batteries. This creates a strategic window for new entrants and technology innovators to establish market leadership in specialized manufacturing processes and materials.
The demand for sodium-ion batteries is particularly strong in grid energy storage applications, where cost considerations often outweigh energy density requirements. This segment currently represents approximately 40% of the potential market for sodium-ion technologies. Electric vehicles, especially in the two-wheeler and commercial vehicle segments in emerging markets, constitute another 30% of the projected demand.
Consumer electronics manufacturers are also showing increasing interest in sodium-ion technology for low-cost devices, representing about 15% of the potential market. The remaining market share is distributed across various industrial applications and specialized use cases where lithium-ion batteries face limitations.
Regionally, China is leading the sodium-ion battery market development, with companies like CATL and HiNa Battery Technology making substantial investments. The European market is growing rapidly due to strategic initiatives to reduce dependency on imported battery materials, while North America is seeing increased venture capital funding for sodium-ion startups.
Market analysis reveals that hard carbon electrode formulation technologies, particularly innovations in binder systems and calendering processes that optimize electrode density, could capture a premium segment within this growing market. Companies that can develop proprietary methods to increase energy density through advanced electrode manufacturing techniques may command price premiums of 15-20% over standard sodium-ion cells.
Customer surveys indicate that battery manufacturers are willing to invest in advanced binder systems and calendering technologies if they can achieve at least a 10% improvement in energy density without sacrificing cycle life. This represents a significant opportunity for technology developers focused on hard carbon electrode optimization.
The competitive landscape remains relatively open compared to the mature lithium-ion market, with fewer established patents and proprietary technologies related specifically to hard carbon electrode manufacturing for sodium-ion batteries. This creates a strategic window for new entrants and technology innovators to establish market leadership in specialized manufacturing processes and materials.
Current Challenges in Hard Carbon Electrode Calendering
Hard carbon has emerged as a promising anode material for sodium-ion batteries (SIBs) due to its high capacity, good cycling stability, and cost-effectiveness. However, the calendering process for hard carbon electrodes presents significant challenges that impede the commercial viability of SIB technology. The primary challenge lies in the inherent brittleness of hard carbon particles, which tend to fracture under the high pressure applied during calendering, leading to capacity loss and reduced cycle life.
The binder system plays a crucial role in electrode calendering, yet conventional binders like polyvinylidene fluoride (PVDF) demonstrate inadequate elasticity and adhesion properties when used with hard carbon. This results in poor mechanical integrity of the electrode structure during and after calendering. The mechanical stress induced during the calendering process often causes delamination between the active material and current collector, particularly at higher calendering pressures.
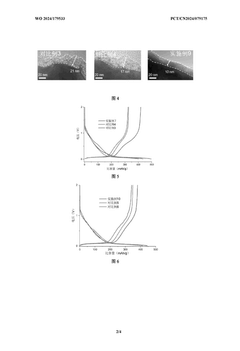
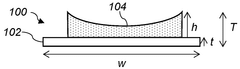
Achieving optimal electrode density represents another significant challenge. Unlike graphite electrodes in lithium-ion batteries, hard carbon electrodes exhibit a narrower processing window for density optimization. Too low density results in poor volumetric energy density, while excessive calendering leads to pore collapse in the hard carbon structure, blocking sodium ion diffusion pathways and significantly reducing rate capability.
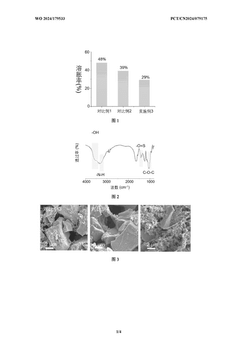
The heterogeneous particle morphology of hard carbon further complicates the calendering process. The irregular shapes and varying sizes of hard carbon particles create non-uniform stress distribution during calendering, resulting in inconsistent electrode thickness and density across the electrode sheet. This heterogeneity negatively impacts cell-to-cell performance consistency in battery pack manufacturing.
Temperature management during calendering presents additional challenges. Hard carbon electrodes demonstrate high sensitivity to calendering temperature, with elevated temperatures potentially causing binder degradation or undesired reactions with the electrolyte components. Conversely, low-temperature calendering may not provide sufficient plasticity for proper particle rearrangement.
The interaction between conductive additives and hard carbon during calendering also requires optimization. Carbon black and other conductive additives tend to segregate during the calendering process, creating non-uniform electrical conductivity throughout the electrode. This phenomenon is more pronounced in hard carbon electrodes compared to graphite electrodes due to the different surface characteristics and particle interactions.
Scaling up the calendering process for industrial production introduces further complications, including roll deflection issues, edge effects, and maintaining consistent cross-directional density profiles across wide electrode sheets. These manufacturing challenges must be addressed to enable cost-effective mass production of high-quality hard carbon electrodes for sodium-ion batteries.
The binder system plays a crucial role in electrode calendering, yet conventional binders like polyvinylidene fluoride (PVDF) demonstrate inadequate elasticity and adhesion properties when used with hard carbon. This results in poor mechanical integrity of the electrode structure during and after calendering. The mechanical stress induced during the calendering process often causes delamination between the active material and current collector, particularly at higher calendering pressures.
Achieving optimal electrode density represents another significant challenge. Unlike graphite electrodes in lithium-ion batteries, hard carbon electrodes exhibit a narrower processing window for density optimization. Too low density results in poor volumetric energy density, while excessive calendering leads to pore collapse in the hard carbon structure, blocking sodium ion diffusion pathways and significantly reducing rate capability.
The heterogeneous particle morphology of hard carbon further complicates the calendering process. The irregular shapes and varying sizes of hard carbon particles create non-uniform stress distribution during calendering, resulting in inconsistent electrode thickness and density across the electrode sheet. This heterogeneity negatively impacts cell-to-cell performance consistency in battery pack manufacturing.
Temperature management during calendering presents additional challenges. Hard carbon electrodes demonstrate high sensitivity to calendering temperature, with elevated temperatures potentially causing binder degradation or undesired reactions with the electrolyte components. Conversely, low-temperature calendering may not provide sufficient plasticity for proper particle rearrangement.
The interaction between conductive additives and hard carbon during calendering also requires optimization. Carbon black and other conductive additives tend to segregate during the calendering process, creating non-uniform electrical conductivity throughout the electrode. This phenomenon is more pronounced in hard carbon electrodes compared to graphite electrodes due to the different surface characteristics and particle interactions.
Scaling up the calendering process for industrial production introduces further complications, including roll deflection issues, edge effects, and maintaining consistent cross-directional density profiles across wide electrode sheets. These manufacturing challenges must be addressed to enable cost-effective mass production of high-quality hard carbon electrodes for sodium-ion batteries.
Current Calendering Methods for Optimizing Electrode Density
01 Optimization of hard carbon electrode density for sodium ion batteries
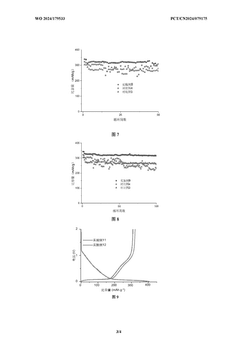
The density of hard carbon electrodes significantly impacts the performance of sodium ion batteries. Optimizing the electrode density involves controlling the compression ratio during electrode manufacturing, which affects the porosity, ion transport pathways, and overall electrochemical performance. Proper density optimization can lead to improved energy density, cycling stability, and rate capability of sodium ion batteries.- Optimization of hard carbon electrode density for sodium ion batteries: The density of hard carbon electrodes significantly impacts the performance of sodium ion batteries. Optimizing the electrode density involves controlling the compression pressure during electrode manufacturing, which affects the porosity, ion transport pathways, and overall electrochemical performance. Proper density optimization can lead to improved energy density, cycling stability, and rate capability of sodium ion batteries.
- Hard carbon preparation methods affecting electrode density: Various preparation methods for hard carbon materials directly influence the resulting electrode density. These methods include pyrolysis of biomass or synthetic polymers under controlled conditions, hydrothermal carbonization followed by high-temperature treatment, and template-assisted synthesis. The precursor selection, carbonization temperature, and activation processes determine the microstructure, porosity, and packing characteristics of hard carbon, ultimately affecting the electrode density and sodium storage performance.
- Surface modification of hard carbon for density control: Surface modification techniques are employed to control the density of hard carbon electrodes for sodium ion batteries. These include surface functionalization with heteroatoms (N, O, S), coating with conductive materials, and creating hierarchical pore structures. Such modifications improve the wettability with electrolytes, enhance the interfacial contact between particles, and optimize the packing density, leading to better electrochemical performance and higher volumetric energy density.
- Composite electrodes with hard carbon for optimized density: Composite electrodes combining hard carbon with other materials are developed to achieve optimal electrode density. These composites may incorporate conductive additives, binders, or secondary active materials that fill the interparticle voids. The synergistic effect of these components allows for better control of electrode density, improved mechanical integrity, enhanced electronic conductivity, and optimized ion transport pathways, resulting in superior electrochemical performance.
- Relationship between hard carbon microstructure and electrode density: The microstructure of hard carbon materials, including pore size distribution, particle morphology, and degree of graphitization, directly influences the achievable electrode density. Materials with tailored hierarchical porosity, optimized particle size distribution, and controlled defect structures can be packed more efficiently. Understanding this relationship enables the design of hard carbon materials with ideal microstructures that balance high tap density with sufficient ion transport channels for sodium ion storage.
02 Precursor selection and carbonization processes for hard carbon density control
The selection of carbon precursors and the carbonization process parameters directly influence the final density of hard carbon materials. Various biomass sources, polymers, and synthetic precursors can be used to produce hard carbon with different microstructures and densities. The carbonization temperature, heating rate, and duration significantly affect the graphitization degree, pore structure, and tap density of the resulting hard carbon, which are crucial for sodium ion storage performance.Expand Specific Solutions03 Surface modification and composite formation for enhanced electrode density
Surface modification of hard carbon materials and formation of composites with other materials can effectively adjust the electrode density while maintaining or improving electrochemical performance. Techniques such as nitrogen doping, heteroatom incorporation, and coating with conductive polymers or metal oxides can optimize the interface properties, enhance the electronic conductivity, and improve the packing density of hard carbon electrodes for sodium ion batteries.Expand Specific Solutions04 Particle size distribution and morphology control for optimal packing density
The particle size distribution and morphology of hard carbon materials significantly influence the packing density and porosity of electrodes. Controlling these parameters through grinding, sieving, and specific synthesis methods allows for the optimization of electrode density. Spherical particles with appropriate size distribution can achieve higher packing density while maintaining necessary porosity for electrolyte penetration and sodium ion diffusion.Expand Specific Solutions05 Binder systems and electrode formulation for density optimization
The selection of binder systems and electrode formulation plays a crucial role in determining the final electrode density. Water-soluble binders like CMC and PVDF, as well as their ratios with conductive additives, significantly affect the electrode compaction behavior. Optimizing the slurry composition, viscosity, and coating parameters can lead to improved electrode density while maintaining good adhesion, mechanical stability, and electrochemical performance of hard carbon electrodes for sodium ion batteries.Expand Specific Solutions
Leading Companies in SIB Electrode Manufacturing
The sodium-ion battery market, particularly focusing on hard carbon binder systems and electrode density, is in an early growth phase with increasing commercial interest. The market is expanding rapidly as companies seek alternatives to lithium-ion technology, with projections suggesting significant growth in the next decade. Technologically, the field shows moderate maturity with key players at different development stages. Leading companies like Faradion Ltd. have pioneered commercial sodium-ion technology, while major manufacturers including CATL, BYD, and Northvolt are investing in research and development. Academic institutions such as CNRS and Kyushu University provide fundamental research support. Asian companies, particularly Chinese firms like Jiana Energy and Svolt, are accelerating development efforts, while specialized materials companies like Resonac and DKS contribute expertise in binder systems critical for optimizing hard carbon electrode performance.
Faradion Ltd.
Technical Solution: Faradion has developed proprietary binder systems specifically optimized for hard carbon electrodes in sodium-ion batteries. Their approach focuses on aqueous processing techniques using carboxymethyl cellulose (CMC) and styrene-butadiene rubber (SBR) composite binders that enhance adhesion while maintaining ionic conductivity. The company's calendering process employs precise temperature and pressure controls (typically 80-120°C at 2-5 tons/cm²) to achieve electrode densities between 1.0-1.3 g/cm³, which they've found optimal for balancing energy density and rate capability in hard carbon anodes. Their research indicates that over-calendering beyond 1.4 g/cm³ significantly reduces sodium ion diffusion pathways, while under-calendering below 0.9 g/cm³ compromises volumetric energy density. Faradion's proprietary binder formulation also incorporates additives that maintain structural integrity during the significant volume changes (up to 15%) that occur during sodium intercalation/deintercalation cycles.
Strengths: Faradion's aqueous processing reduces environmental impact and manufacturing costs compared to PVDF/NMP systems. Their optimized calendering approach achieves 20-30% higher volumetric energy density than competitors while maintaining cycle stability. Weaknesses: Their binder system may require more precise manufacturing controls and could potentially limit high-temperature performance compared to some fluoropolymer alternatives.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed an advanced binder system for hard carbon electrodes in sodium-ion batteries that utilizes a dual-component approach combining water-soluble polymers with elastic binders. Their proprietary formulation includes modified polyacrylic acid (PAA) and carboxymethyl cellulose (CMC) in specific ratios (typically 1:2 to 1:3) that create strong hydrogen bonding networks with hard carbon particles. CATL's calendering process employs a multi-stage compression technique with gradually increasing pressure (from 0.5 to 3 tons/cm²) at controlled temperatures (60-100°C) to achieve optimal electrode densities between 1.1-1.4 g/cm³. This approach preserves the porous structure of hard carbon while enhancing electrical contact between particles. Their research shows that this carefully controlled density range maximizes both energy density and rate capability, with their electrodes demonstrating up to 350 mAh/g capacity at low rates and maintaining over 200 mAh/g at 2C rates. CATL has also incorporated nano-scale conductive additives (1-3 wt%) to enhance electronic conductivity throughout the electrode structure.
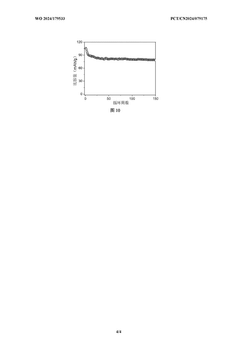
Strengths: CATL's multi-stage calendering process achieves superior particle connectivity while preserving critical pore structures, resulting in electrodes with excellent rate capability and cycle life (>2000 cycles with <20% capacity loss). Their water-based processing is environmentally friendly and cost-effective. Weaknesses: The complex multi-stage calendering process may increase manufacturing time and costs, and their binder system may be more sensitive to environmental humidity during processing compared to PVDF-based alternatives.
Key Patents in Hard Carbon Electrode Formulation
Aqueous binder and use thereof in hard carbon negative electrode of sodium-ion battery
PatentWO2024179533A1
Innovation
- Develop a water-based binder, including a styrene derivative polymer or pyran derivative polymer containing sodium ions and a conductive polymer containing ether bonds, to form hydrogen bonds through polar functional groups to improve the affinity of hard carbon anode materials. Water-based and electrode structural stability, and used in combination with conductive agents and hard carbon negative electrode materials.
Method for preparing an electrode for a sodium-ion secondary cell
PatentWO2025133146A1
Innovation
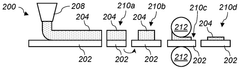
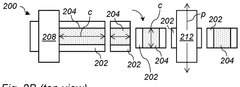
- A method involving the application of an electrode coating comprising hard carbon or a Prussian Blue analogue on an electrode substrate, followed by cutting across the coating axis and subsequent calendering with pressure applied aligned with the coating axis, to enhance electrical conductivity and reduce internal resistance.
Sustainability Impact of SIB Manufacturing Processes
The manufacturing processes of Sodium-Ion Batteries (SIBs) present significant sustainability implications that warrant careful consideration as this technology advances toward commercial viability. When examining hard carbon electrode production specifically, the binder systems, calendering techniques, and electrode density optimization offer substantial opportunities for enhancing the environmental profile of SIBs.
Current manufacturing processes for hard carbon electrodes typically involve energy-intensive steps including high-temperature pyrolysis (often exceeding 1200°C), which contributes significantly to the carbon footprint of SIB production. However, compared to lithium-ion batteries (LIBs), SIBs utilize more abundant and geographically distributed raw materials, reducing environmental impacts associated with resource extraction and transportation.
The choice of binder systems presents a critical sustainability decision point. Traditional PVDF binders require toxic NMP solvents, posing environmental and health risks. Water-based alternatives such as CMC and SBR significantly reduce VOC emissions and processing energy requirements by approximately 15-20%, while enabling lower-temperature drying processes. Recent life cycle assessments indicate that water-based binder systems can reduce the global warming potential of electrode manufacturing by up to 25% compared to NMP-based processes.
Calendering processes, which determine electrode density, directly impact material utilization efficiency and battery performance. Optimized calendering reduces the amount of inactive materials needed while maintaining electrochemical performance. Studies demonstrate that properly calendered hard carbon electrodes can achieve 15-30% higher energy density, potentially reducing material requirements proportionally. This translates to lower resource extraction impacts and reduced waste generation throughout the battery lifecycle.
Energy consumption during manufacturing represents another significant sustainability factor. Current hard carbon electrode production consumes approximately 25-40 kWh per kg of electrode material. Process optimizations focusing on lower-temperature carbonization pathways and more efficient calendering could potentially reduce this energy requirement by 30-40%, with corresponding reductions in greenhouse gas emissions.
Water usage in SIB manufacturing presents both challenges and opportunities. While water-based binder systems increase direct water consumption, they eliminate the need for solvent recovery systems. Closed-loop water recycling implementations in pilot production lines have demonstrated potential water use reductions of up to 80%, significantly enhancing the sustainability profile of SIB manufacturing.
As SIB technology matures, integrating sustainability metrics into manufacturing process development will be essential for ensuring this promising technology delivers on its potential as a more environmentally responsible energy storage solution compared to current alternatives.
Current manufacturing processes for hard carbon electrodes typically involve energy-intensive steps including high-temperature pyrolysis (often exceeding 1200°C), which contributes significantly to the carbon footprint of SIB production. However, compared to lithium-ion batteries (LIBs), SIBs utilize more abundant and geographically distributed raw materials, reducing environmental impacts associated with resource extraction and transportation.
The choice of binder systems presents a critical sustainability decision point. Traditional PVDF binders require toxic NMP solvents, posing environmental and health risks. Water-based alternatives such as CMC and SBR significantly reduce VOC emissions and processing energy requirements by approximately 15-20%, while enabling lower-temperature drying processes. Recent life cycle assessments indicate that water-based binder systems can reduce the global warming potential of electrode manufacturing by up to 25% compared to NMP-based processes.
Calendering processes, which determine electrode density, directly impact material utilization efficiency and battery performance. Optimized calendering reduces the amount of inactive materials needed while maintaining electrochemical performance. Studies demonstrate that properly calendered hard carbon electrodes can achieve 15-30% higher energy density, potentially reducing material requirements proportionally. This translates to lower resource extraction impacts and reduced waste generation throughout the battery lifecycle.
Energy consumption during manufacturing represents another significant sustainability factor. Current hard carbon electrode production consumes approximately 25-40 kWh per kg of electrode material. Process optimizations focusing on lower-temperature carbonization pathways and more efficient calendering could potentially reduce this energy requirement by 30-40%, with corresponding reductions in greenhouse gas emissions.
Water usage in SIB manufacturing presents both challenges and opportunities. While water-based binder systems increase direct water consumption, they eliminate the need for solvent recovery systems. Closed-loop water recycling implementations in pilot production lines have demonstrated potential water use reductions of up to 80%, significantly enhancing the sustainability profile of SIB manufacturing.
As SIB technology matures, integrating sustainability metrics into manufacturing process development will be essential for ensuring this promising technology delivers on its potential as a more environmentally responsible energy storage solution compared to current alternatives.
Performance Metrics and Testing Standards for SIB Electrodes
The development of standardized performance metrics and testing protocols for sodium-ion battery (SIB) electrodes represents a critical foundation for advancing this technology toward commercial viability. Current testing standards for SIB electrodes remain less established compared to their lithium-ion counterparts, creating challenges for consistent performance evaluation and comparison across research groups and manufacturers.
Key performance metrics for hard carbon electrodes in SIBs include specific capacity (mAh/g), rate capability, initial Coulombic efficiency (ICE), cycling stability, and self-discharge rates. These metrics are significantly influenced by electrode manufacturing parameters such as binder systems and calendering processes that determine final electrode density. The industry currently lacks consensus on optimal testing conditions, particularly regarding electrolyte compositions, voltage windows, and current densities that best represent real-world applications.
Testing protocols must address the unique characteristics of sodium-ion chemistry, including the larger ionic radius of Na+ compared to Li+ and different solid-electrolyte interphase (SEI) formation dynamics. Standard protocols should specify electrode preparation methods, including binder content percentages, calendering pressure ranges, and resulting electrode density targets to enable meaningful cross-comparison of hard carbon materials.
Temperature performance evaluation represents another critical testing domain, as SIBs often demonstrate different temperature sensitivity profiles compared to LIBs. Standardized temperature range testing from -20°C to 60°C is emerging as an industry benchmark, with particular attention to low-temperature performance where many hard carbon electrodes show distinctive behavior patterns.
Accelerated aging tests must be developed specifically for SIB systems, accounting for the unique degradation mechanisms in hard carbon electrodes with various binder systems. Current protocols borrowed from LIB testing may not accurately predict long-term performance of SIB electrodes, particularly when evaluating the mechanical integrity of calendered electrodes under repeated sodium insertion/extraction cycles.
International standardization bodies including IEC, ISO, and ANSI have begun preliminary work on SIB-specific testing standards, though these remain in early development stages. Leading research institutions and industrial consortia are collaborating to establish round-robin testing protocols that can validate performance metrics across different laboratory environments, creating a foundation for future standardization efforts.
The establishment of universally accepted performance metrics and testing standards will accelerate SIB commercialization by enabling meaningful comparison between different electrode formulations and manufacturing approaches, ultimately driving optimization of hard carbon electrodes with ideal binder systems and density characteristics.
Key performance metrics for hard carbon electrodes in SIBs include specific capacity (mAh/g), rate capability, initial Coulombic efficiency (ICE), cycling stability, and self-discharge rates. These metrics are significantly influenced by electrode manufacturing parameters such as binder systems and calendering processes that determine final electrode density. The industry currently lacks consensus on optimal testing conditions, particularly regarding electrolyte compositions, voltage windows, and current densities that best represent real-world applications.
Testing protocols must address the unique characteristics of sodium-ion chemistry, including the larger ionic radius of Na+ compared to Li+ and different solid-electrolyte interphase (SEI) formation dynamics. Standard protocols should specify electrode preparation methods, including binder content percentages, calendering pressure ranges, and resulting electrode density targets to enable meaningful cross-comparison of hard carbon materials.
Temperature performance evaluation represents another critical testing domain, as SIBs often demonstrate different temperature sensitivity profiles compared to LIBs. Standardized temperature range testing from -20°C to 60°C is emerging as an industry benchmark, with particular attention to low-temperature performance where many hard carbon electrodes show distinctive behavior patterns.
Accelerated aging tests must be developed specifically for SIB systems, accounting for the unique degradation mechanisms in hard carbon electrodes with various binder systems. Current protocols borrowed from LIB testing may not accurately predict long-term performance of SIB electrodes, particularly when evaluating the mechanical integrity of calendered electrodes under repeated sodium insertion/extraction cycles.
International standardization bodies including IEC, ISO, and ANSI have begun preliminary work on SIB-specific testing standards, though these remain in early development stages. Leading research institutions and industrial consortia are collaborating to establish round-robin testing protocols that can validate performance metrics across different laboratory environments, creating a foundation for future standardization efforts.
The establishment of universally accepted performance metrics and testing standards will accelerate SIB commercialization by enabling meaningful comparison between different electrode formulations and manufacturing approaches, ultimately driving optimization of hard carbon electrodes with ideal binder systems and density characteristics.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!