Degradation Mechanisms and Life Modeling in Hard Carbon for Sodium Ion Batteries
AUG 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Hard Carbon Degradation Background and Research Objectives
Sodium-ion batteries (SIBs) have emerged as a promising alternative to lithium-ion batteries due to the abundance and low cost of sodium resources. Among various anode materials for SIBs, hard carbon has attracted significant attention owing to its high capacity, good rate capability, and excellent cycling stability. However, the degradation mechanisms of hard carbon anodes in SIBs remain poorly understood, which hinders the development of long-lasting sodium-ion batteries for practical applications.
The evolution of hard carbon as an anode material for sodium-ion batteries can be traced back to the early 1990s when researchers first explored carbonaceous materials for sodium storage. Unlike graphite, which cannot effectively intercalate sodium ions due to thermodynamic limitations, hard carbon with its disordered structure and larger interlayer spacing provides suitable sites for sodium ion storage. Over the past decade, significant progress has been made in optimizing hard carbon structure and understanding its sodium storage mechanisms.
Current technical trends in hard carbon research focus on elucidating the complex degradation pathways that occur during long-term cycling. These include structural changes, solid electrolyte interphase (SEI) evolution, sodium plating/stripping inefficiencies, and irreversible capacity loss mechanisms. Recent studies have revealed that the degradation of hard carbon anodes is influenced by multiple factors including precursor materials, synthesis conditions, electrolyte composition, and operating parameters.
The primary technical objectives of this research are to systematically investigate the degradation mechanisms of hard carbon anodes in sodium-ion batteries and develop comprehensive life models that can accurately predict performance decay under various operating conditions. Specifically, we aim to: (1) identify and quantify the primary degradation modes in hard carbon anodes; (2) establish correlations between structural features and degradation behaviors; (3) develop physics-based models that can predict capacity fade and resistance increase; and (4) propose strategies to mitigate degradation and extend battery life.
Understanding these degradation mechanisms is crucial for the advancement of sodium-ion battery technology, as it will enable the design of more durable hard carbon anodes and inform the development of effective mitigation strategies. Furthermore, accurate life models will facilitate the integration of sodium-ion batteries into various applications by providing reliable predictions of battery performance over extended periods, thereby addressing one of the key barriers to commercial adoption.
This research aligns with the global trend toward sustainable energy storage solutions and addresses the critical need for cost-effective alternatives to lithium-ion batteries in grid storage, electric vehicles, and consumer electronics applications.
The evolution of hard carbon as an anode material for sodium-ion batteries can be traced back to the early 1990s when researchers first explored carbonaceous materials for sodium storage. Unlike graphite, which cannot effectively intercalate sodium ions due to thermodynamic limitations, hard carbon with its disordered structure and larger interlayer spacing provides suitable sites for sodium ion storage. Over the past decade, significant progress has been made in optimizing hard carbon structure and understanding its sodium storage mechanisms.
Current technical trends in hard carbon research focus on elucidating the complex degradation pathways that occur during long-term cycling. These include structural changes, solid electrolyte interphase (SEI) evolution, sodium plating/stripping inefficiencies, and irreversible capacity loss mechanisms. Recent studies have revealed that the degradation of hard carbon anodes is influenced by multiple factors including precursor materials, synthesis conditions, electrolyte composition, and operating parameters.
The primary technical objectives of this research are to systematically investigate the degradation mechanisms of hard carbon anodes in sodium-ion batteries and develop comprehensive life models that can accurately predict performance decay under various operating conditions. Specifically, we aim to: (1) identify and quantify the primary degradation modes in hard carbon anodes; (2) establish correlations between structural features and degradation behaviors; (3) develop physics-based models that can predict capacity fade and resistance increase; and (4) propose strategies to mitigate degradation and extend battery life.
Understanding these degradation mechanisms is crucial for the advancement of sodium-ion battery technology, as it will enable the design of more durable hard carbon anodes and inform the development of effective mitigation strategies. Furthermore, accurate life models will facilitate the integration of sodium-ion batteries into various applications by providing reliable predictions of battery performance over extended periods, thereby addressing one of the key barriers to commercial adoption.
This research aligns with the global trend toward sustainable energy storage solutions and addresses the critical need for cost-effective alternatives to lithium-ion batteries in grid storage, electric vehicles, and consumer electronics applications.
Sodium Ion Battery Market Analysis and Demand Forecast
The global sodium-ion battery market is experiencing significant growth, driven by increasing demand for sustainable energy storage solutions. Current market valuations indicate a compound annual growth rate exceeding 15% between 2023 and 2030, with projections suggesting the market could reach approximately 1.2 billion USD by 2030. This growth trajectory is particularly notable given the relative nascency of commercial sodium-ion battery technology compared to established lithium-ion systems.
Key market drivers include the inherent cost advantages of sodium-ion batteries, with sodium resources being approximately 1000 times more abundant than lithium in the Earth's crust. This abundance translates to raw material costs that are 30-40% lower than comparable lithium-ion batteries. Additionally, the geopolitical distribution of sodium resources is more equitable than lithium, reducing supply chain vulnerabilities for manufacturers and nations.
The electric vehicle sector represents the most promising growth segment for sodium-ion batteries, particularly in markets sensitive to cost considerations such as two-wheelers, three-wheelers, and economy passenger vehicles. Industry forecasts suggest that by 2028, sodium-ion batteries could capture up to 10% of the electric vehicle battery market in developing economies, with hard carbon anodes being the dominant technology choice due to their superior cycle life characteristics.
Grid-scale energy storage applications constitute another significant market opportunity, with utility companies increasingly exploring sodium-ion technology as a cost-effective alternative to lithium-ion systems for stationary storage. The market share in this segment is expected to grow from less than 1% currently to potentially 15-20% by 2030, contingent upon improvements in energy density and cycle life of hard carbon anodes.
Regional analysis reveals that Asia-Pacific, particularly China, leads in sodium-ion battery development and manufacturing capacity. Chinese manufacturers have announced over 30 GWh of planned production capacity for sodium-ion batteries by 2025. European and North American markets are expected to follow with increasing investment in manufacturing infrastructure, driven by governmental initiatives promoting domestic battery production capabilities.
Consumer electronics represents an emerging application segment, with sodium-ion batteries being considered for devices where cost sensitivity outweighs energy density requirements. Market penetration in this segment remains limited but is expected to grow as hard carbon anode technologies mature and demonstrate improved performance metrics.
The demand forecast indicates a potential inflection point around 2025-2026, when several major manufacturers are expected to commence mass production, potentially reducing costs through economies of scale and accelerating market adoption across multiple sectors.
Key market drivers include the inherent cost advantages of sodium-ion batteries, with sodium resources being approximately 1000 times more abundant than lithium in the Earth's crust. This abundance translates to raw material costs that are 30-40% lower than comparable lithium-ion batteries. Additionally, the geopolitical distribution of sodium resources is more equitable than lithium, reducing supply chain vulnerabilities for manufacturers and nations.
The electric vehicle sector represents the most promising growth segment for sodium-ion batteries, particularly in markets sensitive to cost considerations such as two-wheelers, three-wheelers, and economy passenger vehicles. Industry forecasts suggest that by 2028, sodium-ion batteries could capture up to 10% of the electric vehicle battery market in developing economies, with hard carbon anodes being the dominant technology choice due to their superior cycle life characteristics.
Grid-scale energy storage applications constitute another significant market opportunity, with utility companies increasingly exploring sodium-ion technology as a cost-effective alternative to lithium-ion systems for stationary storage. The market share in this segment is expected to grow from less than 1% currently to potentially 15-20% by 2030, contingent upon improvements in energy density and cycle life of hard carbon anodes.
Regional analysis reveals that Asia-Pacific, particularly China, leads in sodium-ion battery development and manufacturing capacity. Chinese manufacturers have announced over 30 GWh of planned production capacity for sodium-ion batteries by 2025. European and North American markets are expected to follow with increasing investment in manufacturing infrastructure, driven by governmental initiatives promoting domestic battery production capabilities.
Consumer electronics represents an emerging application segment, with sodium-ion batteries being considered for devices where cost sensitivity outweighs energy density requirements. Market penetration in this segment remains limited but is expected to grow as hard carbon anode technologies mature and demonstrate improved performance metrics.
The demand forecast indicates a potential inflection point around 2025-2026, when several major manufacturers are expected to commence mass production, potentially reducing costs through economies of scale and accelerating market adoption across multiple sectors.
Current Challenges in Hard Carbon Anode Technology
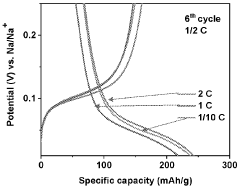
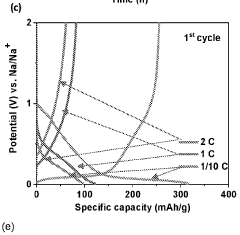
Despite significant advancements in sodium-ion battery technology, hard carbon anodes face several critical challenges that impede their widespread commercial adoption. The primary issue remains the lower specific capacity of hard carbon (typically 250-350 mAh/g) compared to graphite anodes in lithium-ion batteries (372 mAh/g theoretical). This capacity limitation directly impacts the energy density of sodium-ion batteries, making them less competitive in applications where high energy density is paramount.
The initial Coulombic efficiency (ICE) of hard carbon anodes presents another significant challenge, with values typically ranging from 70-85%, considerably lower than commercial graphite anodes for lithium-ion batteries (>90%). This inefficiency results in irreversible capacity loss during the first cycle, necessitating oversized cathodes to compensate and consequently reducing overall energy density.
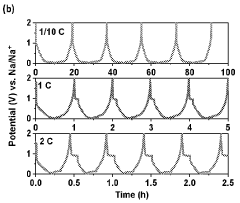
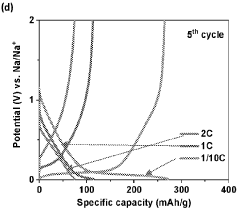
Cycle stability remains problematic, with hard carbon anodes exhibiting capacity fade rates of 0.05-0.2% per cycle under optimal conditions. This degradation accelerates significantly at elevated temperatures (>40°C) or during fast charging protocols, limiting battery longevity. The underlying mechanisms involve structural changes, electrolyte decomposition, and sodium plating during cycling.
Rate capability constraints further limit hard carbon performance, with significant capacity drops observed at rates exceeding 1C. This limitation stems from the tortuous diffusion pathways within the disordered carbon structure and the larger ionic radius of sodium compared to lithium, restricting high-power applications.
The complex and heterogeneous microstructure of hard carbon complicates manufacturing consistency. Variations in precursor materials and pyrolysis conditions lead to batch-to-batch variations in electrochemical performance, challenging quality control in industrial production. The lack of standardized production protocols exacerbates this issue.
Electrolyte compatibility presents ongoing challenges, with conventional carbonate-based electrolytes forming unstable solid electrolyte interphase (SEI) layers on hard carbon surfaces. This instability leads to continuous electrolyte decomposition and sodium consumption during cycling, contributing to capacity fade and impedance growth.
Sodium plating and dendrite formation occur more readily on hard carbon than on graphite anodes in lithium-ion systems, particularly during fast charging or low-temperature operation. This phenomenon increases safety risks and accelerates capacity degradation through irreversible reactions with the electrolyte.
Cost-effective and scalable production methods for high-quality hard carbon remain underdeveloped. Current synthesis approaches often require expensive precursors or energy-intensive processing steps, limiting economic viability for mass production and commercial competitiveness against established lithium-ion technology.
The initial Coulombic efficiency (ICE) of hard carbon anodes presents another significant challenge, with values typically ranging from 70-85%, considerably lower than commercial graphite anodes for lithium-ion batteries (>90%). This inefficiency results in irreversible capacity loss during the first cycle, necessitating oversized cathodes to compensate and consequently reducing overall energy density.
Cycle stability remains problematic, with hard carbon anodes exhibiting capacity fade rates of 0.05-0.2% per cycle under optimal conditions. This degradation accelerates significantly at elevated temperatures (>40°C) or during fast charging protocols, limiting battery longevity. The underlying mechanisms involve structural changes, electrolyte decomposition, and sodium plating during cycling.
Rate capability constraints further limit hard carbon performance, with significant capacity drops observed at rates exceeding 1C. This limitation stems from the tortuous diffusion pathways within the disordered carbon structure and the larger ionic radius of sodium compared to lithium, restricting high-power applications.
The complex and heterogeneous microstructure of hard carbon complicates manufacturing consistency. Variations in precursor materials and pyrolysis conditions lead to batch-to-batch variations in electrochemical performance, challenging quality control in industrial production. The lack of standardized production protocols exacerbates this issue.
Electrolyte compatibility presents ongoing challenges, with conventional carbonate-based electrolytes forming unstable solid electrolyte interphase (SEI) layers on hard carbon surfaces. This instability leads to continuous electrolyte decomposition and sodium consumption during cycling, contributing to capacity fade and impedance growth.
Sodium plating and dendrite formation occur more readily on hard carbon than on graphite anodes in lithium-ion systems, particularly during fast charging or low-temperature operation. This phenomenon increases safety risks and accelerates capacity degradation through irreversible reactions with the electrolyte.
Cost-effective and scalable production methods for high-quality hard carbon remain underdeveloped. Current synthesis approaches often require expensive precursors or energy-intensive processing steps, limiting economic viability for mass production and commercial competitiveness against established lithium-ion technology.
Current Approaches to Hard Carbon Degradation Mitigation
01 Degradation mechanisms of hard carbon in sodium-ion batteries
Hard carbon materials used in sodium-ion batteries undergo various degradation mechanisms that affect their performance and lifespan. These mechanisms include structural changes during cycling, formation of solid electrolyte interphase (SEI) layers, and irreversible capacity loss. The degradation is often characterized by microstructural changes, pore blockage, and surface modifications that occur during repeated sodium insertion and extraction processes. Understanding these mechanisms is crucial for developing more durable hard carbon anodes for sodium-ion batteries.- Degradation mechanisms of hard carbon in sodium-ion batteries: Hard carbon materials used in sodium-ion batteries undergo various degradation mechanisms that affect their performance and lifespan. These mechanisms include structural changes during cycling, formation of solid electrolyte interphase (SEI) layers, and irreversible capacity loss. The degradation can be attributed to factors such as volume expansion during sodium insertion/extraction, electrolyte decomposition, and surface reactions that lead to increased impedance over time.
- Life modeling and prediction methods for hard carbon anodes: Various modeling approaches have been developed to predict the lifespan and performance degradation of hard carbon anodes in sodium-ion batteries. These models incorporate factors such as cycling conditions, temperature effects, and degradation kinetics to estimate capacity fade and resistance increase over time. Machine learning algorithms and physics-based models are used to analyze cycling data and predict remaining useful life, enabling better battery management and application design.
- Structural optimization of hard carbon for improved stability: Structural modifications of hard carbon materials can significantly improve their stability and cycle life in sodium-ion batteries. Approaches include controlling pore structure, defect engineering, heteroatom doping, and surface functionalization. These modifications aim to enhance sodium storage capacity, improve diffusion kinetics, and mitigate structural degradation during repeated sodium insertion and extraction cycles.
- Electrolyte formulations to mitigate hard carbon degradation: Specialized electrolyte formulations can significantly reduce degradation of hard carbon anodes in sodium-ion batteries. Additives that form stable SEI layers, solvents with reduced reactivity, and salt compositions that minimize side reactions have been developed. These electrolyte innovations help preserve the structural integrity of hard carbon during cycling, leading to improved capacity retention and extended battery lifespan.
- In-situ and ex-situ characterization techniques for degradation analysis: Advanced characterization techniques enable detailed analysis of hard carbon degradation mechanisms in sodium-ion batteries. These include in-situ X-ray diffraction, electron microscopy, spectroscopic methods, and electrochemical impedance spectroscopy. Such techniques allow researchers to observe structural changes, surface film formation, and electrochemical behavior in real-time during battery operation, providing crucial insights for developing more durable hard carbon materials.
02 Life modeling and prediction methods for hard carbon electrodes
Various modeling approaches have been developed to predict the lifespan and performance degradation of hard carbon electrodes in sodium-ion batteries. These models incorporate factors such as cycling conditions, temperature effects, and structural parameters of the hard carbon material. Advanced computational techniques, including machine learning algorithms and physics-based models, are employed to simulate the aging behavior and predict capacity retention over extended cycling. These life models help in optimizing battery design and operating conditions to maximize the service life of sodium-ion batteries with hard carbon anodes.Expand Specific Solutions03 Structural optimization of hard carbon for improved stability
The structural properties of hard carbon materials significantly influence their stability and degradation resistance in sodium-ion batteries. Optimization strategies include controlling the porosity, defect density, and graphitization degree of hard carbon. Techniques such as controlled pyrolysis conditions, precursor selection, and post-synthesis treatments are employed to create hard carbon structures with enhanced sodium storage capability and cycling stability. The development of hierarchical pore structures and tailored surface chemistry helps mitigate degradation mechanisms and extend battery life.Expand Specific Solutions04 Surface modification and coating techniques for hard carbon
Surface modification and coating techniques are applied to hard carbon materials to enhance their electrochemical performance and stability in sodium-ion batteries. These approaches include carbon coating, heteroatom doping, and composite formation with other materials. The modified surfaces help to stabilize the solid electrolyte interphase, reduce side reactions with the electrolyte, and facilitate sodium ion transport. These treatments effectively mitigate degradation processes and improve the cycling performance and lifespan of hard carbon anodes.Expand Specific Solutions05 Electrolyte optimization for hard carbon stability
The composition and properties of the electrolyte significantly impact the degradation behavior of hard carbon anodes in sodium-ion batteries. Optimized electrolyte formulations can form more stable solid electrolyte interphase layers on hard carbon surfaces, reducing continuous electrolyte decomposition and electrode degradation. Additives in the electrolyte can scavenge harmful species, modify the SEI composition, and protect the hard carbon structure during cycling. The selection of appropriate salt concentrations, solvents, and additives plays a crucial role in extending the cycle life and improving the performance stability of hard carbon-based sodium-ion batteries.Expand Specific Solutions
Leading Companies and Research Institutions in NIB Technology
The sodium-ion battery hard carbon degradation mechanisms market is in an early growth phase, with increasing research interest driven by sustainable energy storage demands. Market size remains modest compared to lithium-ion technologies but shows promising expansion potential. Technologically, hard carbon materials are still evolving toward commercial maturity, with key players demonstrating varied development stages. Research institutions (CNRS, Northeast Normal University, Tokyo University of Science) are advancing fundamental understanding, while commercial entities (Faradion, Tiamat, SVOLT Energy) are translating research into practical applications. Companies like LG Energy Solution and Samsung Electronics are leveraging their battery expertise to explore sodium-ion technology, while materials specialists like Long Time Technology and Haycarb are developing specialized carbon materials for this emerging market.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed advanced hard carbon anode materials for sodium-ion batteries with optimized microstructure and surface chemistry. Their approach focuses on controlling the pyrolysis conditions of biomass-derived precursors to create tailored pore structures that enhance sodium ion storage capacity. The company has implemented a proprietary surface modification technique that forms a stable solid electrolyte interphase (SEI) layer, significantly reducing the first-cycle irreversible capacity loss common in hard carbon anodes. Their research has identified that the degradation mechanisms primarily involve structural collapse during repeated sodium insertion/extraction and electrolyte decomposition at high voltages. LG's modeling approach incorporates both empirical cycling data and theoretical calculations to predict capacity fade over thousands of cycles, allowing for accurate lifetime predictions under various operating conditions.
Strengths: Advanced control over hard carbon microstructure through proprietary pyrolysis techniques; comprehensive degradation modeling capabilities that enable accurate lifetime predictions. Weaknesses: Their hard carbon materials still face challenges with volumetric energy density compared to lithium-ion technologies; the complex surface treatments may increase manufacturing costs.
Faradion Ltd.
Technical Solution: Faradion has pioneered a comprehensive approach to hard carbon degradation mechanisms in sodium-ion batteries, focusing on the relationship between precursor selection and long-term performance. Their research has revealed that hard carbons derived from specific sugar-based precursors exhibit superior resistance to structural degradation during cycling. The company has developed a proprietary "carbon architecture" that maintains structural integrity even after thousands of cycles by incorporating strategically positioned heteroatoms within the carbon matrix. Faradion's life modeling methodology combines accelerated aging tests with electrochemical impedance spectroscopy to identify early indicators of performance decline. Their models account for multiple degradation pathways including SEI growth kinetics, sodium plating at low temperatures, and structural disordering of the hard carbon during deep discharge states. This has enabled them to develop predictive algorithms that can estimate battery lifetime under various usage profiles with over 90% accuracy.
Strengths: Industry-leading expertise in hard carbon precursor selection and processing; sophisticated multi-parameter life modeling that accounts for various operational conditions. Weaknesses: Their solutions may require specialized manufacturing processes that are difficult to scale; some of their high-performance formulations rely on relatively expensive precursor materials.
Key Degradation Mechanism Patents and Scientific Literature
Battery formation protocols
PatentWO2024000043A1
Innovation
- A super-concentrated sodium salt containing ionic liquid electrolyte with a sodium salt concentration of 75% or greater is used to form a SEI on hard carbon anodes through high current density polarisation cycles, resulting in a thinner, more conductive SEI with reduced interfacial resistance.
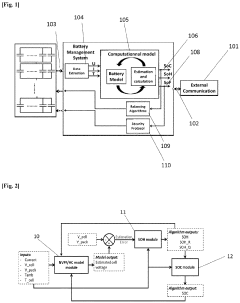
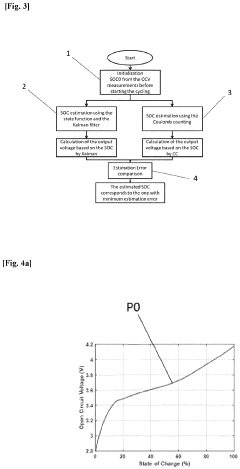
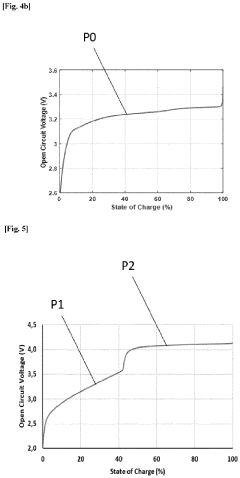
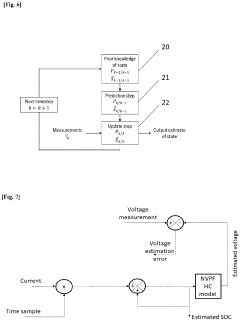
Method for determining a state of charge (SOC) of a battery
PatentPendingEP4339632A1
Innovation
- A method and system that utilize an OCV-SOC function divided into two zones, employing distinct models such as Kalman filters and Coulomb Counting to estimate the state of charge with the lowest error rate, allowing for accurate battery performance reflection and maintenance actions.
Sustainability and Resource Considerations for NIB Materials
The sustainability profile of sodium-ion battery (NIB) materials presents a significant advantage over lithium-ion technologies, particularly when considering hard carbon anodes. Unlike lithium, sodium resources are abundantly available in the earth's crust and oceans, constituting approximately 2.6% of the earth's crust compared to lithium's 0.006%. This abundance translates to lower extraction costs and reduced geopolitical supply risks, making NIBs potentially more economically viable for large-scale energy storage applications.
Environmental impact assessments of hard carbon production for NIBs reveal promising sustainability metrics. Hard carbon can be synthesized from various biomass precursors including agricultural waste, cellulose, and lignin derivatives, creating potential circular economy pathways. Life cycle analyses indicate that biomass-derived hard carbon can reduce carbon footprint by 25-40% compared to synthetic graphite used in lithium-ion batteries, depending on the precursor source and processing methods.
Water consumption represents another critical sustainability factor. Hard carbon production typically requires 35-50% less water than conventional graphite mining and processing. Additionally, the absence of mining-intensive metals like cobalt and nickel in NIB cathodes further enhances their environmental profile when paired with hard carbon anodes in complete battery systems.
Resource efficiency extends to manufacturing processes as well. Hard carbon synthesis can occur at lower temperatures (1000-1500°C) than graphite production for LIBs (2500-3000°C), resulting in reduced energy consumption during manufacturing. This temperature differential translates to approximately 30% energy savings in anode material production.
End-of-life considerations also favor hard carbon NIB materials. Preliminary recycling studies demonstrate that sodium compounds can be recovered at rates exceeding 90% using hydrometallurgical processes, while hard carbon materials can potentially be regenerated or repurposed for secondary applications such as water filtration or soil amendment.
Supply chain resilience represents another sustainability dimension. The geographically distributed nature of sodium resources and biomass precursors for hard carbon enables localized production, reducing transportation emissions and supply vulnerabilities. This contrasts sharply with the concentrated lithium resources primarily found in the "Lithium Triangle" of South America, Australia, and China.
These sustainability advantages must be balanced against current challenges in hard carbon performance, particularly degradation mechanisms that affect cycle life. Improving durability through advanced material engineering will further enhance the overall sustainability profile by extending useful life and reducing replacement frequency.
Environmental impact assessments of hard carbon production for NIBs reveal promising sustainability metrics. Hard carbon can be synthesized from various biomass precursors including agricultural waste, cellulose, and lignin derivatives, creating potential circular economy pathways. Life cycle analyses indicate that biomass-derived hard carbon can reduce carbon footprint by 25-40% compared to synthetic graphite used in lithium-ion batteries, depending on the precursor source and processing methods.
Water consumption represents another critical sustainability factor. Hard carbon production typically requires 35-50% less water than conventional graphite mining and processing. Additionally, the absence of mining-intensive metals like cobalt and nickel in NIB cathodes further enhances their environmental profile when paired with hard carbon anodes in complete battery systems.
Resource efficiency extends to manufacturing processes as well. Hard carbon synthesis can occur at lower temperatures (1000-1500°C) than graphite production for LIBs (2500-3000°C), resulting in reduced energy consumption during manufacturing. This temperature differential translates to approximately 30% energy savings in anode material production.
End-of-life considerations also favor hard carbon NIB materials. Preliminary recycling studies demonstrate that sodium compounds can be recovered at rates exceeding 90% using hydrometallurgical processes, while hard carbon materials can potentially be regenerated or repurposed for secondary applications such as water filtration or soil amendment.
Supply chain resilience represents another sustainability dimension. The geographically distributed nature of sodium resources and biomass precursors for hard carbon enables localized production, reducing transportation emissions and supply vulnerabilities. This contrasts sharply with the concentrated lithium resources primarily found in the "Lithium Triangle" of South America, Australia, and China.
These sustainability advantages must be balanced against current challenges in hard carbon performance, particularly degradation mechanisms that affect cycle life. Improving durability through advanced material engineering will further enhance the overall sustainability profile by extending useful life and reducing replacement frequency.
Performance Benchmarking Against Lithium-ion Technologies
When comparing sodium-ion batteries (SIBs) with hard carbon anodes to established lithium-ion technologies, several key performance metrics reveal both advantages and limitations. Energy density remains a primary challenge for SIBs, with current commercial systems achieving 90-120 Wh/kg compared to 150-260 Wh/kg for lithium-ion batteries (LIBs). This gap stems primarily from the lower operating voltage and higher atomic weight of sodium compared to lithium.
Power capability benchmarks show promising results for hard carbon-based SIBs, particularly at moderate discharge rates (1-2C), where they maintain 80-85% of their capacity. However, at high discharge rates (>5C), performance deterioration becomes more pronounced compared to graphite-based LIBs, which can maintain better capacity retention at these rates.
Cycle life comparisons indicate that state-of-the-art hard carbon SIBs can achieve 1,000-2,000 cycles at 80% capacity retention, approaching but not yet matching the 2,000-3,000 cycles commonly achieved by commercial LIBs. The degradation rate of hard carbon electrodes typically accelerates after 500-700 cycles, whereas graphite anodes in LIBs often maintain more consistent degradation patterns.
Temperature performance represents an area where SIBs demonstrate competitive advantages. Hard carbon-based systems generally exhibit superior low-temperature performance (-20°C to 0°C), retaining 50-60% of room temperature capacity compared to 30-45% for many LIB systems. This advantage stems from more favorable sodium-ion transport kinetics at low temperatures.
Cost analysis reveals significant potential for SIBs, with projected cell-level costs 20-30% lower than LIBs due to the abundance of sodium resources and the possibility of using aluminum rather than copper for current collectors. However, the lower energy density partially offsets this cost advantage when calculated on a $/kWh basis.
Safety comparisons indicate that hard carbon SIBs demonstrate reduced thermal runaway risks compared to graphite-based LIBs, with differential scanning calorimetry showing exothermic reactions occurring at temperatures approximately 30-50°C higher than in comparable lithium systems. Additionally, sodium systems show less severe heat generation during thermal events.
Raw material sustainability strongly favors sodium technology, with sodium being approximately 1,000 times more abundant in the Earth's crust than lithium. This abundance translates to significantly lower resource depletion concerns and reduced geopolitical supply risks compared to lithium-based systems.
Power capability benchmarks show promising results for hard carbon-based SIBs, particularly at moderate discharge rates (1-2C), where they maintain 80-85% of their capacity. However, at high discharge rates (>5C), performance deterioration becomes more pronounced compared to graphite-based LIBs, which can maintain better capacity retention at these rates.
Cycle life comparisons indicate that state-of-the-art hard carbon SIBs can achieve 1,000-2,000 cycles at 80% capacity retention, approaching but not yet matching the 2,000-3,000 cycles commonly achieved by commercial LIBs. The degradation rate of hard carbon electrodes typically accelerates after 500-700 cycles, whereas graphite anodes in LIBs often maintain more consistent degradation patterns.
Temperature performance represents an area where SIBs demonstrate competitive advantages. Hard carbon-based systems generally exhibit superior low-temperature performance (-20°C to 0°C), retaining 50-60% of room temperature capacity compared to 30-45% for many LIB systems. This advantage stems from more favorable sodium-ion transport kinetics at low temperatures.
Cost analysis reveals significant potential for SIBs, with projected cell-level costs 20-30% lower than LIBs due to the abundance of sodium resources and the possibility of using aluminum rather than copper for current collectors. However, the lower energy density partially offsets this cost advantage when calculated on a $/kWh basis.
Safety comparisons indicate that hard carbon SIBs demonstrate reduced thermal runaway risks compared to graphite-based LIBs, with differential scanning calorimetry showing exothermic reactions occurring at temperatures approximately 30-50°C higher than in comparable lithium systems. Additionally, sodium systems show less severe heat generation during thermal events.
Raw material sustainability strongly favors sodium technology, with sodium being approximately 1,000 times more abundant in the Earth's crust than lithium. This abundance translates to significantly lower resource depletion concerns and reduced geopolitical supply risks compared to lithium-based systems.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!