Surface Treatments and Protective Coatings for Hard Carbon in Sodium Ion Batteries
AUG 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Hard Carbon Surface Treatment Background and Objectives
Hard carbon has emerged as a promising anode material for sodium-ion batteries (SIBs) due to its unique structural characteristics and excellent electrochemical performance. The development of surface treatments and protective coatings for hard carbon represents a critical frontier in advancing SIB technology. This research area has evolved significantly over the past decade, transitioning from basic material exploration to sophisticated surface engineering approaches aimed at enhancing battery performance and longevity.
The historical trajectory of hard carbon surface modification began with rudimentary carbon coating techniques in the early 2010s. These initial efforts focused primarily on improving the first cycle coulombic efficiency and reducing irreversible capacity loss. As research progressed, more complex surface treatment methodologies emerged, including heteroatom doping, metal oxide coatings, and polymer-derived protective layers, each addressing specific performance limitations of pristine hard carbon anodes.
Current technological trends indicate a shift toward multifunctional surface treatments that simultaneously address multiple performance parameters. These advanced approaches aim to create synergistic effects between the hard carbon substrate and the protective coating, optimizing interfacial properties while maintaining the intrinsic advantages of hard carbon materials. The integration of nanotechnology and precision surface engineering has further accelerated innovation in this domain.
The primary technical objectives of hard carbon surface treatment research encompass several interconnected goals. First, to mitigate the formation of unstable solid electrolyte interphase (SEI) layers that contribute to capacity fading and reduced cycle life. Second, to enhance sodium ion diffusion kinetics at the electrode-electrolyte interface, thereby improving rate capability and power density. Third, to prevent unwanted side reactions between the electrolyte and hard carbon surface, which can lead to gas evolution and safety concerns.
Additionally, researchers aim to develop scalable and cost-effective surface modification techniques that can be readily implemented in industrial manufacturing processes. This includes the exploration of environmentally friendly coating materials and processes that align with sustainable battery production objectives. The ultimate goal is to establish standardized surface treatment protocols that can be tailored to specific hard carbon precursors and battery application requirements.
Looking forward, the field is expected to evolve toward precision engineering of hard carbon interfaces at the atomic and molecular levels. This will likely involve the development of self-healing coatings, stimuli-responsive surface treatments, and biomimetic approaches inspired by natural protective mechanisms. These advancements will be crucial for enabling next-generation sodium-ion batteries with performance metrics competitive with current lithium-ion technologies.
The historical trajectory of hard carbon surface modification began with rudimentary carbon coating techniques in the early 2010s. These initial efforts focused primarily on improving the first cycle coulombic efficiency and reducing irreversible capacity loss. As research progressed, more complex surface treatment methodologies emerged, including heteroatom doping, metal oxide coatings, and polymer-derived protective layers, each addressing specific performance limitations of pristine hard carbon anodes.
Current technological trends indicate a shift toward multifunctional surface treatments that simultaneously address multiple performance parameters. These advanced approaches aim to create synergistic effects between the hard carbon substrate and the protective coating, optimizing interfacial properties while maintaining the intrinsic advantages of hard carbon materials. The integration of nanotechnology and precision surface engineering has further accelerated innovation in this domain.
The primary technical objectives of hard carbon surface treatment research encompass several interconnected goals. First, to mitigate the formation of unstable solid electrolyte interphase (SEI) layers that contribute to capacity fading and reduced cycle life. Second, to enhance sodium ion diffusion kinetics at the electrode-electrolyte interface, thereby improving rate capability and power density. Third, to prevent unwanted side reactions between the electrolyte and hard carbon surface, which can lead to gas evolution and safety concerns.
Additionally, researchers aim to develop scalable and cost-effective surface modification techniques that can be readily implemented in industrial manufacturing processes. This includes the exploration of environmentally friendly coating materials and processes that align with sustainable battery production objectives. The ultimate goal is to establish standardized surface treatment protocols that can be tailored to specific hard carbon precursors and battery application requirements.
Looking forward, the field is expected to evolve toward precision engineering of hard carbon interfaces at the atomic and molecular levels. This will likely involve the development of self-healing coatings, stimuli-responsive surface treatments, and biomimetic approaches inspired by natural protective mechanisms. These advancements will be crucial for enabling next-generation sodium-ion batteries with performance metrics competitive with current lithium-ion technologies.
Market Analysis for Na-ion Battery Protective Coatings
The global market for sodium-ion battery protective coatings is experiencing significant growth, driven by the increasing demand for sustainable and cost-effective energy storage solutions. As lithium resources face supply constraints and price volatility, sodium-ion batteries have emerged as a promising alternative, particularly for grid storage applications and electric vehicles in price-sensitive markets. The protective coating segment specifically addresses critical performance issues in hard carbon anodes, which are essential components in Na-ion battery technology.
Current market assessments indicate that the Na-ion battery market is projected to grow at a compound annual growth rate of 25-30% through 2030, with the protective coatings segment expected to expand proportionally. This growth is primarily fueled by the inherent advantages of sodium-ion technology, including abundant raw material availability, lower production costs, and enhanced safety profiles compared to lithium-ion counterparts.
Regional analysis reveals that China currently dominates the market landscape, with companies like CATL and HiNa Battery Technology leading commercial deployment efforts. European markets are rapidly developing research capabilities, particularly in countries like France, Germany, and the UK, where academic-industrial partnerships are accelerating commercialization timelines. North America shows increasing interest, though market penetration remains in early stages compared to Asian counterparts.
The protective coating market segment specifically addresses the performance challenges of hard carbon anodes in sodium-ion batteries. These coatings mitigate issues related to solid electrolyte interphase (SEI) formation, electrolyte decomposition, and structural degradation during cycling. Market research indicates that effective surface treatments can extend battery cycle life by 30-50% and improve capacity retention by 15-25%, creating substantial value propositions for manufacturers.
End-user industries driving demand include stationary energy storage systems, which represent approximately 60% of current market applications. The electric vehicle sector, particularly in the two-wheeler and commercial vehicle segments, accounts for roughly 25% of market demand, with consumer electronics and other applications comprising the remainder.
Price sensitivity analysis reveals that protective coating solutions must add no more than 5-8% to overall cell costs to maintain commercial viability. This constraint has created market opportunities for innovative coating technologies that balance performance improvements with cost considerations. Solutions utilizing abundant materials and scalable deposition techniques are gaining particular traction among battery manufacturers.
Market forecasts suggest that as Na-ion battery production scales from current pilot levels to gigawatt-hour capacities by 2025-2027, the protective coating market will experience corresponding growth, potentially reaching several hundred million dollars annually by 2030. This expansion will likely trigger consolidation among coating technology providers and integration with larger battery manufacturing ecosystems.
Current market assessments indicate that the Na-ion battery market is projected to grow at a compound annual growth rate of 25-30% through 2030, with the protective coatings segment expected to expand proportionally. This growth is primarily fueled by the inherent advantages of sodium-ion technology, including abundant raw material availability, lower production costs, and enhanced safety profiles compared to lithium-ion counterparts.
Regional analysis reveals that China currently dominates the market landscape, with companies like CATL and HiNa Battery Technology leading commercial deployment efforts. European markets are rapidly developing research capabilities, particularly in countries like France, Germany, and the UK, where academic-industrial partnerships are accelerating commercialization timelines. North America shows increasing interest, though market penetration remains in early stages compared to Asian counterparts.
The protective coating market segment specifically addresses the performance challenges of hard carbon anodes in sodium-ion batteries. These coatings mitigate issues related to solid electrolyte interphase (SEI) formation, electrolyte decomposition, and structural degradation during cycling. Market research indicates that effective surface treatments can extend battery cycle life by 30-50% and improve capacity retention by 15-25%, creating substantial value propositions for manufacturers.
End-user industries driving demand include stationary energy storage systems, which represent approximately 60% of current market applications. The electric vehicle sector, particularly in the two-wheeler and commercial vehicle segments, accounts for roughly 25% of market demand, with consumer electronics and other applications comprising the remainder.
Price sensitivity analysis reveals that protective coating solutions must add no more than 5-8% to overall cell costs to maintain commercial viability. This constraint has created market opportunities for innovative coating technologies that balance performance improvements with cost considerations. Solutions utilizing abundant materials and scalable deposition techniques are gaining particular traction among battery manufacturers.
Market forecasts suggest that as Na-ion battery production scales from current pilot levels to gigawatt-hour capacities by 2025-2027, the protective coating market will experience corresponding growth, potentially reaching several hundred million dollars annually by 2030. This expansion will likely trigger consolidation among coating technology providers and integration with larger battery manufacturing ecosystems.
Current Challenges in Hard Carbon Surface Modification
Hard carbon materials in sodium-ion batteries (SIBs) face several critical surface-related challenges that impede their commercial viability. The primary issue is the formation of unstable solid electrolyte interphase (SEI) layers during cycling. Unlike lithium-ion batteries, sodium ions interact differently with electrolytes, resulting in thicker, less stable SEI formations on hard carbon surfaces. This instability leads to continuous electrolyte decomposition, increased impedance, and accelerated capacity fading during long-term cycling.
Surface reactivity presents another significant challenge. Hard carbon surfaces contain numerous reactive sites including oxygen-containing functional groups, defects, and edge sites that participate in parasitic reactions with electrolytes. These side reactions not only consume active sodium ions but also generate gas products that compromise cell safety and performance, particularly at elevated temperatures or during fast charging operations.
The inherent hydrophilicity of hard carbon surfaces poses additional complications. These materials readily absorb moisture from the environment, which can subsequently react with sodium and electrolyte components during battery operation. This moisture sensitivity necessitates stringent manufacturing controls and adds complexity to production processes, increasing overall costs.
Sodium ion co-intercalation with solvent molecules represents a unique challenge for hard carbon anodes. The larger ionic radius of sodium compared to lithium makes co-intercalation of solvent molecules more pronounced, causing excessive expansion of the carbon structure and mechanical degradation over multiple cycles. This phenomenon is particularly problematic in ether-based electrolytes, which are otherwise advantageous for their high ionic conductivity.
The non-uniform surface chemistry of hard carbon materials further complicates matters. Variations in surface functionality across different production batches lead to inconsistent electrochemical performance. This heterogeneity makes it difficult to develop standardized surface modification approaches that work universally across different hard carbon sources and preparation methods.
Current surface modification techniques also struggle with scalability issues. Laboratory-scale treatments that show promising results often face significant challenges when scaled to industrial production levels. Many coating processes require complex equipment, precise control of reaction conditions, or environmentally problematic solvents, limiting their commercial viability.
Finally, there exists a fundamental knowledge gap regarding the precise mechanisms of sodium ion storage at hard carbon surfaces versus bulk insertion. This incomplete understanding hampers the development of targeted surface modification strategies that could selectively enhance beneficial surface reactions while suppressing detrimental ones.
Surface reactivity presents another significant challenge. Hard carbon surfaces contain numerous reactive sites including oxygen-containing functional groups, defects, and edge sites that participate in parasitic reactions with electrolytes. These side reactions not only consume active sodium ions but also generate gas products that compromise cell safety and performance, particularly at elevated temperatures or during fast charging operations.
The inherent hydrophilicity of hard carbon surfaces poses additional complications. These materials readily absorb moisture from the environment, which can subsequently react with sodium and electrolyte components during battery operation. This moisture sensitivity necessitates stringent manufacturing controls and adds complexity to production processes, increasing overall costs.
Sodium ion co-intercalation with solvent molecules represents a unique challenge for hard carbon anodes. The larger ionic radius of sodium compared to lithium makes co-intercalation of solvent molecules more pronounced, causing excessive expansion of the carbon structure and mechanical degradation over multiple cycles. This phenomenon is particularly problematic in ether-based electrolytes, which are otherwise advantageous for their high ionic conductivity.
The non-uniform surface chemistry of hard carbon materials further complicates matters. Variations in surface functionality across different production batches lead to inconsistent electrochemical performance. This heterogeneity makes it difficult to develop standardized surface modification approaches that work universally across different hard carbon sources and preparation methods.
Current surface modification techniques also struggle with scalability issues. Laboratory-scale treatments that show promising results often face significant challenges when scaled to industrial production levels. Many coating processes require complex equipment, precise control of reaction conditions, or environmentally problematic solvents, limiting their commercial viability.
Finally, there exists a fundamental knowledge gap regarding the precise mechanisms of sodium ion storage at hard carbon surfaces versus bulk insertion. This incomplete understanding hampers the development of targeted surface modification strategies that could selectively enhance beneficial surface reactions while suppressing detrimental ones.
Current Surface Treatment Solutions for Hard Carbon
01 Surface modification techniques for hard carbon
Various surface modification techniques can be applied to hard carbon materials used in sodium-ion batteries to enhance their electrochemical performance. These techniques include chemical treatments, physical modifications, and coating processes that alter the surface properties of hard carbon. Surface modifications can improve the interface between the hard carbon and the electrolyte, enhance sodium ion diffusion, and reduce unwanted side reactions, ultimately leading to better battery performance and longevity.- Surface modification techniques for hard carbon: Various surface modification techniques can be applied to hard carbon materials used in sodium ion batteries to enhance their electrochemical performance. These techniques include chemical treatments, plasma treatments, and thermal treatments that modify the surface properties of hard carbon. Surface modification can improve the interface between the hard carbon and the electrolyte, enhance sodium ion diffusion, and increase the overall battery efficiency and cycle life.
- Protective coating materials for hard carbon anodes: Protective coatings can be applied to hard carbon anodes in sodium ion batteries to prevent unwanted side reactions with the electrolyte and improve stability. These coating materials include metal oxides, polymers, carbon-based materials, and composite coatings. The protective layers help to suppress the formation of solid electrolyte interphase (SEI), reduce irreversible capacity loss, and enhance the cycling performance of the batteries.
- Doping strategies for hard carbon materials: Doping hard carbon materials with heteroatoms or functional groups can significantly improve their performance in sodium ion batteries. Common dopants include nitrogen, phosphorus, sulfur, and various metals. Doping can create additional active sites for sodium storage, enhance electronic conductivity, and improve the structural stability of hard carbon during charge-discharge cycles, leading to better rate capability and cycling performance.
- Composite structures with hard carbon: Creating composite structures by combining hard carbon with other materials can enhance the performance of sodium ion battery anodes. These composites may include hard carbon/metal oxide hybrids, hard carbon/conductive polymer composites, and hard carbon/graphene structures. The composite approach can leverage the advantages of each component while mitigating their individual limitations, resulting in improved sodium storage capacity, better rate performance, and enhanced cycling stability.
- Electrolyte additives for hard carbon interface optimization: Specific electrolyte additives can be used to optimize the interface between hard carbon anodes and the electrolyte in sodium ion batteries. These additives can form stable protective films on the hard carbon surface, reduce unwanted side reactions, and improve the sodium ion transport properties. The careful selection of electrolyte additives can lead to enhanced coulombic efficiency, reduced irreversible capacity, and improved long-term cycling stability of hard carbon-based sodium ion batteries.
02 Protective coating materials for hard carbon anodes
Protective coatings applied to hard carbon anodes in sodium-ion batteries can significantly improve their stability and performance. These coatings include carbon-based materials, metal oxides, polymers, and composite materials that form a protective layer on the hard carbon surface. Such coatings help prevent direct contact between the hard carbon and the electrolyte, reducing irreversible capacity loss and improving cycling stability while maintaining efficient sodium ion transport.Expand Specific Solutions03 Doping and heteroatom incorporation in hard carbon
Introducing heteroatoms such as nitrogen, sulfur, phosphorus, or boron into hard carbon structures can modify their electronic properties and surface characteristics. These doping strategies create active sites for sodium ion storage, improve the conductivity of hard carbon, and enhance the overall electrochemical performance of sodium-ion batteries. The doped hard carbon materials often exhibit higher capacity, better rate capability, and improved cycling stability compared to pristine hard carbon.Expand Specific Solutions04 Composite structures with hard carbon
Composite structures combining hard carbon with other materials can create synergistic effects that enhance the performance of sodium-ion battery anodes. These composites may include hard carbon with metal oxides, metal sulfides, other carbon allotropes, or conductive polymers. The composite structures can provide additional sodium storage sites, improve electrical conductivity, buffer volume changes during cycling, and enhance the overall electrochemical performance of the anode material.Expand Specific Solutions05 Electrolyte additives and interface engineering
Engineering the interface between hard carbon anodes and electrolytes through specialized additives can significantly improve battery performance. These additives help form stable solid electrolyte interphase (SEI) layers on hard carbon surfaces, reducing irreversible capacity loss and enhancing cycling stability. Interface engineering approaches include using film-forming additives, ionic liquid additives, or functional electrolyte components that modify the surface chemistry of hard carbon and improve its compatibility with the electrolyte system.Expand Specific Solutions
Key Industry Players in Battery Coating Technologies
The surface treatment and protective coating market for hard carbon in sodium-ion batteries is currently in an early growth phase, with expanding market potential driven by the increasing demand for sustainable energy storage solutions. The global market size is projected to grow significantly as sodium-ion technology emerges as a cost-effective alternative to lithium-ion batteries. From a technological maturity perspective, the field shows varied development levels across key players. CATL and SK Innovation are leading commercial development with advanced coating technologies, while academic institutions like Northwestern University and Central South University are pioneering fundamental research. Companies like Furukawa Electric and Resonac Holdings are leveraging their materials expertise to develop specialized coatings, while battery manufacturers such as Jiangsu Zenergy and NEC Energy Devices are integrating these technologies into their production processes.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: Contemporary Amperex Technology Co., Ltd. (CATL) has developed advanced surface treatment technologies for hard carbon anodes in sodium-ion batteries. Their approach involves a multi-step process including fluorination treatment and carbon coating. The fluorination process creates a stable SEI (Solid Electrolyte Interphase) layer on hard carbon particles, significantly reducing irreversible capacity loss during initial cycles. CATL's proprietary carbon coating technology applies a thin, uniform layer of conductive carbon on hard carbon particles through chemical vapor deposition, enhancing electrical conductivity while protecting the underlying material from direct electrolyte contact. Additionally, they've implemented nitrogen-doping techniques that modify the surface functional groups of hard carbon, improving sodium ion diffusion kinetics and storage capacity. Their commercial sodium-ion batteries utilizing these surface-treated hard carbon anodes have demonstrated cycle life exceeding 3,000 cycles with capacity retention above 80%, positioning CATL as a leader in sodium-ion battery commercialization.
Strengths: CATL's surface treatment technologies significantly improve the first cycle efficiency (>85%) and cycling stability of hard carbon anodes. Their integrated manufacturing capabilities allow for scalable production of treated materials. Weaknesses: The fluorination process requires careful handling of hazardous materials and adds manufacturing complexity. The additional processing steps increase production costs compared to untreated hard carbon materials.
SK Innovation Co., Ltd.
Technical Solution: SK Innovation has pioneered a proprietary surface modification technique for hard carbon anodes in sodium-ion batteries focusing on polymer-derived protective coatings. Their approach utilizes a controlled polymerization process that deposits an ultrathin (5-20nm) elastomeric polymer layer on hard carbon particles. This polymer coating undergoes partial carbonization during initial battery formation, creating a flexible yet protective barrier that accommodates the volume changes during sodium insertion/extraction while preventing direct electrolyte contact. The company has further enhanced this technology by incorporating functional additives into the polymer coating, including fluorinated compounds and silicon-based materials that improve SEI stability. SK Innovation's research has demonstrated that these surface-treated hard carbon anodes exhibit significantly reduced irreversible capacity loss (below 15% compared to 25-30% for untreated materials) and improved rate capability. Their latest generation of coatings incorporates ionic conductivity enhancers that facilitate sodium ion transport through the protective layer while maintaining excellent electrolyte resistance properties.
Strengths: The elastomeric nature of SK's coatings provides superior accommodation of volume changes during cycling, reducing mechanical degradation. Their coating process is compatible with existing manufacturing equipment, facilitating industrial implementation. Weaknesses: The polymer-derived coatings may have limited thermal stability at elevated temperatures, potentially restricting application in high-temperature environments. The partial carbonization process requires precise control to achieve optimal protective properties.
Critical Patents in Hard Carbon Protective Coatings
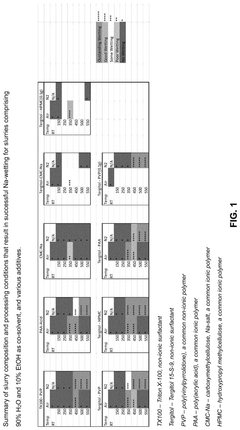
Carbon based surface treatment on substrates to improve wettability
PatentPendingUS20250183362A1
Innovation
- A carbon-based sodiophilic treatment is developed, which forms a meso/nanoporous sodiophilic layer using inexpensive components, easily applied via spray-coating, and provides excellent wetting as low as 110°C, without requiring metals.
Positive electrode active material powder for sodium ion secondary battery
PatentInactiveJP2016173962A
Innovation
- A positive electrode active material powder for sodium ion secondary batteries is coated with carbon, with a specific amount of carbon coating ranging from 0.1 to 1.8 mg/m², and composed of inorganic powders containing transition metals like Cr, Fe, Mn, Co, and Ni, and Na, P, and O, following the general formula Na x M y P 2 O z, where x, y, and z are within specified ranges, to enhance rapid charging and discharging characteristics.
Environmental Impact of Surface Treatment Processes
The surface treatment processes employed for hard carbon materials in sodium-ion batteries present significant environmental considerations that warrant thorough examination. Chemical etching, plasma treatment, and coating deposition methods often involve hazardous substances including strong acids, organic solvents, and volatile compounds that pose risks to both environmental and human health when improperly managed.
Traditional acid treatments utilizing sulfuric, nitric, or hydrofluoric acids generate acidic waste streams requiring neutralization and proper disposal protocols. These processes consume substantial water resources for rinsing and cleaning steps, contributing to industrial wastewater challenges. Similarly, organic solvent-based treatments release volatile organic compounds (VOCs) that contribute to air pollution and potential ozone depletion when emitted without adequate control systems.
Energy consumption represents another critical environmental factor, particularly for high-temperature thermal treatments and plasma-based surface modifications. These energy-intensive processes indirectly contribute to carbon emissions when powered by non-renewable energy sources, potentially offsetting some of the environmental benefits sought through sodium-ion battery development.
Life cycle assessment (LCA) studies indicate that certain coating materials, particularly those containing rare earth elements or precious metals, carry substantial environmental footprints associated with their mining and refinement. The environmental impact extends beyond production to end-of-life considerations, where the presence of these coatings may complicate recycling processes for battery materials.
Recent advancements in green chemistry approaches offer promising alternatives to conventional surface treatments. Water-based processes, supercritical CO2 treatments, and ionic liquid methodologies demonstrate reduced environmental impact while maintaining performance enhancements. Additionally, biomass-derived carbon coatings represent a sustainable alternative to synthetic carbon sources, potentially creating carbon-negative material pathways.
Regulatory frameworks worldwide are increasingly addressing these environmental concerns. The European Union's REACH regulations and similar initiatives in North America and Asia are driving the development of environmentally benign surface treatment alternatives. Industry leaders are responding by implementing closed-loop processing systems that recover and reuse treatment chemicals, significantly reducing waste generation and resource consumption.
The environmental impact assessment of surface treatments must be balanced against performance benefits and battery longevity. Surface modifications that extend battery cycle life effectively reduce the environmental burden through decreased replacement frequency and material consumption over the product lifecycle, potentially justifying moderate environmental impacts during manufacturing.
Traditional acid treatments utilizing sulfuric, nitric, or hydrofluoric acids generate acidic waste streams requiring neutralization and proper disposal protocols. These processes consume substantial water resources for rinsing and cleaning steps, contributing to industrial wastewater challenges. Similarly, organic solvent-based treatments release volatile organic compounds (VOCs) that contribute to air pollution and potential ozone depletion when emitted without adequate control systems.
Energy consumption represents another critical environmental factor, particularly for high-temperature thermal treatments and plasma-based surface modifications. These energy-intensive processes indirectly contribute to carbon emissions when powered by non-renewable energy sources, potentially offsetting some of the environmental benefits sought through sodium-ion battery development.
Life cycle assessment (LCA) studies indicate that certain coating materials, particularly those containing rare earth elements or precious metals, carry substantial environmental footprints associated with their mining and refinement. The environmental impact extends beyond production to end-of-life considerations, where the presence of these coatings may complicate recycling processes for battery materials.
Recent advancements in green chemistry approaches offer promising alternatives to conventional surface treatments. Water-based processes, supercritical CO2 treatments, and ionic liquid methodologies demonstrate reduced environmental impact while maintaining performance enhancements. Additionally, biomass-derived carbon coatings represent a sustainable alternative to synthetic carbon sources, potentially creating carbon-negative material pathways.
Regulatory frameworks worldwide are increasingly addressing these environmental concerns. The European Union's REACH regulations and similar initiatives in North America and Asia are driving the development of environmentally benign surface treatment alternatives. Industry leaders are responding by implementing closed-loop processing systems that recover and reuse treatment chemicals, significantly reducing waste generation and resource consumption.
The environmental impact assessment of surface treatments must be balanced against performance benefits and battery longevity. Surface modifications that extend battery cycle life effectively reduce the environmental burden through decreased replacement frequency and material consumption over the product lifecycle, potentially justifying moderate environmental impacts during manufacturing.
Scalability and Manufacturing Considerations
The scalability of surface treatments and protective coatings for hard carbon in sodium-ion batteries represents a critical factor in their commercial viability. Current laboratory-scale coating processes often employ methods such as atomic layer deposition (ALD), chemical vapor deposition (CVD), or solution-based techniques that demonstrate excellent performance but face significant challenges when transitioning to mass production environments.
Manufacturing considerations must address the cost-effectiveness of coating materials and processes. While certain high-performance coatings like Al2O3 or TiO2 deposited via ALD provide superior protection, the associated equipment costs and slow deposition rates (typically 1-2 nm/hour) create substantial bottlenecks for industrial-scale implementation. Alternative approaches using solution-based methods offer better throughput but may suffer from coating uniformity issues.
Process integration presents another key challenge, as coating steps must be seamlessly incorporated into existing battery manufacturing lines. The ideal coating technology should operate continuously rather than in batch processes to align with high-volume electrode production. Current industry standards for lithium-ion battery manufacturing achieve throughput rates of 20-30 m/min for electrode coating; protective coating processes for hard carbon must approach similar efficiency metrics to be commercially viable.
Environmental and safety considerations also impact scalability. Many coating precursors, particularly those used in vapor deposition techniques, involve toxic or highly reactive chemicals that require specialized handling and waste management systems. Water-based coating alternatives, while more environmentally friendly, often necessitate additional drying steps that increase energy consumption and production time.
Quality control represents a significant manufacturing challenge, as coating thickness uniformity and defect detection must be maintained across large production volumes. In-line monitoring technologies capable of real-time assessment of coating integrity are still developing, with optical methods and electrical testing showing promise but requiring further refinement for industrial deployment.
Cost modeling indicates that surface treatments must add no more than 5-8% to the overall cell cost to remain economically viable. This constraint drives research toward simplified coating architectures and more efficient deposition methods. Recent innovations in plasma-enhanced deposition and roll-to-roll compatible techniques show promising directions for achieving the necessary balance between performance, throughput, and cost-effectiveness required for commercial-scale implementation.
Manufacturing considerations must address the cost-effectiveness of coating materials and processes. While certain high-performance coatings like Al2O3 or TiO2 deposited via ALD provide superior protection, the associated equipment costs and slow deposition rates (typically 1-2 nm/hour) create substantial bottlenecks for industrial-scale implementation. Alternative approaches using solution-based methods offer better throughput but may suffer from coating uniformity issues.
Process integration presents another key challenge, as coating steps must be seamlessly incorporated into existing battery manufacturing lines. The ideal coating technology should operate continuously rather than in batch processes to align with high-volume electrode production. Current industry standards for lithium-ion battery manufacturing achieve throughput rates of 20-30 m/min for electrode coating; protective coating processes for hard carbon must approach similar efficiency metrics to be commercially viable.
Environmental and safety considerations also impact scalability. Many coating precursors, particularly those used in vapor deposition techniques, involve toxic or highly reactive chemicals that require specialized handling and waste management systems. Water-based coating alternatives, while more environmentally friendly, often necessitate additional drying steps that increase energy consumption and production time.
Quality control represents a significant manufacturing challenge, as coating thickness uniformity and defect detection must be maintained across large production volumes. In-line monitoring technologies capable of real-time assessment of coating integrity are still developing, with optical methods and electrical testing showing promise but requiring further refinement for industrial deployment.
Cost modeling indicates that surface treatments must add no more than 5-8% to the overall cell cost to remain economically viable. This constraint drives research toward simplified coating architectures and more efficient deposition methods. Recent innovations in plasma-enhanced deposition and roll-to-roll compatible techniques show promising directions for achieving the necessary balance between performance, throughput, and cost-effectiveness required for commercial-scale implementation.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!