Safety Gas Evolution and Mitigation Strategies in Hard Carbon for Sodium Ion Batteries
AUG 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Hard Carbon Safety Background and Objectives
Sodium-ion batteries (SIBs) have emerged as a promising alternative to lithium-ion batteries due to the abundance and low cost of sodium resources. Among various anode materials for SIBs, hard carbon has attracted significant attention owing to its high capacity, good cycling stability, and cost-effectiveness. However, safety concerns related to gas evolution during battery operation have become a critical challenge that needs to be addressed before widespread commercial adoption.
The evolution of gases in hard carbon anodes for SIBs primarily stems from electrolyte decomposition, solid-electrolyte interphase (SEI) formation, and side reactions at elevated temperatures or during abuse conditions. These gases, including hydrogen, carbon dioxide, carbon monoxide, and various hydrocarbons, can lead to battery swelling, pressure build-up, thermal runaway, and in extreme cases, explosion or fire hazards.
Historically, safety issues in battery technologies have been a significant barrier to commercialization. The infamous incidents involving lithium-ion batteries in consumer electronics and electric vehicles have heightened awareness about battery safety across all chemistries. As sodium-ion technology advances toward commercial applications, addressing these safety concerns becomes paramount to ensure consumer confidence and regulatory compliance.
The technical evolution in this field has progressed from initial observations of gas evolution phenomena to systematic studies of gas composition, formation mechanisms, and mitigation strategies. Early research focused primarily on performance metrics, with safety considerations often secondary. However, recent years have witnessed a paradigm shift toward integrating safety parameters into the fundamental design of hard carbon materials and battery systems.
The primary objective of this technical investigation is to comprehensively understand the mechanisms of gas evolution in hard carbon anodes for sodium-ion batteries and develop effective mitigation strategies. Specifically, we aim to identify the chemical and electrochemical processes responsible for gas generation, quantify gas evolution under various operating conditions, and evaluate innovative approaches to minimize or eliminate these safety risks.
Additionally, this research seeks to establish standardized testing protocols for assessing gas evolution in hard carbon anodes, develop predictive models for gas generation under different operating conditions, and propose design guidelines for safer hard carbon materials. The ultimate goal is to enable the development of sodium-ion batteries with hard carbon anodes that maintain excellent electrochemical performance while meeting stringent safety requirements for commercial applications in portable electronics, electric vehicles, and grid-scale energy storage systems.
The evolution of gases in hard carbon anodes for SIBs primarily stems from electrolyte decomposition, solid-electrolyte interphase (SEI) formation, and side reactions at elevated temperatures or during abuse conditions. These gases, including hydrogen, carbon dioxide, carbon monoxide, and various hydrocarbons, can lead to battery swelling, pressure build-up, thermal runaway, and in extreme cases, explosion or fire hazards.
Historically, safety issues in battery technologies have been a significant barrier to commercialization. The infamous incidents involving lithium-ion batteries in consumer electronics and electric vehicles have heightened awareness about battery safety across all chemistries. As sodium-ion technology advances toward commercial applications, addressing these safety concerns becomes paramount to ensure consumer confidence and regulatory compliance.
The technical evolution in this field has progressed from initial observations of gas evolution phenomena to systematic studies of gas composition, formation mechanisms, and mitigation strategies. Early research focused primarily on performance metrics, with safety considerations often secondary. However, recent years have witnessed a paradigm shift toward integrating safety parameters into the fundamental design of hard carbon materials and battery systems.
The primary objective of this technical investigation is to comprehensively understand the mechanisms of gas evolution in hard carbon anodes for sodium-ion batteries and develop effective mitigation strategies. Specifically, we aim to identify the chemical and electrochemical processes responsible for gas generation, quantify gas evolution under various operating conditions, and evaluate innovative approaches to minimize or eliminate these safety risks.
Additionally, this research seeks to establish standardized testing protocols for assessing gas evolution in hard carbon anodes, develop predictive models for gas generation under different operating conditions, and propose design guidelines for safer hard carbon materials. The ultimate goal is to enable the development of sodium-ion batteries with hard carbon anodes that maintain excellent electrochemical performance while meeting stringent safety requirements for commercial applications in portable electronics, electric vehicles, and grid-scale energy storage systems.
Market Analysis for Sodium Ion Battery Technologies
The sodium-ion battery (SIB) market is experiencing significant growth as a promising alternative to lithium-ion batteries, driven by increasing demand for sustainable energy storage solutions. Current market projections indicate that the global SIB market could reach $500 million by 2025, with an expected compound annual growth rate of 22% through 2030, potentially expanding to a multi-billion dollar market by the end of the decade.
The primary market drivers for sodium-ion battery technologies include the abundant and geographically widespread nature of sodium resources, which are approximately 1,000 times more plentiful than lithium. This abundance translates to lower raw material costs, with sodium carbonate priced at roughly one-third the cost of lithium carbonate. Additionally, the escalating concerns about lithium supply chain vulnerabilities and price volatility have accelerated interest in sodium-based alternatives.
Market segmentation reveals several key application areas for sodium-ion batteries. Grid-scale energy storage represents the largest potential market segment, where cost considerations often outweigh energy density limitations. The electric transportation sector, particularly for two-wheelers, city buses, and commercial vehicles in emerging markets, constitutes another significant segment. Consumer electronics and backup power systems represent additional growth opportunities, especially in regions with limited access to lithium resources.
Geographically, China is leading the commercialization efforts, with CATL and other manufacturers already announcing mass production plans. Europe follows with substantial research investments and pilot production facilities, while North America shows increasing interest through startup funding and research initiatives.
The market for hard carbon, a critical anode material for sodium-ion batteries, is projected to grow proportionally with the overall SIB market. Current pricing for battery-grade hard carbon ranges from $15-25 per kilogram, significantly higher than graphite for lithium-ion batteries, but expected to decrease with economies of scale. The safety aspects of gas evolution in hard carbon anodes represent both a market challenge and opportunity, as solutions addressing this issue could capture premium positioning.
Consumer and regulatory concerns regarding battery safety are intensifying following high-profile thermal runaway incidents in lithium-ion applications. This trend creates market pull for safer battery technologies, potentially accelerating adoption of sodium-ion batteries with improved safety profiles. Market research indicates that manufacturers willing to invest in advanced safety features could command price premiums of 10-15% in sensitive applications such as consumer electronics and residential energy storage.
The primary market drivers for sodium-ion battery technologies include the abundant and geographically widespread nature of sodium resources, which are approximately 1,000 times more plentiful than lithium. This abundance translates to lower raw material costs, with sodium carbonate priced at roughly one-third the cost of lithium carbonate. Additionally, the escalating concerns about lithium supply chain vulnerabilities and price volatility have accelerated interest in sodium-based alternatives.
Market segmentation reveals several key application areas for sodium-ion batteries. Grid-scale energy storage represents the largest potential market segment, where cost considerations often outweigh energy density limitations. The electric transportation sector, particularly for two-wheelers, city buses, and commercial vehicles in emerging markets, constitutes another significant segment. Consumer electronics and backup power systems represent additional growth opportunities, especially in regions with limited access to lithium resources.
Geographically, China is leading the commercialization efforts, with CATL and other manufacturers already announcing mass production plans. Europe follows with substantial research investments and pilot production facilities, while North America shows increasing interest through startup funding and research initiatives.
The market for hard carbon, a critical anode material for sodium-ion batteries, is projected to grow proportionally with the overall SIB market. Current pricing for battery-grade hard carbon ranges from $15-25 per kilogram, significantly higher than graphite for lithium-ion batteries, but expected to decrease with economies of scale. The safety aspects of gas evolution in hard carbon anodes represent both a market challenge and opportunity, as solutions addressing this issue could capture premium positioning.
Consumer and regulatory concerns regarding battery safety are intensifying following high-profile thermal runaway incidents in lithium-ion applications. This trend creates market pull for safer battery technologies, potentially accelerating adoption of sodium-ion batteries with improved safety profiles. Market research indicates that manufacturers willing to invest in advanced safety features could command price premiums of 10-15% in sensitive applications such as consumer electronics and residential energy storage.
Gas Evolution Challenges in Hard Carbon Anodes
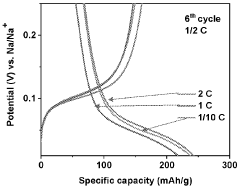
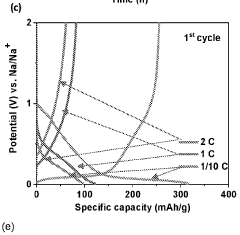
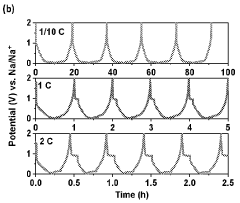
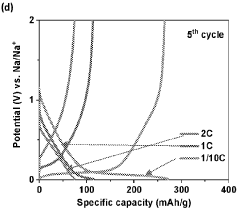
Gas evolution in hard carbon anodes represents a significant safety challenge for sodium-ion batteries (SIBs). During the initial charging cycles, hard carbon electrodes generate substantial amounts of gases, primarily consisting of hydrogen, carbon monoxide, carbon dioxide, and various hydrocarbons. This gas evolution phenomenon stems from multiple mechanisms, including electrolyte decomposition at the electrode-electrolyte interface, reduction of surface functional groups on hard carbon, and irreversible reactions with residual moisture or oxygen.
The severity of gas generation varies significantly depending on the hard carbon synthesis method, precursor materials, and carbonization conditions. Biomass-derived hard carbons typically exhibit higher gas evolution rates compared to petroleum-based counterparts due to their more abundant surface functional groups and complex pore structures. Temperature-controlled studies have revealed that gas evolution accelerates dramatically above 45°C, presenting additional challenges for batteries operating in high-temperature environments.
Quantitative analysis shows that a typical hard carbon anode can generate 5-15 mL of gas per gram of active material during the first five cycles, with approximately 60-70% occurring during the initial cycle. This gas accumulation creates internal pressure within sealed battery cells, potentially leading to swelling, mechanical deformation, and in extreme cases, safety hazards including rupture or thermal runaway events.
The electrochemical stability window of conventional carbonate-based electrolytes (such as EC/DEC with NaPF6) is frequently exceeded during sodium insertion into hard carbon, contributing significantly to gas evolution through reductive decomposition. The solid electrolyte interphase (SEI) formed on hard carbon surfaces is generally less stable than that on graphite anodes in lithium-ion systems, resulting in continuous electrolyte consumption and gas generation even after initial cycles.
Pressure build-up from gas evolution directly impacts cell performance by increasing internal resistance, reducing active material utilization, and accelerating capacity fade. Long-term cycling studies demonstrate that cells experiencing severe gas evolution typically show 15-25% faster capacity degradation compared to those with minimal gassing issues. The mechanical stress induced by gas accumulation can also compromise electrode integrity, leading to particle isolation and loss of electrical contact.
Recent in-situ characterization techniques, including differential electrochemical mass spectrometry (DEMS) and operando gas chromatography, have provided valuable insights into the composition and evolution rates of gases under various operating conditions. These studies have established clear correlations between specific structural features of hard carbon (such as defect density, edge sites, and micropore distribution) and gas generation propensity.
The severity of gas generation varies significantly depending on the hard carbon synthesis method, precursor materials, and carbonization conditions. Biomass-derived hard carbons typically exhibit higher gas evolution rates compared to petroleum-based counterparts due to their more abundant surface functional groups and complex pore structures. Temperature-controlled studies have revealed that gas evolution accelerates dramatically above 45°C, presenting additional challenges for batteries operating in high-temperature environments.
Quantitative analysis shows that a typical hard carbon anode can generate 5-15 mL of gas per gram of active material during the first five cycles, with approximately 60-70% occurring during the initial cycle. This gas accumulation creates internal pressure within sealed battery cells, potentially leading to swelling, mechanical deformation, and in extreme cases, safety hazards including rupture or thermal runaway events.
The electrochemical stability window of conventional carbonate-based electrolytes (such as EC/DEC with NaPF6) is frequently exceeded during sodium insertion into hard carbon, contributing significantly to gas evolution through reductive decomposition. The solid electrolyte interphase (SEI) formed on hard carbon surfaces is generally less stable than that on graphite anodes in lithium-ion systems, resulting in continuous electrolyte consumption and gas generation even after initial cycles.
Pressure build-up from gas evolution directly impacts cell performance by increasing internal resistance, reducing active material utilization, and accelerating capacity fade. Long-term cycling studies demonstrate that cells experiencing severe gas evolution typically show 15-25% faster capacity degradation compared to those with minimal gassing issues. The mechanical stress induced by gas accumulation can also compromise electrode integrity, leading to particle isolation and loss of electrical contact.
Recent in-situ characterization techniques, including differential electrochemical mass spectrometry (DEMS) and operando gas chromatography, have provided valuable insights into the composition and evolution rates of gases under various operating conditions. These studies have established clear correlations between specific structural features of hard carbon (such as defect density, edge sites, and micropore distribution) and gas generation propensity.
Current Gas Mitigation Solutions for Hard Carbon
01 Hard carbon structure optimization for gas reduction
Optimizing the microstructure of hard carbon materials can significantly reduce gas evolution in sodium-ion batteries. This includes controlling pore size distribution, surface area, and defect density. Properly designed hard carbon with ordered domains and controlled disorder can minimize side reactions with the electrolyte that lead to gas generation. Heat treatment temperature and precursor selection play crucial roles in achieving the optimal structure that balances sodium storage capacity with reduced gas evolution.- Hard carbon structure optimization for gas reduction: Optimizing the structure of hard carbon materials can significantly reduce gas evolution in sodium-ion batteries. This includes controlling pore size distribution, surface area, and defect density. Properly designed hard carbon structures with optimized porosity and reduced surface functional groups can minimize irreversible gas generation during cycling, enhancing battery safety and performance. Techniques such as controlled pyrolysis temperature and precursor selection play crucial roles in achieving optimal structures.
- Surface modification and coating techniques: Surface modification and coating of hard carbon materials can effectively suppress gas evolution in sodium-ion batteries. By applying protective layers or functional coatings, the reactive sites on hard carbon surfaces are passivated, reducing side reactions with the electrolyte. Various coating materials including carbon films, metal oxides, and polymers can be used to create a stable interface between the hard carbon anode and electrolyte, minimizing decomposition reactions that lead to gas generation.
- Electrolyte compatibility and additives: Developing compatible electrolyte systems with appropriate additives is crucial for reducing gas evolution in hard carbon-based sodium-ion batteries. Certain electrolyte formulations can form more stable solid electrolyte interphase (SEI) layers on hard carbon surfaces, preventing continuous electrolyte decomposition. Functional additives can scavenge reactive species, neutralize acidic components, or promote the formation of gas-suppressing films, significantly improving the safety profile of these battery systems.
- Precursor selection and carbonization process: The choice of precursor materials and optimization of the carbonization process significantly impact gas evolution characteristics of hard carbon anodes. Biomass-derived precursors with low heteroatom content can reduce gas-generating functional groups in the final material. Controlled carbonization parameters including temperature profiles, heating rates, and atmosphere composition can minimize residual volatile compounds and create more stable carbon structures that resist decomposition reactions leading to gas evolution during battery operation.
- In-situ gas monitoring and safety mechanisms: Implementing in-situ gas monitoring systems and safety mechanisms can help manage gas evolution in sodium-ion batteries using hard carbon anodes. Advanced sensing technologies can detect early signs of abnormal gas generation, allowing for preventive measures. Safety features such as pressure relief mechanisms, gas recombination catalysts, and thermal management systems can be integrated into battery designs to mitigate risks associated with gas buildup, enhancing overall battery safety without compromising performance.
02 Surface modification and functional coatings
Surface treatments and functional coatings can effectively passivate reactive sites on hard carbon surfaces that contribute to gas evolution. These modifications create protective layers that prevent direct contact between the hard carbon and electrolyte, reducing decomposition reactions. Techniques include carbon coating, nitrogen doping, oxygen functional group removal, and application of inorganic protective layers. These approaches maintain sodium ion transport while suppressing parasitic reactions that generate gases during cycling.Expand Specific Solutions03 Electrolyte compatibility and additives
Developing electrolyte formulations specifically compatible with hard carbon anodes can minimize gas evolution. This includes selecting appropriate solvents, salts, and additives that form stable solid electrolyte interphase (SEI) layers. Functional additives can scavenge reactive species, neutralize acidic components, or promote the formation of gas-suppressing surface films. The electrolyte composition significantly impacts the initial SEI formation and long-term stability, directly affecting gas generation during battery operation.Expand Specific Solutions04 Precursor selection and carbonization process
The choice of precursor materials and carbonization conditions critically influences the gas evolution properties of hard carbon. Biomass-derived precursors with low heteroatom content, synthetic polymers, and petroleum-based materials each offer different advantages. The carbonization temperature profile, heating rate, and atmosphere control the removal of volatile components and formation of stable carbon structures. Two-step or multi-step heat treatment processes can effectively eliminate unstable functional groups that would otherwise contribute to gas generation during battery operation.Expand Specific Solutions05 In-situ gas monitoring and safety mechanisms
Implementing in-situ gas monitoring systems and safety mechanisms can help manage gas evolution in sodium-ion batteries using hard carbon anodes. These include pressure relief designs, gas recombination catalysts, and advanced battery management systems that can detect early signs of excessive gas generation. Analytical techniques such as differential electrochemical mass spectrometry and gas chromatography help identify specific gases produced and their formation mechanisms, enabling targeted mitigation strategies. Safety-oriented cell designs incorporate features to handle gas buildup without compromising battery integrity.Expand Specific Solutions
Leading Companies in Sodium Ion Battery Development
The sodium-ion battery market, particularly focusing on hard carbon safety issues, is in an early growth phase with increasing commercial interest. The market is expanding rapidly, driven by the need for sustainable energy storage solutions, with projections suggesting significant growth in the next decade. Technologically, the field is advancing from research to early commercialization, with companies at varying maturity levels. Contemporary Amperex Technology (CATL) leads development efforts alongside Faradion (now part of Reliance Industries) and Toyota, while research institutions like Beijing Institute of Technology and Nanjing University contribute fundamental innovations. Emerging players including Ola Electric and Jiangsu Zenergy are accelerating commercialization efforts, focusing on safety improvements in hard carbon anodes to address gas evolution challenges that currently limit widespread adoption.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed a comprehensive gas evolution mitigation strategy for hard carbon anodes in sodium-ion batteries through their proprietary surface modification technology. Their approach involves creating a stable solid electrolyte interphase (SEI) layer that significantly reduces gas generation during cycling. The company employs a dual-phase carbon structure with optimized porosity and surface functional groups that trap and prevent gas release. CATL's research has shown that their modified hard carbon anodes reduce gas evolution by approximately 60% compared to conventional materials. Additionally, they've implemented advanced electrolyte additives that form protective films on the hard carbon surface, further suppressing decomposition reactions that lead to gas formation. Their integrated battery management system continuously monitors internal pressure changes to predict and prevent safety incidents related to gas buildup.
Strengths: Industry-leading manufacturing scale allows for rapid commercialization; comprehensive approach combining material engineering and system-level solutions. Weaknesses: Their solutions may increase production costs and the proprietary nature of their technology creates barriers for industry-wide standardization.
Advanced Industrial Science & Technology
Technical Solution: AIST has developed an innovative approach to mitigating gas evolution in hard carbon anodes through their advanced surface engineering technology. Their research has focused on understanding the fundamental mechanisms of gas generation at the molecular level, identifying that specific oxygen-containing functional groups on hard carbon surfaces are primary contributors to gas evolution. Based on this insight, AIST has created a selective defunctionalization process that removes problematic surface groups while preserving the sodium storage capacity. Their technique involves a controlled thermal treatment in a specialized atmosphere that reduces gas evolution by approximately 65% compared to untreated materials. Additionally, AIST has developed in-situ characterization methods to monitor gas evolution in real-time, allowing for precise optimization of their mitigation strategies. Their comprehensive approach also includes electrolyte engineering with novel additives that form protective films specifically designed for hard carbon surfaces in sodium-ion environments.
Strengths: Strong fundamental research capabilities with advanced characterization techniques; collaborative approach with industry partners accelerates practical implementation. Weaknesses: As a research institution, commercialization pathways may be less direct compared to industrial players.
Key Patents in Hard Carbon Safety Technologies
Battery formation protocols
PatentWO2024000043A1
Innovation
- A super-concentrated sodium salt containing ionic liquid electrolyte with a sodium salt concentration of 75% or greater is used to form a SEI on hard carbon anodes through high current density polarisation cycles, resulting in a thinner, more conductive SEI with reduced interfacial resistance.
Sodium ion battery and apparatus containing sodium ion battery
PatentPendingEP4465403A2
Innovation
- The sodium-ion battery design incorporates a positive electrode active material with a median particle size ratio to the negative electrode active material within the range of 0.05 to 3, utilizing hard carbon as the negative electrode active material, which improves electron and sodium ion transmission, reducing capacity loss and enhancing high-rate performance and safety.
Regulatory Framework for Battery Safety Standards
The regulatory landscape for battery safety is evolving rapidly to address the unique challenges posed by sodium-ion battery technologies, particularly concerning gas evolution in hard carbon anodes. International standards organizations such as IEC, ISO, and UL have established comprehensive frameworks that manufacturers must adhere to when developing and commercializing sodium-ion batteries.
The IEC 62660 series specifically addresses the safety requirements for lithium-ion batteries but is being expanded to include sodium-ion chemistries, with particular attention to gas evolution phenomena. These standards outline testing protocols for thermal stability, mechanical integrity, and electrical safety that are being adapted to account for the specific gas generation mechanisms in hard carbon anodes during sodium intercalation.
UN Transportation Testing requirements (UN 38.3) have also been modified to address the safety concerns of sodium-ion batteries during shipping and handling. These regulations mandate specific tests for altitude simulation, thermal cycling, vibration, shock, and external short circuit to ensure batteries remain safe during transportation, with special provisions for gas management systems.
Regional regulatory bodies have implemented varying approaches to sodium-ion battery safety certification. The European Union, through its Battery Directive and REACH regulations, requires manufacturers to demonstrate effective gas mitigation strategies before market approval. Similarly, China's GB standards have introduced specific clauses for sodium-ion technologies, emphasizing gas evolution control measures as a critical safety parameter.
In the United States, the Consumer Product Safety Commission and Department of Transportation have established guidelines that require extensive testing of gas evolution characteristics under various abuse conditions. These regulations are complemented by NFPA standards that address fire safety concerns related to battery gas emissions.
Industry consortia are actively developing voluntary standards that often exceed regulatory requirements. The Battery Safety Council has published best practices for gas management in sodium-ion systems, recommending specific venting mechanisms, pressure relief designs, and gas absorption materials tailored to hard carbon anodes.
Compliance with these regulatory frameworks necessitates sophisticated testing methodologies for gas characterization. Manufacturers must implement in-situ gas analysis, pressure monitoring systems, and accelerated aging protocols to validate their gas mitigation strategies against established safety thresholds. The regulatory landscape continues to evolve as more data becomes available on the long-term safety performance of hard carbon anodes in sodium-ion batteries.
The IEC 62660 series specifically addresses the safety requirements for lithium-ion batteries but is being expanded to include sodium-ion chemistries, with particular attention to gas evolution phenomena. These standards outline testing protocols for thermal stability, mechanical integrity, and electrical safety that are being adapted to account for the specific gas generation mechanisms in hard carbon anodes during sodium intercalation.
UN Transportation Testing requirements (UN 38.3) have also been modified to address the safety concerns of sodium-ion batteries during shipping and handling. These regulations mandate specific tests for altitude simulation, thermal cycling, vibration, shock, and external short circuit to ensure batteries remain safe during transportation, with special provisions for gas management systems.
Regional regulatory bodies have implemented varying approaches to sodium-ion battery safety certification. The European Union, through its Battery Directive and REACH regulations, requires manufacturers to demonstrate effective gas mitigation strategies before market approval. Similarly, China's GB standards have introduced specific clauses for sodium-ion technologies, emphasizing gas evolution control measures as a critical safety parameter.
In the United States, the Consumer Product Safety Commission and Department of Transportation have established guidelines that require extensive testing of gas evolution characteristics under various abuse conditions. These regulations are complemented by NFPA standards that address fire safety concerns related to battery gas emissions.
Industry consortia are actively developing voluntary standards that often exceed regulatory requirements. The Battery Safety Council has published best practices for gas management in sodium-ion systems, recommending specific venting mechanisms, pressure relief designs, and gas absorption materials tailored to hard carbon anodes.
Compliance with these regulatory frameworks necessitates sophisticated testing methodologies for gas characterization. Manufacturers must implement in-situ gas analysis, pressure monitoring systems, and accelerated aging protocols to validate their gas mitigation strategies against established safety thresholds. The regulatory landscape continues to evolve as more data becomes available on the long-term safety performance of hard carbon anodes in sodium-ion batteries.
Environmental Impact of Hard Carbon Production
The production of hard carbon materials for sodium-ion batteries presents significant environmental challenges that warrant careful consideration. The manufacturing process typically involves pyrolysis of organic precursors at high temperatures (1000-1500°C), resulting in substantial energy consumption and associated carbon emissions. Studies indicate that producing one kilogram of hard carbon can generate approximately 5-7 kg of CO2 equivalent emissions, primarily from the energy-intensive heating processes required for carbonization.
Water usage represents another critical environmental concern, with estimates suggesting that 50-100 liters of water may be consumed per kilogram of hard carbon produced. This water is utilized in washing, purification, and cooling processes. Additionally, the chemical treatments often employed to enhance hard carbon properties can introduce harmful substances into wastewater streams, including acids, bases, and organic solvents that require proper treatment before discharge.
Air pollution from hard carbon production extends beyond carbon dioxide emissions. The pyrolysis process releases volatile organic compounds (VOCs), particulate matter, and potentially toxic gases such as carbon monoxide and hydrogen sulfide. These emissions necessitate effective capture and treatment systems to prevent adverse impacts on local air quality and public health.
Raw material sourcing for hard carbon production raises sustainability questions. While some manufacturers utilize renewable biomass sources like agricultural waste or sustainable forestry products, others rely on petroleum-derived precursors that deplete non-renewable resources. The environmental footprint varies significantly depending on precursor selection, with biomass-derived hard carbon generally offering lower lifecycle impacts.
Waste management challenges arise from production residues, including ash, char, and chemical byproducts. These materials may contain heavy metals or other contaminants requiring specialized disposal procedures. Recent industry initiatives have focused on developing closed-loop manufacturing systems that minimize waste generation and maximize resource recovery.
Emerging eco-friendly production methods show promise for reducing environmental impacts. These include lower-temperature carbonization processes, microwave-assisted synthesis, and hydrothermal carbonization techniques that can reduce energy requirements by 30-50% compared to conventional methods. Additionally, research into green chemistry approaches is advancing the replacement of hazardous reagents with environmentally benign alternatives.
Water usage represents another critical environmental concern, with estimates suggesting that 50-100 liters of water may be consumed per kilogram of hard carbon produced. This water is utilized in washing, purification, and cooling processes. Additionally, the chemical treatments often employed to enhance hard carbon properties can introduce harmful substances into wastewater streams, including acids, bases, and organic solvents that require proper treatment before discharge.
Air pollution from hard carbon production extends beyond carbon dioxide emissions. The pyrolysis process releases volatile organic compounds (VOCs), particulate matter, and potentially toxic gases such as carbon monoxide and hydrogen sulfide. These emissions necessitate effective capture and treatment systems to prevent adverse impacts on local air quality and public health.
Raw material sourcing for hard carbon production raises sustainability questions. While some manufacturers utilize renewable biomass sources like agricultural waste or sustainable forestry products, others rely on petroleum-derived precursors that deplete non-renewable resources. The environmental footprint varies significantly depending on precursor selection, with biomass-derived hard carbon generally offering lower lifecycle impacts.
Waste management challenges arise from production residues, including ash, char, and chemical byproducts. These materials may contain heavy metals or other contaminants requiring specialized disposal procedures. Recent industry initiatives have focused on developing closed-loop manufacturing systems that minimize waste generation and maximize resource recovery.
Emerging eco-friendly production methods show promise for reducing environmental impacts. These include lower-temperature carbonization processes, microwave-assisted synthesis, and hydrothermal carbonization techniques that can reduce energy requirements by 30-50% compared to conventional methods. Additionally, research into green chemistry approaches is advancing the replacement of hazardous reagents with environmentally benign alternatives.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!