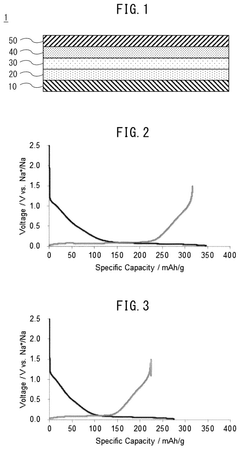
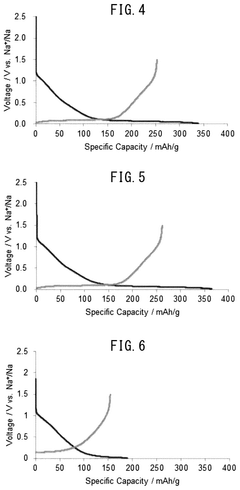
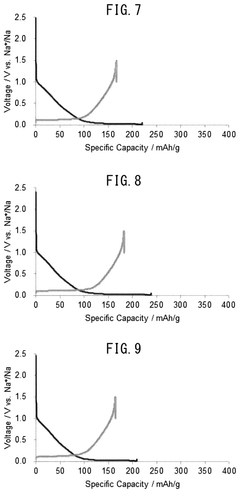
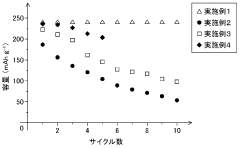
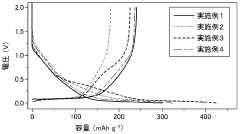
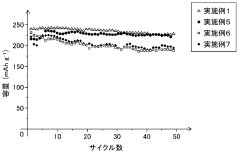
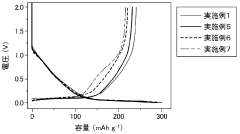
Fast Charging Protocols and Trade Offs in Hard Carbon for Sodium Ion Batteries
AUG 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Fast Charging Protocols Evolution and Objectives
Fast charging technology for sodium-ion batteries (SIBs) has evolved significantly over the past decade, driven by the increasing demand for rapid energy storage solutions. The initial charging protocols for SIBs were largely adapted from lithium-ion battery technologies, with constant current-constant voltage (CC-CV) being the predominant method. However, these early approaches failed to address the unique electrochemical characteristics of sodium ions and hard carbon anodes, resulting in suboptimal charging performance and accelerated degradation.
By 2015, researchers began developing sodium-specific charging protocols that accounted for the larger ionic radius of sodium (102 pm) compared to lithium (76 pm), which affects diffusion kinetics within hard carbon structures. This period saw the emergence of multi-stage charging protocols that carefully managed current densities to prevent sodium plating and structural damage to hard carbon anodes during fast charging operations.
The period from 2018 to 2020 marked a significant advancement with the introduction of pulse charging techniques specifically optimized for hard carbon materials in SIBs. These protocols alternated between high-current pulses and relaxation periods, allowing for better ion distribution within the carbon structure and heat dissipation, which proved crucial for maintaining structural integrity during rapid charging cycles.
Recent developments have focused on adaptive charging protocols that utilize machine learning algorithms to dynamically adjust charging parameters based on real-time battery state and environmental conditions. These smart protocols can predict optimal charging strategies for hard carbon anodes by analyzing voltage response patterns, temperature fluctuations, and impedance measurements, thereby maximizing charging speed while minimizing degradation effects.
The primary objectives of fast charging protocol development for hard carbon-based SIBs include reducing charging time to under 15 minutes for 80% capacity while maintaining cycle life above 1,000 cycles. Additionally, researchers aim to mitigate the trade-offs between charging speed and capacity retention, which has been a persistent challenge due to the complex pore structure of hard carbon materials and their interaction with sodium ions during rapid intercalation.
Another critical objective is to develop universal charging standards that can be applied across different hard carbon formulations and cell designs, facilitating broader commercial adoption of SIB technology. This standardization effort requires comprehensive understanding of how various hard carbon microstructures respond to different charging regimes, and how these responses change throughout battery lifetime.
Future protocol development aims to integrate thermal management considerations directly into charging algorithms, as temperature control has proven essential for optimizing the performance-degradation balance in fast-charging scenarios for hard carbon anodes in sodium-ion batteries.
By 2015, researchers began developing sodium-specific charging protocols that accounted for the larger ionic radius of sodium (102 pm) compared to lithium (76 pm), which affects diffusion kinetics within hard carbon structures. This period saw the emergence of multi-stage charging protocols that carefully managed current densities to prevent sodium plating and structural damage to hard carbon anodes during fast charging operations.
The period from 2018 to 2020 marked a significant advancement with the introduction of pulse charging techniques specifically optimized for hard carbon materials in SIBs. These protocols alternated between high-current pulses and relaxation periods, allowing for better ion distribution within the carbon structure and heat dissipation, which proved crucial for maintaining structural integrity during rapid charging cycles.
Recent developments have focused on adaptive charging protocols that utilize machine learning algorithms to dynamically adjust charging parameters based on real-time battery state and environmental conditions. These smart protocols can predict optimal charging strategies for hard carbon anodes by analyzing voltage response patterns, temperature fluctuations, and impedance measurements, thereby maximizing charging speed while minimizing degradation effects.
The primary objectives of fast charging protocol development for hard carbon-based SIBs include reducing charging time to under 15 minutes for 80% capacity while maintaining cycle life above 1,000 cycles. Additionally, researchers aim to mitigate the trade-offs between charging speed and capacity retention, which has been a persistent challenge due to the complex pore structure of hard carbon materials and their interaction with sodium ions during rapid intercalation.
Another critical objective is to develop universal charging standards that can be applied across different hard carbon formulations and cell designs, facilitating broader commercial adoption of SIB technology. This standardization effort requires comprehensive understanding of how various hard carbon microstructures respond to different charging regimes, and how these responses change throughout battery lifetime.
Future protocol development aims to integrate thermal management considerations directly into charging algorithms, as temperature control has proven essential for optimizing the performance-degradation balance in fast-charging scenarios for hard carbon anodes in sodium-ion batteries.
Market Demand Analysis for Rapid-Charging Sodium Ion Batteries
The global energy storage market is witnessing a significant shift towards sodium-ion batteries (SIBs) as a viable alternative to lithium-ion batteries (LIBs), with rapid charging capabilities emerging as a critical market differentiator. Market research indicates that the global sodium-ion battery market is projected to grow at a CAGR of 18-20% from 2023 to 2030, driven primarily by increasing demand for cost-effective energy storage solutions with fast charging capabilities.
The rapid charging segment within the SIB market is experiencing particularly strong growth due to several converging market factors. Electric vehicle manufacturers are actively seeking battery technologies that can reduce charging times to under 15 minutes, comparable to traditional refueling experiences, to overcome consumer adoption barriers related to range anxiety and charging convenience.
Grid-scale energy storage represents another significant market opportunity for fast-charging SIBs. Utility companies require storage solutions that can rapidly absorb excess renewable energy during production peaks and quickly discharge during demand surges. The ability of hard carbon-based SIBs to handle rapid charging protocols makes them increasingly attractive for these applications, with market demand expected to reach 30 GWh by 2025 for grid applications alone.
Consumer electronics manufacturers are also driving demand for rapid-charging sodium-ion batteries. The portable electronics sector values batteries that can reach 80% capacity in under 30 minutes while maintaining cycle life and safety. This segment is expected to adopt SIB technology as performance improvements continue, with market penetration beginning in power tools and expanding to mobile devices.
Regional analysis reveals that Asia-Pacific currently dominates the market demand for rapid-charging SIBs, with China leading in both production capacity and consumption. European markets show the fastest growth rate, driven by stringent sustainability regulations and ambitious renewable energy targets that necessitate advanced energy storage solutions.
Price sensitivity analysis indicates that consumers and industrial buyers are willing to pay a 15-20% premium for batteries with verified fast-charging capabilities compared to standard charging options, provided cycle life is not significantly compromised. This premium pricing potential creates a compelling business case for continued R&D investment in hard carbon optimization for sodium-ion batteries.
Market forecasts suggest that by 2028, rapid-charging sodium-ion batteries could capture up to 12% of the total battery market in specific applications where the combination of cost-effectiveness and charging speed outweighs the slightly lower energy density compared to premium lithium-ion solutions.
The rapid charging segment within the SIB market is experiencing particularly strong growth due to several converging market factors. Electric vehicle manufacturers are actively seeking battery technologies that can reduce charging times to under 15 minutes, comparable to traditional refueling experiences, to overcome consumer adoption barriers related to range anxiety and charging convenience.
Grid-scale energy storage represents another significant market opportunity for fast-charging SIBs. Utility companies require storage solutions that can rapidly absorb excess renewable energy during production peaks and quickly discharge during demand surges. The ability of hard carbon-based SIBs to handle rapid charging protocols makes them increasingly attractive for these applications, with market demand expected to reach 30 GWh by 2025 for grid applications alone.
Consumer electronics manufacturers are also driving demand for rapid-charging sodium-ion batteries. The portable electronics sector values batteries that can reach 80% capacity in under 30 minutes while maintaining cycle life and safety. This segment is expected to adopt SIB technology as performance improvements continue, with market penetration beginning in power tools and expanding to mobile devices.
Regional analysis reveals that Asia-Pacific currently dominates the market demand for rapid-charging SIBs, with China leading in both production capacity and consumption. European markets show the fastest growth rate, driven by stringent sustainability regulations and ambitious renewable energy targets that necessitate advanced energy storage solutions.
Price sensitivity analysis indicates that consumers and industrial buyers are willing to pay a 15-20% premium for batteries with verified fast-charging capabilities compared to standard charging options, provided cycle life is not significantly compromised. This premium pricing potential creates a compelling business case for continued R&D investment in hard carbon optimization for sodium-ion batteries.
Market forecasts suggest that by 2028, rapid-charging sodium-ion batteries could capture up to 12% of the total battery market in specific applications where the combination of cost-effectiveness and charging speed outweighs the slightly lower energy density compared to premium lithium-ion solutions.
Technical Challenges in Hard Carbon Fast Charging
Hard carbon materials face significant technical challenges when subjected to fast charging protocols in sodium-ion batteries (SIBs). The primary obstacle lies in the complex microstructure of hard carbon, characterized by a combination of graphitic domains and disordered regions. This heterogeneous structure creates varied sodium ion diffusion pathways, leading to inconsistent charging behavior across the material.
The sodium ion insertion mechanism in hard carbon occurs through a two-stage process: first, sodium ions intercalate between graphene-like layers, and subsequently occupy nanopores. During fast charging, this dual-mechanism creates bottlenecks as the rates of these processes differ substantially. The intercalation stage typically proceeds more rapidly than the nanopore filling stage, creating an imbalance that limits overall charging speed.
Electrolyte penetration presents another significant challenge. The tortuous pore network in hard carbon restricts electrolyte access to internal active sites during rapid charging. This limitation becomes particularly pronounced in larger hard carbon particles, where the diffusion distance for both sodium ions and electrolytes increases substantially, creating concentration gradients that reduce charging efficiency.
The solid-electrolyte interphase (SEI) formation dynamics also complicate fast charging protocols. Under high current densities, the SEI layer forms unevenly and often incompletely, leading to continuous electrolyte decomposition and sodium consumption. This not only reduces coulombic efficiency but also accelerates capacity fade during repeated fast charging cycles.
Thermal management emerges as a critical concern during fast charging. The increased current density generates substantial heat through both resistive heating and exothermic electrode reactions. Hard carbon's relatively low thermal conductivity exacerbates this issue, creating localized hotspots that accelerate side reactions and potentially trigger thermal runaway under extreme conditions.
Mechanical stability represents another significant hurdle. The rapid insertion of sodium ions during fast charging induces substantial volume changes in hard carbon structures. These volume fluctuations generate mechanical stress that can lead to particle cracking, electrode delamination, and loss of electrical contact between active material particles and current collectors.
Electronic conductivity limitations further constrain fast charging capabilities. Hard carbon typically exhibits lower electronic conductivity compared to graphite, creating resistance to electron flow during high-rate operation. This resistance manifests as increased polarization and voltage hysteresis, directly limiting the practical fast charging rates achievable with hard carbon electrodes.
The sodium ion insertion mechanism in hard carbon occurs through a two-stage process: first, sodium ions intercalate between graphene-like layers, and subsequently occupy nanopores. During fast charging, this dual-mechanism creates bottlenecks as the rates of these processes differ substantially. The intercalation stage typically proceeds more rapidly than the nanopore filling stage, creating an imbalance that limits overall charging speed.
Electrolyte penetration presents another significant challenge. The tortuous pore network in hard carbon restricts electrolyte access to internal active sites during rapid charging. This limitation becomes particularly pronounced in larger hard carbon particles, where the diffusion distance for both sodium ions and electrolytes increases substantially, creating concentration gradients that reduce charging efficiency.
The solid-electrolyte interphase (SEI) formation dynamics also complicate fast charging protocols. Under high current densities, the SEI layer forms unevenly and often incompletely, leading to continuous electrolyte decomposition and sodium consumption. This not only reduces coulombic efficiency but also accelerates capacity fade during repeated fast charging cycles.
Thermal management emerges as a critical concern during fast charging. The increased current density generates substantial heat through both resistive heating and exothermic electrode reactions. Hard carbon's relatively low thermal conductivity exacerbates this issue, creating localized hotspots that accelerate side reactions and potentially trigger thermal runaway under extreme conditions.
Mechanical stability represents another significant hurdle. The rapid insertion of sodium ions during fast charging induces substantial volume changes in hard carbon structures. These volume fluctuations generate mechanical stress that can lead to particle cracking, electrode delamination, and loss of electrical contact between active material particles and current collectors.
Electronic conductivity limitations further constrain fast charging capabilities. Hard carbon typically exhibits lower electronic conductivity compared to graphite, creating resistance to electron flow during high-rate operation. This resistance manifests as increased polarization and voltage hysteresis, directly limiting the practical fast charging rates achievable with hard carbon electrodes.
Current Fast Charging Solutions for Hard Carbon Anodes
01 Hard carbon material composition and structure for fast charging
Hard carbon materials with specific compositions and structures can enhance fast charging capabilities in sodium-ion batteries. These materials typically feature optimized pore structures, particle sizes, and carbon arrangements that facilitate rapid sodium ion insertion and extraction. Modifications such as doping with heteroatoms, controlling defect density, and tailoring the degree of graphitization can significantly improve the rate performance of hard carbon anodes.- Hard carbon material composition and preparation methods: Hard carbon materials with specific compositions and preparation methods can enhance fast charging capabilities in sodium-ion batteries. These materials often involve carbon precursors derived from biomass or synthetic polymers that undergo carbonization processes at controlled temperatures. The resulting hard carbon structures feature optimized pore distributions, surface areas, and d-spacing characteristics that facilitate rapid sodium ion insertion and extraction during charging cycles.
- Surface modification and doping strategies: Surface modifications and heteroatom doping of hard carbon materials can significantly improve fast charging performance in sodium-ion batteries. Techniques include nitrogen, phosphorus, or sulfur doping, surface functionalization, and creation of defect sites. These modifications enhance electronic conductivity, create additional sodium storage sites, and optimize the solid-electrolyte interphase formation, collectively enabling faster ion transport and improved rate capability during rapid charging protocols.
- Composite structures with conductive additives: Composite structures combining hard carbon with conductive additives create synergistic effects that enhance fast charging capabilities. These composites often incorporate materials such as graphene, carbon nanotubes, or conductive polymers that form interconnected networks within the hard carbon matrix. The resulting hierarchical structures provide efficient electron transport pathways and shortened ion diffusion distances, enabling higher current densities during fast charging while maintaining structural stability.
- Optimized electrolyte systems for fast charging: Specialized electrolyte formulations designed specifically for hard carbon electrodes can enable faster charging protocols in sodium-ion batteries. These electrolytes often feature optimized salt concentrations, solvent mixtures, and additives that enhance ionic conductivity and sodium ion desolvation kinetics at the electrode interface. Some formulations also incorporate flame-retardant components or stabilizing additives that maintain performance and safety during high-current charging operations.
- Advanced charging protocols and battery management systems: Specialized charging protocols and battery management systems tailored for hard carbon-based sodium-ion batteries can maximize fast charging capabilities while preserving battery life. These approaches often employ multi-stage charging algorithms that adjust current rates based on state-of-charge, temperature monitoring, and impedance feedback. Some systems incorporate pulse charging techniques or variable current profiles that mitigate dendrite formation and heat generation during rapid charging, optimizing the balance between charging speed and battery longevity.
02 Surface modification and coating techniques
Surface treatments and coating strategies can improve the fast charging performance of hard carbon anodes in sodium-ion batteries. These include carbon coating, metal oxide deposition, polymer modification, and formation of protective layers that enhance conductivity and stability during rapid charging. Such modifications reduce interfacial resistance, prevent unwanted side reactions, and maintain structural integrity during high-rate cycling.Expand Specific Solutions03 Precursor selection and carbonization processes
The choice of precursor materials and carbonization methods significantly impacts the fast-charging capability of hard carbon anodes. Biomass-derived precursors, synthetic polymers, and various carbon-rich compounds can be processed through controlled pyrolysis, hydrothermal carbonization, or template-assisted methods. Optimizing temperature profiles, heating rates, and activation procedures during carbonization creates hard carbon structures with enhanced sodium storage and transport properties suitable for rapid charging.Expand Specific Solutions04 Composite and hybrid electrode designs
Composite electrodes combining hard carbon with other materials can significantly enhance fast charging performance. These designs incorporate conductive additives, secondary active materials, or structural reinforcements to improve electron transport, mechanical stability, and ion diffusion pathways. Hierarchical structures, 3D architectures, and strategic material combinations create synergistic effects that enable higher charging rates while maintaining capacity and cycle life.Expand Specific Solutions05 Electrolyte optimization and charging protocol design
Specialized electrolyte formulations and charging protocols can maximize the fast-charging capabilities of hard carbon anodes. Advanced electrolytes with optimized salt concentrations, solvent mixtures, and additives improve sodium ion transport and solid-electrolyte interphase properties. Pulse charging techniques, multi-stage protocols, and temperature-controlled charging strategies can be implemented to achieve optimal charging speeds while minimizing degradation mechanisms and extending battery lifetime.Expand Specific Solutions
Key Industry Players in Na-ion Battery Development
The sodium-ion battery fast charging landscape is evolving rapidly, currently in the early growth phase with market projections showing significant expansion potential. While the technology remains less mature than lithium-ion counterparts, hard carbon optimization for fast charging represents a critical development area. Key players like BYD, StoreDot, and SVOLT are advancing commercial solutions, while research institutions including Chinese Academy of Sciences and University of Electronic Science & Technology of China are developing fundamental innovations. Phillips 66 and Toyota are exploring industrial applications, with Faradion (acquired by Reliance) pioneering sodium-ion chemistry commercialization. The competitive landscape features established battery manufacturers diversifying their portfolios alongside specialized startups focusing exclusively on sodium-ion technology optimization.
Faradion Ltd.
Technical Solution: Faradion has pioneered advanced hard carbon anode materials specifically engineered for sodium-ion batteries with fast charging capabilities. Their proprietary technology involves a controlled pyrolysis process of organic precursors to create optimized pore structures in hard carbon, enabling rapid Na+ ion intercalation. Faradion's fast charging protocol utilizes a multi-stage charging approach with variable current densities (up to 3C) that adapts based on the state of charge, significantly reducing charging times while minimizing structural degradation. Their hard carbon materials feature tailored surface functional groups and controlled microstructure that facilitate faster Na+ diffusion at the solid-electrolyte interface. The company has demonstrated charging capabilities reaching 80% capacity in under 20 minutes while maintaining cycle stability through precise thermal management systems that prevent localized heating during rapid charging.
Strengths: Superior Na+ diffusion kinetics through engineered pore structures; excellent cycle stability even at high charging rates; cost-effective manufacturing process using sustainable precursors. Weaknesses: Slightly lower energy density compared to some competitors; temperature sensitivity requiring sophisticated thermal management systems.
BYD Co., Ltd.
Technical Solution: BYD has developed an integrated fast charging solution for sodium-ion batteries utilizing advanced hard carbon materials derived from sustainable biomass sources. Their technology employs a hierarchical porous structure in hard carbon anodes with optimized pore size distribution (predominantly mesopores of 2-5nm) that facilitates rapid Na+ transport while maintaining high capacity. BYD's fast charging protocol implements a proprietary pulse charging technique that alternates between high current pulses and rest periods, allowing for thermal relaxation and preventing localized overheating. Their hard carbon materials undergo a specialized activation process that creates additional active sites for Na+ storage while maintaining structural stability during rapid charge/discharge cycles. The company has integrated their fast-charging sodium-ion technology with thermal management systems that utilize phase-change materials to absorb excess heat generated during rapid charging, enabling consistent performance across varying environmental conditions.
Strengths: Vertically integrated manufacturing capabilities from raw materials to battery packs; excellent thermal management during fast charging; cost-effective production using renewable biomass precursors. Weaknesses: Slightly lower energy density compared to lithium-ion alternatives; limited performance data in extreme temperature conditions.
Critical Patents in Na-ion Fast Charging Technologies
Negative electrode mixture for sodium ion battery, and sodium ion battery and method for manufacturing thereof
PatentPendingUS20250158064A1
Innovation
- A negative electrode mixture for sodium ion batteries is developed, comprising hard carbon as the negative electrode active material and zirconium oxide, with a mass ratio of zirconium oxide to the total mass of hard carbon and zirconium oxide ranging from 1% to 20%. This mixture is used to form a negative electrode active material layer in the battery.
Sodium ion secondary battery
PatentWO2010109889A1
Innovation
- The use of hard carbon as the negative electrode active material in conjunction with a non-aqueous solvent comprising substantially saturated cyclic carbonate or a mixed solvent of saturated cyclic and chain carbonates, such as propylene carbonate and diethyl carbonate, enhances the battery's charge/discharge efficiency and allows operation at room temperature while minimizing performance degradation.
Safety and Thermal Management Considerations
Safety and thermal management are critical considerations in the development of fast charging protocols for hard carbon-based sodium-ion batteries (SIBs). The rapid charging process inherently generates more heat than standard charging rates, creating potential safety hazards that must be carefully managed. During fast charging, the increased current density can lead to accelerated temperature rise within the cell, particularly at the electrode-electrolyte interface where most electrochemical reactions occur.
Hard carbon materials present specific thermal challenges during fast charging due to their unique microstructure and sodium storage mechanisms. The turbostratic structure of hard carbon, while beneficial for sodium ion storage, can create localized hotspots during rapid charging as sodium ions navigate through the disordered carbon layers. These temperature gradients can trigger thermal runaway if not properly managed, leading to potential cell failure or safety incidents.
Electrolyte decomposition represents another significant concern during fast charging of hard carbon electrodes. The elevated temperatures can accelerate side reactions at the electrode-electrolyte interface, forming a thicker and more resistive solid electrolyte interphase (SEI) layer. This not only reduces battery efficiency but also generates additional heat, creating a potentially dangerous feedback loop that must be interrupted through effective thermal management systems.
Advanced cooling strategies have emerged as essential components of fast charging protocols for hard carbon SIBs. These include active liquid cooling systems that can rapidly dissipate heat from the cell surface, phase change materials that absorb excess thermal energy, and intelligent thermal management algorithms that adjust charging parameters based on real-time temperature monitoring. The integration of these cooling technologies with battery management systems enables dynamic control of charging rates to prevent dangerous temperature excursions.
Safety mechanisms such as thermal fuses, pressure relief valves, and current interrupt devices provide additional protection layers for fast-charging hard carbon batteries. These components are designed to activate under abnormal conditions, preventing catastrophic failure by isolating the battery or releasing pressure before dangerous thresholds are reached. The placement and sensitivity of these safety features must be carefully engineered to respond appropriately to the thermal profiles generated during fast charging.
Cell design optimization plays a crucial role in thermal management during fast charging. Factors such as electrode thickness, tab placement, and current collector design significantly impact heat distribution and dissipation pathways. Thinner electrodes generally allow for better heat transfer but reduce energy density, representing one of many trade-offs engineers must navigate when designing hard carbon cells for fast charging applications.
Hard carbon materials present specific thermal challenges during fast charging due to their unique microstructure and sodium storage mechanisms. The turbostratic structure of hard carbon, while beneficial for sodium ion storage, can create localized hotspots during rapid charging as sodium ions navigate through the disordered carbon layers. These temperature gradients can trigger thermal runaway if not properly managed, leading to potential cell failure or safety incidents.
Electrolyte decomposition represents another significant concern during fast charging of hard carbon electrodes. The elevated temperatures can accelerate side reactions at the electrode-electrolyte interface, forming a thicker and more resistive solid electrolyte interphase (SEI) layer. This not only reduces battery efficiency but also generates additional heat, creating a potentially dangerous feedback loop that must be interrupted through effective thermal management systems.
Advanced cooling strategies have emerged as essential components of fast charging protocols for hard carbon SIBs. These include active liquid cooling systems that can rapidly dissipate heat from the cell surface, phase change materials that absorb excess thermal energy, and intelligent thermal management algorithms that adjust charging parameters based on real-time temperature monitoring. The integration of these cooling technologies with battery management systems enables dynamic control of charging rates to prevent dangerous temperature excursions.
Safety mechanisms such as thermal fuses, pressure relief valves, and current interrupt devices provide additional protection layers for fast-charging hard carbon batteries. These components are designed to activate under abnormal conditions, preventing catastrophic failure by isolating the battery or releasing pressure before dangerous thresholds are reached. The placement and sensitivity of these safety features must be carefully engineered to respond appropriately to the thermal profiles generated during fast charging.
Cell design optimization plays a crucial role in thermal management during fast charging. Factors such as electrode thickness, tab placement, and current collector design significantly impact heat distribution and dissipation pathways. Thinner electrodes generally allow for better heat transfer but reduce energy density, representing one of many trade-offs engineers must navigate when designing hard carbon cells for fast charging applications.
Sustainability and Resource Availability Assessment
The sustainability profile of sodium-ion batteries (SIBs) represents a significant advantage over lithium-ion alternatives, particularly when considering hard carbon anodes in fast-charging applications. Sodium resources are abundantly available in the earth's crust (2.6%) and oceans (1.1%), making them approximately 1,000 times more plentiful than lithium. This abundance translates directly to lower extraction costs and reduced geopolitical supply risks, positioning SIBs as a more sustainable energy storage solution for large-scale applications.
The extraction of sodium compounds, primarily from seawater and mineral deposits, requires substantially less water consumption compared to lithium extraction from brine pools. This reduced water footprint is particularly significant in water-stressed regions where lithium mining operations have faced increasing environmental scrutiny. Additionally, the carbon footprint associated with sodium extraction and processing is estimated to be 25-30% lower than comparable lithium supply chains.
Hard carbon materials used in SIB anodes can be derived from renewable biomass sources such as agricultural waste, cellulose, and lignin. This circular economy approach provides dual environmental benefits: waste valorization and reduced dependency on mineral extraction. Life cycle assessments indicate that biowaste-derived hard carbon production can reduce greenhouse gas emissions by up to 40% compared to synthetic carbon materials used in conventional battery technologies.
The fast-charging protocols being developed for hard carbon anodes must balance performance optimization with resource efficiency considerations. Current research indicates that optimized charging protocols can extend battery cycle life by 30-40%, thereby reducing the frequency of battery replacement and associated resource consumption. However, aggressive fast-charging regimes may accelerate degradation mechanisms, potentially offsetting sustainability gains through shortened operational lifespans.
From a manufacturing perspective, hard carbon production requires lower processing temperatures (1000-1500°C) compared to graphite anode materials (2500-3000°C), resulting in reduced energy consumption during production. This energy advantage compounds when considering the entire battery manufacturing process, with SIBs demonstrating approximately 20% lower embodied energy compared to lithium-ion equivalents.
End-of-life considerations also favor SIBs, as they contain fewer toxic materials and critical minerals, simplifying recycling processes. Emerging recycling technologies specifically designed for sodium-ion batteries show recovery rates exceeding 90% for key materials, creating a more sustainable closed-loop system that further enhances their environmental credentials in fast-charging applications.
The extraction of sodium compounds, primarily from seawater and mineral deposits, requires substantially less water consumption compared to lithium extraction from brine pools. This reduced water footprint is particularly significant in water-stressed regions where lithium mining operations have faced increasing environmental scrutiny. Additionally, the carbon footprint associated with sodium extraction and processing is estimated to be 25-30% lower than comparable lithium supply chains.
Hard carbon materials used in SIB anodes can be derived from renewable biomass sources such as agricultural waste, cellulose, and lignin. This circular economy approach provides dual environmental benefits: waste valorization and reduced dependency on mineral extraction. Life cycle assessments indicate that biowaste-derived hard carbon production can reduce greenhouse gas emissions by up to 40% compared to synthetic carbon materials used in conventional battery technologies.
The fast-charging protocols being developed for hard carbon anodes must balance performance optimization with resource efficiency considerations. Current research indicates that optimized charging protocols can extend battery cycle life by 30-40%, thereby reducing the frequency of battery replacement and associated resource consumption. However, aggressive fast-charging regimes may accelerate degradation mechanisms, potentially offsetting sustainability gains through shortened operational lifespans.
From a manufacturing perspective, hard carbon production requires lower processing temperatures (1000-1500°C) compared to graphite anode materials (2500-3000°C), resulting in reduced energy consumption during production. This energy advantage compounds when considering the entire battery manufacturing process, with SIBs demonstrating approximately 20% lower embodied energy compared to lithium-ion equivalents.
End-of-life considerations also favor SIBs, as they contain fewer toxic materials and critical minerals, simplifying recycling processes. Emerging recycling technologies specifically designed for sodium-ion batteries show recovery rates exceeding 90% for key materials, creating a more sustainable closed-loop system that further enhances their environmental credentials in fast-charging applications.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!