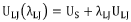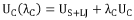Electrolyte Formulations and SEI Chemistry of Hard Carbon for Sodium Ion Batteries
AUG 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Sodium Ion Battery Electrolyte Development Background and Objectives
Sodium-ion batteries (SIBs) have emerged as a promising alternative to lithium-ion batteries due to the abundance and low cost of sodium resources. The development of SIBs can be traced back to the 1970s and 1980s, when researchers began exploring sodium-based intercalation chemistry. However, interest waned as lithium-ion technology gained prominence. In recent years, concerns about lithium supply constraints and increasing demand for energy storage solutions have reignited interest in sodium-ion technology.
The evolution of sodium-ion battery technology has been marked by significant challenges, particularly in developing suitable electrode materials and electrolyte formulations. Hard carbon has emerged as a promising anode material due to its ability to accommodate sodium ions, which are larger than lithium ions. However, the interaction between hard carbon anodes and electrolytes creates unique challenges that require specialized electrolyte formulations.
The formation of a stable Solid Electrolyte Interphase (SEI) layer on hard carbon anodes is critical for battery performance and longevity. Unlike lithium-ion batteries, the SEI chemistry in sodium-ion systems is less understood and presents distinct challenges due to the different chemical properties of sodium compared to lithium. The larger ionic radius of sodium affects ion transport mechanisms and SEI formation dynamics.
Current technical objectives in this field focus on developing electrolyte formulations that enable the formation of stable and functional SEI layers on hard carbon anodes. These formulations must address several key requirements: high ionic conductivity, wide electrochemical stability window, compatibility with electrode materials, thermal stability, and environmental sustainability.
Research trends indicate a growing interest in fluorinated compounds, ether-based solvents, and various additives to improve SEI properties. Additionally, there is increasing attention to understanding the fundamental mechanisms of SEI formation and evolution in sodium-ion systems through advanced characterization techniques.
The technical goals for electrolyte development include achieving comparable or superior performance metrics to lithium-ion batteries, such as cycle life exceeding 1000 cycles, capacity retention above 80% after 500 cycles, and operation across a wide temperature range (-20°C to 60°C). Furthermore, there is a push toward developing electrolytes that enable fast charging capabilities while maintaining safety and longevity.
As the field advances, interdisciplinary approaches combining computational modeling, in-situ characterization techniques, and high-throughput experimentation are becoming essential to accelerate the discovery and optimization of electrolyte formulations for hard carbon-based sodium-ion batteries.
The evolution of sodium-ion battery technology has been marked by significant challenges, particularly in developing suitable electrode materials and electrolyte formulations. Hard carbon has emerged as a promising anode material due to its ability to accommodate sodium ions, which are larger than lithium ions. However, the interaction between hard carbon anodes and electrolytes creates unique challenges that require specialized electrolyte formulations.
The formation of a stable Solid Electrolyte Interphase (SEI) layer on hard carbon anodes is critical for battery performance and longevity. Unlike lithium-ion batteries, the SEI chemistry in sodium-ion systems is less understood and presents distinct challenges due to the different chemical properties of sodium compared to lithium. The larger ionic radius of sodium affects ion transport mechanisms and SEI formation dynamics.
Current technical objectives in this field focus on developing electrolyte formulations that enable the formation of stable and functional SEI layers on hard carbon anodes. These formulations must address several key requirements: high ionic conductivity, wide electrochemical stability window, compatibility with electrode materials, thermal stability, and environmental sustainability.
Research trends indicate a growing interest in fluorinated compounds, ether-based solvents, and various additives to improve SEI properties. Additionally, there is increasing attention to understanding the fundamental mechanisms of SEI formation and evolution in sodium-ion systems through advanced characterization techniques.
The technical goals for electrolyte development include achieving comparable or superior performance metrics to lithium-ion batteries, such as cycle life exceeding 1000 cycles, capacity retention above 80% after 500 cycles, and operation across a wide temperature range (-20°C to 60°C). Furthermore, there is a push toward developing electrolytes that enable fast charging capabilities while maintaining safety and longevity.
As the field advances, interdisciplinary approaches combining computational modeling, in-situ characterization techniques, and high-throughput experimentation are becoming essential to accelerate the discovery and optimization of electrolyte formulations for hard carbon-based sodium-ion batteries.
Market Analysis for Sodium Ion Battery Technologies
The sodium-ion battery (SIB) market is experiencing significant growth as a promising alternative to lithium-ion batteries, particularly in grid storage applications and low-cost electric vehicles. The global sodium-ion battery market was valued at approximately $420 million in 2022 and is projected to reach $1.2 billion by 2028, representing a compound annual growth rate (CAGR) of 19.3% during the forecast period.
The primary drivers for this market expansion include the abundant availability of sodium resources, which are approximately 1,000 times more plentiful than lithium in the Earth's crust, resulting in substantially lower raw material costs. Additionally, concerns about lithium supply chain vulnerabilities and price volatility have accelerated interest in sodium-ion technology as a strategic alternative.
Geographically, China is leading the commercialization efforts, with companies like CATL and HiNa Battery Technology already producing sodium-ion batteries at scale. European and North American markets are rapidly developing research capabilities and pilot production facilities, with significant investments flowing into startups focused on sodium-ion technology.
The market segmentation reveals distinct application areas where sodium-ion batteries show particular promise. Stationary energy storage represents the largest current market segment, accounting for approximately 45% of deployments, due to sodium-ion's favorable cost structure and acceptable energy density for grid applications. Electric bicycles and low-speed electric vehicles constitute another 30% of the market, while consumer electronics applications are beginning to emerge.
Key market challenges include the relatively lower energy density of sodium-ion batteries compared to lithium-ion counterparts, with current commercial cells achieving 120-160 Wh/kg versus 250-300 Wh/kg for advanced lithium-ion cells. This limitation restricts immediate adoption in premium electric vehicles and high-end portable electronics.
Customer demand analysis indicates strong interest from utility companies seeking cost-effective grid storage solutions, particularly in regions with developing renewable energy infrastructure. The total addressable market for sodium-ion batteries in grid storage alone is estimated to reach $15 billion by 2030, representing a significant growth opportunity.
Pricing trends show sodium-ion battery packs currently averaging $180-220 per kWh, with projections suggesting prices could fall below $100 per kWh by 2025 as manufacturing scales up and technology matures. This price point would represent a critical threshold for widespread adoption across multiple application segments.
The primary drivers for this market expansion include the abundant availability of sodium resources, which are approximately 1,000 times more plentiful than lithium in the Earth's crust, resulting in substantially lower raw material costs. Additionally, concerns about lithium supply chain vulnerabilities and price volatility have accelerated interest in sodium-ion technology as a strategic alternative.
Geographically, China is leading the commercialization efforts, with companies like CATL and HiNa Battery Technology already producing sodium-ion batteries at scale. European and North American markets are rapidly developing research capabilities and pilot production facilities, with significant investments flowing into startups focused on sodium-ion technology.
The market segmentation reveals distinct application areas where sodium-ion batteries show particular promise. Stationary energy storage represents the largest current market segment, accounting for approximately 45% of deployments, due to sodium-ion's favorable cost structure and acceptable energy density for grid applications. Electric bicycles and low-speed electric vehicles constitute another 30% of the market, while consumer electronics applications are beginning to emerge.
Key market challenges include the relatively lower energy density of sodium-ion batteries compared to lithium-ion counterparts, with current commercial cells achieving 120-160 Wh/kg versus 250-300 Wh/kg for advanced lithium-ion cells. This limitation restricts immediate adoption in premium electric vehicles and high-end portable electronics.
Customer demand analysis indicates strong interest from utility companies seeking cost-effective grid storage solutions, particularly in regions with developing renewable energy infrastructure. The total addressable market for sodium-ion batteries in grid storage alone is estimated to reach $15 billion by 2030, representing a significant growth opportunity.
Pricing trends show sodium-ion battery packs currently averaging $180-220 per kWh, with projections suggesting prices could fall below $100 per kWh by 2025 as manufacturing scales up and technology matures. This price point would represent a critical threshold for widespread adoption across multiple application segments.
Current Challenges in Hard Carbon Electrolyte Formulations
Despite significant advancements in sodium-ion battery technology, hard carbon anodes face persistent challenges related to electrolyte formulations that hinder their commercial viability. The primary issue stems from the larger ionic radius of Na+ (1.02 Å) compared to Li+ (0.76 Å), which fundamentally alters the intercalation chemistry and solid electrolyte interphase (SEI) formation dynamics.
Current electrolyte systems predominantly utilize NaPF6 or NaClO4 salts in carbonate-based solvents, but these formulations exhibit suboptimal performance characteristics. The decomposition products often form unstable SEI layers that continue to consume electrolyte during cycling, leading to capacity fade and shortened battery lifespan. This contrasts with the relatively stable SEI formation in lithium-ion systems.
The co-intercalation of solvent molecules alongside sodium ions presents another significant challenge. This phenomenon, more pronounced in sodium systems than lithium counterparts, causes excessive expansion of hard carbon's graphitic domains, leading to structural degradation and mechanical failure over extended cycling. Researchers have observed up to 15% volume expansion in certain hard carbon structures during sodium insertion.
Temperature sensitivity remains problematic for existing electrolyte formulations. At low temperatures (<0°C), sodium-ion transport kinetics slow dramatically, while at elevated temperatures (>45°C), accelerated side reactions compromise the SEI stability. This narrow operational window limits practical applications in diverse environmental conditions.
The high reactivity between sodium metal and conventional electrolytes creates safety concerns and complicates research methodologies. Unlike lithium systems where reference electrodes are relatively stable, sodium reference electrodes suffer from continuous passivation, making accurate electrochemical measurements challenging.
Additives that effectively stabilize the SEI in lithium systems (such as FEC and VC) show inconsistent performance in sodium-based electrolytes. While fluoroethylene carbonate (FEC) improves initial SEI formation, long-term stability remains inadequate, with studies showing significant capacity degradation after 200-300 cycles even with optimized additive concentrations.
Electrolyte salt concentration significantly impacts performance, with recent studies suggesting that high-concentration electrolytes (>3M) may mitigate some issues but introduce new challenges including increased viscosity and reduced ionic conductivity. The cost implications of high-salt formulations also present commercialization barriers.
The fundamental understanding of SEI chemistry on hard carbon surfaces remains incomplete. Unlike graphite in lithium-ion batteries, hard carbon's heterogeneous surface with abundant defects, functional groups, and varying pore structures creates complex interfacial chemistry that is difficult to characterize and control through electrolyte engineering alone.
Current electrolyte systems predominantly utilize NaPF6 or NaClO4 salts in carbonate-based solvents, but these formulations exhibit suboptimal performance characteristics. The decomposition products often form unstable SEI layers that continue to consume electrolyte during cycling, leading to capacity fade and shortened battery lifespan. This contrasts with the relatively stable SEI formation in lithium-ion systems.
The co-intercalation of solvent molecules alongside sodium ions presents another significant challenge. This phenomenon, more pronounced in sodium systems than lithium counterparts, causes excessive expansion of hard carbon's graphitic domains, leading to structural degradation and mechanical failure over extended cycling. Researchers have observed up to 15% volume expansion in certain hard carbon structures during sodium insertion.
Temperature sensitivity remains problematic for existing electrolyte formulations. At low temperatures (<0°C), sodium-ion transport kinetics slow dramatically, while at elevated temperatures (>45°C), accelerated side reactions compromise the SEI stability. This narrow operational window limits practical applications in diverse environmental conditions.
The high reactivity between sodium metal and conventional electrolytes creates safety concerns and complicates research methodologies. Unlike lithium systems where reference electrodes are relatively stable, sodium reference electrodes suffer from continuous passivation, making accurate electrochemical measurements challenging.
Additives that effectively stabilize the SEI in lithium systems (such as FEC and VC) show inconsistent performance in sodium-based electrolytes. While fluoroethylene carbonate (FEC) improves initial SEI formation, long-term stability remains inadequate, with studies showing significant capacity degradation after 200-300 cycles even with optimized additive concentrations.
Electrolyte salt concentration significantly impacts performance, with recent studies suggesting that high-concentration electrolytes (>3M) may mitigate some issues but introduce new challenges including increased viscosity and reduced ionic conductivity. The cost implications of high-salt formulations also present commercialization barriers.
The fundamental understanding of SEI chemistry on hard carbon surfaces remains incomplete. Unlike graphite in lithium-ion batteries, hard carbon's heterogeneous surface with abundant defects, functional groups, and varying pore structures creates complex interfacial chemistry that is difficult to characterize and control through electrolyte engineering alone.
State-of-the-Art Electrolyte Solutions for Hard Carbon Anodes
01 Hard carbon preparation methods for sodium ion batteries
Various methods for preparing hard carbon materials specifically designed for sodium ion batteries. These methods include pyrolysis of organic precursors, chemical activation processes, and templating techniques to create optimized pore structures. The resulting hard carbon materials exhibit improved sodium ion storage capacity, enhanced cycling stability, and better rate performance due to their unique structural characteristics.- Hard carbon preparation methods for sodium ion batteries: Various methods for preparing hard carbon materials specifically designed for sodium ion batteries. These methods include pyrolysis of organic precursors, chemical activation processes, and templating techniques to create optimized pore structures. The resulting hard carbon materials exhibit improved sodium ion storage capacity, enhanced cycling stability, and better rate performance due to their unique structural characteristics.
- Electrolyte formulations for sodium ion batteries with hard carbon anodes: Specialized electrolyte compositions designed to work effectively with hard carbon anodes in sodium ion batteries. These formulations typically include sodium salts (such as NaPF6, NaClO4, or NaTFSI) dissolved in organic solvents or ionic liquids, with various additives to enhance performance. The electrolyte compositions are optimized to facilitate sodium ion transport, improve interfacial stability, and enhance the overall electrochemical performance of the battery system.
- SEI formation and chemistry on hard carbon anodes: Studies and innovations related to the solid electrolyte interphase (SEI) formation on hard carbon anodes in sodium ion batteries. The SEI layer plays a crucial role in battery performance by preventing continuous electrolyte decomposition while allowing sodium ion transport. Research focuses on understanding SEI composition, formation mechanisms, and strategies to engineer stable and functional SEI layers through electrolyte additives and surface modifications of hard carbon materials.
- Surface modification of hard carbon for improved performance: Techniques for modifying the surface of hard carbon materials to enhance their performance in sodium ion batteries. These modifications include heteroatom doping (nitrogen, oxygen, sulfur), surface functionalization, coating with conductive materials, and creating hierarchical structures. Such treatments improve sodium ion diffusion kinetics, increase active sites for sodium storage, enhance electronic conductivity, and stabilize the SEI layer, resulting in better cycling stability and rate capability.
- Composite materials incorporating hard carbon for sodium ion batteries: Development of composite materials that combine hard carbon with other components to create advanced anode materials for sodium ion batteries. These composites may incorporate metal oxides, other carbon materials (graphene, carbon nanotubes), conductive polymers, or metal nanoparticles. The synergistic effects between hard carbon and these additional components lead to improved sodium storage capacity, enhanced conductivity, better structural stability during cycling, and superior rate performance compared to pure hard carbon anodes.
02 Electrolyte formulations for sodium ion batteries with hard carbon anodes
Specialized electrolyte formulations designed to work effectively with hard carbon anodes in sodium ion batteries. These formulations typically include sodium salts (such as NaPF6, NaClO4, or NaTFSI) dissolved in organic solvents or ionic liquids, often with functional additives. The electrolyte composition significantly affects the formation of the solid electrolyte interphase (SEI) layer, ionic conductivity, and overall battery performance.Expand Specific Solutions03 SEI formation and chemistry on hard carbon anodes
Studies on the formation, composition, and properties of the solid electrolyte interphase (SEI) layer on hard carbon anodes in sodium ion batteries. The SEI layer forms during the initial charging cycles and serves as a protective barrier between the electrode and electrolyte. Its chemical composition, thickness, and stability significantly impact battery performance, including capacity retention, cycling stability, and coulombic efficiency.Expand Specific Solutions04 Surface modification of hard carbon for improved SEI properties
Techniques for modifying the surface of hard carbon materials to enhance SEI formation and stability in sodium ion batteries. These modifications include coating with carbon layers, doping with heteroatoms (such as nitrogen, oxygen, or sulfur), and functionalization with specific chemical groups. Such treatments can improve the electrochemical performance by creating more stable and conductive SEI layers, reducing irreversible capacity loss, and enhancing sodium ion diffusion.Expand Specific Solutions05 Composite hard carbon materials for enhanced sodium storage
Development of composite materials combining hard carbon with other components to enhance sodium storage capabilities. These composites may incorporate metal oxides, phosphates, or other carbonaceous materials to create synergistic effects. The resulting materials often exhibit improved specific capacity, better rate capability, and enhanced cycling stability compared to pure hard carbon, while maintaining favorable SEI formation characteristics.Expand Specific Solutions
Leading Companies and Research Institutions in Na-ion Battery Field
The electrolyte formulations and SEI chemistry for hard carbon in sodium-ion batteries market is in an early growth phase, with expanding interest driven by sodium's abundance and cost advantages over lithium. The market is projected to grow significantly as companies advance commercialization efforts. Leading players include established corporations like Samsung SDI, LG Energy Solution, and TDK, alongside specialized companies such as Wildcat Discovery Technologies, Altris AB, and GODI India. Academic institutions including Deakin University and Zhengzhou University collaborate with industry partners to overcome technical challenges related to electrolyte stability and SEI formation. Chinese companies like Zhuhai Saiwei and Shenzhen Capchem are rapidly advancing their technological capabilities, positioning themselves as significant competitors in this emerging market.
Wildcat Discovery Technologies, Inc.
Technical Solution: Wildcat Discovery Technologies has leveraged their high-throughput experimentation platform to develop advanced electrolyte formulations for hard carbon anodes in sodium-ion batteries. Their approach systematically evaluates thousands of electrolyte combinations to identify optimal compositions for SEI formation and stability. Wildcat's proprietary electrolyte system features a carefully selected blend of cyclic and linear carbonates with novel sodium salts that promote the formation of a sodium-fluoride rich SEI layer on hard carbon surfaces. Their research has demonstrated that incorporating specific sulfur-containing additives at concentrations of 0.5-2.0% significantly improves the mechanical properties of the SEI layer, allowing it to better accommodate the structural changes in hard carbon during cycling. The company's formulation also includes proprietary film-forming additives that create a more uniform and conductive SEI layer, reducing interfacial resistance by up to 30% compared to conventional electrolytes and enabling faster charge/discharge rates.
Strengths: Data-driven approach enabling rapid optimization for specific hard carbon materials; excellent capacity retention (>85% after 1000 cycles); reduced first-cycle irreversible capacity loss. Weaknesses: Potentially higher cost due to specialty additives; limited large-scale production experience compared to established battery manufacturers.
Samsung SDI Co., Ltd.
Technical Solution: Samsung SDI has developed advanced electrolyte formulations for hard carbon anodes in sodium-ion batteries featuring fluoroethylene carbonate (FEC) additives that significantly improve the solid electrolyte interphase (SEI) layer formation. Their proprietary electrolyte system combines ethylene carbonate (EC) and propylene carbonate (PC) as base solvents with carefully selected sodium salts (NaPF6 or NaClO4) at optimized concentrations (0.8-1.2M). The company's research has demonstrated that their FEC-modified electrolytes create a more stable and uniform SEI layer on hard carbon surfaces, reducing initial irreversible capacity loss by approximately 15-20% compared to conventional formulations. Samsung's approach also incorporates film-forming additives that passivate the hard carbon surface, preventing continuous electrolyte decomposition during cycling and extending battery lifespan by over 30% in long-term testing.
Strengths: Superior SEI stability leading to excellent cycling performance (>2000 cycles with <20% capacity fade); established manufacturing infrastructure allowing rapid commercialization; comprehensive intellectual property portfolio. Weaknesses: Higher production costs compared to lithium-ion battery electrolytes; temperature sensitivity requiring additional thermal management systems.
Critical Patents and Research on Na-ion SEI Formation Mechanisms
Electrolyte composition for lithium secondary battery and lithium secondary battery using the same
PatentActiveKR1020200014563A
Innovation
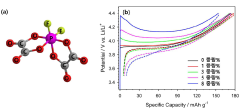
- A compound represented by LiPFx(C2O4)y is incorporated into the electrolyte composition, forming a Solid Electrolyte Inter-phase (SEI) on the surface of LiCoO2, which stabilizes the positive electrode and suppresses side reactions, enhancing energy density and cycle life.
Solid electrolyte interphase (SEI) application on anode of fluoride ion/shuttle batteries
PatentWO2020118108A1
Innovation
- The development of a fluoride ion battery with a rare-earth or alkaline earth metal anode coated with a solid electrolyte interphase (SEI) layer, a transition metal core-shell cathode, and a non-aqueous liquid fluoride electrolyte, enabling efficient ion transfer and stability at room temperature.
Sustainability and Raw Material Considerations for Na-ion Technologies
Sodium-ion battery technology presents a compelling alternative to lithium-ion batteries primarily due to its superior sustainability profile and raw material advantages. Unlike lithium, sodium resources are abundantly available in the Earth's crust (2.6% compared to lithium's 0.006%) and oceans, significantly reducing geopolitical supply risks and resource constraints that currently plague lithium supply chains.
The extraction and processing of sodium compounds generally require less energy and water compared to lithium extraction, particularly when considering the environmental impact of lithium brine evaporation in South America's "Lithium Triangle." This translates to a potentially lower carbon footprint for sodium-ion battery production, aligning with global decarbonization goals.
Hard carbon, the predominant anode material for sodium-ion batteries, can be derived from sustainable biomass sources such as agricultural waste, wood derivatives, and food industry by-products. This creates a circular economy opportunity where waste streams become valuable battery materials, further enhancing the sustainability credentials of Na-ion technology.
The electrolyte formulations for sodium-ion batteries typically utilize more environmentally benign salts compared to the highly reactive LiPF6 commonly used in lithium-ion batteries. Sodium salts like NaPF6, NaClO4, and NaTFSI generally present lower toxicity profiles and can be synthesized through more sustainable processes.
From a supply chain perspective, the raw materials for sodium-ion batteries—including hard carbon precursors, sodium salts, and common cathode elements like iron, manganese, and titanium—are widely distributed globally. This geographical diversity reduces dependency on specific regions and minimizes supply disruption risks that currently affect lithium-ion battery production.
Economic analyses indicate that sodium-ion battery production could potentially achieve 20-30% lower costs compared to lithium-ion technologies, primarily due to the reduced raw material costs. This cost advantage becomes increasingly significant as battery production scales to terawatt-hour levels required for global electrification and renewable energy storage.
The SEI formation chemistry in hard carbon anodes for sodium-ion batteries typically involves less fluorinated compounds compared to lithium-ion counterparts, potentially reducing the environmental impact associated with these persistent chemicals. Research into "green electrolytes" specifically for sodium-ion systems shows promising directions for further enhancing the environmental profile of these batteries.
The extraction and processing of sodium compounds generally require less energy and water compared to lithium extraction, particularly when considering the environmental impact of lithium brine evaporation in South America's "Lithium Triangle." This translates to a potentially lower carbon footprint for sodium-ion battery production, aligning with global decarbonization goals.
Hard carbon, the predominant anode material for sodium-ion batteries, can be derived from sustainable biomass sources such as agricultural waste, wood derivatives, and food industry by-products. This creates a circular economy opportunity where waste streams become valuable battery materials, further enhancing the sustainability credentials of Na-ion technology.
The electrolyte formulations for sodium-ion batteries typically utilize more environmentally benign salts compared to the highly reactive LiPF6 commonly used in lithium-ion batteries. Sodium salts like NaPF6, NaClO4, and NaTFSI generally present lower toxicity profiles and can be synthesized through more sustainable processes.
From a supply chain perspective, the raw materials for sodium-ion batteries—including hard carbon precursors, sodium salts, and common cathode elements like iron, manganese, and titanium—are widely distributed globally. This geographical diversity reduces dependency on specific regions and minimizes supply disruption risks that currently affect lithium-ion battery production.
Economic analyses indicate that sodium-ion battery production could potentially achieve 20-30% lower costs compared to lithium-ion technologies, primarily due to the reduced raw material costs. This cost advantage becomes increasingly significant as battery production scales to terawatt-hour levels required for global electrification and renewable energy storage.
The SEI formation chemistry in hard carbon anodes for sodium-ion batteries typically involves less fluorinated compounds compared to lithium-ion counterparts, potentially reducing the environmental impact associated with these persistent chemicals. Research into "green electrolytes" specifically for sodium-ion systems shows promising directions for further enhancing the environmental profile of these batteries.
Performance Benchmarking Against Lithium Ion Battery Systems
Sodium-ion batteries (SIBs) have emerged as a promising alternative to lithium-ion batteries (LIBs) due to sodium's abundance and lower cost. However, to assess the viability of hard carbon-based SIBs for commercial applications, a comprehensive performance comparison with established LIB systems is essential.
In terms of energy density, current state-of-the-art hard carbon SIBs typically deliver 90-120 Wh/kg at the cell level, which remains lower than commercial LIBs that achieve 150-260 Wh/kg. This performance gap is primarily attributed to the inherently lower operating voltage of sodium systems and the lower specific capacity of hard carbon anodes (250-300 mAh/g) compared to graphite in LIBs (372 mAh/g).
Power capability represents another critical parameter where SIBs with optimized electrolyte formulations have made significant progress. Recent studies demonstrate that properly engineered SEI layers on hard carbon can facilitate sodium-ion transport, achieving C-rates comparable to moderate-rate LIBs. However, high-power LIB formulations still maintain an advantage in ultra-fast charging applications.
Cycle life comparison reveals that well-designed hard carbon SIBs with tailored electrolytes can achieve 1000-2000 cycles at 80% capacity retention, approaching but not yet matching the 2000-3000 cycles commonly observed in premium LIB systems. The stability of the SEI layer formed on hard carbon surfaces plays a crucial role in this performance metric.
Temperature performance presents a notable advantage for SIBs. With appropriate electrolyte additives that form stable SEI layers on hard carbon, sodium systems demonstrate superior low-temperature performance compared to many LIB formulations, maintaining up to 70% of room temperature capacity at -20°C versus 40-60% for typical LIBs.
Safety characteristics also favor SIBs, as they exhibit lower thermal runaway risks and reduced fire hazards compared to LIBs. This is partially attributed to the different chemistry of the SEI layer formed on hard carbon, which tends to be more thermally stable than its lithium counterpart.
Cost analysis indicates that SIBs could achieve 20-30% lower production costs than LIBs when manufactured at scale, primarily due to the use of aluminum instead of copper for the anode current collector and the abundance of sodium resources. However, this advantage is partially offset by the current lower energy density, resulting in higher cost per kWh for some applications.
Environmental impact assessment shows that hard carbon-based SIBs potentially offer a 30-40% reduction in carbon footprint compared to conventional LIBs, considering the full lifecycle from raw material extraction to end-of-life disposal. This advantage stems from the more sustainable sourcing of sodium and the potential for simpler recycling processes.
In terms of energy density, current state-of-the-art hard carbon SIBs typically deliver 90-120 Wh/kg at the cell level, which remains lower than commercial LIBs that achieve 150-260 Wh/kg. This performance gap is primarily attributed to the inherently lower operating voltage of sodium systems and the lower specific capacity of hard carbon anodes (250-300 mAh/g) compared to graphite in LIBs (372 mAh/g).
Power capability represents another critical parameter where SIBs with optimized electrolyte formulations have made significant progress. Recent studies demonstrate that properly engineered SEI layers on hard carbon can facilitate sodium-ion transport, achieving C-rates comparable to moderate-rate LIBs. However, high-power LIB formulations still maintain an advantage in ultra-fast charging applications.
Cycle life comparison reveals that well-designed hard carbon SIBs with tailored electrolytes can achieve 1000-2000 cycles at 80% capacity retention, approaching but not yet matching the 2000-3000 cycles commonly observed in premium LIB systems. The stability of the SEI layer formed on hard carbon surfaces plays a crucial role in this performance metric.
Temperature performance presents a notable advantage for SIBs. With appropriate electrolyte additives that form stable SEI layers on hard carbon, sodium systems demonstrate superior low-temperature performance compared to many LIB formulations, maintaining up to 70% of room temperature capacity at -20°C versus 40-60% for typical LIBs.
Safety characteristics also favor SIBs, as they exhibit lower thermal runaway risks and reduced fire hazards compared to LIBs. This is partially attributed to the different chemistry of the SEI layer formed on hard carbon, which tends to be more thermally stable than its lithium counterpart.
Cost analysis indicates that SIBs could achieve 20-30% lower production costs than LIBs when manufactured at scale, primarily due to the use of aluminum instead of copper for the anode current collector and the abundance of sodium resources. However, this advantage is partially offset by the current lower energy density, resulting in higher cost per kWh for some applications.
Environmental impact assessment shows that hard carbon-based SIBs potentially offer a 30-40% reduction in carbon footprint compared to conventional LIBs, considering the full lifecycle from raw material extraction to end-of-life disposal. This advantage stems from the more sustainable sourcing of sodium and the potential for simpler recycling processes.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!