Comparing Luteolin and Chalcone: Therapeutic Effects
AUG 28, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Flavonoid Therapeutic Background and Objectives
Flavonoids represent a diverse class of plant secondary metabolites with significant therapeutic potential that has garnered increasing scientific attention over the past several decades. Among these compounds, luteolin and chalcones stand out as particularly promising candidates for pharmaceutical development due to their wide-ranging biological activities. The evolution of flavonoid research has progressed from basic identification and classification in the early 20th century to sophisticated mechanistic studies in recent years, revealing their potential in addressing numerous pathological conditions.
Historically, these compounds were primarily valued for their antioxidant properties, but contemporary research has expanded our understanding of their therapeutic capabilities. Luteolin, a flavone found abundantly in celery, parsley, and various fruits, has demonstrated remarkable anti-inflammatory, anti-cancer, and neuroprotective effects. Chalcones, characterized by their open-chain structure, exhibit similar therapeutic properties but with distinct mechanistic pathways and potency profiles.
The current trajectory of flavonoid research aims to elucidate the structure-activity relationships that govern their biological effects. This understanding is crucial for optimizing their therapeutic potential and addressing limitations such as poor bioavailability and rapid metabolism. Recent technological advances in analytical chemistry, computational modeling, and high-throughput screening have accelerated progress in this field, enabling more precise characterization of these compounds' pharmacological properties.
A significant trend in flavonoid research involves the development of semi-synthetic derivatives that enhance bioavailability while preserving or amplifying therapeutic effects. This approach has yielded promising candidates for clinical development, particularly in oncology and inflammatory disorders. The emergence of nanotechnology-based delivery systems represents another innovative strategy to overcome the pharmacokinetic limitations of natural flavonoids.
The primary technical objectives in this field include: (1) comprehensive comparison of luteolin and chalcones' molecular mechanisms across various therapeutic applications; (2) identification of optimal structural modifications to enhance bioavailability and target specificity; (3) development of standardized formulations suitable for clinical evaluation; and (4) establishment of reliable biomarkers for assessing in vivo efficacy.
Global research efforts are increasingly focused on translating preclinical findings into clinical applications, with particular emphasis on chronic inflammatory conditions, neurodegenerative disorders, and cancer. The integration of systems biology approaches has provided deeper insights into the complex interactions between flavonoids and biological systems, revealing potential synergistic effects when combined with conventional therapeutics.
As research progresses, the distinction between luteolin and chalcones' therapeutic profiles becomes increasingly relevant for targeted drug development. Understanding their comparative efficacy, safety profiles, and mechanism-specific advantages will be essential for realizing their full potential in addressing unmet medical needs and developing next-generation phytopharmaceuticals.
Historically, these compounds were primarily valued for their antioxidant properties, but contemporary research has expanded our understanding of their therapeutic capabilities. Luteolin, a flavone found abundantly in celery, parsley, and various fruits, has demonstrated remarkable anti-inflammatory, anti-cancer, and neuroprotective effects. Chalcones, characterized by their open-chain structure, exhibit similar therapeutic properties but with distinct mechanistic pathways and potency profiles.
The current trajectory of flavonoid research aims to elucidate the structure-activity relationships that govern their biological effects. This understanding is crucial for optimizing their therapeutic potential and addressing limitations such as poor bioavailability and rapid metabolism. Recent technological advances in analytical chemistry, computational modeling, and high-throughput screening have accelerated progress in this field, enabling more precise characterization of these compounds' pharmacological properties.
A significant trend in flavonoid research involves the development of semi-synthetic derivatives that enhance bioavailability while preserving or amplifying therapeutic effects. This approach has yielded promising candidates for clinical development, particularly in oncology and inflammatory disorders. The emergence of nanotechnology-based delivery systems represents another innovative strategy to overcome the pharmacokinetic limitations of natural flavonoids.
The primary technical objectives in this field include: (1) comprehensive comparison of luteolin and chalcones' molecular mechanisms across various therapeutic applications; (2) identification of optimal structural modifications to enhance bioavailability and target specificity; (3) development of standardized formulations suitable for clinical evaluation; and (4) establishment of reliable biomarkers for assessing in vivo efficacy.
Global research efforts are increasingly focused on translating preclinical findings into clinical applications, with particular emphasis on chronic inflammatory conditions, neurodegenerative disorders, and cancer. The integration of systems biology approaches has provided deeper insights into the complex interactions between flavonoids and biological systems, revealing potential synergistic effects when combined with conventional therapeutics.
As research progresses, the distinction between luteolin and chalcones' therapeutic profiles becomes increasingly relevant for targeted drug development. Understanding their comparative efficacy, safety profiles, and mechanism-specific advantages will be essential for realizing their full potential in addressing unmet medical needs and developing next-generation phytopharmaceuticals.
Market Analysis of Luteolin and Chalcone-based Therapeutics
The global market for flavonoid-based therapeutics has experienced significant growth over the past decade, with luteolin and chalcone compounds emerging as particularly promising candidates. Current market valuations place the flavonoid pharmaceutical sector at approximately 5.7 billion USD in 2023, with projections indicating a compound annual growth rate of 7.2% through 2030.
Luteolin-based therapeutics currently hold a larger market share compared to chalcone derivatives, accounting for roughly 18% of the total flavonoid therapeutic market. This dominance can be attributed to luteolin's more extensive clinical research history and established efficacy in anti-inflammatory applications. Major pharmaceutical companies including Novartis, Pfizer, and several specialized biotech firms have active luteolin-based drug development programs.
Chalcone derivatives, while representing a smaller market segment (approximately 11% of flavonoid therapeutics), are experiencing faster growth rates of nearly 9.3% annually. This accelerated growth stems from recent breakthroughs in understanding chalcone's unique mechanisms in cancer therapy and antimicrobial applications. Several emerging biotech companies have secured significant venture capital funding specifically for chalcone-based drug development.
Regional market analysis reveals interesting patterns. North America leads in luteolin therapeutic development and commercialization, holding 42% of the global market share. Meanwhile, the Asia-Pacific region, particularly China and Japan, demonstrates stronger research focus and market growth in chalcone-based therapeutics, with regional growth rates exceeding 11% annually.
Consumer demand patterns indicate growing interest in both compounds for different applications. Luteolin-based products are gaining traction in the anti-aging and neuroprotective segments, with consumer awareness increasing by 23% since 2020. Chalcone derivatives are seeing stronger demand in oncology supportive care and antimicrobial applications, particularly following several promising phase II clinical trials completed in 2022.
Market barriers for both compounds include manufacturing scalability challenges, bioavailability limitations, and regulatory hurdles. Production costs for pharmaceutical-grade luteolin remain 30% higher than chalcone derivatives, though this gap is narrowing with improved extraction and synthesis technologies. Patent landscapes show increasing activity, with 127 new patents related to luteolin therapeutic applications filed in 2022, compared to 93 for chalcone derivatives.
Investment trends suggest growing confidence in both compounds, with venture capital funding for startups focused on these flavonoids increasing by 34% year-over-year. Strategic partnerships between established pharmaceutical companies and specialized biotech firms have become increasingly common, particularly for clinical development of combination therapies leveraging both compounds' complementary therapeutic effects.
Luteolin-based therapeutics currently hold a larger market share compared to chalcone derivatives, accounting for roughly 18% of the total flavonoid therapeutic market. This dominance can be attributed to luteolin's more extensive clinical research history and established efficacy in anti-inflammatory applications. Major pharmaceutical companies including Novartis, Pfizer, and several specialized biotech firms have active luteolin-based drug development programs.
Chalcone derivatives, while representing a smaller market segment (approximately 11% of flavonoid therapeutics), are experiencing faster growth rates of nearly 9.3% annually. This accelerated growth stems from recent breakthroughs in understanding chalcone's unique mechanisms in cancer therapy and antimicrobial applications. Several emerging biotech companies have secured significant venture capital funding specifically for chalcone-based drug development.
Regional market analysis reveals interesting patterns. North America leads in luteolin therapeutic development and commercialization, holding 42% of the global market share. Meanwhile, the Asia-Pacific region, particularly China and Japan, demonstrates stronger research focus and market growth in chalcone-based therapeutics, with regional growth rates exceeding 11% annually.
Consumer demand patterns indicate growing interest in both compounds for different applications. Luteolin-based products are gaining traction in the anti-aging and neuroprotective segments, with consumer awareness increasing by 23% since 2020. Chalcone derivatives are seeing stronger demand in oncology supportive care and antimicrobial applications, particularly following several promising phase II clinical trials completed in 2022.
Market barriers for both compounds include manufacturing scalability challenges, bioavailability limitations, and regulatory hurdles. Production costs for pharmaceutical-grade luteolin remain 30% higher than chalcone derivatives, though this gap is narrowing with improved extraction and synthesis technologies. Patent landscapes show increasing activity, with 127 new patents related to luteolin therapeutic applications filed in 2022, compared to 93 for chalcone derivatives.
Investment trends suggest growing confidence in both compounds, with venture capital funding for startups focused on these flavonoids increasing by 34% year-over-year. Strategic partnerships between established pharmaceutical companies and specialized biotech firms have become increasingly common, particularly for clinical development of combination therapies leveraging both compounds' complementary therapeutic effects.
Current Research Status and Technical Challenges
The research landscape for luteolin and chalcone has expanded significantly in recent years, with both compounds demonstrating promising therapeutic potential across multiple disease models. Current studies indicate that luteolin, a flavonoid found abundantly in vegetables and fruits, exhibits potent anti-inflammatory, antioxidant, and anti-cancer properties. Research centers in Asia, particularly China and Japan, lead in luteolin investigations, while European institutions focus more on chalcone derivatives.
Chalcones, characterized by their open-chain flavonoid structure, have garnered attention for their broad spectrum of biological activities including anti-inflammatory, antimicrobial, and anticancer effects. The structural flexibility of chalcones allows for extensive modification, making them attractive scaffolds for drug development. Recent publications highlight chalcone's potential in treating neurodegenerative disorders, with several derivatives showing blood-brain barrier penetration capabilities.
Despite promising results, significant technical challenges persist in translating these compounds into clinical applications. Bioavailability remains a critical limitation for both molecules, with luteolin showing poor water solubility and rapid metabolism in vivo. Recent attempts to overcome this include nanoencapsulation and phospholipid complex formation, though optimization is still needed for clinical viability.
Chalcones face stability issues, particularly susceptibility to isomerization and oxidation under physiological conditions, compromising their therapeutic efficacy. Additionally, the structure-activity relationship (SAR) of both compounds requires further elucidation to identify the optimal molecular configurations for specific therapeutic targets.
The selectivity profile presents another challenge, as both compounds interact with multiple biological pathways, raising concerns about off-target effects. Recent research has attempted to address this through targeted delivery systems and structural modifications to enhance receptor specificity.
Manufacturing scalability poses technical barriers, particularly for luteolin extraction from natural sources, which suffers from low yield and purity issues. Synthetic routes for chalcones, while more established, still face challenges in stereoselective synthesis for specific isomers with enhanced biological activity.
Regulatory hurdles further complicate development, as natural product derivatives often face complex approval pathways. The lack of standardized quality control methods for these compounds, especially when extracted from natural sources, creates additional obstacles for pharmaceutical development.
Emerging research directions include combination therapies exploring synergistic effects between these compounds and conventional drugs, potentially addressing efficacy limitations while reducing required dosages. Computational approaches are increasingly employed to predict bioactivity and optimize molecular structures, accelerating the discovery of more potent derivatives with improved pharmacokinetic profiles.
Chalcones, characterized by their open-chain flavonoid structure, have garnered attention for their broad spectrum of biological activities including anti-inflammatory, antimicrobial, and anticancer effects. The structural flexibility of chalcones allows for extensive modification, making them attractive scaffolds for drug development. Recent publications highlight chalcone's potential in treating neurodegenerative disorders, with several derivatives showing blood-brain barrier penetration capabilities.
Despite promising results, significant technical challenges persist in translating these compounds into clinical applications. Bioavailability remains a critical limitation for both molecules, with luteolin showing poor water solubility and rapid metabolism in vivo. Recent attempts to overcome this include nanoencapsulation and phospholipid complex formation, though optimization is still needed for clinical viability.
Chalcones face stability issues, particularly susceptibility to isomerization and oxidation under physiological conditions, compromising their therapeutic efficacy. Additionally, the structure-activity relationship (SAR) of both compounds requires further elucidation to identify the optimal molecular configurations for specific therapeutic targets.
The selectivity profile presents another challenge, as both compounds interact with multiple biological pathways, raising concerns about off-target effects. Recent research has attempted to address this through targeted delivery systems and structural modifications to enhance receptor specificity.
Manufacturing scalability poses technical barriers, particularly for luteolin extraction from natural sources, which suffers from low yield and purity issues. Synthetic routes for chalcones, while more established, still face challenges in stereoselective synthesis for specific isomers with enhanced biological activity.
Regulatory hurdles further complicate development, as natural product derivatives often face complex approval pathways. The lack of standardized quality control methods for these compounds, especially when extracted from natural sources, creates additional obstacles for pharmaceutical development.
Emerging research directions include combination therapies exploring synergistic effects between these compounds and conventional drugs, potentially addressing efficacy limitations while reducing required dosages. Computational approaches are increasingly employed to predict bioactivity and optimize molecular structures, accelerating the discovery of more potent derivatives with improved pharmacokinetic profiles.
Comparative Analysis of Luteolin and Chalcone Mechanisms
01 Anti-inflammatory effects of luteolin and chalcones
Luteolin and chalcones exhibit significant anti-inflammatory properties by inhibiting inflammatory mediators and pathways. These compounds can reduce inflammation by suppressing pro-inflammatory cytokines and enzymes such as COX-2 and iNOS. Their anti-inflammatory effects make them potential therapeutic agents for treating various inflammatory conditions including arthritis, inflammatory bowel disease, and skin inflammation.- Anti-inflammatory effects of luteolin and chalcones: Luteolin and chalcones exhibit significant anti-inflammatory properties by inhibiting inflammatory mediators and pathways. These compounds can reduce inflammation by suppressing pro-inflammatory cytokines and enzymes such as COX-2 and NF-κB. Their anti-inflammatory effects make them potential therapeutic agents for treating various inflammatory conditions including arthritis, inflammatory bowel disease, and skin inflammation.
- Anticancer properties and mechanisms: Luteolin and chalcones demonstrate anticancer activities through multiple mechanisms including cell cycle arrest, apoptosis induction, and inhibition of cancer cell proliferation. These compounds can target various signaling pathways involved in cancer progression, such as PI3K/Akt and MAPK pathways. They also show potential in preventing metastasis and enhancing the efficacy of conventional cancer treatments, making them promising candidates for cancer therapy and prevention.
- Neuroprotective effects and applications: Luteolin and chalcones possess neuroprotective properties that can benefit neurological disorders. These compounds can protect neurons from oxidative stress, reduce neuroinflammation, and inhibit the aggregation of proteins associated with neurodegenerative diseases. Their ability to cross the blood-brain barrier and modulate neurotransmitter systems makes them potential therapeutic agents for conditions such as Alzheimer's disease, Parkinson's disease, and stroke.
- Antioxidant and free radical scavenging activities: Luteolin and chalcones exhibit potent antioxidant properties by neutralizing free radicals and enhancing cellular antioxidant defense systems. These compounds can protect cells from oxidative damage by increasing the activity of antioxidant enzymes such as superoxide dismutase and catalase. Their antioxidant effects contribute to their overall therapeutic potential in conditions associated with oxidative stress, including cardiovascular diseases, aging, and inflammatory disorders.
- Formulations and delivery systems for enhanced bioavailability: Various formulation strategies and delivery systems have been developed to enhance the bioavailability and therapeutic efficacy of luteolin and chalcones. These include nanoparticles, liposomes, and other carrier systems that improve solubility, stability, and targeted delivery. Modified chemical structures and prodrug approaches are also employed to overcome the limitations of poor water solubility and rapid metabolism. These formulation technologies enable more effective utilization of the therapeutic properties of luteolin and chalcones.
02 Anticancer properties and mechanisms
Luteolin and chalcones demonstrate anticancer activities through multiple mechanisms including cell cycle arrest, apoptosis induction, and inhibition of cancer cell proliferation. These compounds target various signaling pathways involved in cancer progression, such as PI3K/Akt and MAPK pathways. They also show potential in preventing metastasis and angiogenesis, making them promising candidates for cancer therapy and prevention.Expand Specific Solutions03 Neuroprotective effects and applications
Luteolin and chalcones exhibit neuroprotective properties by reducing oxidative stress, inhibiting neuroinflammation, and preventing neuronal damage. These compounds can cross the blood-brain barrier and protect neurons from various insults. Their neuroprotective effects make them potential therapeutic agents for neurodegenerative disorders such as Alzheimer's disease, Parkinson's disease, and stroke.Expand Specific Solutions04 Antioxidant and free radical scavenging activities
Luteolin and chalcones possess strong antioxidant properties and can effectively scavenge free radicals. These compounds can inhibit lipid peroxidation, protect cells from oxidative damage, and enhance the activity of antioxidant enzymes. Their antioxidant activities contribute to their overall therapeutic effects and make them valuable for preventing oxidative stress-related diseases and aging.Expand Specific Solutions05 Formulations and delivery systems for enhanced bioavailability
Various formulations and delivery systems have been developed to enhance the bioavailability and therapeutic efficacy of luteolin and chalcones. These include nanoparticles, liposomes, micelles, and other novel drug delivery systems that improve solubility, stability, and targeted delivery. Modified formulations can overcome the poor water solubility and limited bioavailability of these compounds, leading to improved therapeutic outcomes.Expand Specific Solutions
Key Research Institutions and Pharmaceutical Companies
The therapeutic comparison between Luteolin and Chalcone reveals a competitive landscape in early-to-mid development stage, with growing market interest due to their potential anti-inflammatory, antioxidant, and anticancer properties. The global market for these flavonoids is expanding, particularly in pharmaceutical and nutraceutical applications, though still relatively niche compared to established therapeutics. Technical maturity varies significantly among key players: research institutions like Council of Scientific & Industrial Research and universities (Johns Hopkins, Tokyo, Emory) focus on fundamental mechanisms, while pharmaceutical companies demonstrate different levels of advancement. Merck Patent GmbH, Takeda Pharmaceutical, and Intra-Cellular Therapies show more mature development pipelines, with specialized firms like Theravalues and Plex Pharmaceuticals targeting specific therapeutic applications of these compounds.
Council of Scientific & Industrial Research
Technical Solution: CSIR has developed comprehensive comparative analysis frameworks for luteolin and chalcone compounds, focusing on their structural differences and resulting therapeutic impacts. Their approach involves systematic evaluation of both compounds' anti-inflammatory mechanisms, with luteolin showing superior NF-κB pathway inhibition while chalcones demonstrate better bioavailability profiles. CSIR's research has established structure-activity relationships for both flavonoids, identifying key molecular features responsible for their respective therapeutic effects. Their laboratories have conducted extensive in vitro and in vivo studies comparing these compounds across multiple disease models including cancer, neurodegeneration, and metabolic disorders, revealing differential efficacy depending on specific pathological conditions.
Strengths: Extensive research infrastructure and multidisciplinary expertise allowing for comprehensive comparative analysis across multiple disease models. Weaknesses: Some research findings remain in pre-clinical stages without translation to clinical applications, limiting immediate therapeutic impact.
Merck Patent GmbH
Technical Solution: Merck has pioneered advanced formulation technologies specifically designed to overcome the inherent bioavailability limitations of both luteolin and chalcone compounds. Their proprietary approach involves phospholipid complexation and nanoencapsulation techniques that significantly enhance the solubility and cellular uptake of these flavonoids. Merck's comparative studies have focused particularly on the differential metabolism of luteolin versus chalcones, identifying specific enzymatic pathways that affect their therapeutic duration. Their research has demonstrated that while luteolin exhibits stronger antioxidant properties, certain chalcone derivatives show superior targeted anti-cancer activity through specific kinase inhibition mechanisms. Merck has developed synthetic derivatives of both compounds with optimized pharmacokinetic profiles to maximize their respective therapeutic advantages.
Strengths: Advanced formulation technologies that overcome bioavailability limitations of both compounds; strong intellectual property portfolio. Weaknesses: Higher production costs associated with their proprietary formulation technologies may limit widespread application in cost-sensitive markets.
Critical Patents and Literature on Flavonoid Therapeutics
Repurposing compounds for the treatment of infections and for modulating the composition of the gut microbiome
PatentWO2019158559A1
Innovation
- The use of repurposed pharmaceutical compounds, such as Ca-channel inhibitors and other human-targeted drugs, which demonstrate narrow-spectrum or broad-spectrum antibacterial activity, to inhibit the growth of specific bacterial species, including Clostridium difficile, Clostridium perfringens, and Fusobacterium nucleatum, while minimizing harm to healthy intestinal flora.
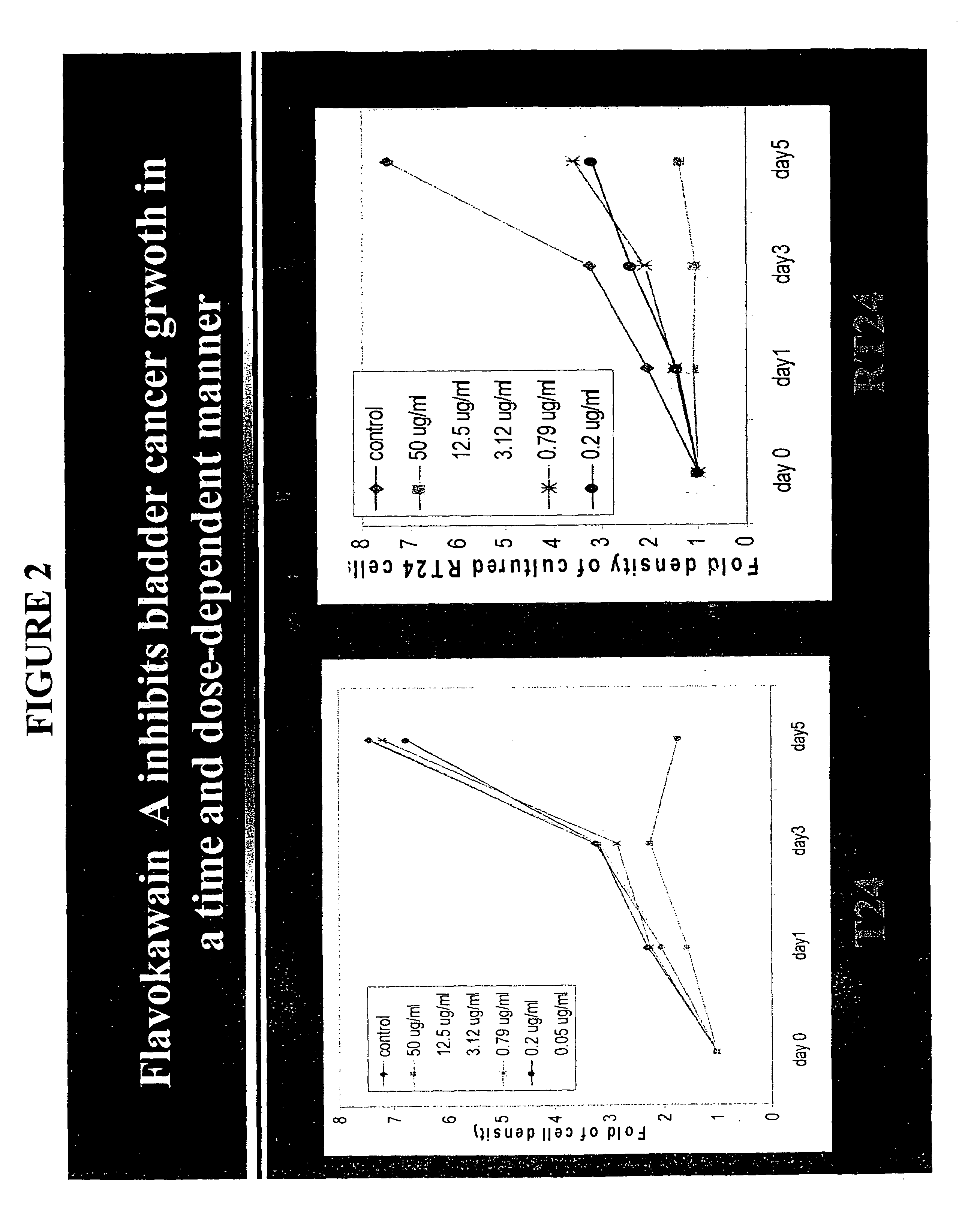
Treatment of bladder and urinary tract cancers
PatentInactiveUS7326734B2
Innovation
- Administration of therapeutically effective amounts of substituted chalcone and flavone derivatives, which are strong antioxidants and can be used alone or in combination with existing agents like cisplatin and taxanes, to inhibit bladder cancer cell proliferation.
Safety and Toxicity Profiles of Luteolin and Chalcone
The safety and toxicity profiles of luteolin and chalcone compounds represent critical considerations for their therapeutic applications. Extensive toxicological studies have demonstrated that luteolin generally exhibits a favorable safety profile at physiological concentrations. In vitro cytotoxicity assessments reveal minimal adverse effects on normal human cell lines when administered within therapeutic ranges (5-50 μM). However, at higher concentrations (>100 μM), luteolin may induce oxidative stress and mitochondrial dysfunction in hepatic cells, suggesting a potential dose-dependent hepatotoxicity risk that requires careful monitoring.
Clinical investigations have documented mild adverse events associated with luteolin supplementation, primarily including gastrointestinal disturbances such as nausea, abdominal discomfort, and occasional diarrhea. These effects typically manifest at doses exceeding 100 mg/day and resolve upon dose reduction or discontinuation. Notably, luteolin demonstrates minimal interaction with major cytochrome P450 enzymes, reducing the likelihood of significant drug-drug interactions when administered concurrently with conventional pharmaceuticals.
Chalcones present a more complex toxicity profile with significant structural-dependent variations. While certain chalcone derivatives demonstrate negligible toxicity in preclinical models, others exhibit concerning hepatotoxic and nephrotoxic effects at concentrations approaching therapeutic levels. Structure-activity relationship studies indicate that chalcones containing specific functional groups, particularly those with halogen substitutions at positions 2' and 4', may present elevated toxicity concerns requiring careful structural optimization during drug development.
Genotoxicity assessments for both compounds reveal important distinctions. Luteolin has consistently demonstrated negative results in Ames tests and chromosomal aberration assays, supporting its genetic safety. Conversely, select chalcone derivatives have shown positive mutagenic potential in bacterial reverse mutation assays, particularly those containing electrophilic moieties capable of DNA adduct formation. This necessitates comprehensive genotoxicity screening during chalcone-based drug development.
Reproductive and developmental toxicity studies present another critical safety dimension. Current evidence suggests luteolin possesses minimal reproductive toxicity at therapeutic doses, though comprehensive multi-generational studies remain limited. Chalcones demonstrate greater variability, with certain derivatives showing embryotoxic effects in animal models at doses approaching therapeutic ranges, warranting caution in applications targeting reproductive-age populations.
Long-term safety data remains a significant gap for both compounds. While acute and subchronic toxicity profiles are reasonably well-characterized, chronic exposure studies exceeding 12 months are notably scarce. This represents a critical research priority, particularly as these compounds gain interest for chronic disease management applications requiring extended administration periods.
Clinical investigations have documented mild adverse events associated with luteolin supplementation, primarily including gastrointestinal disturbances such as nausea, abdominal discomfort, and occasional diarrhea. These effects typically manifest at doses exceeding 100 mg/day and resolve upon dose reduction or discontinuation. Notably, luteolin demonstrates minimal interaction with major cytochrome P450 enzymes, reducing the likelihood of significant drug-drug interactions when administered concurrently with conventional pharmaceuticals.
Chalcones present a more complex toxicity profile with significant structural-dependent variations. While certain chalcone derivatives demonstrate negligible toxicity in preclinical models, others exhibit concerning hepatotoxic and nephrotoxic effects at concentrations approaching therapeutic levels. Structure-activity relationship studies indicate that chalcones containing specific functional groups, particularly those with halogen substitutions at positions 2' and 4', may present elevated toxicity concerns requiring careful structural optimization during drug development.
Genotoxicity assessments for both compounds reveal important distinctions. Luteolin has consistently demonstrated negative results in Ames tests and chromosomal aberration assays, supporting its genetic safety. Conversely, select chalcone derivatives have shown positive mutagenic potential in bacterial reverse mutation assays, particularly those containing electrophilic moieties capable of DNA adduct formation. This necessitates comprehensive genotoxicity screening during chalcone-based drug development.
Reproductive and developmental toxicity studies present another critical safety dimension. Current evidence suggests luteolin possesses minimal reproductive toxicity at therapeutic doses, though comprehensive multi-generational studies remain limited. Chalcones demonstrate greater variability, with certain derivatives showing embryotoxic effects in animal models at doses approaching therapeutic ranges, warranting caution in applications targeting reproductive-age populations.
Long-term safety data remains a significant gap for both compounds. While acute and subchronic toxicity profiles are reasonably well-characterized, chronic exposure studies exceeding 12 months are notably scarce. This represents a critical research priority, particularly as these compounds gain interest for chronic disease management applications requiring extended administration periods.
Regulatory Pathway for Flavonoid-based Drug Development
The regulatory landscape for flavonoid-based drug development presents a complex pathway that requires careful navigation through various international regulatory frameworks. For luteolin and chalcone derivatives, developers must first determine the intended regulatory classification - whether as a pharmaceutical drug, dietary supplement, or functional food ingredient - as this fundamentally shapes the approval process.
In the United States, the FDA's Center for Drug Evaluation and Research (CDER) oversees pharmaceutical applications for flavonoid compounds. For luteolin and chalcone as therapeutic agents, developers must submit an Investigational New Drug (IND) application before proceeding to clinical trials. This requires comprehensive preclinical data demonstrating safety profiles and potential efficacy mechanisms, with particular attention to the unique pharmacokinetic properties of these flavonoids.
The European Medicines Agency (EMA) employs a somewhat different approach, offering scientific advice consultations earlier in the development process. For flavonoid-based therapeutics, the EMA's Committee for Medicinal Products for Human Use (CHMP) provides specific guidelines regarding required toxicology studies and bioavailability assessments, recognizing the challenges in demonstrating consistent bioavailability of plant-derived compounds like luteolin and chalcone.
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has established specialized pathways for natural product-derived therapeutics, potentially offering accelerated review processes for flavonoid compounds with substantial traditional use documentation. This may present strategic advantages for luteolin-based therapies with historical usage in Asian traditional medicine systems.
Regulatory bodies increasingly recognize the need for specialized approaches to botanical-derived compounds. The FDA's Botanical Drug Development Guidance provides specific considerations for plant-derived therapeutics, addressing issues particularly relevant to flavonoid compounds like extraction standardization, chemical characterization requirements, and batch-to-batch consistency demonstrations.
Clinical trial design for flavonoid-based therapeutics presents unique regulatory challenges. Authorities typically require robust bioanalytical methods for detecting both parent compounds and metabolites, with validation of these methods being a critical regulatory hurdle. For luteolin and chalcone specifically, their extensive metabolism and relatively low bioavailability necessitate specialized pharmacokinetic study designs to satisfy regulatory requirements.
Intellectual property protection strategies must be carefully integrated with regulatory pathways. While natural compounds themselves may face patentability challenges, novel formulations enhancing bioavailability, specific synthetic derivatives, or unique therapeutic applications of luteolin or chalcone may secure stronger patent protection, thereby supporting the commercial viability of the regulatory investment.
In the United States, the FDA's Center for Drug Evaluation and Research (CDER) oversees pharmaceutical applications for flavonoid compounds. For luteolin and chalcone as therapeutic agents, developers must submit an Investigational New Drug (IND) application before proceeding to clinical trials. This requires comprehensive preclinical data demonstrating safety profiles and potential efficacy mechanisms, with particular attention to the unique pharmacokinetic properties of these flavonoids.
The European Medicines Agency (EMA) employs a somewhat different approach, offering scientific advice consultations earlier in the development process. For flavonoid-based therapeutics, the EMA's Committee for Medicinal Products for Human Use (CHMP) provides specific guidelines regarding required toxicology studies and bioavailability assessments, recognizing the challenges in demonstrating consistent bioavailability of plant-derived compounds like luteolin and chalcone.
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has established specialized pathways for natural product-derived therapeutics, potentially offering accelerated review processes for flavonoid compounds with substantial traditional use documentation. This may present strategic advantages for luteolin-based therapies with historical usage in Asian traditional medicine systems.
Regulatory bodies increasingly recognize the need for specialized approaches to botanical-derived compounds. The FDA's Botanical Drug Development Guidance provides specific considerations for plant-derived therapeutics, addressing issues particularly relevant to flavonoid compounds like extraction standardization, chemical characterization requirements, and batch-to-batch consistency demonstrations.
Clinical trial design for flavonoid-based therapeutics presents unique regulatory challenges. Authorities typically require robust bioanalytical methods for detecting both parent compounds and metabolites, with validation of these methods being a critical regulatory hurdle. For luteolin and chalcone specifically, their extensive metabolism and relatively low bioavailability necessitate specialized pharmacokinetic study designs to satisfy regulatory requirements.
Intellectual property protection strategies must be carefully integrated with regulatory pathways. While natural compounds themselves may face patentability challenges, novel formulations enhancing bioavailability, specific synthetic derivatives, or unique therapeutic applications of luteolin or chalcone may secure stronger patent protection, thereby supporting the commercial viability of the regulatory investment.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!