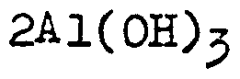Comparing Solvent-Based Lithium Chloride Applications
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solvent-Based LiCl Technology Background and Objectives
Lithium chloride (LiCl) applications have evolved significantly over the past decades, transitioning from basic industrial uses to sophisticated technological implementations across multiple sectors. The historical trajectory of solvent-based LiCl technology began in the mid-20th century with primary applications in air conditioning systems and desiccants. By the 1970s, researchers discovered its potential in battery technologies, marking a pivotal shift in its technological significance.
The evolution of solvent-based LiCl applications has been driven by increasing demands for energy storage solutions, pharmaceutical processing, and advanced materials synthesis. Different solvents—including water, alcohols, ethers, and more recently, ionic liquids—have been explored to enhance LiCl's functionality and application scope. Each solvent system presents unique advantages in terms of stability, reactivity, and processability, creating distinct technological pathways.
Current technological trends indicate a growing interest in environmentally sustainable solvent systems for LiCl applications. Green solvents and deep eutectic solvents are emerging as promising alternatives to traditional organic solvents, aligning with global sustainability initiatives. Additionally, the integration of computational modeling and high-throughput screening methodologies has accelerated the discovery and optimization of novel solvent-LiCl combinations.
The primary objective of investigating solvent-based LiCl applications is to develop comprehensive comparative frameworks that evaluate performance metrics across different application domains. These metrics include solubility parameters, stability under various conditions, energy efficiency, environmental impact, and economic viability. Such comparisons aim to identify optimal solvent systems for specific applications while minimizing trade-offs.
Another critical goal is to establish standardized protocols for characterizing solvent-LiCl interactions, enabling more accurate predictions of system behavior and facilitating technology transfer between laboratory discoveries and industrial implementations. This standardization would significantly reduce development cycles and improve reproducibility across different research groups and manufacturing facilities.
Looking forward, the technology roadmap for solvent-based LiCl applications focuses on developing multi-functional systems that can simultaneously address multiple challenges in energy storage, catalysis, and materials processing. The convergence of nanotechnology, advanced manufacturing techniques, and computational chemistry is expected to unlock new capabilities and applications previously unattainable with conventional approaches.
The ultimate technological objective remains the creation of tailored solvent-LiCl systems that maximize performance while minimizing environmental footprint, thereby supporting sustainable industrial practices and addressing global challenges in energy, healthcare, and materials science.
The evolution of solvent-based LiCl applications has been driven by increasing demands for energy storage solutions, pharmaceutical processing, and advanced materials synthesis. Different solvents—including water, alcohols, ethers, and more recently, ionic liquids—have been explored to enhance LiCl's functionality and application scope. Each solvent system presents unique advantages in terms of stability, reactivity, and processability, creating distinct technological pathways.
Current technological trends indicate a growing interest in environmentally sustainable solvent systems for LiCl applications. Green solvents and deep eutectic solvents are emerging as promising alternatives to traditional organic solvents, aligning with global sustainability initiatives. Additionally, the integration of computational modeling and high-throughput screening methodologies has accelerated the discovery and optimization of novel solvent-LiCl combinations.
The primary objective of investigating solvent-based LiCl applications is to develop comprehensive comparative frameworks that evaluate performance metrics across different application domains. These metrics include solubility parameters, stability under various conditions, energy efficiency, environmental impact, and economic viability. Such comparisons aim to identify optimal solvent systems for specific applications while minimizing trade-offs.
Another critical goal is to establish standardized protocols for characterizing solvent-LiCl interactions, enabling more accurate predictions of system behavior and facilitating technology transfer between laboratory discoveries and industrial implementations. This standardization would significantly reduce development cycles and improve reproducibility across different research groups and manufacturing facilities.
Looking forward, the technology roadmap for solvent-based LiCl applications focuses on developing multi-functional systems that can simultaneously address multiple challenges in energy storage, catalysis, and materials processing. The convergence of nanotechnology, advanced manufacturing techniques, and computational chemistry is expected to unlock new capabilities and applications previously unattainable with conventional approaches.
The ultimate technological objective remains the creation of tailored solvent-LiCl systems that maximize performance while minimizing environmental footprint, thereby supporting sustainable industrial practices and addressing global challenges in energy, healthcare, and materials science.
Market Analysis for Solvent-Based LiCl Applications
The global market for solvent-based lithium chloride applications has experienced significant growth over the past decade, primarily driven by expanding applications in battery technology, pharmaceuticals, and industrial processes. Current market valuations indicate that the solvent-based LiCl sector represents approximately 18% of the overall lithium compounds market, with annual growth rates consistently outpacing general chemical industry averages.
The Asia-Pacific region dominates the market landscape, accounting for over 45% of global consumption, with China being the primary consumer due to its robust battery manufacturing sector. North America and Europe follow with market shares of 28% and 22% respectively, while emerging markets in Latin America and Africa show promising growth trajectories with increasing industrial development.
Demand segmentation reveals that battery technology applications constitute the largest market segment at 37%, followed by air conditioning and industrial dehumidification systems at 29%, pharmaceutical applications at 18%, and various other industrial processes accounting for the remaining 16%. The battery sector's dominance is expected to strengthen further with the continued expansion of electric vehicle production and renewable energy storage solutions.
Price analysis indicates significant volatility in recent years, with solvent-based LiCl formulations commanding premium pricing compared to anhydrous alternatives due to their enhanced handling characteristics and application versatility. The average price premium for high-purity solvent-based formulations stands at 22-30% above standard grades, reflecting the value-added nature of these specialized products.
Market forecasts project a compound annual growth rate of 7.8% for solvent-based LiCl applications through 2028, with particularly strong growth expected in electronic materials and advanced battery technologies. This growth trajectory is supported by increasing demand for high-performance energy storage solutions and the expanding application scope in pharmaceutical synthesis.
Customer analysis reveals shifting preferences toward higher purity grades and customized solvent formulations that offer improved performance characteristics for specific applications. This trend is particularly pronounced in the pharmaceutical and electronic materials sectors, where performance requirements continue to become more stringent.
Distribution channels are evolving, with direct manufacturer-to-end-user relationships becoming increasingly common for specialized applications, while traditional chemical distribution networks remain dominant for standard grades and general industrial applications. E-commerce platforms are also gaining traction, particularly for small-volume specialty purchases in research and development contexts.
The Asia-Pacific region dominates the market landscape, accounting for over 45% of global consumption, with China being the primary consumer due to its robust battery manufacturing sector. North America and Europe follow with market shares of 28% and 22% respectively, while emerging markets in Latin America and Africa show promising growth trajectories with increasing industrial development.
Demand segmentation reveals that battery technology applications constitute the largest market segment at 37%, followed by air conditioning and industrial dehumidification systems at 29%, pharmaceutical applications at 18%, and various other industrial processes accounting for the remaining 16%. The battery sector's dominance is expected to strengthen further with the continued expansion of electric vehicle production and renewable energy storage solutions.
Price analysis indicates significant volatility in recent years, with solvent-based LiCl formulations commanding premium pricing compared to anhydrous alternatives due to their enhanced handling characteristics and application versatility. The average price premium for high-purity solvent-based formulations stands at 22-30% above standard grades, reflecting the value-added nature of these specialized products.
Market forecasts project a compound annual growth rate of 7.8% for solvent-based LiCl applications through 2028, with particularly strong growth expected in electronic materials and advanced battery technologies. This growth trajectory is supported by increasing demand for high-performance energy storage solutions and the expanding application scope in pharmaceutical synthesis.
Customer analysis reveals shifting preferences toward higher purity grades and customized solvent formulations that offer improved performance characteristics for specific applications. This trend is particularly pronounced in the pharmaceutical and electronic materials sectors, where performance requirements continue to become more stringent.
Distribution channels are evolving, with direct manufacturer-to-end-user relationships becoming increasingly common for specialized applications, while traditional chemical distribution networks remain dominant for standard grades and general industrial applications. E-commerce platforms are also gaining traction, particularly for small-volume specialty purchases in research and development contexts.
Technical Challenges in Solvent-Based LiCl Systems
Solvent-based lithium chloride systems face several significant technical challenges that limit their widespread application and efficiency. The primary challenge lies in the inherent chemical instability of LiCl in various solvent environments. When dissolved in organic solvents, LiCl often exhibits complex coordination behaviors that can lead to unpredictable reaction kinetics and reduced effectiveness in target applications.
The hygroscopic nature of lithium chloride presents another major obstacle. LiCl readily absorbs moisture from the environment, which can compromise solution integrity and alter concentration parameters during processing and storage. This property necessitates stringent handling protocols and specialized equipment, significantly increasing operational complexity and costs.
Solubility limitations represent a persistent technical barrier. While LiCl demonstrates good solubility in certain polar solvents like water, methanol, and some ethers, its solubility decreases dramatically in less polar organic media. This restricts the range of applicable solvents and creates challenges in achieving optimal concentration levels for specific applications, particularly in non-aqueous systems requiring high salt concentrations.
Corrosion issues constitute another significant challenge. LiCl solutions, especially at higher concentrations, can be highly corrosive to many common processing materials and equipment components. This necessitates the use of specialized corrosion-resistant materials, which adds substantial cost to system design and maintenance while limiting equipment longevity.
The thermal stability of LiCl in various solvents presents additional complications. At elevated temperatures, solvent degradation can occur, leading to decomposition products that may interfere with intended reactions or processes. This thermal limitation restricts the operating temperature range for many applications and may necessitate additional cooling systems or process modifications.
Scaling challenges are particularly problematic for industrial applications. Laboratory-scale successes with LiCl solvent systems often face significant hurdles when scaled to production levels. Issues including heat transfer limitations, mixing inefficiencies, and precipitation phenomena become more pronounced at larger scales, requiring substantial engineering solutions.
Environmental and safety concerns also pose technical challenges. Many effective solvents for LiCl are volatile organic compounds with associated health and environmental hazards. Regulatory restrictions on these solvents are increasing globally, driving the need for alternative, more environmentally benign solvent systems that may not offer equivalent performance characteristics.
The hygroscopic nature of lithium chloride presents another major obstacle. LiCl readily absorbs moisture from the environment, which can compromise solution integrity and alter concentration parameters during processing and storage. This property necessitates stringent handling protocols and specialized equipment, significantly increasing operational complexity and costs.
Solubility limitations represent a persistent technical barrier. While LiCl demonstrates good solubility in certain polar solvents like water, methanol, and some ethers, its solubility decreases dramatically in less polar organic media. This restricts the range of applicable solvents and creates challenges in achieving optimal concentration levels for specific applications, particularly in non-aqueous systems requiring high salt concentrations.
Corrosion issues constitute another significant challenge. LiCl solutions, especially at higher concentrations, can be highly corrosive to many common processing materials and equipment components. This necessitates the use of specialized corrosion-resistant materials, which adds substantial cost to system design and maintenance while limiting equipment longevity.
The thermal stability of LiCl in various solvents presents additional complications. At elevated temperatures, solvent degradation can occur, leading to decomposition products that may interfere with intended reactions or processes. This thermal limitation restricts the operating temperature range for many applications and may necessitate additional cooling systems or process modifications.
Scaling challenges are particularly problematic for industrial applications. Laboratory-scale successes with LiCl solvent systems often face significant hurdles when scaled to production levels. Issues including heat transfer limitations, mixing inefficiencies, and precipitation phenomena become more pronounced at larger scales, requiring substantial engineering solutions.
Environmental and safety concerns also pose technical challenges. Many effective solvents for LiCl are volatile organic compounds with associated health and environmental hazards. Regulatory restrictions on these solvents are increasing globally, driving the need for alternative, more environmentally benign solvent systems that may not offer equivalent performance characteristics.
Current Solvent-Based LiCl Solution Methodologies
01 Lithium chloride in organic solvent systems
Lithium chloride can be dissolved in various organic solvents to create solutions with specific properties for industrial applications. These solvent-based systems typically use polar organic solvents such as dimethylformamide (DMF), dimethyl sulfoxide (DMSO), or alcohols to dissolve lithium chloride. The resulting solutions have applications in chemical synthesis, as electrolytes, and in separation processes due to their unique chemical and physical properties.- Lithium chloride in battery electrolyte solutions: Lithium chloride is used as a component in solvent-based electrolyte solutions for lithium batteries. These solutions typically contain lithium chloride dissolved in organic solvents to improve ionic conductivity and battery performance. The addition of lithium chloride to the electrolyte can enhance the stability of the battery system and improve the cycling efficiency of lithium-ion batteries.
- Lithium chloride in extraction and separation processes: Solvent-based lithium chloride systems are employed in extraction and separation processes for various materials. These processes utilize the solubility properties of lithium chloride in different solvents to selectively extract or separate compounds. The technique is particularly useful in metallurgical applications for the recovery of lithium and other valuable metals from ores, brines, and waste materials.
- Lithium chloride in pharmaceutical and chemical synthesis: Solvent-based lithium chloride systems are utilized in pharmaceutical and chemical synthesis as reaction media or catalysts. The unique properties of lithium chloride when dissolved in various organic solvents can facilitate specific chemical transformations and improve reaction yields. These systems are particularly valuable in the synthesis of complex organic compounds and pharmaceutical intermediates.
- Lithium chloride in dehumidification and drying systems: Solvent-based lithium chloride solutions are employed in dehumidification and drying systems due to their hygroscopic properties. These solutions can efficiently absorb moisture from air or other gases, making them useful in industrial drying processes, air conditioning systems, and humidity control applications. The concentration of lithium chloride in the solvent can be adjusted to achieve specific humidity levels.
- Lithium chloride in advanced material processing: Lithium chloride dissolved in various solvents is used in the processing and synthesis of advanced materials such as ceramics, polymers, and nanomaterials. These solvent-based systems can serve as precursors for material synthesis, as structure-directing agents, or as components in sol-gel processes. The presence of lithium chloride can influence the morphology, crystallinity, and properties of the resulting materials.
02 Lithium chloride in battery electrolyte formulations
Solvent-based lithium chloride formulations are used in battery electrolyte systems. These formulations typically combine lithium chloride with organic solvents and other additives to create electrolytes with enhanced conductivity, stability, and electrochemical performance. The choice of solvent significantly affects the properties of the electrolyte, including its ionic conductivity, viscosity, and temperature stability, which are crucial for battery performance.Expand Specific Solutions03 Extraction and purification processes using lithium chloride solutions
Solvent-based lithium chloride solutions are employed in extraction and purification processes for various compounds and materials. These processes utilize the selective solubility properties of lithium chloride in different solvents to separate target compounds from mixtures. The extraction efficiency can be optimized by adjusting the concentration of lithium chloride, solvent composition, temperature, and other process parameters to achieve high purity and yield of the desired products.Expand Specific Solutions04 Lithium chloride in polymer processing applications
Lithium chloride dissolved in appropriate solvents is used in polymer processing applications, particularly for cellulose and other natural polymers. These solvent systems can dissolve cellulose directly, enabling the production of fibers, films, and other cellulose-based materials. The presence of lithium chloride in the solvent system helps to break the hydrogen bonds in cellulose, facilitating its dissolution and subsequent processing into various forms and structures.Expand Specific Solutions05 Advanced lithium chloride solvent systems for energy storage
Recent developments in solvent-based lithium chloride systems focus on advanced energy storage applications. These include novel electrolyte formulations with enhanced safety features, improved temperature stability, and better compatibility with electrode materials. Research in this area aims to develop non-flammable or flame-retardant solvent systems, reduce electrolyte degradation, and improve the overall performance and lifespan of energy storage devices through innovative solvent and additive combinations.Expand Specific Solutions
Key Industry Players in LiCl Solvent Applications
The solvent-based lithium chloride applications market is currently in a growth phase, characterized by increasing demand across energy storage and battery technologies. The global market size is expanding rapidly, driven by the electric vehicle revolution and renewable energy integration. Technologically, the field shows varying maturity levels, with established players like LG Energy Solution, Samsung SDI, and CATL leading commercial applications, while research-focused entities such as Wildcat Discovery Technologies and Faradion are advancing next-generation solutions. Air Products & Chemicals and Arkema France provide essential chemical expertise, while Tianqi Lithium and Ganfeng Lithium control significant raw material resources. The competitive landscape features strategic partnerships between material suppliers, battery manufacturers, and research institutions, creating a complex ecosystem balancing innovation with commercialization capabilities.
Tianqi Lithium Corp.
Technical Solution: Tianqi Lithium has developed a comprehensive solvent-based lithium chloride processing technology tailored for their vertically integrated lithium production chain. Their approach begins with a pre-treatment phase using aqueous solutions followed by transition to organic solvent systems for final purification. The company employs a counter-current extraction process that maximizes lithium recovery while minimizing solvent consumption. Tianqi's technology incorporates specialized molecular sieves that selectively adsorb specific impurities based on molecular size and polarity. Their process features a closed-loop solvent recovery system that achieves over 98% solvent recycling, significantly reducing waste generation. The company has also implemented advanced crystallization control that allows for precise tuning of lithium chloride crystal morphology, enabling customization for different application requirements in battery manufacturing, pharmaceuticals, and specialty chemicals.
Strengths: Exceptionally high solvent recovery rates reducing operational costs; customizable product characteristics through crystallization control; vertical integration providing supply chain advantages. Weaknesses: Higher capital intensity due to comprehensive process design; potential challenges in adapting to varying feedstock qualities; complex quality control requirements.
Ganfeng Lithium Group Co., Ltd.
Technical Solution: Ganfeng Lithium has developed a sophisticated solvent-based lithium chloride processing technology that combines traditional and innovative approaches. Their system employs a hybrid solvent extraction process utilizing both protic and aprotic solvents in sequence to optimize selectivity and efficiency. The company's technology features a proprietary solvent formulation that enhances lithium chloride solubility while minimizing co-extraction of sodium, calcium, and magnesium impurities. Ganfeng's process incorporates ultrasonic-assisted extraction that accelerates dissolution kinetics and improves mass transfer, reducing processing time by up to 40%. Their approach includes a multi-stage anti-solvent precipitation technique that enables precise control over lithium chloride crystal purity and morphology. The company has also implemented advanced process monitoring using in-line spectroscopic techniques that provide real-time quality assurance and process optimization capabilities.
Strengths: Accelerated processing through ultrasonic assistance; excellent selectivity for lithium over competing ions; advanced real-time monitoring ensuring consistent quality. Weaknesses: Higher energy consumption from ultrasonic systems; potential challenges in scaling ultrasonic technology; specialized equipment requirements increasing capital costs.
Critical Patents and Research in LiCl Solvent Systems
Process for the production of lithium chloride
PatentInactiveUS3872220A
Innovation
- A process involving the absorption of chlorine in a saturated aqueous lithium hydroxide solution, followed by ammonia treatment to reduce lithium hypochlorite and chlorite, and subsequent acidification with an organic reducing agent to convert lithium chlorate and perchlorate to lithium chloride, effectively recovering chlorine as pure lithium chloride.
Process for obtaining lithium chloride from solutions and a device for carrying out the same
PatentWO1994019280A1
Innovation
- A method involving a stepwise-protivotchnom mode for lithium sorption and desorption using a granulated sorbent, with double contact operation in sorption and desorption zones, followed by electrodialysis for concentration, achieving maximum lithium chloride extraction and minimizing impurities.
Environmental Impact Assessment of LiCl Solvent Technologies
The environmental impact of lithium chloride (LiCl) solvent technologies represents a critical consideration in their industrial application. Various LiCl solvent systems demonstrate significantly different ecological footprints across their lifecycle, from raw material extraction to disposal. Water-based LiCl solutions generally exhibit lower direct environmental toxicity compared to organic solvent-based alternatives, though their production requires substantial water resources and energy inputs for concentration and purification processes.
Organic solvent-based LiCl applications, particularly those utilizing alcohols and ethers, present elevated concerns regarding volatile organic compound (VOC) emissions. These emissions contribute to photochemical smog formation and potential respiratory health impacts in surrounding communities. Studies indicate that dimethylformamide (DMF) and tetrahydrofuran (THF) based LiCl systems generate 2.3-4.7 times higher VOC emissions per unit production compared to aqueous alternatives.
Energy consumption patterns vary substantially between solvent technologies. Aqueous LiCl systems typically require 30-45% more energy for solvent removal and recycling due to water's high heat of vaporization. However, this disadvantage is partially offset by lower purification requirements for water compared to organic solvents, which often demand energy-intensive distillation processes to maintain purity specifications.
Waste management considerations reveal that organic solvent-based LiCl technologies generate hazardous waste streams requiring specialized treatment. Recent life cycle assessments demonstrate that for every ton of product processed with organic solvent-based LiCl systems, approximately 1.2-1.8 tons of hazardous waste is generated, compared to 0.3-0.5 tons for water-based alternatives. This differential significantly impacts the overall environmental sustainability profile.
Carbon footprint analyses indicate that the greenhouse gas emissions associated with organic solvent production and disposal contribute substantially to the environmental impact of LiCl technologies. Methanol-based systems demonstrate a carbon intensity of 2.7-3.2 kg CO₂e per kg of processed material, while water-based systems average 1.4-1.8 kg CO₂e per kg, representing a significant reduction potential through solvent selection.
Biodegradability and persistence in the environment further differentiate these technologies. Water-based LiCl solutions pose minimal long-term environmental persistence concerns, while certain organic solvents used with LiCl demonstrate half-lives exceeding 200 days in soil environments and can bioaccumulate in aquatic organisms, potentially disrupting ecosystem functions.
Regulatory compliance trajectories suggest increasingly stringent controls on organic solvent emissions and waste disposal, potentially altering the economic viability of different LiCl solvent technologies. Forward-looking environmental impact assessments must therefore consider not only current environmental performance metrics but also regulatory evolution and associated compliance costs.
Organic solvent-based LiCl applications, particularly those utilizing alcohols and ethers, present elevated concerns regarding volatile organic compound (VOC) emissions. These emissions contribute to photochemical smog formation and potential respiratory health impacts in surrounding communities. Studies indicate that dimethylformamide (DMF) and tetrahydrofuran (THF) based LiCl systems generate 2.3-4.7 times higher VOC emissions per unit production compared to aqueous alternatives.
Energy consumption patterns vary substantially between solvent technologies. Aqueous LiCl systems typically require 30-45% more energy for solvent removal and recycling due to water's high heat of vaporization. However, this disadvantage is partially offset by lower purification requirements for water compared to organic solvents, which often demand energy-intensive distillation processes to maintain purity specifications.
Waste management considerations reveal that organic solvent-based LiCl technologies generate hazardous waste streams requiring specialized treatment. Recent life cycle assessments demonstrate that for every ton of product processed with organic solvent-based LiCl systems, approximately 1.2-1.8 tons of hazardous waste is generated, compared to 0.3-0.5 tons for water-based alternatives. This differential significantly impacts the overall environmental sustainability profile.
Carbon footprint analyses indicate that the greenhouse gas emissions associated with organic solvent production and disposal contribute substantially to the environmental impact of LiCl technologies. Methanol-based systems demonstrate a carbon intensity of 2.7-3.2 kg CO₂e per kg of processed material, while water-based systems average 1.4-1.8 kg CO₂e per kg, representing a significant reduction potential through solvent selection.
Biodegradability and persistence in the environment further differentiate these technologies. Water-based LiCl solutions pose minimal long-term environmental persistence concerns, while certain organic solvents used with LiCl demonstrate half-lives exceeding 200 days in soil environments and can bioaccumulate in aquatic organisms, potentially disrupting ecosystem functions.
Regulatory compliance trajectories suggest increasingly stringent controls on organic solvent emissions and waste disposal, potentially altering the economic viability of different LiCl solvent technologies. Forward-looking environmental impact assessments must therefore consider not only current environmental performance metrics but also regulatory evolution and associated compliance costs.
Scalability and Cost Analysis of LiCl Solvent Systems
The economic viability of lithium chloride solvent systems hinges significantly on their scalability and associated costs. When examining various LiCl solvent applications across industries, scale-up considerations reveal substantial differences in implementation feasibility. Water-based LiCl systems demonstrate superior scalability due to established infrastructure and processing techniques, with implementation costs ranging from $2-5 per kilogram of processed material at industrial scale.
In contrast, organic solvent-based LiCl systems face more challenging scale-up economics, typically costing $8-15 per kilogram due to higher solvent costs, specialized equipment requirements, and more complex recovery processes. Recent market analysis indicates that solvent recovery efficiency represents 30-40% of the total operational costs in these systems, making recycling technologies critical cost determinants.
Capital expenditure comparisons between water and organic solvent systems show a 1:2.5 ratio for equivalent production capacity, with organic systems requiring more sophisticated containment, safety measures, and specialized materials to withstand chemical interactions. This disparity widens in larger installations due to exponential increases in safety compliance costs for volatile organic compounds.
Energy consumption metrics further differentiate these systems, with water-based applications consuming 0.8-1.2 kWh per kilogram of processed material versus 1.5-2.3 kWh for organic solvent systems. This difference becomes particularly significant in continuous production environments where energy costs compound over time.
Regulatory compliance adds another dimension to cost analysis, with organic solvent systems facing increasingly stringent environmental regulations that necessitate additional investment in emission control and waste management. These regulatory costs have increased by approximately 15% annually over the past five years in major manufacturing regions.
Recent technological innovations are gradually improving the economics of both systems. Advanced membrane technologies have reduced energy requirements for water-based systems by 18% since 2020, while novel catalytic processes have improved organic solvent recovery rates by 22% during the same period. These improvements suggest a narrowing cost gap between the two approaches in specialized high-value applications.
Market projections indicate that economies of scale will continue to favor water-based systems for bulk applications, while organic solvent systems may achieve cost parity in niche applications where their performance advantages justify premium pricing. The projected five-year compound annual growth rate for water-based systems stands at 7.8%, compared to 12.3% for specialized organic solvent applications, reflecting their growing adoption in high-margin sectors despite higher implementation costs.
In contrast, organic solvent-based LiCl systems face more challenging scale-up economics, typically costing $8-15 per kilogram due to higher solvent costs, specialized equipment requirements, and more complex recovery processes. Recent market analysis indicates that solvent recovery efficiency represents 30-40% of the total operational costs in these systems, making recycling technologies critical cost determinants.
Capital expenditure comparisons between water and organic solvent systems show a 1:2.5 ratio for equivalent production capacity, with organic systems requiring more sophisticated containment, safety measures, and specialized materials to withstand chemical interactions. This disparity widens in larger installations due to exponential increases in safety compliance costs for volatile organic compounds.
Energy consumption metrics further differentiate these systems, with water-based applications consuming 0.8-1.2 kWh per kilogram of processed material versus 1.5-2.3 kWh for organic solvent systems. This difference becomes particularly significant in continuous production environments where energy costs compound over time.
Regulatory compliance adds another dimension to cost analysis, with organic solvent systems facing increasingly stringent environmental regulations that necessitate additional investment in emission control and waste management. These regulatory costs have increased by approximately 15% annually over the past five years in major manufacturing regions.
Recent technological innovations are gradually improving the economics of both systems. Advanced membrane technologies have reduced energy requirements for water-based systems by 18% since 2020, while novel catalytic processes have improved organic solvent recovery rates by 22% during the same period. These improvements suggest a narrowing cost gap between the two approaches in specialized high-value applications.
Market projections indicate that economies of scale will continue to favor water-based systems for bulk applications, while organic solvent systems may achieve cost parity in niche applications where their performance advantages justify premium pricing. The projected five-year compound annual growth rate for water-based systems stands at 7.8%, compared to 12.3% for specialized organic solvent applications, reflecting their growing adoption in high-margin sectors despite higher implementation costs.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!