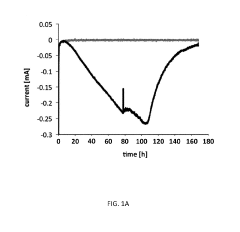
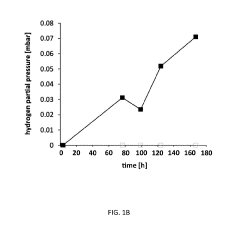
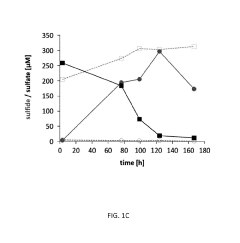
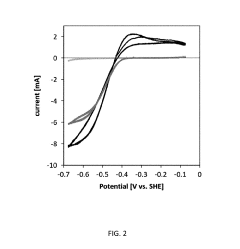
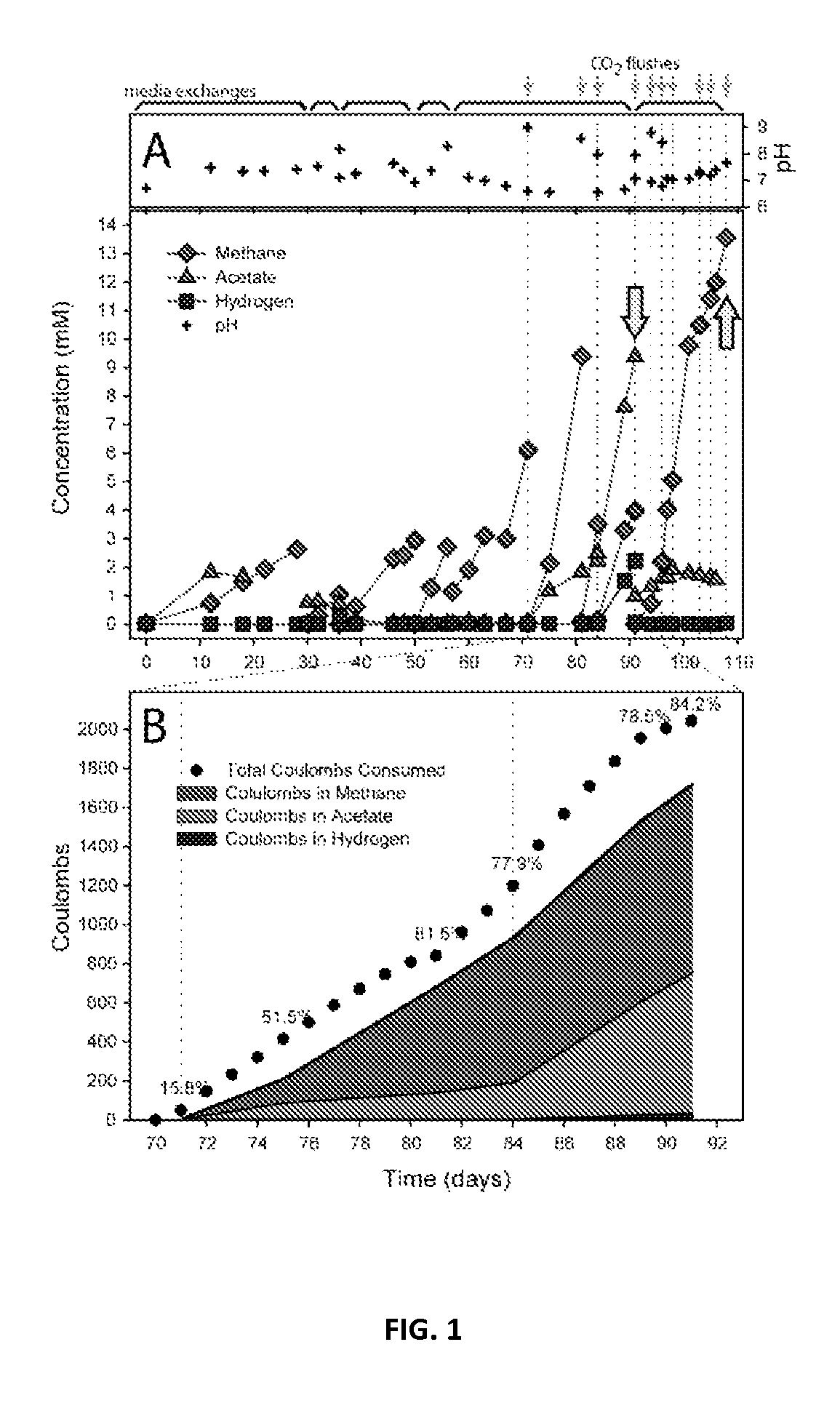
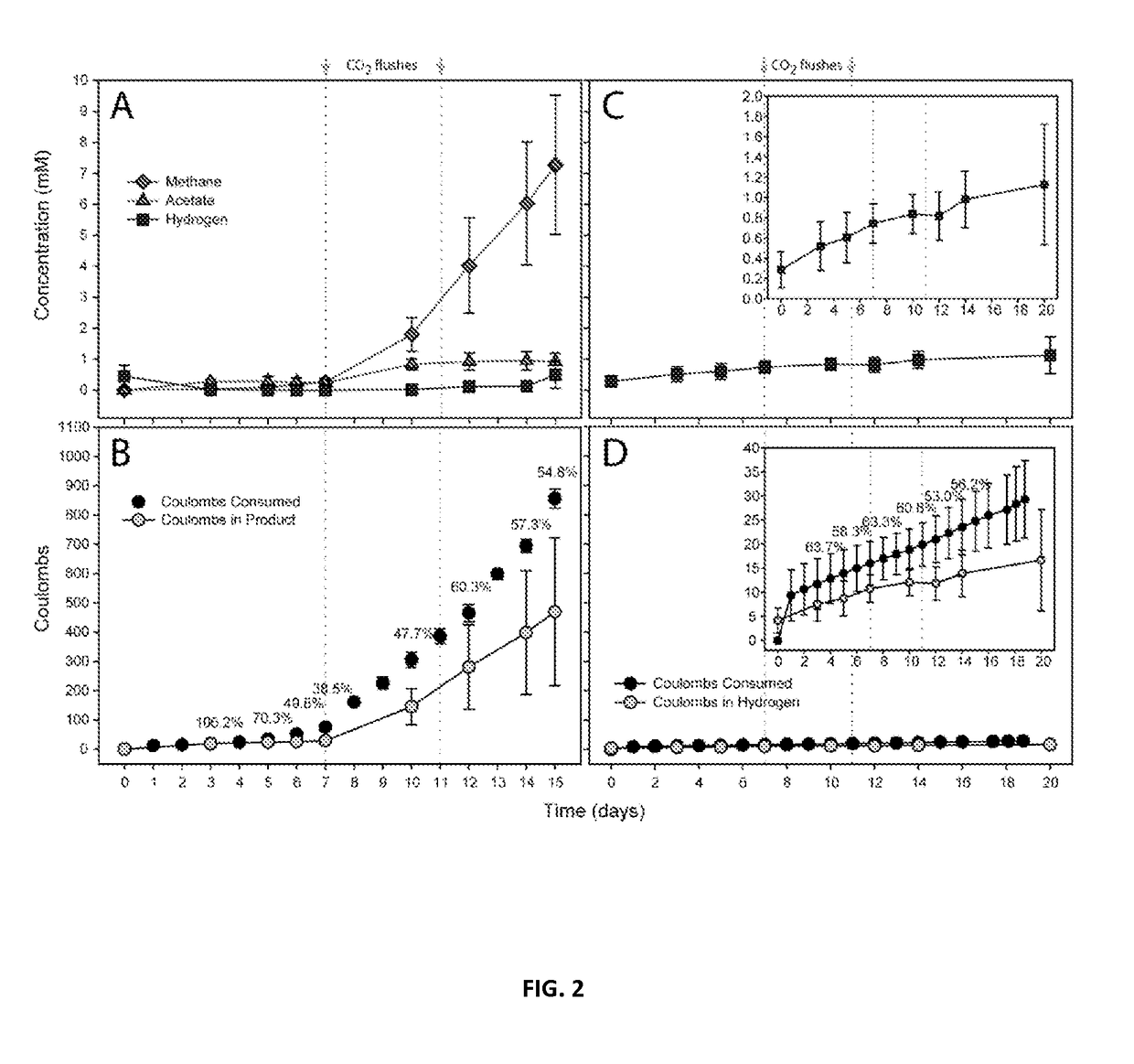
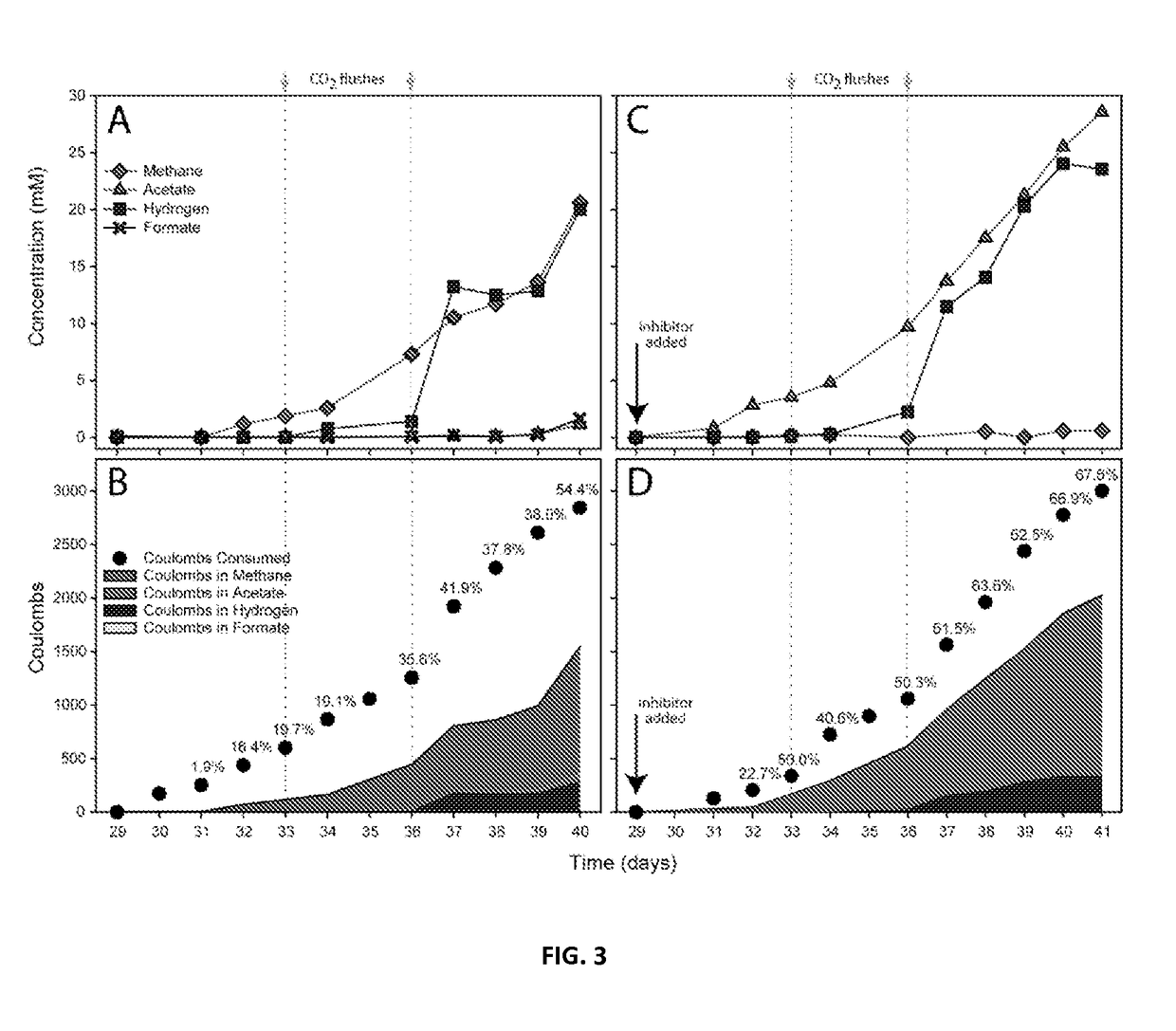
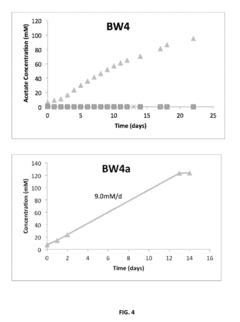
Electron Balance Analysis In Microbial Electrosynthesis
SEP 4, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Microbial Electrosynthesis Background and Objectives
Microbial Electrosynthesis (MES) represents a groundbreaking biotechnological approach that has evolved significantly over the past decade. This innovative technology harnesses the metabolic capabilities of microorganisms to convert electrical energy into valuable chemical compounds, effectively bridging the gap between renewable electricity and sustainable chemical production. The concept emerged from the broader field of bioelectrochemical systems, gaining momentum as researchers recognized its potential for carbon capture and utilization.
The evolution of MES technology can be traced back to early studies on microbial fuel cells, which demonstrated that certain microorganisms could interact with electrodes. This discovery led to the reverse application - using electricity to drive microbial metabolism toward the production of desired compounds. Since its conceptualization in the early 2000s, MES has progressed from proof-of-concept laboratory experiments to more sophisticated systems with improved efficiency and product specificity.
Current technological trends in MES focus on enhancing electron transfer mechanisms between electrodes and microorganisms, optimizing reactor designs, and developing more robust microbial catalysts. The field is witnessing a shift toward mixed culture approaches and the integration of synthetic biology tools to engineer more efficient electrosynthetic pathways. Additionally, there is growing interest in coupling MES with renewable energy sources to create truly sustainable bioprocesses.
The electron balance analysis in MES represents a critical aspect of this technology, as it provides insights into the efficiency of energy conversion from electricity to chemical bonds. Understanding electron flow and distribution is essential for optimizing the overall process and maximizing product yields. This analysis involves tracking electrons from their source (cathode) through various microbial metabolic pathways to final products.
The primary objectives of advancing electron balance analysis in MES include: developing more accurate methods for quantifying electron transfer rates; identifying and mitigating competing electron sinks; enhancing coulombic efficiency; and establishing standardized protocols for performance evaluation. These objectives align with the broader goal of transforming MES into a commercially viable technology for sustainable chemical production.
As climate change concerns intensify and renewable electricity becomes more abundant, MES offers a promising platform for carbon-neutral or even carbon-negative manufacturing of chemicals and fuels. The technology aims to eventually replace conventional petrochemical processes with bioelectrochemical alternatives that operate at ambient conditions with minimal environmental impact, potentially revolutionizing how we produce essential chemicals in a post-fossil fuel economy.
The evolution of MES technology can be traced back to early studies on microbial fuel cells, which demonstrated that certain microorganisms could interact with electrodes. This discovery led to the reverse application - using electricity to drive microbial metabolism toward the production of desired compounds. Since its conceptualization in the early 2000s, MES has progressed from proof-of-concept laboratory experiments to more sophisticated systems with improved efficiency and product specificity.
Current technological trends in MES focus on enhancing electron transfer mechanisms between electrodes and microorganisms, optimizing reactor designs, and developing more robust microbial catalysts. The field is witnessing a shift toward mixed culture approaches and the integration of synthetic biology tools to engineer more efficient electrosynthetic pathways. Additionally, there is growing interest in coupling MES with renewable energy sources to create truly sustainable bioprocesses.
The electron balance analysis in MES represents a critical aspect of this technology, as it provides insights into the efficiency of energy conversion from electricity to chemical bonds. Understanding electron flow and distribution is essential for optimizing the overall process and maximizing product yields. This analysis involves tracking electrons from their source (cathode) through various microbial metabolic pathways to final products.
The primary objectives of advancing electron balance analysis in MES include: developing more accurate methods for quantifying electron transfer rates; identifying and mitigating competing electron sinks; enhancing coulombic efficiency; and establishing standardized protocols for performance evaluation. These objectives align with the broader goal of transforming MES into a commercially viable technology for sustainable chemical production.
As climate change concerns intensify and renewable electricity becomes more abundant, MES offers a promising platform for carbon-neutral or even carbon-negative manufacturing of chemicals and fuels. The technology aims to eventually replace conventional petrochemical processes with bioelectrochemical alternatives that operate at ambient conditions with minimal environmental impact, potentially revolutionizing how we produce essential chemicals in a post-fossil fuel economy.
Market Applications and Demand Analysis
The market for Microbial Electrosynthesis (MES) technology is experiencing significant growth driven by increasing demand for sustainable production methods across multiple industries. The global push towards carbon neutrality has positioned MES as a promising platform technology for converting CO2 into value-added chemicals and fuels using renewable electricity. This creates a circular carbon economy that addresses both waste management and sustainable production challenges.
The chemical manufacturing sector represents the largest potential market for MES applications, particularly for producing platform chemicals such as acetate, ethanol, butyrate, and more complex organic compounds. Traditional petrochemical production methods face mounting pressure from environmental regulations and carbon pricing mechanisms, creating market pull for alternative production technologies like MES.
Biopharmaceutical companies have shown increasing interest in MES for producing high-value pharmaceutical precursors and specialty chemicals. The precision and selectivity offered by bioelectrochemical systems provide advantages over traditional fermentation processes, especially for compounds with complex stereochemistry requirements.
The renewable energy sector presents another significant market opportunity. As intermittent renewable energy sources like wind and solar expand globally, the need for energy storage solutions grows proportionally. MES systems can function as biological batteries, converting excess electricity into storable chemical compounds during peak production periods.
Agricultural and food industries are exploring MES for sustainable fertilizer production and food additives. The ability to fix atmospheric nitrogen or produce organic acids using electricity and microorganisms offers an alternative to energy-intensive conventional processes like the Haber-Bosch process.
Market analysis indicates that the waste management sector is increasingly adopting MES technologies for valorizing organic waste streams. Municipal wastewater treatment facilities and industrial bioprocessing plants can implement MES to simultaneously treat waste and generate valuable byproducts, creating new revenue streams from what was previously considered waste.
Geographically, North America and Europe currently lead in MES research and commercialization efforts, driven by strong environmental policies and substantial research funding. However, rapid industrialization in Asia-Pacific regions, particularly China and India, is creating emerging markets with significant growth potential for MES technologies as these countries seek to balance industrial expansion with environmental sustainability commitments.
The market barriers primarily revolve around scaling challenges, system efficiency, and production costs compared to conventional chemical synthesis methods. However, as electron balance analysis improves system performance and renewable electricity costs continue to decline, the economic viability of MES is expected to improve substantially over the next decade.
The chemical manufacturing sector represents the largest potential market for MES applications, particularly for producing platform chemicals such as acetate, ethanol, butyrate, and more complex organic compounds. Traditional petrochemical production methods face mounting pressure from environmental regulations and carbon pricing mechanisms, creating market pull for alternative production technologies like MES.
Biopharmaceutical companies have shown increasing interest in MES for producing high-value pharmaceutical precursors and specialty chemicals. The precision and selectivity offered by bioelectrochemical systems provide advantages over traditional fermentation processes, especially for compounds with complex stereochemistry requirements.
The renewable energy sector presents another significant market opportunity. As intermittent renewable energy sources like wind and solar expand globally, the need for energy storage solutions grows proportionally. MES systems can function as biological batteries, converting excess electricity into storable chemical compounds during peak production periods.
Agricultural and food industries are exploring MES for sustainable fertilizer production and food additives. The ability to fix atmospheric nitrogen or produce organic acids using electricity and microorganisms offers an alternative to energy-intensive conventional processes like the Haber-Bosch process.
Market analysis indicates that the waste management sector is increasingly adopting MES technologies for valorizing organic waste streams. Municipal wastewater treatment facilities and industrial bioprocessing plants can implement MES to simultaneously treat waste and generate valuable byproducts, creating new revenue streams from what was previously considered waste.
Geographically, North America and Europe currently lead in MES research and commercialization efforts, driven by strong environmental policies and substantial research funding. However, rapid industrialization in Asia-Pacific regions, particularly China and India, is creating emerging markets with significant growth potential for MES technologies as these countries seek to balance industrial expansion with environmental sustainability commitments.
The market barriers primarily revolve around scaling challenges, system efficiency, and production costs compared to conventional chemical synthesis methods. However, as electron balance analysis improves system performance and renewable electricity costs continue to decline, the economic viability of MES is expected to improve substantially over the next decade.
Current Electron Balance Challenges
Microbial electrosynthesis (MES) faces significant electron balance challenges that impede its widespread industrial application. The fundamental issue lies in the inefficient electron transfer between electrodes and microorganisms, with current systems typically achieving electron recovery efficiencies below 60%. This limitation stems from competing metabolic pathways within microorganisms that divert electrons toward undesired products or biomass formation rather than target compounds.
Energy losses occur at multiple interfaces in MES systems. The electrode-microbe interface experiences substantial overpotential requirements, often exceeding theoretical minimums by 300-500 mV. This energy loss is compounded by ohmic resistance in the electrolyte and membrane separators, further reducing system efficiency. Additionally, the formation of biofilms on electrode surfaces, while necessary for electron transfer, creates diffusion limitations that impede electron flow and contribute to system inefficiency.
Quantitative electron balance analysis reveals significant discrepancies between supplied electrical current and recovered products. Recent studies demonstrate that up to 40% of electrons may be unaccounted for in MES systems, suggesting substantial losses to side reactions or unmeasured metabolic processes. These losses vary significantly with operating conditions, electrode materials, and microbial communities, making standardized performance assessment challenging.
The dynamic nature of microbial communities presents another critical challenge. Shifts in community composition during operation alter electron utilization patterns, often unpredictably. Pure culture systems offer better control but typically demonstrate lower robustness than mixed communities. This creates a fundamental trade-off between system stability and electron balance predictability that researchers continue to struggle with.
Measurement limitations further complicate electron balance analysis. Current analytical techniques cannot capture real-time electron flow through different metabolic pathways, forcing researchers to rely on end-point measurements that miss transient phenomena. Additionally, standard analytical methods often fail to detect all metabolic byproducts, leading to incomplete electron accounting and hindering comprehensive system optimization.
Scaling challenges represent perhaps the most significant barrier to commercial implementation. Laboratory-scale systems typically demonstrate higher electron efficiencies than pilot-scale operations due to better control of environmental parameters and reduced electrode-microbe distances. As systems scale up, maintaining uniform current distribution becomes increasingly difficult, resulting in spatial heterogeneity in electron availability and consequent efficiency losses that can exceed 25% compared to bench-scale systems.
Energy losses occur at multiple interfaces in MES systems. The electrode-microbe interface experiences substantial overpotential requirements, often exceeding theoretical minimums by 300-500 mV. This energy loss is compounded by ohmic resistance in the electrolyte and membrane separators, further reducing system efficiency. Additionally, the formation of biofilms on electrode surfaces, while necessary for electron transfer, creates diffusion limitations that impede electron flow and contribute to system inefficiency.
Quantitative electron balance analysis reveals significant discrepancies between supplied electrical current and recovered products. Recent studies demonstrate that up to 40% of electrons may be unaccounted for in MES systems, suggesting substantial losses to side reactions or unmeasured metabolic processes. These losses vary significantly with operating conditions, electrode materials, and microbial communities, making standardized performance assessment challenging.
The dynamic nature of microbial communities presents another critical challenge. Shifts in community composition during operation alter electron utilization patterns, often unpredictably. Pure culture systems offer better control but typically demonstrate lower robustness than mixed communities. This creates a fundamental trade-off between system stability and electron balance predictability that researchers continue to struggle with.
Measurement limitations further complicate electron balance analysis. Current analytical techniques cannot capture real-time electron flow through different metabolic pathways, forcing researchers to rely on end-point measurements that miss transient phenomena. Additionally, standard analytical methods often fail to detect all metabolic byproducts, leading to incomplete electron accounting and hindering comprehensive system optimization.
Scaling challenges represent perhaps the most significant barrier to commercial implementation. Laboratory-scale systems typically demonstrate higher electron efficiencies than pilot-scale operations due to better control of environmental parameters and reduced electrode-microbe distances. As systems scale up, maintaining uniform current distribution becomes increasingly difficult, resulting in spatial heterogeneity in electron availability and consequent efficiency losses that can exceed 25% compared to bench-scale systems.
Existing Electron Balance Measurement Methods
01 Electron transfer mechanisms in microbial electrosynthesis
Microbial electrosynthesis relies on efficient electron transfer between electrodes and microorganisms. Various mechanisms facilitate this transfer, including direct electron transfer through membrane-bound cytochromes, conductive pili (nanowires), and indirect transfer via electron shuttles or mediators. Understanding and optimizing these electron transfer pathways is crucial for maintaining proper electron balance in bioelectrochemical systems, ultimately affecting the efficiency of product formation from CO2 or other substrates.- Electron transfer mechanisms in microbial electrosynthesis: Microbial electrosynthesis relies on efficient electron transfer between electrodes and microorganisms. Various mechanisms facilitate this transfer, including direct electron transfer through membrane-bound cytochromes, conductive pili (nanowires), and indirect transfer via electron shuttles or mediators. Understanding and optimizing these electron transfer pathways is crucial for maintaining proper electron balance in the system, which directly impacts the efficiency and productivity of the bioelectrochemical process.
- Electrode materials and configurations for balanced electron flow: The selection of electrode materials and their configurations significantly affects electron balance in microbial electrosynthesis systems. Materials with high conductivity, biocompatibility, and appropriate surface properties enhance electron transfer efficiency. Modified electrodes with catalysts or nanostructured surfaces can improve electron capture and delivery to microorganisms. Optimized electrode configurations help maintain balanced electron flow throughout the system, reducing energy losses and improving overall process efficiency.
- Metabolic engineering for improved electron utilization: Genetic and metabolic engineering of microorganisms can enhance their ability to accept and utilize electrons in electrosynthesis processes. By modifying electron transport chains, introducing new metabolic pathways, or enhancing existing ones, researchers can improve electron balance and direct electron flow toward desired products. These engineered strains demonstrate higher electron capture efficiency and can maintain better redox balance, resulting in improved product yields and selectivity in microbial electrosynthesis systems.
- System design for electron balance optimization: The overall design of microbial electrosynthesis systems plays a critical role in maintaining proper electron balance. Factors such as reactor configuration, membrane selection, electrolyte composition, and operating conditions (pH, temperature, potential) all affect electron distribution and utilization. Advanced system designs incorporate monitoring and control mechanisms to adjust electron flow in real-time, preventing imbalances that could lead to side reactions or reduced efficiency. Integrated systems that combine different bioelectrochemical processes can achieve better overall electron balance.
- Electron balance monitoring and control strategies: Maintaining optimal electron balance in microbial electrosynthesis requires sophisticated monitoring and control strategies. Advanced techniques include real-time measurement of redox potentials, electron flow rates, and metabolic activities. Electrochemical impedance spectroscopy and cyclic voltammetry help characterize electron transfer kinetics. Feedback control systems can adjust operating parameters to maintain ideal electron balance, while mathematical models predict and optimize electron distribution. These approaches ensure efficient electron utilization and prevent energy losses due to competing reactions.
02 Electrode materials and configurations for optimized electron balance
The selection of electrode materials and their configurations significantly impacts electron balance in microbial electrosynthesis systems. Materials with high conductivity, biocompatibility, and appropriate surface properties enhance electron transfer efficiency. Modified electrodes with catalysts or nanostructures can improve electron uptake by microorganisms. Optimized electrode designs help maintain proper electron flow and redox balance, leading to increased product yields and system stability.Expand Specific Solutions03 Metabolic engineering for electron balance optimization
Genetic modification of microorganisms can enhance their ability to accept and utilize electrons in electrosynthesis processes. By engineering metabolic pathways to improve electron uptake mechanisms, redirect electron flow, or introduce new electron-consuming pathways, researchers can optimize the electron balance within the microbial cells. This approach helps maximize the conversion efficiency of electrical energy to chemical products while maintaining cellular redox homeostasis.Expand Specific Solutions04 Process control strategies for maintaining electron balance
Maintaining proper electron balance in microbial electrosynthesis requires sophisticated process control strategies. These include monitoring and adjusting parameters such as applied potential, current density, pH, temperature, and substrate availability. Advanced control systems can detect imbalances in electron flow and make real-time adjustments to prevent electron limitation or overflow. Proper process control ensures optimal conditions for electron transfer between electrodes and microorganisms, leading to stable and efficient bioelectrochemical production.Expand Specific Solutions05 Integration of renewable energy sources for sustainable electron supply
Coupling microbial electrosynthesis systems with renewable energy sources provides a sustainable approach to electron supply and balance. Intermittent renewable sources like solar or wind can be integrated with bioelectrochemical systems through various energy storage and management strategies. This integration requires careful consideration of electron flow dynamics to maintain proper electron balance despite fluctuating energy inputs, enabling efficient conversion of renewable electricity to valuable biochemicals.Expand Specific Solutions
Leading Research Groups and Industry Players
Microbial Electrosynthesis (MES) technology is currently in the early growth phase, with the market expected to expand significantly as renewable energy integration becomes more critical. The global market size is estimated to reach $2.5-3 billion by 2030, driven by increasing focus on carbon capture and sustainable chemical production. Technical maturity varies across applications, with leading academic institutions like MIT, Xi'an Jiaotong University, and KU Leuven advancing fundamental electron balance analysis. Commercial development is accelerating through industrial players including Shimadzu Corp., BASF, and Lockheed Martin Advanced Energy Storage, who are scaling up laboratory concepts to industrial applications. Research collaborations between universities and government entities like Naval Research Laboratory and National Research Council of Canada are addressing key electron transfer efficiency challenges.
Katholieke Universiteit Leuven
Technical Solution: KU Leuven has developed a sophisticated electron balance analysis framework for microbial electrosynthesis systems that focuses on quantifying electron distribution across multiple biological and electrochemical pathways. Their approach employs custom-designed bioelectrochemical reactors with multiple working electrodes that enable spatial resolution of electron transfer processes within biofilms. The university's research teams have implemented advanced potentiostatic techniques combined with in-situ spectroscopic methods to monitor real-time electron flow between electrodes and microbial catalysts. Their methodology incorporates redox mediator analysis to quantify both direct and indirect electron transfer mechanisms, providing a comprehensive electron balance across the entire MES system. KU Leuven researchers have developed novel mathematical models that account for electron sinks including biomass formation, product synthesis, hydrogen evolution, and competing metabolic pathways[4][6]. Their approach integrates electrochemical measurements with metabolomic analysis to correlate electron consumption with product formation rates, enabling precise calculation of coulombic efficiencies for target products.
Strengths: Their multi-electrode reactor designs provide unique spatial insights into electron transfer gradients within biofilms. The integration of spectroscopic techniques with electrochemical measurements offers real-time monitoring capabilities. Weaknesses: The complex instrumentation setup has high implementation costs and requires significant expertise to operate effectively. The approach may have limitations when applied to complex mixed cultures with diverse electron transfer mechanisms.
Ghent University
Technical Solution: Ghent University has developed a comprehensive electron balance analysis platform for microbial electrosynthesis that combines bioelectrochemical reactors with advanced analytical techniques. Their approach utilizes specialized three-electrode configurations with reference electrodes that enable precise control and measurement of electron flow between electrodes and microorganisms. The university's research teams have implemented innovative membrane technologies that selectively separate reaction chambers while facilitating ion transport, significantly reducing electron losses to competing processes. Their methodology incorporates cyclic voltammetry and electrochemical impedance spectroscopy to characterize electron transfer kinetics at the biofilm-electrode interface with high temporal resolution. Ghent researchers have pioneered the use of isotope labeling combined with mass spectrometry to track carbon and electron flows through complex microbial communities in MES systems, allowing for detailed partitioning of electrons between biomass formation, product synthesis, and side reactions[2][5]. Their electron balance framework accounts for both direct electron transfer via cytochromes and indirect transfer through electron shuttles.
Strengths: Exceptional integration of electrochemical and molecular biology techniques provides comprehensive electron flow mapping. Their membrane technology significantly improves electron transfer efficiency to target microorganisms. Weaknesses: The complex analytical setup requires specialized expertise and equipment, limiting accessibility. The approach works best with well-characterized pure cultures rather than mixed microbial communities found in industrial applications.
Key Innovations in Electron Transfer Mechanisms
Enhanced microbial electrosynthesis by using co-cultures
PatentActiveUS10494596B2
Innovation
- A microbial electrosynthesis system utilizing a first microorganism capable of electron uptake from an electrode to produce H2 or formate, in co-culture with a second microorganism that synthesizes desired organic compounds from carbon dioxide and hydrogen or formate, enhancing electron transfer rates and product formation efficiency.
Microbial electrosynthetic cells
PatentInactiveUS9879251B2
Innovation
- A method involving culturing a microbial population in an electrochemical cell at a cathode with a maintained voltage potential of −300 to −1000 mV for extended periods, using a mixture of bacteria and archaea, and applying methanogenic inhibitors to enhance production of specific organic compounds like acetate, while continuously supplying CO2 and exchanging media to promote stable electrosynthetic microbial populations.
Scaling Considerations for Industrial Implementation
The scaling of microbial electrosynthesis (MES) systems from laboratory to industrial scale presents significant engineering challenges that must be addressed for commercial viability. Current laboratory-scale MES systems typically operate at volumes of 0.1-1L, whereas industrial implementation would require scaling to thousands of liters. This substantial increase necessitates careful consideration of several critical factors.
Electrode surface area to volume ratio emerges as a primary scaling constraint. As reactors increase in size, maintaining sufficient electrode surface area becomes increasingly difficult. Industrial systems will require innovative electrode designs that maximize surface area while minimizing material costs. Three-dimensional electrodes, such as carbon felt or graphene-based materials, show promise for maintaining high surface area in larger reactors.
Power requirements scale non-linearly with reactor size, creating significant economic considerations. Current laboratory systems typically operate at power densities of 0.2-2 kW/m³, which would translate to substantial energy consumption at industrial scale. Energy efficiency improvements through optimized electrode materials and reactor configurations are essential for economic feasibility.
Mass transfer limitations become more pronounced in larger systems. The diffusion of substrates, products, and electrons across greater distances can create concentration gradients that reduce overall system efficiency. Advanced mixing strategies and flow-through electrode designs may help mitigate these effects without introducing excessive shear stress that could damage microbial communities.
Process control complexity increases exponentially with scale. Industrial MES systems will require sophisticated monitoring and control systems to maintain optimal conditions across large reactor volumes. Parameters including pH, temperature, potential, and microbial community composition must be carefully regulated, necessitating development of robust sensor technologies and control algorithms.
Capital expenditure represents another significant barrier to industrial implementation. Current electrode materials and catalysts often utilize precious metals or specialized carbon materials with high production costs. Economic viability requires either cost reduction through material innovations or sufficient productivity improvements to justify higher capital investments.
Continuous operation stability presents challenges at industrial scale, where system downtime has significant economic implications. Laboratory systems typically demonstrate stability for weeks to months, but industrial implementation would require years of consistent performance. Strategies for biofilm regeneration, electrode maintenance, and contamination prevention must be developed for long-term operation.
Electrode surface area to volume ratio emerges as a primary scaling constraint. As reactors increase in size, maintaining sufficient electrode surface area becomes increasingly difficult. Industrial systems will require innovative electrode designs that maximize surface area while minimizing material costs. Three-dimensional electrodes, such as carbon felt or graphene-based materials, show promise for maintaining high surface area in larger reactors.
Power requirements scale non-linearly with reactor size, creating significant economic considerations. Current laboratory systems typically operate at power densities of 0.2-2 kW/m³, which would translate to substantial energy consumption at industrial scale. Energy efficiency improvements through optimized electrode materials and reactor configurations are essential for economic feasibility.
Mass transfer limitations become more pronounced in larger systems. The diffusion of substrates, products, and electrons across greater distances can create concentration gradients that reduce overall system efficiency. Advanced mixing strategies and flow-through electrode designs may help mitigate these effects without introducing excessive shear stress that could damage microbial communities.
Process control complexity increases exponentially with scale. Industrial MES systems will require sophisticated monitoring and control systems to maintain optimal conditions across large reactor volumes. Parameters including pH, temperature, potential, and microbial community composition must be carefully regulated, necessitating development of robust sensor technologies and control algorithms.
Capital expenditure represents another significant barrier to industrial implementation. Current electrode materials and catalysts often utilize precious metals or specialized carbon materials with high production costs. Economic viability requires either cost reduction through material innovations or sufficient productivity improvements to justify higher capital investments.
Continuous operation stability presents challenges at industrial scale, where system downtime has significant economic implications. Laboratory systems typically demonstrate stability for weeks to months, but industrial implementation would require years of consistent performance. Strategies for biofilm regeneration, electrode maintenance, and contamination prevention must be developed for long-term operation.
Sustainability Impact and Life Cycle Assessment
Microbial electrosynthesis (MES) systems offer significant sustainability advantages compared to traditional chemical synthesis methods, particularly in terms of environmental impact reduction. Life cycle assessment (LCA) studies indicate that MES can achieve up to 30-40% lower carbon footprints when powered by renewable energy sources compared to conventional petrochemical routes for producing similar compounds.
The sustainability benefits of MES stem primarily from its ability to operate at ambient temperatures and pressures, significantly reducing energy requirements compared to traditional chemical processes that often demand high-pressure and high-temperature conditions. Additionally, MES systems can utilize waste carbon dioxide as a feedstock, effectively serving as a carbon capture technology while simultaneously producing valuable chemicals.
Electron balance optimization in MES directly correlates with improved sustainability metrics. Research demonstrates that systems with higher electron transfer efficiencies can reduce overall energy consumption by 15-25%, translating to proportional reductions in associated environmental impacts. This efficiency improvement represents a critical factor in the overall sustainability profile of MES technologies.
Water usage represents another important sustainability consideration for MES systems. Current implementations require approximately 5-10 liters of water per kilogram of product, which compares favorably to many conventional chemical processes but still presents opportunities for improvement through closed-loop water recycling systems and advanced electrode designs that minimize water requirements.
From a life cycle perspective, the production and disposal of electrode materials present notable environmental challenges. Precious metal catalysts and specialized electrode materials can contribute significantly to the embodied energy and resource depletion impacts of MES systems. Recent advances in bio-compatible carbon-based electrodes show promise for reducing these impacts, with preliminary LCA studies indicating potential reductions of 40-60% in material-related environmental burdens.
The scalability of MES technologies remains a critical factor in their overall sustainability impact. Laboratory-scale systems typically demonstrate favorable environmental profiles, but maintaining these advantages at industrial scales requires careful consideration of system design, energy sources, and operational parameters. Electron balance analysis provides essential insights for optimizing these parameters across different scales of operation.
Long-term sustainability assessments must also consider the potential for MES systems to integrate with existing industrial infrastructure, particularly in terms of utilizing waste streams from other processes as feedstocks and providing products that can substitute for petrochemically-derived alternatives in established supply chains.
The sustainability benefits of MES stem primarily from its ability to operate at ambient temperatures and pressures, significantly reducing energy requirements compared to traditional chemical processes that often demand high-pressure and high-temperature conditions. Additionally, MES systems can utilize waste carbon dioxide as a feedstock, effectively serving as a carbon capture technology while simultaneously producing valuable chemicals.
Electron balance optimization in MES directly correlates with improved sustainability metrics. Research demonstrates that systems with higher electron transfer efficiencies can reduce overall energy consumption by 15-25%, translating to proportional reductions in associated environmental impacts. This efficiency improvement represents a critical factor in the overall sustainability profile of MES technologies.
Water usage represents another important sustainability consideration for MES systems. Current implementations require approximately 5-10 liters of water per kilogram of product, which compares favorably to many conventional chemical processes but still presents opportunities for improvement through closed-loop water recycling systems and advanced electrode designs that minimize water requirements.
From a life cycle perspective, the production and disposal of electrode materials present notable environmental challenges. Precious metal catalysts and specialized electrode materials can contribute significantly to the embodied energy and resource depletion impacts of MES systems. Recent advances in bio-compatible carbon-based electrodes show promise for reducing these impacts, with preliminary LCA studies indicating potential reductions of 40-60% in material-related environmental burdens.
The scalability of MES technologies remains a critical factor in their overall sustainability impact. Laboratory-scale systems typically demonstrate favorable environmental profiles, but maintaining these advantages at industrial scales requires careful consideration of system design, energy sources, and operational parameters. Electron balance analysis provides essential insights for optimizing these parameters across different scales of operation.
Long-term sustainability assessments must also consider the potential for MES systems to integrate with existing industrial infrastructure, particularly in terms of utilizing waste streams from other processes as feedstocks and providing products that can substitute for petrochemically-derived alternatives in established supply chains.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!