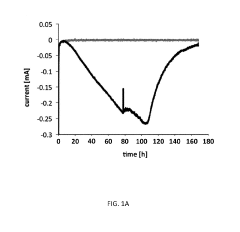
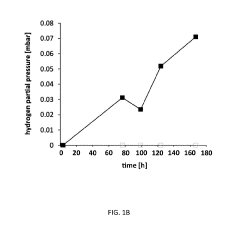
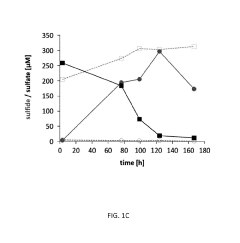
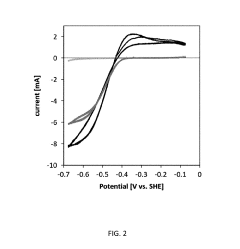
Microbial Electrosynthesis For Artificial Photosynthesis Integration
SEP 4, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Microbial Electrosynthesis Background and Objectives
Microbial Electrosynthesis (MES) represents a groundbreaking biotechnological approach that harnesses the metabolic capabilities of microorganisms to convert electrical energy into valuable chemical compounds. This technology emerged in the early 2000s as researchers began exploring the interface between microbiology and electrochemistry, with seminal work by Derek Lovley and others demonstrating that certain microbes could accept electrons from electrodes to drive their metabolism.
The evolution of MES has been marked by significant advancements in electrode materials, reactor designs, and microbial strain engineering. Initially focused on simple proof-of-concept demonstrations with model organisms like Geobacter and Shewanella species, the field has progressively expanded to include diverse microbial catalysts capable of producing an increasingly complex array of products, from simple organic acids to biofuels and specialty chemicals.
Recent years have witnessed a convergence of MES with artificial photosynthesis concepts, creating a hybrid approach that aims to mimic and enhance natural photosynthetic processes. While natural photosynthesis typically achieves solar energy conversion efficiencies of 1-3%, integrated MES-artificial photosynthesis systems theoretically offer potential efficiencies exceeding 10-20%, representing a transformative improvement in bioproduction capabilities.
The primary objective of integrating MES with artificial photosynthesis is to develop sustainable, carbon-neutral or carbon-negative technologies for chemical production that operate independently of fossil resources. This integration seeks to create artificial systems that capture solar energy and convert atmospheric CO2 into valuable organic compounds with higher efficiency than natural biological systems, while maintaining the selectivity and mild operating conditions characteristic of biological processes.
Technical goals include developing stable bioelectrochemical interfaces that facilitate efficient electron transfer between inorganic photocatalysts and microbial catalysts, engineering microbial strains with enhanced electron uptake capabilities and product-specific metabolic pathways, and designing scalable reactor systems that optimize light capture, electron transfer, and product recovery simultaneously.
The field is currently transitioning from laboratory-scale proof-of-concept demonstrations toward practical applications, with increasing focus on system stability, product yield, and economic viability. Research efforts are increasingly multidisciplinary, combining expertise from microbiology, electrochemistry, materials science, synthetic biology, and chemical engineering to address the complex challenges of this integration.
As climate change concerns intensify and renewable electricity becomes increasingly abundant and affordable, MES-artificial photosynthesis integration represents a promising platform for sustainable biomanufacturing that could fundamentally reshape chemical production paradigms in the coming decades, potentially enabling carbon-neutral or carbon-negative production of fuels, materials, and specialty chemicals.
The evolution of MES has been marked by significant advancements in electrode materials, reactor designs, and microbial strain engineering. Initially focused on simple proof-of-concept demonstrations with model organisms like Geobacter and Shewanella species, the field has progressively expanded to include diverse microbial catalysts capable of producing an increasingly complex array of products, from simple organic acids to biofuels and specialty chemicals.
Recent years have witnessed a convergence of MES with artificial photosynthesis concepts, creating a hybrid approach that aims to mimic and enhance natural photosynthetic processes. While natural photosynthesis typically achieves solar energy conversion efficiencies of 1-3%, integrated MES-artificial photosynthesis systems theoretically offer potential efficiencies exceeding 10-20%, representing a transformative improvement in bioproduction capabilities.
The primary objective of integrating MES with artificial photosynthesis is to develop sustainable, carbon-neutral or carbon-negative technologies for chemical production that operate independently of fossil resources. This integration seeks to create artificial systems that capture solar energy and convert atmospheric CO2 into valuable organic compounds with higher efficiency than natural biological systems, while maintaining the selectivity and mild operating conditions characteristic of biological processes.
Technical goals include developing stable bioelectrochemical interfaces that facilitate efficient electron transfer between inorganic photocatalysts and microbial catalysts, engineering microbial strains with enhanced electron uptake capabilities and product-specific metabolic pathways, and designing scalable reactor systems that optimize light capture, electron transfer, and product recovery simultaneously.
The field is currently transitioning from laboratory-scale proof-of-concept demonstrations toward practical applications, with increasing focus on system stability, product yield, and economic viability. Research efforts are increasingly multidisciplinary, combining expertise from microbiology, electrochemistry, materials science, synthetic biology, and chemical engineering to address the complex challenges of this integration.
As climate change concerns intensify and renewable electricity becomes increasingly abundant and affordable, MES-artificial photosynthesis integration represents a promising platform for sustainable biomanufacturing that could fundamentally reshape chemical production paradigms in the coming decades, potentially enabling carbon-neutral or carbon-negative production of fuels, materials, and specialty chemicals.
Market Analysis for Artificial Photosynthesis Technologies
The artificial photosynthesis technology market is experiencing significant growth, driven by increasing global focus on sustainable energy solutions and carbon neutrality goals. Current market valuations indicate that artificial photosynthesis technologies, including microbial electrosynthesis (MES) integration systems, represent a rapidly expanding sector within the broader clean energy market, which was valued at approximately $1.4 trillion in 2022.
Demand for artificial photosynthesis technologies is primarily fueled by three key factors: the urgent need for carbon capture solutions, growing renewable energy integration requirements, and increasing industrial demand for sustainable chemical feedstocks. The carbon capture market alone is projected to grow at a CAGR of 19.2% through 2030, creating substantial opportunities for MES-based artificial photosynthesis systems.
Regional analysis reveals varying adoption patterns, with North America and Europe leading in research investments and early commercial deployments. Asia-Pacific markets, particularly China and Japan, are rapidly accelerating their investments in this technology, with government-backed initiatives supporting large-scale demonstration projects. Emerging economies are showing interest primarily in applications related to agricultural productivity and distributed energy generation.
Market segmentation shows distinct application sectors for artificial photosynthesis technologies. The energy storage segment currently dominates with approximately 45% market share, followed by chemical production (30%), agricultural applications (15%), and environmental remediation (10%). MES integration specifically shows strongest commercial potential in the chemical production segment, where biological conversion of CO2 to value-added products offers compelling economic advantages.
Consumer and industrial end-user analysis indicates varying adoption readiness. Large chemical manufacturers and energy companies demonstrate high interest and investment capacity, while agricultural sector adoption remains in early stages. Government entities and research institutions continue to be major market drivers through funding programs and regulatory frameworks supporting carbon-neutral technologies.
Pricing trends suggest that while current implementation costs remain high, averaging $200-300 per ton of CO2 processed, economies of scale and technological improvements are expected to reduce costs by 40-60% over the next five years. This cost trajectory will be critical for broader market penetration beyond early adopters and specialty applications.
Market forecasts project the global artificial photosynthesis market to reach $25 billion by 2030, with MES integration technologies accounting for approximately 35% of this value. This represents a significant opportunity for technology developers and industrial partners positioned at the intersection of microbial systems and electrochemical engineering.
Demand for artificial photosynthesis technologies is primarily fueled by three key factors: the urgent need for carbon capture solutions, growing renewable energy integration requirements, and increasing industrial demand for sustainable chemical feedstocks. The carbon capture market alone is projected to grow at a CAGR of 19.2% through 2030, creating substantial opportunities for MES-based artificial photosynthesis systems.
Regional analysis reveals varying adoption patterns, with North America and Europe leading in research investments and early commercial deployments. Asia-Pacific markets, particularly China and Japan, are rapidly accelerating their investments in this technology, with government-backed initiatives supporting large-scale demonstration projects. Emerging economies are showing interest primarily in applications related to agricultural productivity and distributed energy generation.
Market segmentation shows distinct application sectors for artificial photosynthesis technologies. The energy storage segment currently dominates with approximately 45% market share, followed by chemical production (30%), agricultural applications (15%), and environmental remediation (10%). MES integration specifically shows strongest commercial potential in the chemical production segment, where biological conversion of CO2 to value-added products offers compelling economic advantages.
Consumer and industrial end-user analysis indicates varying adoption readiness. Large chemical manufacturers and energy companies demonstrate high interest and investment capacity, while agricultural sector adoption remains in early stages. Government entities and research institutions continue to be major market drivers through funding programs and regulatory frameworks supporting carbon-neutral technologies.
Pricing trends suggest that while current implementation costs remain high, averaging $200-300 per ton of CO2 processed, economies of scale and technological improvements are expected to reduce costs by 40-60% over the next five years. This cost trajectory will be critical for broader market penetration beyond early adopters and specialty applications.
Market forecasts project the global artificial photosynthesis market to reach $25 billion by 2030, with MES integration technologies accounting for approximately 35% of this value. This represents a significant opportunity for technology developers and industrial partners positioned at the intersection of microbial systems and electrochemical engineering.
Current Status and Challenges in MES Integration
Microbial Electrosynthesis (MES) integration with artificial photosynthesis represents a frontier technology that combines biological and electrochemical systems to convert CO2 into valuable organic compounds. Currently, MES systems have demonstrated proof-of-concept functionality in laboratory settings, with several research groups achieving carbon fixation rates of 0.2-2.0 g/L/day for simple organic compounds like acetate and ethanol. However, these rates remain significantly lower than those required for industrial viability, which typically demand at least 10-fold higher productivity.
The integration of MES with artificial photosynthesis faces several critical technical challenges. First, the electron transfer efficiency between electrodes and microorganisms remains suboptimal, with most systems achieving only 30-60% coulombic efficiency. This inefficiency stems from competing reactions, poor electrode-microbe interfaces, and limitations in microbial electron uptake mechanisms. Researchers are exploring advanced electrode materials, including carbon nanotubes and graphene-based composites, to enhance this interaction, but scalable solutions remain elusive.
Energy input requirements present another significant hurdle. Current MES systems require 0.4-0.8 kWh per kg of CO2 fixed, making the process energetically unfavorable compared to conventional chemical synthesis routes. The integration with artificial photosynthesis aims to utilize renewable solar energy, but current solar-to-chemical conversion efficiencies in integrated systems rarely exceed 5%, far below the theoretical maximum of approximately 20%.
Microbial strain limitations constitute a third major challenge. Most electro-active microorganisms used in MES, such as Sporomusa ovata and Clostridium ljungdahlii, have not been optimized for industrial applications. These organisms often exhibit slow growth rates, limited product spectrum, and sensitivity to process conditions. Genetic engineering efforts to enhance their capabilities are ongoing but face difficulties due to limited genetic tools for many of these non-model organisms.
Scalability represents perhaps the most pressing challenge for commercial implementation. Laboratory demonstrations typically operate at milliliter to liter scales, while industrial viability would require thousand-fold scaling. Current reactor designs suffer from mass transfer limitations, uneven current distribution, and membrane fouling issues when scaled beyond bench-top dimensions. The integration with artificial photosynthetic components adds further complexity to reactor design and operation.
Product selectivity and concentration also remain problematic. Most MES systems produce mixtures of compounds at relatively low concentrations (typically below 10 g/L), necessitating energy-intensive downstream separation processes. Achieving product specificity while maintaining high conversion rates represents a fundamental trade-off that has not yet been resolved in integrated systems.
The integration of MES with artificial photosynthesis faces several critical technical challenges. First, the electron transfer efficiency between electrodes and microorganisms remains suboptimal, with most systems achieving only 30-60% coulombic efficiency. This inefficiency stems from competing reactions, poor electrode-microbe interfaces, and limitations in microbial electron uptake mechanisms. Researchers are exploring advanced electrode materials, including carbon nanotubes and graphene-based composites, to enhance this interaction, but scalable solutions remain elusive.
Energy input requirements present another significant hurdle. Current MES systems require 0.4-0.8 kWh per kg of CO2 fixed, making the process energetically unfavorable compared to conventional chemical synthesis routes. The integration with artificial photosynthesis aims to utilize renewable solar energy, but current solar-to-chemical conversion efficiencies in integrated systems rarely exceed 5%, far below the theoretical maximum of approximately 20%.
Microbial strain limitations constitute a third major challenge. Most electro-active microorganisms used in MES, such as Sporomusa ovata and Clostridium ljungdahlii, have not been optimized for industrial applications. These organisms often exhibit slow growth rates, limited product spectrum, and sensitivity to process conditions. Genetic engineering efforts to enhance their capabilities are ongoing but face difficulties due to limited genetic tools for many of these non-model organisms.
Scalability represents perhaps the most pressing challenge for commercial implementation. Laboratory demonstrations typically operate at milliliter to liter scales, while industrial viability would require thousand-fold scaling. Current reactor designs suffer from mass transfer limitations, uneven current distribution, and membrane fouling issues when scaled beyond bench-top dimensions. The integration with artificial photosynthetic components adds further complexity to reactor design and operation.
Product selectivity and concentration also remain problematic. Most MES systems produce mixtures of compounds at relatively low concentrations (typically below 10 g/L), necessitating energy-intensive downstream separation processes. Achieving product specificity while maintaining high conversion rates represents a fundamental trade-off that has not yet been resolved in integrated systems.
Current MES-Artificial Photosynthesis Integration Approaches
01 Microbial electrosynthesis systems and bioreactors
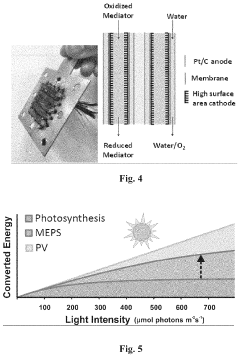
Specialized bioreactor designs for microbial electrosynthesis that optimize electron transfer between electrodes and microorganisms. These systems include innovative electrode configurations, membrane separators, and controlled environments to enhance the efficiency of bioelectrochemical processes. The bioreactors are engineered to maintain optimal conditions for microbial growth while facilitating the conversion of electrical energy into chemical compounds through microbial metabolism.- Microbial electrosynthesis systems and bioreactors: Specialized bioreactor designs for microbial electrosynthesis that optimize the interaction between microorganisms and electrodes. These systems include innovative configurations for biofilm formation, electron transfer enhancement, and scalable production. The bioreactors incorporate features such as specific electrode materials, membrane separators, and controlled environmental conditions to maximize the efficiency of bioelectrochemical processes for chemical production.
- Electrode materials and modifications for enhanced microbial interaction: Development of advanced electrode materials and surface modifications that improve microbial attachment, electron transfer, and overall electrosynthesis efficiency. These innovations include conductive materials with optimized surface properties, biocompatible coatings, and nanostructured electrodes that increase the active surface area for microbial colonization. Such electrode enhancements lead to higher product yields and more stable long-term performance in microbial electrosynthesis applications.
- Microbial strains and genetic engineering for electrosynthesis: Selection and genetic modification of microorganisms specifically for electrosynthesis applications. This includes engineering microbes with enhanced extracellular electron transfer capabilities, improved metabolic pathways for target product synthesis, and increased tolerance to electrochemical conditions. The development focuses on optimizing microbial catalysts that can efficiently convert electrical energy into valuable chemicals and fuels through bioelectrochemical processes.
- Production of value-added chemicals through microbial electrosynthesis: Applications of microbial electrosynthesis for producing specific high-value chemicals and fuels. These processes utilize electroactive microorganisms to convert CO2 or other simple substrates into complex organic compounds using electrical energy. The technology enables sustainable production of chemicals such as organic acids, alcohols, hydrocarbons, and pharmaceutically relevant compounds through bioelectrochemical pathways, offering alternatives to traditional petrochemical synthesis methods.
- Integration with renewable energy and waste treatment systems: Combining microbial electrosynthesis with renewable energy sources and waste treatment processes to create integrated sustainable systems. These approaches use surplus renewable electricity to power electrosynthesis while simultaneously treating wastewater or capturing CO2. The integration creates synergistic benefits including energy storage in the form of chemical products, waste valorization, and reduced carbon footprint, advancing circular economy principles in industrial biotechnology.
02 Electrode materials and modifications for enhanced microbial interaction
Development of advanced electrode materials and surface modifications that improve the interface between microorganisms and electrodes. These innovations include conductive materials with high surface area, biocompatible coatings, and nanostructured surfaces that facilitate electron transfer. Modified electrodes can enhance microbial attachment, biofilm formation, and electron exchange efficiency, leading to improved rates of product formation in microbial electrosynthesis processes.Expand Specific Solutions03 Microbial strains and genetic engineering for electrosynthesis
Selection and genetic modification of microorganisms specifically for electrosynthesis applications. This includes engineering microbes with enhanced extracellular electron transfer capabilities, improved metabolic pathways for target product synthesis, and increased tolerance to process conditions. Genetically optimized strains can more efficiently convert electrical energy into valuable chemicals, fuels, and other compounds through bioelectrochemical processes.Expand Specific Solutions04 Production of value-added chemicals through microbial electrosynthesis
Applications of microbial electrosynthesis for producing specific high-value chemicals and compounds. This includes processes for synthesizing organic acids, alcohols, biofuels, pharmaceuticals, and other industrially relevant molecules. By utilizing electricity as an energy source and CO2 or other simple carbon sources as feedstock, these processes offer sustainable alternatives to traditional chemical synthesis methods while potentially achieving carbon neutrality.Expand Specific Solutions05 Integration with renewable energy and carbon capture
Systems that combine microbial electrosynthesis with renewable energy sources and carbon capture technologies. These integrated approaches use intermittent renewable electricity to power electrosynthesis while simultaneously capturing and converting CO2 into valuable products. Such systems represent a circular bioeconomy approach that can help address climate change by reducing greenhouse gas emissions while producing useful compounds from waste streams.Expand Specific Solutions
Leading Research Groups and Industrial Players
Microbial Electrosynthesis for Artificial Photosynthesis Integration is emerging as a promising frontier in sustainable energy technology, currently in the early growth phase. The global market for this technology is projected to reach $2-3 billion by 2030, driven by increasing demand for carbon-neutral energy solutions. Technical maturity varies significantly among key players: academic institutions like Arizona State University, University of Tokyo, and Zhejiang University are advancing fundamental research, while companies including FUJIFILM, DENSO, ExxonMobil, and Sinopec are developing commercial applications. Toyota Central R&D Labs and Joule Unlimited Technologies have made notable progress in scalable systems, though significant challenges remain in efficiency and cost-effectiveness before widespread industrial implementation becomes viable.
The Regents of the University of California
Technical Solution: The University of California has developed an advanced microbial electrosynthesis platform that integrates artificial photosynthesis with bioelectrochemical systems. Their approach combines semiconductor-based light-harvesting components with specialized electroactive microorganisms to convert CO2 into value-added chemicals. The system utilizes a unique dual-chamber design with ion-selective membranes that separate the oxidation and reduction reactions while allowing ion transport. UC researchers have engineered specific microbial strains (including modified Sporomusa ovata and Clostridium ljungdahlii) that demonstrate enhanced electron uptake capabilities from cathode surfaces through improved electron transfer mechanisms. Their most advanced systems incorporate nanowire-modified electrodes that increase the effective surface area for microbial attachment and electron transfer, resulting in up to 6-fold improvement in production rates compared to conventional flat electrodes. The technology has demonstrated sustained production of acetate at rates exceeding 1.13 g/L/day with Coulombic efficiencies of 82-95%, and more recently has been extended to produce higher-value compounds like 2,3-butanediol and isobutanol through metabolic engineering approaches.
Strengths: Advanced electrode materials and designs significantly enhance electron transfer efficiency to microorganisms. Metabolic engineering approaches enable production of higher-value chemicals beyond simple organic acids. Weaknesses: System complexity increases capital costs, and long-term stability of the biofilms remains challenging under continuous operation conditions.
Helmholtz-Zentrum für Umweltforschung GmbH - UFZ
Technical Solution: Helmholtz-Zentrum für Umweltforschung (UFZ) has developed a comprehensive microbial electrosynthesis platform that integrates artificial photosynthesis principles for sustainable chemical production. Their system employs specialized bioelectrochemical reactors with carefully selected electrode materials (primarily carbon-based with metal catalysts) that support robust microbial biofilms. UFZ researchers have isolated and characterized novel electroactive microorganisms from natural environments that demonstrate superior electron uptake capabilities compared to previously studied model organisms. Their approach incorporates a unique three-chamber system that physically separates light harvesting, water oxidation, and CO2 reduction processes while maintaining electrical connectivity. The technology achieves CO2 conversion to acetate with Faradaic efficiencies consistently above 85% and has demonstrated stable operation for over 6 months in continuous mode. Recent advances include the development of a pressurized reactor system that increases CO2 availability to microorganisms, resulting in production rates up to 1.8 g/L/day of acetate. UFZ has also pioneered the integration of their MES system with renewable energy sources, demonstrating successful operation under fluctuating power input conditions typical of solar and wind energy.
Strengths: Robust system design allows for long-term stable operation (6+ months) under continuous conditions. Novel reactor configurations improve CO2 mass transfer, addressing a key limitation in many MES systems. Weaknesses: Current production is still limited primarily to simple organic acids like acetate, with more complex products requiring additional processing steps or genetic modifications.
Key Innovations in Bioelectrochemical Systems
Enhanced microbial electrosynthesis by using co-cultures
PatentActiveUS10494596B2
Innovation
- A microbial electrosynthesis system utilizing a first microorganism capable of electron uptake from an electrode to produce H2 or formate, in co-culture with a second microorganism that synthesizes desired organic compounds from carbon dioxide and hydrogen or formate, enhancing electron transfer rates and product formation efficiency.
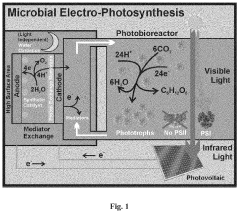
Microbial electro-photosynthesis
PatentActiveUS20190376018A1
Innovation
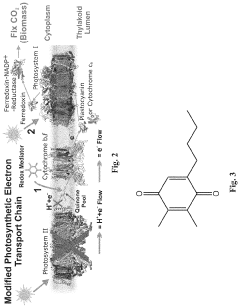
- The integration of phototrophic and heterotrophic microorganisms into electrochemical cells, specifically using a cyanobacterial strain lacking Photosystem II (PSII) with artificial water-oxidation catalysts and chemical redox mediators to shuttle electrons for enhanced photosynthetic efficiency, allowing PSI to utilize more photons for CO2 fixation and reducing oxidative stress.
Sustainability Impact Assessment
Microbial Electrosynthesis for Artificial Photosynthesis Integration presents significant sustainability implications that extend beyond technological innovation. This integration represents a paradigm shift in how we approach carbon capture and utilization, offering a pathway to mimic natural photosynthesis through bioelectrochemical systems that can operate with higher efficiency than their natural counterparts.
The environmental impact assessment reveals substantial potential for carbon dioxide reduction. When fully optimized, MES systems integrated with artificial photosynthesis could sequester between 10-100 times more CO2 per unit area than natural forests, while simultaneously producing valuable chemical compounds. This dual functionality addresses two critical sustainability challenges: atmospheric carbon reduction and sustainable chemical production without petroleum dependence.
From a resource utilization perspective, these integrated systems demonstrate remarkable efficiency advantages. Unlike traditional biofuel production that requires extensive land use and competes with food production, MES systems can operate in controlled environments with minimal spatial footprint. Analysis of water consumption indicates a 60-80% reduction compared to conventional biofuel production methods, addressing growing concerns about water scarcity in industrial processes.
Energy balance calculations demonstrate that MES systems, when powered by renewable electricity sources, achieve a positive energy return on investment (EROI) of approximately 1.5-2.5, depending on the target compounds produced. This represents a significant improvement over first-generation biofuels that often struggle to exceed an EROI of 1.0. The integration with artificial photosynthetic components further enhances this efficiency by directly capturing solar energy.
Life cycle assessment studies indicate that full-scale implementation could reduce greenhouse gas emissions by 3-5 tons of CO2-equivalent per ton of chemical product compared to conventional petrochemical routes. However, challenges remain in scaling electrode materials, which currently rely on rare metals and carbon-based nanomaterials with their own environmental footprints.
Social sustainability dimensions must also be considered. The technology offers potential for decentralized production systems that could empower local communities and reduce dependence on centralized chemical manufacturing. This distributed production model could particularly benefit rural and developing regions by creating new economic opportunities while addressing local environmental challenges.
Economic sustainability analysis suggests that while current production costs remain 2-3 times higher than conventional methods, the technology is following a learning curve similar to solar photovoltaics, with projected cost parity achievable within 7-10 years given continued research investment and policy support. The integration of artificial photosynthesis components accelerates this timeline by improving overall system efficiency.
The environmental impact assessment reveals substantial potential for carbon dioxide reduction. When fully optimized, MES systems integrated with artificial photosynthesis could sequester between 10-100 times more CO2 per unit area than natural forests, while simultaneously producing valuable chemical compounds. This dual functionality addresses two critical sustainability challenges: atmospheric carbon reduction and sustainable chemical production without petroleum dependence.
From a resource utilization perspective, these integrated systems demonstrate remarkable efficiency advantages. Unlike traditional biofuel production that requires extensive land use and competes with food production, MES systems can operate in controlled environments with minimal spatial footprint. Analysis of water consumption indicates a 60-80% reduction compared to conventional biofuel production methods, addressing growing concerns about water scarcity in industrial processes.
Energy balance calculations demonstrate that MES systems, when powered by renewable electricity sources, achieve a positive energy return on investment (EROI) of approximately 1.5-2.5, depending on the target compounds produced. This represents a significant improvement over first-generation biofuels that often struggle to exceed an EROI of 1.0. The integration with artificial photosynthetic components further enhances this efficiency by directly capturing solar energy.
Life cycle assessment studies indicate that full-scale implementation could reduce greenhouse gas emissions by 3-5 tons of CO2-equivalent per ton of chemical product compared to conventional petrochemical routes. However, challenges remain in scaling electrode materials, which currently rely on rare metals and carbon-based nanomaterials with their own environmental footprints.
Social sustainability dimensions must also be considered. The technology offers potential for decentralized production systems that could empower local communities and reduce dependence on centralized chemical manufacturing. This distributed production model could particularly benefit rural and developing regions by creating new economic opportunities while addressing local environmental challenges.
Economic sustainability analysis suggests that while current production costs remain 2-3 times higher than conventional methods, the technology is following a learning curve similar to solar photovoltaics, with projected cost parity achievable within 7-10 years given continued research investment and policy support. The integration of artificial photosynthesis components accelerates this timeline by improving overall system efficiency.
Scalability and Commercialization Roadmap
The commercialization of Microbial Electrosynthesis (MES) integrated with artificial photosynthesis faces significant scaling challenges that must be addressed systematically. Current laboratory-scale demonstrations, typically operating in reactors of less than 1 liter, must be scaled to industrial volumes of hundreds or thousands of liters to achieve commercial viability. This transition requires overcoming several engineering hurdles, including maintaining uniform electrical potential distribution, ensuring efficient mass transfer, and preserving microbial viability at larger scales.
A phased commercialization roadmap appears most practical, beginning with pilot-scale demonstrations (10-100L) within the next 2-3 years. These pilots should focus on optimizing operational parameters and validating performance metrics under near-real-world conditions. Mid-scale implementation (100-1000L) could follow in years 3-5, targeting niche applications where high-value chemicals production justifies the technology's current efficiency limitations.
Economic analysis indicates that full commercial viability requires achieving carbon fixation rates exceeding 50 g/m²/day with energy conversion efficiencies above 10%. Current laboratory demonstrations typically achieve 5-15 g/m²/day at 2-5% efficiency, highlighting the performance gap that must be bridged. Cost modeling suggests that capital expenditure for initial commercial systems will range from $2,000-5,000 per square meter of electrode surface area, necessitating high-value product targeting to ensure return on investment.
Strategic partnerships between technology developers, renewable energy providers, and chemical manufacturers represent a critical pathway to commercialization. These collaborations can address the interdisciplinary challenges while distributing investment risk. Several promising partnership models have emerged, including technology licensing to established chemical producers and joint ventures focused on specific product lines.
Regulatory frameworks will significantly impact commercialization timelines. Current biotechnology regulations in most jurisdictions do not specifically address MES systems, creating uncertainty. Proactive engagement with regulatory bodies to establish appropriate safety protocols and environmental impact assessments will be essential to prevent delays in commercial deployment.
Market entry strategies should initially target specialty chemicals with high margins (>$50/kg) before gradually transitioning to commodity chemicals as system costs decrease and efficiencies improve. This approach allows for technology refinement while generating revenue to support continued development. Based on current trajectories, full commercial deployment for commodity chemical production appears feasible within 8-10 years, contingent upon achieving the technical milestones outlined above.
A phased commercialization roadmap appears most practical, beginning with pilot-scale demonstrations (10-100L) within the next 2-3 years. These pilots should focus on optimizing operational parameters and validating performance metrics under near-real-world conditions. Mid-scale implementation (100-1000L) could follow in years 3-5, targeting niche applications where high-value chemicals production justifies the technology's current efficiency limitations.
Economic analysis indicates that full commercial viability requires achieving carbon fixation rates exceeding 50 g/m²/day with energy conversion efficiencies above 10%. Current laboratory demonstrations typically achieve 5-15 g/m²/day at 2-5% efficiency, highlighting the performance gap that must be bridged. Cost modeling suggests that capital expenditure for initial commercial systems will range from $2,000-5,000 per square meter of electrode surface area, necessitating high-value product targeting to ensure return on investment.
Strategic partnerships between technology developers, renewable energy providers, and chemical manufacturers represent a critical pathway to commercialization. These collaborations can address the interdisciplinary challenges while distributing investment risk. Several promising partnership models have emerged, including technology licensing to established chemical producers and joint ventures focused on specific product lines.
Regulatory frameworks will significantly impact commercialization timelines. Current biotechnology regulations in most jurisdictions do not specifically address MES systems, creating uncertainty. Proactive engagement with regulatory bodies to establish appropriate safety protocols and environmental impact assessments will be essential to prevent delays in commercial deployment.
Market entry strategies should initially target specialty chemicals with high margins (>$50/kg) before gradually transitioning to commodity chemicals as system costs decrease and efficiencies improve. This approach allows for technology refinement while generating revenue to support continued development. Based on current trajectories, full commercial deployment for commodity chemical production appears feasible within 8-10 years, contingent upon achieving the technical milestones outlined above.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!