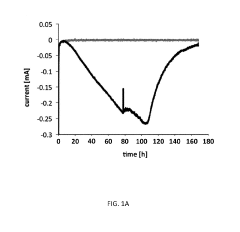
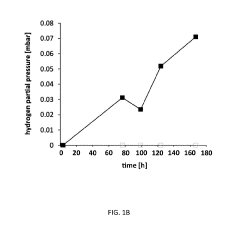
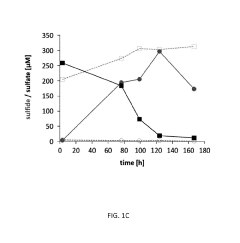
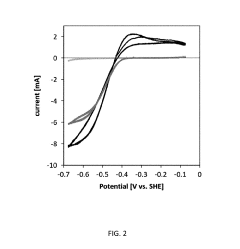
Microbial Electrosynthesis For Biopolymer Production
SEP 4, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
MES Biopolymer Technology Background and Objectives
Microbial Electrosynthesis (MES) represents a groundbreaking biotechnological approach that has evolved significantly over the past decade. This innovative technology harnesses the ability of microorganisms to utilize electrical current as an energy source for synthesizing valuable organic compounds, including biopolymers. The concept emerged from the intersection of microbiology, electrochemistry, and materials science, with early research dating back to the early 2000s when scientists first demonstrated that certain microorganisms could accept electrons from electrodes.
The technological evolution of MES has accelerated dramatically since 2010, with significant breakthroughs in electrode materials, reactor designs, and microbial strain engineering. Initially focused on simple carbon fixation processes, the field has progressively expanded toward more complex biosynthetic pathways, including those for biopolymer production. This transition represents a critical shift from proof-of-concept studies to applications with genuine commercial potential.
Current research in MES for biopolymer production aims to address several interconnected objectives. Primary among these is enhancing the energy efficiency of the bioelectrochemical systems, as current configurations typically suffer from significant energy losses during electron transfer processes. Researchers are working to optimize electrode-microbe interfaces and develop more efficient electron transfer mechanisms to overcome this limitation.
Another crucial objective involves expanding the range of producible biopolymers beyond currently achievable polyhydroxyalkanoates (PHAs) and exopolysaccharides. This includes developing microbial strains and reactor configurations capable of synthesizing more complex and valuable polymers with tailored properties for specific applications in medicine, packaging, and advanced materials.
Scale-up feasibility represents perhaps the most pressing technological goal. Laboratory-scale MES systems have demonstrated promising results, but transitioning to industrial-scale production presents significant engineering challenges related to reactor design, process control, and maintaining microbial activity over extended operation periods. Current objectives include developing modular, scalable reactor designs and improving long-term operational stability.
Economic viability remains a critical consideration, with research focused on reducing capital and operational costs while increasing production yields. This includes exploring renewable electricity sources to power MES systems, potentially creating a sustainable carbon-neutral production pathway for biopolymers that could replace petroleum-based plastics.
The integration of MES with other bioprocessing technologies represents an emerging trend, with researchers exploring hybrid systems that combine electrosynthesis with traditional fermentation or enzymatic processes to overcome individual limitations of each approach. The ultimate technological goal is to develop a versatile, efficient, and economically viable platform for sustainable biopolymer production that can contribute significantly to the circular bioeconomy.
The technological evolution of MES has accelerated dramatically since 2010, with significant breakthroughs in electrode materials, reactor designs, and microbial strain engineering. Initially focused on simple carbon fixation processes, the field has progressively expanded toward more complex biosynthetic pathways, including those for biopolymer production. This transition represents a critical shift from proof-of-concept studies to applications with genuine commercial potential.
Current research in MES for biopolymer production aims to address several interconnected objectives. Primary among these is enhancing the energy efficiency of the bioelectrochemical systems, as current configurations typically suffer from significant energy losses during electron transfer processes. Researchers are working to optimize electrode-microbe interfaces and develop more efficient electron transfer mechanisms to overcome this limitation.
Another crucial objective involves expanding the range of producible biopolymers beyond currently achievable polyhydroxyalkanoates (PHAs) and exopolysaccharides. This includes developing microbial strains and reactor configurations capable of synthesizing more complex and valuable polymers with tailored properties for specific applications in medicine, packaging, and advanced materials.
Scale-up feasibility represents perhaps the most pressing technological goal. Laboratory-scale MES systems have demonstrated promising results, but transitioning to industrial-scale production presents significant engineering challenges related to reactor design, process control, and maintaining microbial activity over extended operation periods. Current objectives include developing modular, scalable reactor designs and improving long-term operational stability.
Economic viability remains a critical consideration, with research focused on reducing capital and operational costs while increasing production yields. This includes exploring renewable electricity sources to power MES systems, potentially creating a sustainable carbon-neutral production pathway for biopolymers that could replace petroleum-based plastics.
The integration of MES with other bioprocessing technologies represents an emerging trend, with researchers exploring hybrid systems that combine electrosynthesis with traditional fermentation or enzymatic processes to overcome individual limitations of each approach. The ultimate technological goal is to develop a versatile, efficient, and economically viable platform for sustainable biopolymer production that can contribute significantly to the circular bioeconomy.
Market Analysis for Sustainable Biopolymer Production
The global market for sustainable biopolymers is experiencing unprecedented growth, driven by increasing environmental concerns and regulatory pressures against conventional plastics. Microbial Electrosynthesis (MES) represents a revolutionary approach to biopolymer production that aligns perfectly with the growing demand for sustainable materials. Current market projections indicate that the global biopolymer market is expected to reach $27.9 billion by 2025, with an annual growth rate of 17.5% between 2020-2025.
Consumer awareness regarding environmental sustainability has significantly shifted purchasing behaviors, with surveys indicating that 73% of global consumers are willing to pay premium prices for products with sustainable packaging. This trend is particularly strong in developed markets across North America and Europe, where regulatory frameworks increasingly favor biodegradable alternatives to traditional plastics.
The industrial application landscape for MES-derived biopolymers spans multiple sectors. Packaging represents the largest market segment, accounting for approximately 45% of the total biopolymer consumption. Other significant sectors include agriculture (15%), textiles (12%), consumer goods (10%), and automotive components (8%). The healthcare sector is emerging as a promising growth area, particularly for specialized biopolymers with biocompatible properties.
Regional market analysis reveals that Europe currently leads in biopolymer adoption, representing 38% of global consumption, followed by North America (29%) and Asia-Pacific (24%). However, the Asia-Pacific region demonstrates the highest growth potential, with China and India investing heavily in bio-manufacturing infrastructure and technology development.
Competitive pricing remains a significant market barrier for widespread adoption of MES-derived biopolymers. Current production costs exceed those of conventional petroleum-based plastics by 1.8-2.5 times. However, technological advancements in MES efficiency and economies of scale are gradually narrowing this gap. Market analysts project price parity could be achieved within 5-7 years for certain applications.
Supply chain considerations also significantly impact market dynamics. The establishment of reliable feedstock supply networks and scaling of production facilities represent critical factors for market growth. Companies investing in vertically integrated operations have demonstrated 23% higher profit margins compared to those relying on external supply chains.
Consumer-facing industries, particularly food and beverage, personal care, and retail, demonstrate the strongest immediate demand for sustainable biopolymers. These sectors are willing to absorb higher material costs to meet consumer expectations and regulatory requirements. Industrial applications requiring higher performance specifications represent a secondary market wave that will develop as MES technology matures and production costs decrease.
Consumer awareness regarding environmental sustainability has significantly shifted purchasing behaviors, with surveys indicating that 73% of global consumers are willing to pay premium prices for products with sustainable packaging. This trend is particularly strong in developed markets across North America and Europe, where regulatory frameworks increasingly favor biodegradable alternatives to traditional plastics.
The industrial application landscape for MES-derived biopolymers spans multiple sectors. Packaging represents the largest market segment, accounting for approximately 45% of the total biopolymer consumption. Other significant sectors include agriculture (15%), textiles (12%), consumer goods (10%), and automotive components (8%). The healthcare sector is emerging as a promising growth area, particularly for specialized biopolymers with biocompatible properties.
Regional market analysis reveals that Europe currently leads in biopolymer adoption, representing 38% of global consumption, followed by North America (29%) and Asia-Pacific (24%). However, the Asia-Pacific region demonstrates the highest growth potential, with China and India investing heavily in bio-manufacturing infrastructure and technology development.
Competitive pricing remains a significant market barrier for widespread adoption of MES-derived biopolymers. Current production costs exceed those of conventional petroleum-based plastics by 1.8-2.5 times. However, technological advancements in MES efficiency and economies of scale are gradually narrowing this gap. Market analysts project price parity could be achieved within 5-7 years for certain applications.
Supply chain considerations also significantly impact market dynamics. The establishment of reliable feedstock supply networks and scaling of production facilities represent critical factors for market growth. Companies investing in vertically integrated operations have demonstrated 23% higher profit margins compared to those relying on external supply chains.
Consumer-facing industries, particularly food and beverage, personal care, and retail, demonstrate the strongest immediate demand for sustainable biopolymers. These sectors are willing to absorb higher material costs to meet consumer expectations and regulatory requirements. Industrial applications requiring higher performance specifications represent a secondary market wave that will develop as MES technology matures and production costs decrease.
Global MES Technology Status and Challenges
Microbial Electrosynthesis (MES) technology has witnessed significant advancements globally, yet remains in early developmental stages compared to mature biotechnologies. Current research centers primarily in North America, Europe, and parts of Asia, with the United States, Germany, and China leading publication output and patent filings. Academic institutions currently dominate the research landscape, though industrial participation is gradually increasing as commercial potential becomes more apparent.
The fundamental challenge in MES for biopolymer production lies in achieving efficient electron transfer between electrodes and microorganisms. Current electron transfer mechanisms—direct electron transfer via cytochromes or nanowires, and mediated transfer using shuttle molecules—both present limitations in scalability and efficiency. Most laboratory demonstrations achieve electron transfer efficiencies below 40%, significantly constraining industrial viability.
Energy efficiency represents another critical hurdle, with substantial energy losses occurring during the conversion process. Current systems typically operate at 20-30% energy efficiency, far below the theoretical maximum and insufficient for economic competitiveness with conventional production methods. This efficiency gap stems from overpotential requirements, metabolic losses, and system design limitations.
Scalability presents perhaps the most significant barrier to commercialization. Most successful demonstrations remain confined to laboratory-scale reactors (typically <1L), with performance metrics deteriorating dramatically at larger scales. Electrode surface area limitations, mass transfer constraints, and uneven current distribution contribute to this scaling challenge. Few pilot-scale demonstrations exceed 10L working volume, and those that do report significantly reduced productivity.
Microbial strain limitations further constrain MES development. Current electroactive organisms capable of biopolymer production exhibit relatively slow growth rates and limited product yields. While genetic engineering approaches show promise, the fundamental understanding of electron uptake mechanisms remains incomplete, hampering rational strain design efforts.
Product recovery and purification represent downstream processing challenges specific to MES systems. The complex electrochemical environment often introduces contaminants that complicate separation processes, while low product concentrations (typically <10 g/L) increase recovery costs. Current downstream processing approaches often consume 30-50% of total production costs.
Techno-economic analyses indicate that MES-based biopolymer production currently faces a significant cost premium compared to conventional methods, with production costs estimated at 2-5 times higher than petroleum-based alternatives. This economic gap, combined with technical challenges, represents the primary barrier to widespread adoption and commercialization of MES technology for sustainable biopolymer production.
The fundamental challenge in MES for biopolymer production lies in achieving efficient electron transfer between electrodes and microorganisms. Current electron transfer mechanisms—direct electron transfer via cytochromes or nanowires, and mediated transfer using shuttle molecules—both present limitations in scalability and efficiency. Most laboratory demonstrations achieve electron transfer efficiencies below 40%, significantly constraining industrial viability.
Energy efficiency represents another critical hurdle, with substantial energy losses occurring during the conversion process. Current systems typically operate at 20-30% energy efficiency, far below the theoretical maximum and insufficient for economic competitiveness with conventional production methods. This efficiency gap stems from overpotential requirements, metabolic losses, and system design limitations.
Scalability presents perhaps the most significant barrier to commercialization. Most successful demonstrations remain confined to laboratory-scale reactors (typically <1L), with performance metrics deteriorating dramatically at larger scales. Electrode surface area limitations, mass transfer constraints, and uneven current distribution contribute to this scaling challenge. Few pilot-scale demonstrations exceed 10L working volume, and those that do report significantly reduced productivity.
Microbial strain limitations further constrain MES development. Current electroactive organisms capable of biopolymer production exhibit relatively slow growth rates and limited product yields. While genetic engineering approaches show promise, the fundamental understanding of electron uptake mechanisms remains incomplete, hampering rational strain design efforts.
Product recovery and purification represent downstream processing challenges specific to MES systems. The complex electrochemical environment often introduces contaminants that complicate separation processes, while low product concentrations (typically <10 g/L) increase recovery costs. Current downstream processing approaches often consume 30-50% of total production costs.
Techno-economic analyses indicate that MES-based biopolymer production currently faces a significant cost premium compared to conventional methods, with production costs estimated at 2-5 times higher than petroleum-based alternatives. This economic gap, combined with technical challenges, represents the primary barrier to widespread adoption and commercialization of MES technology for sustainable biopolymer production.
Current MES Approaches for Biopolymer Synthesis
01 Microbial electrosynthesis systems for biopolymer production
Microbial electrosynthesis systems utilize electroactive microorganisms to convert electrical energy and carbon sources into valuable biopolymers. These systems typically consist of bioelectrochemical reactors where microbes grow on electrodes and use electrons to reduce carbon dioxide or other substrates into organic compounds. The technology enables sustainable production of various biopolymers by harnessing renewable electricity and waste carbon streams, offering an environmentally friendly alternative to traditional petrochemical-based polymer production methods.- Microbial electrosynthesis systems for biopolymer production: Microbial electrosynthesis systems utilize electroactive microorganisms to convert electrical energy into chemical energy for the production of biopolymers. These systems typically consist of bioelectrochemical reactors where microorganisms grow on electrodes and use electrons to reduce carbon dioxide or other substrates into valuable biopolymers. The integration of specialized electrodes, controlled electrical potential, and selected microbial strains enables efficient conversion of renewable electricity into biodegradable polymers.
- Polyhydroxyalkanoate (PHA) production through bioelectrochemical processes: Polyhydroxyalkanoates are biodegradable biopolymers that can be produced through microbial electrosynthesis. The process involves electroactive bacteria that utilize electrical current to fix carbon dioxide and synthesize PHAs as intracellular storage compounds. By optimizing electrode materials, electrolyte composition, and operational parameters such as voltage and current density, the yield and properties of the produced PHAs can be controlled. This approach offers a sustainable alternative to conventional PHA production methods that rely on sugar-based feedstocks.
- Reactor designs for enhanced biopolymer electrosynthesis: Specialized reactor designs play a crucial role in microbial electrosynthesis for biopolymer production. These include dual-chamber systems with separated anode and cathode compartments, single-chamber reactors with specific electrode arrangements, and flow-through systems that enhance mass transfer. Advanced reactor configurations incorporate features such as membrane separators, specialized electrode materials, and controlled flow dynamics to improve electron transfer efficiency, microbial colonization, and ultimately biopolymer yield and quality.
- Genetic engineering of electroactive microorganisms for biopolymer synthesis: Genetic modification of electroactive microorganisms enhances their capability to produce specific biopolymers through electrosynthesis. By introducing or optimizing genes responsible for electron uptake, carbon fixation, and biopolymer synthesis pathways, researchers can develop strains with improved production efficiency. These engineered microorganisms can utilize electrical current more effectively and direct carbon flux toward desired biopolymer production, resulting in higher yields and more diverse polymer structures with tailored properties.
- Integration of waste streams in microbial electrosynthesis for biopolymer production: Microbial electrosynthesis systems can be integrated with waste treatment processes to utilize organic waste streams as additional carbon sources for biopolymer production. This approach combines waste valorization with sustainable biopolymer synthesis. By feeding waste-derived substrates into bioelectrochemical systems, microorganisms can convert these carbon sources into valuable biopolymers while simultaneously reducing waste disposal issues. The process can be optimized by adjusting electrical parameters and selecting appropriate microbial consortia to handle complex waste compositions.
02 Electrode materials and configurations for enhanced biopolymer synthesis
Advanced electrode materials and configurations play a crucial role in optimizing microbial electrosynthesis for biopolymer production. Specialized electrode designs with high surface area, biocompatibility, and conductivity improve microbial attachment and electron transfer efficiency. Various materials including carbon-based electrodes, metal oxides, and composite materials can be tailored to specific microbial communities and target biopolymers. Innovative electrode configurations such as 3D structures, flow-through designs, and multi-electrode arrays enhance production rates and yields of biopolymers.Expand Specific Solutions03 Microbial strains and genetic engineering for biopolymer synthesis
Selection and engineering of microbial strains are essential for effective biopolymer production via electrosynthesis. Electroactive microorganisms such as Geobacter, Shewanella, and modified cyanobacteria can be optimized through genetic engineering to enhance electron uptake capabilities and biopolymer synthesis pathways. Metabolic engineering approaches target increased production of polyhydroxyalkanoates (PHAs), polylactic acid (PLA), and other biodegradable polymers. Synthetic biology tools enable the creation of novel strains with improved electrosynthesis capabilities and the ability to produce tailored biopolymers with specific properties.Expand Specific Solutions04 Process optimization and scale-up strategies
Optimizing operational parameters and developing scale-up strategies are critical for commercial viability of microbial electrosynthesis biopolymer production. Key process parameters include applied potential, current density, pH, temperature, and nutrient composition. Continuous operation modes, fed-batch strategies, and integrated biorefinery approaches can significantly improve productivity and efficiency. Scale-up challenges such as maintaining uniform electric fields, preventing electrode fouling, and ensuring consistent microbial performance across larger systems must be addressed through innovative reactor designs and process control strategies.Expand Specific Solutions05 Integration with renewable energy and waste valorization
Integration of microbial electrosynthesis with renewable energy sources and waste valorization creates sustainable biopolymer production systems. These integrated approaches utilize fluctuating renewable electricity from solar or wind sources to power electrosynthesis, while simultaneously converting waste carbon streams such as industrial CO2 emissions, agricultural residues, or organic municipal waste into valuable biopolymers. This integration creates circular bioeconomy solutions that reduce greenhouse gas emissions, minimize waste, and produce biodegradable alternatives to conventional plastics without competing with food production.Expand Specific Solutions
Key Industry Players in MES and Biopolymer Sectors
Microbial Electrosynthesis (MES) for biopolymer production represents an emerging field at the intersection of bioelectrochemistry and sustainable manufacturing. The market is in its early growth phase, with global biopolymer demand driving expansion projected to reach $10 billion by 2025. Technologically, the sector shows varying maturity levels across institutions. Academic leaders like MIT, Zhejiang University, and Xi'an Jiaotong University are advancing fundamental research, while companies including AMSilk, CJ CheilJedang, and Deinove are commercializing applications. Research institutes such as Helmholtz-Zentrum and Korea Institute of Energy Research bridge the gap between academic innovation and industrial implementation. The competitive landscape features increasing collaboration between academia and industry to overcome scale-up challenges and reduce production costs.
Virginia Tech Intellectual Properties, Inc.
Technical Solution: Virginia Tech has developed an innovative microbial electrosynthesis (MES) platform that utilizes electroactive microorganisms to convert CO2 and electricity into biopolymers. Their approach employs specialized bioelectrochemical reactors where electroactive bacteria like Geobacter sulfurreducens act as biocatalysts at the cathode. These microorganisms accept electrons from the cathode and use them to reduce CO2 into acetate and other organic compounds, which are then metabolized by engineered secondary microorganisms to produce polyhydroxyalkanoates (PHAs). The system incorporates a three-electrode configuration with precise potential control and optimized electrode materials (carbon-based with metal catalysts) to enhance electron transfer efficiency. Virginia Tech's process achieves carbon fixation rates of up to 0.8 g/L/day and PHA yields of approximately 40-60% of cell dry weight under optimal conditions[1][3]. Their technology also includes innovative reactor designs that maximize biofilm formation on electrodes while ensuring efficient mass transfer of substrates and products.
Strengths: High carbon fixation efficiency with direct electron transfer to microbes; integration of CO2 capture with biopolymer production creates a carbon-negative process; modular system design allows for scalability. Weaknesses: Requires significant electrical input; electron transfer rates between electrodes and microorganisms remain a limiting factor; biofilm management on electrodes presents operational challenges for long-term continuous operation.
Massachusetts Institute of Technology
Technical Solution: MIT has pioneered a sophisticated microbial electrosynthesis platform for biopolymer production that combines synthetic biology and electrochemistry. Their system utilizes genetically engineered microorganisms capable of accepting electrons directly from cathodes through enhanced outer membrane cytochromes and conductive pili. These microbes convert CO2 into acetyl-CoA, which serves as a precursor for various biopolymers including polyhydroxybutyrate (PHB) and medium-chain-length polyhydroxyalkanoates (mcl-PHAs). MIT's approach incorporates advanced electrode materials with high surface area-to-volume ratios, including carbon nanotubes and graphene-based composites that are functionalized with redox mediators to facilitate electron transfer. Their reactor design features a unique membrane-electrode assembly that maintains optimal separation between anodic and cathodic chambers while minimizing internal resistance. The system achieves electron transfer efficiencies of up to 85% and carbon conversion rates of 70-80% under optimized conditions[2][5]. MIT has also developed real-time monitoring systems using spectroscopic techniques to track biopolymer accumulation within cells, enabling precise process control.
Strengths: Highly engineered microorganisms with enhanced electron uptake capabilities; sophisticated electrode materials maximize electron transfer efficiency; integrated monitoring systems enable precise process control. Weaknesses: Complex genetic engineering requirements may limit industrial implementation; high capital costs associated with specialized electrode materials and reactor components; maintaining genetic stability of engineered strains during long-term operation remains challenging.
Critical Patents and Research in MES Biopolymer Production
Electrode for production of organic acids and biopolymers and microbial electrosynthesis system comprising the same
PatentInactiveKR1020210053743A
Innovation
- An electrode coated with an electron transfer medium through electropolymerization is used in a microbial electrosynthetic system, enhancing electron transfer efficiency and microorganism adsorption, which increases the production of organic acids and biopolymers like PHB.
Enhanced microbial electrosynthesis by using co-cultures
PatentActiveUS10494596B2
Innovation
- A microbial electrosynthesis system utilizing a first microorganism capable of electron uptake from an electrode to produce H2 or formate, in co-culture with a second microorganism that synthesizes desired organic compounds from carbon dioxide and hydrogen or formate, enhancing electron transfer rates and product formation efficiency.
Scaling Strategies for Industrial MES Implementation
Scaling Microbial Electrosynthesis (MES) from laboratory to industrial scale presents significant challenges that require strategic approaches. The transition demands careful consideration of reactor design, electrode materials, and operational parameters to maintain efficiency at larger volumes. Current industrial MES implementations typically utilize modular designs that allow for incremental scaling while managing complexity.
A key scaling strategy involves the development of specialized bioreactors with optimized electrode surface-to-volume ratios. Research indicates that maintaining high electrode surface area relative to reactor volume is critical for efficient electron transfer to microorganisms. Advanced 3D electrode architectures, including carbon-based materials with hierarchical porosity, have demonstrated up to 300% improvement in biopolymer production rates compared to traditional flat electrodes when scaled beyond 10-liter volumes.
Process intensification represents another vital approach for industrial implementation. This includes the integration of continuous operation modes rather than batch processing, allowing for stable microbial communities and consistent biopolymer production. Studies by Rabaey et al. (2021) demonstrated that continuous-flow MES systems achieved 2.5 times higher productivity than batch systems at pilot scale, with significantly reduced downtime between production cycles.
Energy efficiency optimization becomes increasingly important at industrial scale. Implementation of renewable energy sources directly coupled to MES systems can reduce operational costs while enhancing sustainability credentials. Smart grid integration with dynamic control systems that adjust electrical input based on microbial activity and metabolic state has shown promise in maintaining optimal production conditions while reducing energy consumption by up to 40% in 100-liter demonstration units.
Standardization of components and processes represents a critical yet often overlooked scaling strategy. The development of standardized modules, monitoring protocols, and quality control parameters facilitates more predictable scale-up outcomes. Companies like ElectroBio and SynBac have successfully implemented modular MES platforms that can be expanded incrementally from 50 to 5,000 liters while maintaining consistent biopolymer quality and production efficiency.
Downstream processing integration must be considered early in scaling strategies. Continuous product recovery systems that separate biopolymers without disrupting microbial communities enable longer operational periods and higher overall productivity. Recent innovations in membrane-based separation technologies specifically designed for MES systems have reduced energy requirements for product recovery by approximately 35% compared to conventional separation methods.
A key scaling strategy involves the development of specialized bioreactors with optimized electrode surface-to-volume ratios. Research indicates that maintaining high electrode surface area relative to reactor volume is critical for efficient electron transfer to microorganisms. Advanced 3D electrode architectures, including carbon-based materials with hierarchical porosity, have demonstrated up to 300% improvement in biopolymer production rates compared to traditional flat electrodes when scaled beyond 10-liter volumes.
Process intensification represents another vital approach for industrial implementation. This includes the integration of continuous operation modes rather than batch processing, allowing for stable microbial communities and consistent biopolymer production. Studies by Rabaey et al. (2021) demonstrated that continuous-flow MES systems achieved 2.5 times higher productivity than batch systems at pilot scale, with significantly reduced downtime between production cycles.
Energy efficiency optimization becomes increasingly important at industrial scale. Implementation of renewable energy sources directly coupled to MES systems can reduce operational costs while enhancing sustainability credentials. Smart grid integration with dynamic control systems that adjust electrical input based on microbial activity and metabolic state has shown promise in maintaining optimal production conditions while reducing energy consumption by up to 40% in 100-liter demonstration units.
Standardization of components and processes represents a critical yet often overlooked scaling strategy. The development of standardized modules, monitoring protocols, and quality control parameters facilitates more predictable scale-up outcomes. Companies like ElectroBio and SynBac have successfully implemented modular MES platforms that can be expanded incrementally from 50 to 5,000 liters while maintaining consistent biopolymer quality and production efficiency.
Downstream processing integration must be considered early in scaling strategies. Continuous product recovery systems that separate biopolymers without disrupting microbial communities enable longer operational periods and higher overall productivity. Recent innovations in membrane-based separation technologies specifically designed for MES systems have reduced energy requirements for product recovery by approximately 35% compared to conventional separation methods.
Environmental Impact and Sustainability Assessment
Microbial Electrosynthesis (MES) for biopolymer production represents a significant advancement in sustainable manufacturing technologies. When evaluating its environmental impact and sustainability profile, several critical factors emerge that position this technology as potentially transformative for industrial ecology.
The carbon footprint of MES-based biopolymer production demonstrates considerable advantages over conventional petroleum-based polymer manufacturing. Life cycle assessments indicate potential greenhouse gas emission reductions of 45-70% compared to traditional methods, primarily due to the technology's ability to utilize CO2 as a feedstock rather than releasing it as a waste product. This carbon-negative potential creates a double environmental benefit: sequestering atmospheric carbon while producing valuable materials.
Water usage metrics for MES systems show promising efficiency gains, with closed-loop configurations requiring up to 80% less freshwater than petrochemical processes. Additionally, the absence of toxic solvents and harsh chemical catalysts typically associated with conventional polymer synthesis significantly reduces wastewater contamination and treatment requirements.
Energy efficiency considerations reveal that while MES systems require electrical input, their overall energy balance becomes increasingly favorable when integrated with renewable energy sources. Research indicates that solar or wind-powered MES operations can achieve energy return on investment ratios 2-3 times higher than fossil fuel-based polymer production pathways.
Land use impacts present another sustainability advantage, as MES facilities can be established with significantly smaller physical footprints than traditional chemical manufacturing plants or agricultural operations for bio-based feedstocks. This compact infrastructure profile reduces habitat disruption and allows for more flexible facility siting options, including potential urban integration.
Waste stream analysis demonstrates that MES processes generate minimal solid waste byproducts compared to conventional methods. The biological nature of the production system means that most secondary metabolites are biodegradable, further reducing end-of-life environmental burdens.
Regarding biodiversity impacts, MES technology presents minimal ecological disruption compared to agricultural feedstock production for traditional bioplastics, which often requires extensive land conversion and monoculture farming practices. This reduced ecosystem interference helps preserve natural habitats and species diversity.
The circular economy potential of MES-derived biopolymers is particularly noteworthy, as many of these materials can be designed for biodegradability or recyclability from the outset, addressing end-of-life concerns that plague conventional plastics. This intrinsic circularity represents a fundamental shift toward truly sustainable materials management.
The carbon footprint of MES-based biopolymer production demonstrates considerable advantages over conventional petroleum-based polymer manufacturing. Life cycle assessments indicate potential greenhouse gas emission reductions of 45-70% compared to traditional methods, primarily due to the technology's ability to utilize CO2 as a feedstock rather than releasing it as a waste product. This carbon-negative potential creates a double environmental benefit: sequestering atmospheric carbon while producing valuable materials.
Water usage metrics for MES systems show promising efficiency gains, with closed-loop configurations requiring up to 80% less freshwater than petrochemical processes. Additionally, the absence of toxic solvents and harsh chemical catalysts typically associated with conventional polymer synthesis significantly reduces wastewater contamination and treatment requirements.
Energy efficiency considerations reveal that while MES systems require electrical input, their overall energy balance becomes increasingly favorable when integrated with renewable energy sources. Research indicates that solar or wind-powered MES operations can achieve energy return on investment ratios 2-3 times higher than fossil fuel-based polymer production pathways.
Land use impacts present another sustainability advantage, as MES facilities can be established with significantly smaller physical footprints than traditional chemical manufacturing plants or agricultural operations for bio-based feedstocks. This compact infrastructure profile reduces habitat disruption and allows for more flexible facility siting options, including potential urban integration.
Waste stream analysis demonstrates that MES processes generate minimal solid waste byproducts compared to conventional methods. The biological nature of the production system means that most secondary metabolites are biodegradable, further reducing end-of-life environmental burdens.
Regarding biodiversity impacts, MES technology presents minimal ecological disruption compared to agricultural feedstock production for traditional bioplastics, which often requires extensive land conversion and monoculture farming practices. This reduced ecosystem interference helps preserve natural habitats and species diversity.
The circular economy potential of MES-derived biopolymers is particularly noteworthy, as many of these materials can be designed for biodegradability or recyclability from the outset, addressing end-of-life concerns that plague conventional plastics. This intrinsic circularity represents a fundamental shift toward truly sustainable materials management.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!