Evaluating ICP-MS Sample Introduction Techniques for Optimal Results
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
ICP-MS Technology Evolution and Objectives
Inductively Coupled Plasma Mass Spectrometry (ICP-MS) has evolved significantly since its commercial introduction in the early 1980s. This analytical technique combines the high-temperature ICP source with a mass spectrometer, enabling precise detection of metals and several non-metals at concentrations as low as one part per trillion. The evolution of ICP-MS technology has been driven by the increasing demand for ultra-trace elemental analysis across various industries including environmental monitoring, pharmaceuticals, semiconductor manufacturing, and clinical research.
The historical development of ICP-MS sample introduction techniques represents a fascinating journey of technological innovation. Initially, conventional nebulizers were the primary means of introducing samples into the plasma. However, these early systems suffered from significant limitations including low transport efficiency, high sample consumption, and susceptibility to matrix effects. These challenges prompted researchers to explore alternative sample introduction methodologies that could enhance analytical performance while minimizing interferences.
By the 1990s, significant advancements emerged with the development of microconcentric nebulizers, ultrasonic nebulizers, and desolvation systems. These innovations addressed many of the limitations of conventional systems by improving sample transport efficiency and reducing oxide formation. The early 2000s witnessed further refinements with the introduction of collision/reaction cell technology, which substantially mitigated polyatomic interferences that had previously limited the application scope of ICP-MS.
Recent technological trends have focused on enhancing sample introduction efficiency while minimizing sample consumption. Microflow nebulizers capable of operating at flow rates below 100 μL/min have gained prominence, particularly in applications where sample volume is limited. Parallel to this, laser ablation systems have revolutionized solid sample analysis, enabling direct sampling without dissolution and preserving spatial information within the sample.
The primary objectives of current ICP-MS sample introduction research center on several key areas: reducing matrix effects that compromise analytical accuracy, minimizing polyatomic and isobaric interferences, improving transport efficiency to enhance sensitivity, decreasing sample and reagent consumption for more sustainable laboratory practices, and developing robust systems capable of handling complex matrices such as high-salt solutions or organic solvents.
Looking forward, the field aims to develop intelligent sample introduction systems that can automatically adapt to varying sample matrices, further miniaturize components to reduce resource consumption, and integrate with automated sample preparation workflows. These advancements will be crucial in expanding the application scope of ICP-MS and meeting the increasingly stringent analytical requirements across scientific disciplines and industries.
The historical development of ICP-MS sample introduction techniques represents a fascinating journey of technological innovation. Initially, conventional nebulizers were the primary means of introducing samples into the plasma. However, these early systems suffered from significant limitations including low transport efficiency, high sample consumption, and susceptibility to matrix effects. These challenges prompted researchers to explore alternative sample introduction methodologies that could enhance analytical performance while minimizing interferences.
By the 1990s, significant advancements emerged with the development of microconcentric nebulizers, ultrasonic nebulizers, and desolvation systems. These innovations addressed many of the limitations of conventional systems by improving sample transport efficiency and reducing oxide formation. The early 2000s witnessed further refinements with the introduction of collision/reaction cell technology, which substantially mitigated polyatomic interferences that had previously limited the application scope of ICP-MS.
Recent technological trends have focused on enhancing sample introduction efficiency while minimizing sample consumption. Microflow nebulizers capable of operating at flow rates below 100 μL/min have gained prominence, particularly in applications where sample volume is limited. Parallel to this, laser ablation systems have revolutionized solid sample analysis, enabling direct sampling without dissolution and preserving spatial information within the sample.
The primary objectives of current ICP-MS sample introduction research center on several key areas: reducing matrix effects that compromise analytical accuracy, minimizing polyatomic and isobaric interferences, improving transport efficiency to enhance sensitivity, decreasing sample and reagent consumption for more sustainable laboratory practices, and developing robust systems capable of handling complex matrices such as high-salt solutions or organic solvents.
Looking forward, the field aims to develop intelligent sample introduction systems that can automatically adapt to varying sample matrices, further miniaturize components to reduce resource consumption, and integrate with automated sample preparation workflows. These advancements will be crucial in expanding the application scope of ICP-MS and meeting the increasingly stringent analytical requirements across scientific disciplines and industries.
Market Analysis of ICP-MS Applications
The global ICP-MS (Inductively Coupled Plasma Mass Spectrometry) market continues to demonstrate robust growth, with a current market valuation exceeding $1.2 billion and projected to reach $1.8 billion by 2027, growing at a CAGR of approximately 7.5%. This growth is primarily driven by increasing applications across multiple sectors including environmental monitoring, pharmaceutical analysis, food safety testing, and clinical diagnostics.
Environmental testing represents the largest application segment, accounting for nearly 30% of the total ICP-MS market. This dominance stems from stringent regulatory requirements for monitoring heavy metals and trace elements in water, soil, and air samples across developed and developing regions. The pharmaceutical sector follows closely, utilizing ICP-MS for impurity profiling and quality control processes.
Geographically, North America leads the market with approximately 35% share, followed by Europe (30%) and Asia-Pacific (25%). The Asia-Pacific region, particularly China, India, and South Korea, is experiencing the fastest growth due to expanding industrial activities, increasing environmental concerns, and growing investments in healthcare infrastructure.
The clinical diagnostics segment is emerging as the fastest-growing application area, with a CAGR exceeding 9%. This growth is fueled by the increasing adoption of ICP-MS for biomonitoring, toxicology studies, and clinical research. The technique's ability to detect multiple elements simultaneously at ultra-trace levels makes it invaluable for analyzing biological samples.
End-user analysis reveals that research institutions and academic laboratories constitute approximately 40% of the market, followed by environmental testing laboratories (25%), pharmaceutical companies (20%), and clinical diagnostic centers (15%). This distribution highlights the technique's versatility and broad applicability across different sectors.
Market trends indicate a growing demand for automated sample introduction systems that can handle high-throughput requirements while maintaining analytical precision. Additionally, there is increasing interest in specialized sample introduction techniques for challenging matrices such as high-salt solutions, organic solvents, and biological fluids, which traditionally pose difficulties for conventional nebulization systems.
The competitive landscape features major analytical instrument manufacturers including Agilent Technologies, PerkinElmer, Thermo Fisher Scientific, and Shimadzu Corporation, collectively holding over 75% of the market share. These companies are increasingly focusing on developing integrated solutions that combine advanced sample introduction techniques with sophisticated software for data analysis and interpretation.
Environmental testing represents the largest application segment, accounting for nearly 30% of the total ICP-MS market. This dominance stems from stringent regulatory requirements for monitoring heavy metals and trace elements in water, soil, and air samples across developed and developing regions. The pharmaceutical sector follows closely, utilizing ICP-MS for impurity profiling and quality control processes.
Geographically, North America leads the market with approximately 35% share, followed by Europe (30%) and Asia-Pacific (25%). The Asia-Pacific region, particularly China, India, and South Korea, is experiencing the fastest growth due to expanding industrial activities, increasing environmental concerns, and growing investments in healthcare infrastructure.
The clinical diagnostics segment is emerging as the fastest-growing application area, with a CAGR exceeding 9%. This growth is fueled by the increasing adoption of ICP-MS for biomonitoring, toxicology studies, and clinical research. The technique's ability to detect multiple elements simultaneously at ultra-trace levels makes it invaluable for analyzing biological samples.
End-user analysis reveals that research institutions and academic laboratories constitute approximately 40% of the market, followed by environmental testing laboratories (25%), pharmaceutical companies (20%), and clinical diagnostic centers (15%). This distribution highlights the technique's versatility and broad applicability across different sectors.
Market trends indicate a growing demand for automated sample introduction systems that can handle high-throughput requirements while maintaining analytical precision. Additionally, there is increasing interest in specialized sample introduction techniques for challenging matrices such as high-salt solutions, organic solvents, and biological fluids, which traditionally pose difficulties for conventional nebulization systems.
The competitive landscape features major analytical instrument manufacturers including Agilent Technologies, PerkinElmer, Thermo Fisher Scientific, and Shimadzu Corporation, collectively holding over 75% of the market share. These companies are increasingly focusing on developing integrated solutions that combine advanced sample introduction techniques with sophisticated software for data analysis and interpretation.
Current Sample Introduction Challenges
Despite significant advancements in ICP-MS technology, sample introduction remains a critical bottleneck that limits analytical performance. Current systems face several persistent challenges that affect measurement accuracy, precision, and detection limits. The conventional nebulizer-spray chamber combination, while widely adopted, suffers from low transport efficiency with typically only 1-3% of the sample reaching the plasma. This inefficiency necessitates larger sample volumes and extends analysis time, particularly problematic when sample availability is limited.
Matrix effects present another significant challenge, especially with complex environmental and biological samples. High dissolved solid content often leads to signal suppression, drift, and physical blockages in sample introduction components. Salt deposits accumulating on sampling cones and interface regions require frequent maintenance interventions, reducing instrument availability and increasing operational costs.
Volatile organic solvents introduce additional complications, causing plasma instability and carbon deposition on interface components. The resulting signal fluctuations compromise measurement reproducibility, while carbon buildup necessitates more frequent cleaning cycles. These issues are particularly pronounced in applications involving organic extracts or when analyzing samples prepared in organic matrices.
Memory effects constitute another persistent challenge, especially when analyzing elements like mercury, boron, and iodine. Carryover between samples can lead to false positives and compromised quantitative results, requiring extended washout periods that reduce sample throughput and analytical efficiency.
The introduction of nanoparticles and single-cell analysis applications has exposed limitations in conventional sample introduction systems. Traditional nebulizers often fail to maintain particle integrity or provide consistent transport of individual cells, limiting the applicability of ICP-MS in these emerging fields.
Temperature stability represents another critical challenge, as fluctuations in the spray chamber temperature can significantly affect aerosol generation and transport efficiency. These variations introduce additional measurement uncertainty, particularly problematic for high-precision isotope ratio determinations.
Automation compatibility issues also persist, with many specialized sample introduction systems lacking seamless integration with autosampler systems. This limitation restricts throughput capabilities and increases the manual intervention required, particularly challenging for laboratories processing large sample batches.
These challenges collectively highlight the need for innovative approaches to sample introduction that can overcome efficiency limitations while maintaining robustness across diverse sample types and analytical conditions.
Matrix effects present another significant challenge, especially with complex environmental and biological samples. High dissolved solid content often leads to signal suppression, drift, and physical blockages in sample introduction components. Salt deposits accumulating on sampling cones and interface regions require frequent maintenance interventions, reducing instrument availability and increasing operational costs.
Volatile organic solvents introduce additional complications, causing plasma instability and carbon deposition on interface components. The resulting signal fluctuations compromise measurement reproducibility, while carbon buildup necessitates more frequent cleaning cycles. These issues are particularly pronounced in applications involving organic extracts or when analyzing samples prepared in organic matrices.
Memory effects constitute another persistent challenge, especially when analyzing elements like mercury, boron, and iodine. Carryover between samples can lead to false positives and compromised quantitative results, requiring extended washout periods that reduce sample throughput and analytical efficiency.
The introduction of nanoparticles and single-cell analysis applications has exposed limitations in conventional sample introduction systems. Traditional nebulizers often fail to maintain particle integrity or provide consistent transport of individual cells, limiting the applicability of ICP-MS in these emerging fields.
Temperature stability represents another critical challenge, as fluctuations in the spray chamber temperature can significantly affect aerosol generation and transport efficiency. These variations introduce additional measurement uncertainty, particularly problematic for high-precision isotope ratio determinations.
Automation compatibility issues also persist, with many specialized sample introduction systems lacking seamless integration with autosampler systems. This limitation restricts throughput capabilities and increases the manual intervention required, particularly challenging for laboratories processing large sample batches.
These challenges collectively highlight the need for innovative approaches to sample introduction that can overcome efficiency limitations while maintaining robustness across diverse sample types and analytical conditions.
Comparative Analysis of Introduction Techniques
01 Nebulization and aerosol generation techniques
Various nebulization techniques are employed in ICP-MS to convert liquid samples into aerosols for analysis. These include pneumatic nebulizers, ultrasonic nebulizers, and microflow nebulizers that optimize sample delivery to the plasma. Advanced nebulization systems improve sensitivity by creating uniform droplet sizes and enhancing transport efficiency, which leads to better detection limits and more consistent results. These techniques are particularly important for samples with limited volume or high dissolved solid content.- Nebulizer and spray chamber optimization for ICP-MS: Optimizing nebulizer design and spray chamber configurations is crucial for efficient sample introduction in ICP-MS analysis. Advanced nebulizers can improve aerosol generation quality, while specialized spray chambers enhance droplet selection and reduce signal noise. These components work together to control sample flow rate, droplet size distribution, and transport efficiency, ultimately improving sensitivity and reproducibility of analytical results.
- Laser ablation techniques for solid sample introduction: Laser ablation systems provide direct solid sample introduction for ICP-MS without requiring dissolution. These systems use focused laser beams to vaporize small portions of solid samples, generating fine particles that are transported to the plasma. Optimizing laser parameters such as wavelength, pulse duration, and energy density is essential for consistent ablation and accurate results. This technique is particularly valuable for spatially resolved analysis and samples difficult to dissolve.
- Microfluidic and flow injection sample introduction systems: Microfluidic and flow injection systems offer precise control over sample introduction in ICP-MS. These techniques use small-volume sample handling with automated injection, reducing sample consumption and contamination risks. Integration with separation techniques allows for speciation analysis. The systems can be optimized for high-throughput analysis while maintaining analytical performance through controlled flow rates and precise timing of sample introduction.
- Desolvation and aerosol modification techniques: Desolvation systems and aerosol modification techniques improve ICP-MS sensitivity by removing solvents and optimizing particle size distribution before plasma introduction. These methods include membrane desolvation, heated spray chambers, and condensers that reduce solvent load on the plasma. By minimizing polyatomic interferences from solvent molecules and improving ionization efficiency, these techniques enhance detection limits and stability, particularly for challenging matrices and high-precision isotope ratio measurements.
- Automated sample preparation and introduction interfaces: Automated sample preparation and introduction interfaces streamline ICP-MS workflows by integrating sample handling, preparation, and introduction steps. These systems incorporate robotic sample changers, automated dilution, and intelligent sample queuing to reduce human error and increase throughput. Advanced interfaces can monitor and adjust sample introduction parameters in real-time based on feedback from the instrument, ensuring optimal performance across varying sample types and maintaining consistent analytical conditions throughout extended analytical runs.
02 Sample preparation and pretreatment methods
Effective sample preparation is crucial for optimal ICP-MS results. This includes digestion methods for solid samples, dilution protocols for high-concentration samples, and matrix modification techniques to reduce interferences. Pretreatment steps such as filtration, centrifugation, and chemical separation help eliminate particulates and matrix components that could affect instrument performance. Proper sample preparation ensures accurate quantification, reduces signal suppression, and extends the life of instrument components.Expand Specific Solutions03 Automated sample introduction systems
Automated sample introduction systems enhance ICP-MS analysis by improving precision and throughput. These systems include autosamplers, flow injection systems, and integrated sample preparation platforms that minimize human error and contamination risks. Automation allows for consistent sample delivery rates, precise volume control, and programmable dilution sequences. These features are particularly valuable for large sample batches, quality control procedures, and applications requiring high reproducibility.Expand Specific Solutions04 Specialized interfaces for challenging samples
Specialized interfaces have been developed for challenging sample types in ICP-MS analysis. These include desolvation systems for organic solvents, collision/reaction cells for polyatomic interference reduction, and high-temperature introduction systems for samples with high dissolved solids. Advanced interface designs improve plasma stability when analyzing complex matrices and enhance sensitivity for trace element detection. These specialized interfaces extend the application range of ICP-MS to previously difficult sample types.Expand Specific Solutions05 Hyphenated techniques and specialized sample delivery
Hyphenated techniques combine ICP-MS with separation methods to enhance analytical capabilities. These include coupling with chromatography systems (LC-ICP-MS, GC-ICP-MS), laser ablation for direct solid sampling, and electrothermal vaporization for improved sensitivity. These integrated approaches allow for speciation analysis, spatial distribution mapping, and enhanced detection of ultra-trace elements. Specialized delivery systems optimize the interface between the separation technique and the mass spectrometer, maintaining signal integrity throughout the analytical process.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The ICP-MS sample introduction market is in a mature growth phase, characterized by established technologies and ongoing innovations. The global market size for ICP-MS technologies is substantial, driven by increasing demands in environmental monitoring, pharmaceutical analysis, and materials science. Technologically, the field shows high maturity with companies like Agilent Technologies, Thermo Fisher Scientific, and PerkinElmer leading with advanced solutions. Elemental Scientific and Kimia Analytics are innovating with specialized sample introduction systems and high-performance ICP torches, respectively. Academic institutions including China University of Geosciences, ETH Zurich, and EPFL contribute significantly to research advancements. The competitive landscape features both established analytical instrumentation corporations and specialized firms focusing on niche applications, creating a dynamic ecosystem of innovation.
Elemental Scientific, Inc.
Technical Solution: Elemental Scientific specializes exclusively in sample introduction systems for ICP-MS, offering their flagship prepFAST automated sample introduction platform that integrates inline dilution, internal standard addition, and matrix matching capabilities. Their patented Apex Ω desolvation system achieves 95% solvent removal while maintaining sample integrity, resulting in 5-10× sensitivity enhancement for most elements and >50× for selenium and arsenic. The company's microFAST system operates at ultra-low flow rates (5-200 μL/min) while maintaining precision of <1% RSD, enabling analysis of precious samples including biological fluids and nanoparticle suspensions. Their dual-valve discrete sampling technology reduces sample carryover to <0.1%, significantly outperforming conventional systems (typically 0.1-1%). Elemental Scientific has also developed specialized introduction systems for specific applications, including their seaFAST system for seawater analysis that incorporates inline matrix elimination and preconcentration, achieving sub-ppt detection limits for trace metals in high-salt matrices without requiring separate sample preparation steps.
Strengths: Purpose-built specialized systems for specific applications yield optimal performance; modular design allows customization for different analytical needs; superior washout characteristics minimize cross-contamination. Weaknesses: Limited integration with some ICP-MS platforms requires additional interface development; specialized focus means laboratories need multiple vendors for complete analytical solutions; higher cost compared to standard nebulizers.
Thermo Fisher Scientific (Bremen) GmbH
Technical Solution: Thermo Fisher Scientific has developed advanced sample introduction systems for ICP-MS featuring their patented Total Consumption Nebulizer (TCN) technology that achieves nearly 100% sample utilization efficiency compared to conventional nebulizers' 1-3% efficiency. Their integrated cyclonic spray chambers with temperature control (-10°C to +20°C) significantly reduce oxide formation and minimize matrix effects. The company's PrepFAST autosampling system incorporates inline dilution capabilities, allowing automated preparation of calibration standards and dynamic adjustment of sample concentrations. Their latest innovation includes the Argon Gas Dilution (AGD) technology that enables analysis of samples with total dissolved solids (TDS) up to 25%, far exceeding the conventional 0.2% limit without requiring physical dilution. Additionally, their specialized desolvation systems remove over 98% of solvent before sample introduction, dramatically reducing polyatomic interferences.
Strengths: Industry-leading sample utilization efficiency reducing sample volume requirements and waste; superior interference management through temperature-controlled spray chambers; high-throughput capabilities with automated inline dilution. Weaknesses: Premium pricing structure places systems beyond reach of smaller laboratories; proprietary consumables create vendor lock-in; complex systems require specialized training and maintenance.
Key Patents and Innovations in Sample Introduction
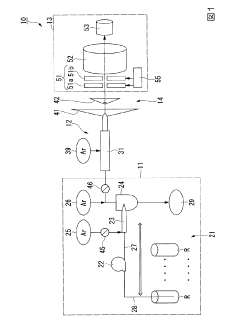
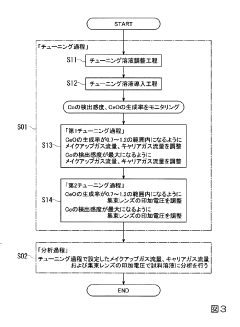
Inductively coupled plasma mass spectrometry
PatentInactiveJP2020027038A
Innovation
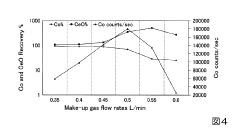
- The method involves tuning the ICP-MS system using a sample solution with a high-concentration acid matrix as a tuning liquid, adjusting carrier gas flow rates and focusing lens settings to control the production rate of coexisting element oxides within a specific range, thereby maximizing detection sensitivity.
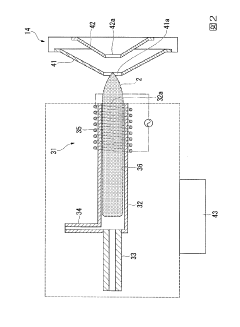
Temperature-controlled sample introduction system for analysis of viscous samples
PatentWO2021127481A1
Innovation
- A temperature-controlled sample introduction system that includes a temperature-controlled autosampler deck, heated transfer line, and a sample introduction system with a heated environment to maintain samples in a fluid state from autosampler to analysis device, using heating elements and thermocouples to regulate temperatures and prevent viscosity increases.
Environmental Impact and Sustainability Considerations
The environmental impact of ICP-MS sample introduction techniques represents a critical consideration in modern analytical laboratories. Traditional nebulization systems generate significant waste, with typical sample consumption rates of 1-2 mL/min while only 1-2% of the sample actually reaches the plasma. This inefficiency results in substantial chemical waste production, particularly problematic when analyzing samples containing toxic elements or requiring hazardous preparation reagents such as hydrofluoric acid.
Newer introduction technologies demonstrate promising sustainability improvements. Micronebulizers and microflow systems operate at 50-200 μL/min, reducing sample consumption by up to 95% compared to conventional systems. This reduction directly translates to decreased waste generation and minimized exposure to hazardous materials for laboratory personnel. Additionally, these systems require less argon gas, addressing both environmental and economic concerns as argon production is energy-intensive.
The environmental footprint extends beyond waste generation to energy consumption. ICP-MS instruments typically require 2-5 kW of power during operation, with sample introduction systems contributing significantly to this demand. High-efficiency introduction techniques can reduce plasma load, potentially decreasing power requirements by 10-15% and subsequently reducing carbon emissions associated with laboratory operations.
Water consumption presents another environmental challenge, particularly in desolvation systems that require cooling water. Recirculating chillers offer a solution, reducing water usage by up to 98% compared to open-loop cooling systems. This adaptation is especially valuable in regions facing water scarcity issues.
Laboratory waste management protocols must evolve alongside introduction techniques. Acid digestion procedures commonly used in sample preparation generate acidic waste requiring neutralization before disposal. Advanced introduction systems compatible with smaller sample volumes and lower acid concentrations can significantly reduce this environmental burden.
The life cycle assessment of consumables associated with sample introduction—such as nebulizers, spray chambers, and tubing—reveals additional sustainability concerns. Durable materials that extend component lifespan reduce replacement frequency and associated waste. Some manufacturers now offer recycling programs for certain components, though this practice remains limited in scope.
Future developments should prioritize green chemistry principles, focusing on systems that minimize reagent use, reduce energy consumption, and incorporate recyclable or biodegradable components. The integration of these considerations into method development and validation protocols will be essential for establishing truly sustainable analytical practices in ICP-MS applications.
Newer introduction technologies demonstrate promising sustainability improvements. Micronebulizers and microflow systems operate at 50-200 μL/min, reducing sample consumption by up to 95% compared to conventional systems. This reduction directly translates to decreased waste generation and minimized exposure to hazardous materials for laboratory personnel. Additionally, these systems require less argon gas, addressing both environmental and economic concerns as argon production is energy-intensive.
The environmental footprint extends beyond waste generation to energy consumption. ICP-MS instruments typically require 2-5 kW of power during operation, with sample introduction systems contributing significantly to this demand. High-efficiency introduction techniques can reduce plasma load, potentially decreasing power requirements by 10-15% and subsequently reducing carbon emissions associated with laboratory operations.
Water consumption presents another environmental challenge, particularly in desolvation systems that require cooling water. Recirculating chillers offer a solution, reducing water usage by up to 98% compared to open-loop cooling systems. This adaptation is especially valuable in regions facing water scarcity issues.
Laboratory waste management protocols must evolve alongside introduction techniques. Acid digestion procedures commonly used in sample preparation generate acidic waste requiring neutralization before disposal. Advanced introduction systems compatible with smaller sample volumes and lower acid concentrations can significantly reduce this environmental burden.
The life cycle assessment of consumables associated with sample introduction—such as nebulizers, spray chambers, and tubing—reveals additional sustainability concerns. Durable materials that extend component lifespan reduce replacement frequency and associated waste. Some manufacturers now offer recycling programs for certain components, though this practice remains limited in scope.
Future developments should prioritize green chemistry principles, focusing on systems that minimize reagent use, reduce energy consumption, and incorporate recyclable or biodegradable components. The integration of these considerations into method development and validation protocols will be essential for establishing truly sustainable analytical practices in ICP-MS applications.
Quality Control and Validation Protocols
Quality control and validation protocols are essential components of any ICP-MS analytical workflow, ensuring reliable and reproducible results across different sample introduction techniques. Establishing a comprehensive QC framework begins with the implementation of daily performance checks that evaluate key parameters such as sensitivity, oxide formation rates, and doubly charged ion ratios. These checks provide critical baseline information about instrument performance and help identify potential issues before sample analysis begins.
Method validation protocols must be tailored to specific sample introduction techniques, as each presents unique challenges. For conventional nebulizer systems, validation should focus on nebulizer efficiency, spray chamber temperature stability, and aerosol transport characteristics. When using laser ablation systems, validation protocols must additionally address laser energy stability, ablation cell design efficiency, and particle size distribution monitoring.
Reference materials play a pivotal role in quality assurance, with certified reference materials (CRMs) serving as the gold standard for method validation. For liquid sample introduction, multi-element standards with certified concentrations provide the necessary benchmarks, while solid reference materials with homogeneous composition are essential for laser ablation techniques. Regular analysis of these materials throughout analytical runs helps detect and correct for instrumental drift.
Statistical process control tools enhance quality monitoring by establishing control charts for key performance indicators. These charts typically track internal standard recovery rates, calibration curve stability, and blank contamination levels. Upper and lower control limits, typically set at ±2σ or ±3σ from the mean, provide objective criteria for identifying out-of-control situations requiring corrective action.
Uncertainty estimation represents another critical aspect of validation, requiring thorough assessment of all potential error sources. For sample introduction techniques, this includes quantifying uncertainties from sample transport efficiency, matrix effects, signal stability, and calibration procedures. Combined uncertainty calculations following ISO guidelines provide a comprehensive picture of result reliability.
Inter-laboratory comparison studies offer an external validation mechanism, allowing laboratories to benchmark their performance against peers using similar sample introduction techniques. Participation in proficiency testing schemes helps identify systematic biases and provides valuable insights into method robustness across different laboratory environments and instrument configurations.
Method validation protocols must be tailored to specific sample introduction techniques, as each presents unique challenges. For conventional nebulizer systems, validation should focus on nebulizer efficiency, spray chamber temperature stability, and aerosol transport characteristics. When using laser ablation systems, validation protocols must additionally address laser energy stability, ablation cell design efficiency, and particle size distribution monitoring.
Reference materials play a pivotal role in quality assurance, with certified reference materials (CRMs) serving as the gold standard for method validation. For liquid sample introduction, multi-element standards with certified concentrations provide the necessary benchmarks, while solid reference materials with homogeneous composition are essential for laser ablation techniques. Regular analysis of these materials throughout analytical runs helps detect and correct for instrumental drift.
Statistical process control tools enhance quality monitoring by establishing control charts for key performance indicators. These charts typically track internal standard recovery rates, calibration curve stability, and blank contamination levels. Upper and lower control limits, typically set at ±2σ or ±3σ from the mean, provide objective criteria for identifying out-of-control situations requiring corrective action.
Uncertainty estimation represents another critical aspect of validation, requiring thorough assessment of all potential error sources. For sample introduction techniques, this includes quantifying uncertainties from sample transport efficiency, matrix effects, signal stability, and calibration procedures. Combined uncertainty calculations following ISO guidelines provide a comprehensive picture of result reliability.
Inter-laboratory comparison studies offer an external validation mechanism, allowing laboratories to benchmark their performance against peers using similar sample introduction techniques. Participation in proficiency testing schemes helps identify systematic biases and provides valuable insights into method robustness across different laboratory environments and instrument configurations.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!