Using ICP-MS for Quality Control in Pharmaceutical Composition Analysis
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
ICP-MS Technology Evolution and Objectives
Inductively Coupled Plasma Mass Spectrometry (ICP-MS) has evolved significantly since its commercial introduction in the early 1980s. Initially developed as an analytical technique for geological samples, ICP-MS has undergone remarkable technological advancements that have expanded its application scope considerably. The evolution trajectory shows a clear progression from basic elemental analysis to sophisticated multi-element detection systems capable of measuring trace elements at parts-per-trillion levels.
The fundamental principle of ICP-MS involves ionizing samples using an inductively coupled plasma and then separating and quantifying ions based on their mass-to-charge ratios. Early systems faced challenges with interference, limited sensitivity, and complex operation requirements. However, technological innovations in the 1990s, including the introduction of collision/reaction cells, significantly improved analytical capabilities by reducing polyatomic interferences.
By the early 2000s, high-resolution ICP-MS systems emerged, offering unprecedented sensitivity and selectivity. The integration of time-of-flight mass analyzers further enhanced multi-element detection capabilities. Recent advancements include the development of triple quadrupole ICP-MS systems, which provide superior interference removal and enhanced detection limits critical for pharmaceutical applications.
In the pharmaceutical industry specifically, ICP-MS technology has evolved from a specialized research tool to an essential quality control instrument. This transition has been driven by increasingly stringent regulatory requirements for elemental impurity testing in drug products, particularly following the implementation of ICH Q3D guidelines and USP <232>/<233> standards.
The primary technological objectives for ICP-MS in pharmaceutical composition analysis include achieving lower detection limits for toxic elements, enhancing sample throughput capabilities, improving instrument robustness for routine quality control environments, and developing simplified methods for complex pharmaceutical matrices. Additionally, there is a growing focus on miniaturization and automation to facilitate integration into continuous manufacturing processes.
Future technological goals include the development of more user-friendly interfaces that require less specialized training, improved software for data processing and compliance documentation, and enhanced coupling with separation techniques such as HPLC for speciation analysis. There is also significant interest in developing portable or benchtop ICP-MS systems that maintain high analytical performance while requiring less laboratory infrastructure, potentially enabling at-line testing in pharmaceutical production environments.
The convergence of ICP-MS with other analytical technologies, particularly with chromatographic separation methods and automated sample preparation systems, represents a key evolutionary direction aimed at providing comprehensive elemental analysis solutions for pharmaceutical quality control applications.
The fundamental principle of ICP-MS involves ionizing samples using an inductively coupled plasma and then separating and quantifying ions based on their mass-to-charge ratios. Early systems faced challenges with interference, limited sensitivity, and complex operation requirements. However, technological innovations in the 1990s, including the introduction of collision/reaction cells, significantly improved analytical capabilities by reducing polyatomic interferences.
By the early 2000s, high-resolution ICP-MS systems emerged, offering unprecedented sensitivity and selectivity. The integration of time-of-flight mass analyzers further enhanced multi-element detection capabilities. Recent advancements include the development of triple quadrupole ICP-MS systems, which provide superior interference removal and enhanced detection limits critical for pharmaceutical applications.
In the pharmaceutical industry specifically, ICP-MS technology has evolved from a specialized research tool to an essential quality control instrument. This transition has been driven by increasingly stringent regulatory requirements for elemental impurity testing in drug products, particularly following the implementation of ICH Q3D guidelines and USP <232>/<233> standards.
The primary technological objectives for ICP-MS in pharmaceutical composition analysis include achieving lower detection limits for toxic elements, enhancing sample throughput capabilities, improving instrument robustness for routine quality control environments, and developing simplified methods for complex pharmaceutical matrices. Additionally, there is a growing focus on miniaturization and automation to facilitate integration into continuous manufacturing processes.
Future technological goals include the development of more user-friendly interfaces that require less specialized training, improved software for data processing and compliance documentation, and enhanced coupling with separation techniques such as HPLC for speciation analysis. There is also significant interest in developing portable or benchtop ICP-MS systems that maintain high analytical performance while requiring less laboratory infrastructure, potentially enabling at-line testing in pharmaceutical production environments.
The convergence of ICP-MS with other analytical technologies, particularly with chromatographic separation methods and automated sample preparation systems, represents a key evolutionary direction aimed at providing comprehensive elemental analysis solutions for pharmaceutical quality control applications.
Pharmaceutical QC Market Analysis
The pharmaceutical quality control market has been experiencing robust growth, driven by stringent regulatory requirements and increasing focus on drug safety. The global pharmaceutical QC market was valued at approximately $5.3 billion in 2022 and is projected to reach $8.9 billion by 2028, growing at a CAGR of 9.1% during the forecast period. This growth trajectory is particularly evident in advanced analytical technologies like ICP-MS (Inductively Coupled Plasma Mass Spectrometry).
Within the pharmaceutical QC segment, elemental analysis technologies account for roughly 18% of the total market share, with ICP-MS representing a significant and growing portion of this subsegment. The demand for ICP-MS in pharmaceutical composition analysis has been increasing at an annual rate of 11.3%, outpacing the overall QC market growth.
North America currently dominates the pharmaceutical QC market with approximately 38% market share, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region is witnessing the fastest growth rate at 12.7% annually, primarily driven by the expansion of pharmaceutical manufacturing in China and India, coupled with evolving regulatory frameworks in these regions.
The pharmaceutical industry's increasing focus on contamination control and elemental impurity analysis has been a major market driver. The implementation of ICH Q3D guidelines for elemental impurities has significantly boosted the adoption of ICP-MS technology across pharmaceutical manufacturing facilities worldwide. Additionally, the rising complexity of drug formulations, particularly biologics and gene therapies, necessitates more sophisticated analytical methods for quality control.
Contract manufacturing organizations (CMOs) and contract research organizations (CROs) represent the fastest-growing end-user segment for ICP-MS technology, expanding at 13.2% annually. This trend reflects the pharmaceutical industry's increasing reliance on outsourcing for specialized analytical testing services.
Key challenges in the market include the high initial investment cost for ICP-MS instrumentation, which ranges from $150,000 to $500,000 depending on specifications, and the technical expertise required for operation and data interpretation. These factors have limited adoption among smaller pharmaceutical companies and emerging markets, creating potential opportunities for equipment leasing models and analytical service providers.
The market is also witnessing a shift toward integrated analytical platforms that combine ICP-MS with other techniques such as chromatography, offering comprehensive solutions for pharmaceutical composition analysis. This integration trend is expected to reshape the competitive landscape over the next five years.
Within the pharmaceutical QC segment, elemental analysis technologies account for roughly 18% of the total market share, with ICP-MS representing a significant and growing portion of this subsegment. The demand for ICP-MS in pharmaceutical composition analysis has been increasing at an annual rate of 11.3%, outpacing the overall QC market growth.
North America currently dominates the pharmaceutical QC market with approximately 38% market share, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region is witnessing the fastest growth rate at 12.7% annually, primarily driven by the expansion of pharmaceutical manufacturing in China and India, coupled with evolving regulatory frameworks in these regions.
The pharmaceutical industry's increasing focus on contamination control and elemental impurity analysis has been a major market driver. The implementation of ICH Q3D guidelines for elemental impurities has significantly boosted the adoption of ICP-MS technology across pharmaceutical manufacturing facilities worldwide. Additionally, the rising complexity of drug formulations, particularly biologics and gene therapies, necessitates more sophisticated analytical methods for quality control.
Contract manufacturing organizations (CMOs) and contract research organizations (CROs) represent the fastest-growing end-user segment for ICP-MS technology, expanding at 13.2% annually. This trend reflects the pharmaceutical industry's increasing reliance on outsourcing for specialized analytical testing services.
Key challenges in the market include the high initial investment cost for ICP-MS instrumentation, which ranges from $150,000 to $500,000 depending on specifications, and the technical expertise required for operation and data interpretation. These factors have limited adoption among smaller pharmaceutical companies and emerging markets, creating potential opportunities for equipment leasing models and analytical service providers.
The market is also witnessing a shift toward integrated analytical platforms that combine ICP-MS with other techniques such as chromatography, offering comprehensive solutions for pharmaceutical composition analysis. This integration trend is expected to reshape the competitive landscape over the next five years.
Current Capabilities and Limitations of ICP-MS in Pharma
Inductively Coupled Plasma Mass Spectrometry (ICP-MS) has emerged as a powerful analytical technique in pharmaceutical quality control, offering exceptional sensitivity for elemental analysis. Current ICP-MS systems can detect elements at concentrations as low as parts per trillion (ppt), making them ideal for monitoring trace elemental impurities in pharmaceutical products as mandated by ICH Q3D guidelines and USP <232>/<233> standards.
The technique excels in multi-elemental analysis capabilities, allowing simultaneous detection of numerous elements across the periodic table in a single analytical run. Modern instruments achieve impressive throughput, processing up to 30-40 samples per hour with minimal sample preparation requirements compared to older techniques like atomic absorption spectroscopy.
Despite these advantages, ICP-MS faces several limitations in pharmaceutical applications. Sample introduction challenges persist, particularly with organic-rich matrices common in pharmaceutical formulations. These matrices can cause signal suppression, plasma instability, and carbon deposition on instrument components, necessitating specialized sample preparation protocols or alternative introduction systems.
Polyatomic interferences remain problematic, where molecular species formed in the plasma can have the same mass-to-charge ratio as analytes of interest. While collision/reaction cell technologies have mitigated many interferences, complete elimination remains elusive for certain element combinations, potentially affecting accuracy in complex pharmaceutical matrices.
Cost considerations present significant barriers to widespread adoption. High-end ICP-MS systems typically range from $200,000 to $500,000, with substantial ongoing expenses for argon gas, maintenance, and specialized operator training. This financial burden limits accessibility for smaller pharmaceutical companies and contract laboratories.
Sample preparation remains labor-intensive, requiring digestion procedures that can introduce contamination or analyte loss. The destructive nature of ICP-MS analysis means samples cannot be recovered, and the technique provides limited information about chemical speciation—a growing concern in pharmaceutical analysis where the toxicity of elements often depends on their chemical form rather than total concentration.
Regulatory compliance presents another challenge, with evolving standards requiring continuous method validation and verification. While ICP-MS meets current requirements for elemental impurity testing, pharmaceutical companies must maintain robust quality systems to ensure ongoing compliance with changing regulatory expectations and method performance requirements.
The technique excels in multi-elemental analysis capabilities, allowing simultaneous detection of numerous elements across the periodic table in a single analytical run. Modern instruments achieve impressive throughput, processing up to 30-40 samples per hour with minimal sample preparation requirements compared to older techniques like atomic absorption spectroscopy.
Despite these advantages, ICP-MS faces several limitations in pharmaceutical applications. Sample introduction challenges persist, particularly with organic-rich matrices common in pharmaceutical formulations. These matrices can cause signal suppression, plasma instability, and carbon deposition on instrument components, necessitating specialized sample preparation protocols or alternative introduction systems.
Polyatomic interferences remain problematic, where molecular species formed in the plasma can have the same mass-to-charge ratio as analytes of interest. While collision/reaction cell technologies have mitigated many interferences, complete elimination remains elusive for certain element combinations, potentially affecting accuracy in complex pharmaceutical matrices.
Cost considerations present significant barriers to widespread adoption. High-end ICP-MS systems typically range from $200,000 to $500,000, with substantial ongoing expenses for argon gas, maintenance, and specialized operator training. This financial burden limits accessibility for smaller pharmaceutical companies and contract laboratories.
Sample preparation remains labor-intensive, requiring digestion procedures that can introduce contamination or analyte loss. The destructive nature of ICP-MS analysis means samples cannot be recovered, and the technique provides limited information about chemical speciation—a growing concern in pharmaceutical analysis where the toxicity of elements often depends on their chemical form rather than total concentration.
Regulatory compliance presents another challenge, with evolving standards requiring continuous method validation and verification. While ICP-MS meets current requirements for elemental impurity testing, pharmaceutical companies must maintain robust quality systems to ensure ongoing compliance with changing regulatory expectations and method performance requirements.
Contemporary ICP-MS Protocols for Pharmaceutical Analysis
01 Calibration and standardization methods for ICP-MS
Various calibration and standardization methods are employed to ensure accurate and reliable results in ICP-MS analysis. These methods include the use of internal standards, external calibration curves, and standard reference materials. Proper calibration helps to compensate for matrix effects, instrument drift, and other factors that can affect measurement accuracy. Regular calibration verification is essential for maintaining quality control in ICP-MS analysis.- Calibration and standardization methods for ICP-MS: Various calibration and standardization techniques are essential for ensuring accurate and reliable ICP-MS measurements. These methods include the use of internal standards, external calibration curves, and standard addition methods. Proper calibration helps to compensate for matrix effects, instrument drift, and other factors that can affect measurement accuracy. Regular calibration verification using certified reference materials is crucial for maintaining quality control in ICP-MS analysis.
- Sample preparation and introduction systems: Effective sample preparation and introduction systems are critical for ICP-MS quality control. These include methods for sample digestion, dilution, and matrix modification to minimize interferences. Automated sample introduction systems can improve precision and reduce contamination risks. Techniques such as flow injection, laser ablation, and chromatographic separation coupled with ICP-MS provide enhanced sample handling capabilities and improved analytical performance for complex matrices.
- Interference reduction and elimination strategies: Various strategies are employed to reduce or eliminate spectral and non-spectral interferences in ICP-MS analysis. These include collision/reaction cell technology, cool plasma conditions, mathematical correction models, and high-resolution mass spectrometry. Effective interference management is essential for accurate trace element analysis, particularly in complex matrices. Quality control protocols typically include specific procedures for identifying and addressing common interferences encountered in different sample types.
- Automated quality control systems and software: Automated quality control systems and specialized software play a crucial role in modern ICP-MS analysis. These systems monitor instrument performance, validate results, and flag potential issues in real-time. Advanced software solutions incorporate statistical process control, automated calibration verification, and comprehensive data management capabilities. Integration with laboratory information management systems (LIMS) enhances traceability and compliance with regulatory requirements for quality control in analytical laboratories.
- Method validation and performance verification: Comprehensive method validation and regular performance verification are fundamental aspects of ICP-MS quality control. This includes assessment of method detection limits, quantification limits, linearity, accuracy, precision, and robustness. Proficiency testing, analysis of certified reference materials, and inter-laboratory comparisons are used to verify method performance. Quality control charts and statistical tools help monitor long-term instrument stability and method reliability, ensuring consistent analytical results across different samples and operating conditions.
02 Sample preparation techniques for ICP-MS quality control
Effective sample preparation is crucial for reliable ICP-MS analysis. This includes methods for digestion, dilution, and matrix modification to ensure samples are suitable for analysis. Techniques such as microwave digestion, acid dissolution, and filtration help to minimize interferences and contamination. Quality control measures during sample preparation include the use of procedural blanks, duplicate samples, and spiked samples to monitor recovery rates and identify potential sources of error.Expand Specific Solutions03 Interference reduction and elimination strategies
Various strategies are employed to reduce or eliminate interferences in ICP-MS analysis. These include the use of collision/reaction cells, mathematical correction models, and optimized instrument parameters. Spectral interferences can be addressed through high-resolution mass spectrometry or chemical separation techniques. Non-spectral interferences, such as matrix effects, can be minimized through sample dilution, matrix matching, or the use of internal standards. These approaches are essential for maintaining high quality control standards in ICP-MS analysis.Expand Specific Solutions04 Automated quality control systems and software
Automated systems and specialized software play a significant role in ICP-MS quality control. These systems can monitor instrument performance, flag anomalous results, and apply correction algorithms in real-time. Quality control software can track long-term instrument stability, generate control charts, and provide statistical analysis of quality control samples. Automation reduces human error and increases throughput while maintaining analytical quality. Integration with laboratory information management systems (LIMS) further enhances data integrity and traceability.Expand Specific Solutions05 Quality control protocols and reference materials
Comprehensive quality control protocols are essential for reliable ICP-MS analysis. These include the regular analysis of certified reference materials, quality control samples, and performance check solutions. Protocols typically specify the frequency of quality checks, acceptance criteria, and corrective actions when quality control failures occur. The use of certified reference materials that match sample matrices helps to validate analytical methods and verify instrument performance. Documentation of quality control results provides evidence of data reliability and facilitates regulatory compliance.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The ICP-MS pharmaceutical quality control market is in a growth phase, with increasing adoption driven by regulatory requirements for precise elemental analysis. The market size is expanding steadily, projected to reach significant value as pharmaceutical companies enhance their analytical capabilities. Technologically, the field shows varying maturity levels across players. Industry leaders Thermo Fisher Scientific and Agilent Technologies demonstrate advanced capabilities with comprehensive ICP-MS solutions, while Revvity Health Sciences and FUJIFILM are developing specialized applications. Pharmaceutical companies like Novartis, Chugai, and Genentech are integrating these technologies into their quality control workflows. Academic institutions including Zhejiang University and ETH Zurich contribute through research advancements, creating a competitive ecosystem balancing established providers and emerging specialized applications.
Thermo Fisher Scientific (Bremen) GmbH
Technical Solution: Thermo Fisher Scientific has developed advanced ICP-MS systems specifically optimized for pharmaceutical quality control, including their iCAP RQ and iCAP TQ ICP-MS platforms. These systems feature collision/reaction cell technology that effectively removes polyatomic interferences common in pharmaceutical matrices. Their proprietary QCell technology utilizes helium collision mode and kinetic energy discrimination to achieve detection limits in the ppt (parts-per-trillion) range for most elements. The company has also integrated specialized software solutions like Qtegra ISDS that streamline compliance with 21 CFR Part 11 requirements and provide comprehensive audit trails essential for pharmaceutical quality control. Their systems incorporate intelligent autodilution capabilities that automatically handle samples exceeding calibration ranges, reducing manual intervention and potential contamination risks. Thermo Fisher's ICP-MS solutions also feature specialized sample introduction systems designed to handle the diverse matrices encountered in pharmaceutical analysis, including organic solvents and high-salt solutions.
Strengths: Industry-leading sensitivity and detection limits; comprehensive compliance features for pharmaceutical regulations; robust interference removal technology; extensive application support specifically for pharmaceutical matrices. Weaknesses: Higher initial investment cost compared to some competitors; complex systems may require specialized training; consumable costs can be significant for routine high-volume testing.
Agilent Technologies, Inc.
Technical Solution: Agilent Technologies has pioneered several innovations in ICP-MS technology for pharmaceutical quality control, most notably their 7900 and 8900 ICP-MS systems with patented HMI (High Matrix Introduction) technology. This approach allows for direct analysis of samples with up to 3% total dissolved solids without significant dilution, addressing a key challenge in pharmaceutical sample preparation. Their unique Triple Quadrupole ICP-MS technology enables MS/MS mode operation that provides unprecedented control over polyatomic and isobaric interferences through reaction chemistry. Agilent has developed specialized pharmaceutical workflows incorporating their ICP-MS MassHunter software that streamlines method development, validation, and reporting according to USP <232>, <233>, and ICH Q3D guidelines. The company's Ultra High Matrix Introduction (UHMI) technology dilutes only the sample matrix while maintaining analyte signal strength, improving detection limits in complex pharmaceutical formulations. Additionally, Agilent offers integrated speciation analysis capabilities through HPLC-ICP-MS hyphenation, critical for differentiating between toxic and non-toxic forms of elements like arsenic and mercury in pharmaceutical products.
Strengths: Superior matrix tolerance capabilities; excellent interference management through triple quadrupole technology; comprehensive software solutions specifically designed for pharmaceutical compliance; extensive application support network. Weaknesses: Premium pricing structure; complex systems require significant expertise to fully utilize advanced features; higher operating costs for some consumables compared to competitors.
Critical Patents and Innovations in ICP-MS Technology
Inductively coupled plasma mass spectrometer
PatentActiveUS7671329B2
Innovation
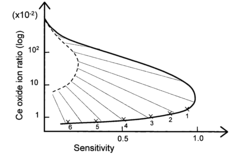
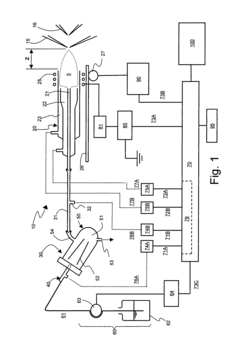
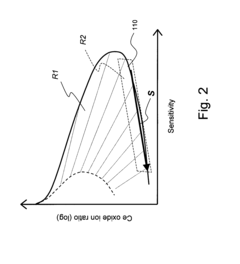
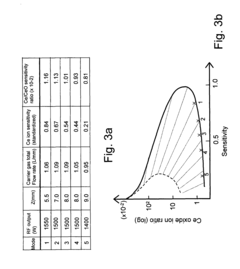
- An ICP-MS system with a control device that adjusts the amount of liquid drops, carrier gas flow rate, RF power output, and plasma torch position to optimize ion sensitivity, allowing for continuous analysis of samples with varying concentrations without additional dilution equipment or lengthy procedures, thereby maintaining high sensitivity and precision.
Inductively coupled plasma mass spectrometry (ICP-MS) with ion trapping
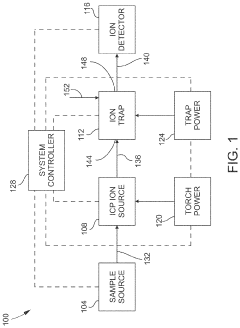
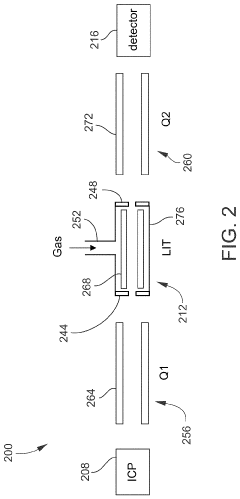
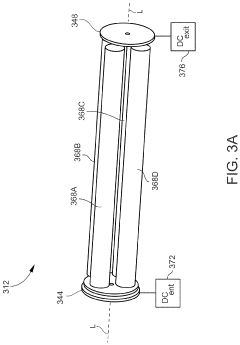
PatentActiveUS11443933B1
Innovation
- Incorporating an ion trap, such as a linear ion trap, into the ICP-MS system to confine and mass-selectively eject ions, allowing for the simultaneous analysis of multiple elements from transient signals by preventing ion exit and entry during a confinement period and transmitting selected ions to a detector for measurement.
Regulatory Compliance and Validation Requirements
Pharmaceutical manufacturers utilizing ICP-MS technology must navigate a complex regulatory landscape that varies by region but maintains consistent core principles. The FDA in the United States, the EMA in Europe, and the NMPA in China all require strict adherence to Good Manufacturing Practices (GMP) for analytical methods used in quality control. For ICP-MS specifically, compliance with USP <232>, <233>, and ICH Q3D guidelines is mandatory when analyzing elemental impurities in pharmaceutical products.
Method validation represents a critical regulatory requirement for ICP-MS implementation. Pharmaceutical companies must demonstrate that their analytical procedures are suitable for their intended purpose through comprehensive validation protocols. These protocols typically include assessments of specificity, linearity, range, accuracy, precision, detection limit, quantitation limit, and robustness. The validation process must follow guidelines outlined in ICH Q2(R1) and be thoroughly documented to withstand regulatory scrutiny during inspections.
System suitability testing constitutes another essential regulatory component, requiring regular verification that the ICP-MS instrument performs within established parameters before sample analysis. This includes daily calibration checks, sensitivity tests, and mass calibration verification. Manufacturers must establish acceptance criteria for these tests and maintain detailed records of all system suitability results.
Data integrity requirements have become increasingly stringent in recent years, with regulatory bodies focusing on the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). ICP-MS systems must incorporate appropriate electronic records management with audit trails, user access controls, and data backup procedures that comply with 21 CFR Part 11 or EU Annex 11 requirements.
Ongoing performance verification represents another regulatory expectation, requiring pharmaceutical manufacturers to implement regular proficiency testing, participate in interlaboratory comparison programs, and conduct periodic revalidation of their ICP-MS methods. This ensures continued reliability of analytical results and demonstrates commitment to maintaining compliance with evolving regulatory standards.
Change control procedures must be established to manage modifications to validated ICP-MS methods, including changes to equipment, software, reagents, or analytical parameters. Any significant change requires appropriate validation and regulatory notification depending on the nature and impact of the modification.
Method validation represents a critical regulatory requirement for ICP-MS implementation. Pharmaceutical companies must demonstrate that their analytical procedures are suitable for their intended purpose through comprehensive validation protocols. These protocols typically include assessments of specificity, linearity, range, accuracy, precision, detection limit, quantitation limit, and robustness. The validation process must follow guidelines outlined in ICH Q2(R1) and be thoroughly documented to withstand regulatory scrutiny during inspections.
System suitability testing constitutes another essential regulatory component, requiring regular verification that the ICP-MS instrument performs within established parameters before sample analysis. This includes daily calibration checks, sensitivity tests, and mass calibration verification. Manufacturers must establish acceptance criteria for these tests and maintain detailed records of all system suitability results.
Data integrity requirements have become increasingly stringent in recent years, with regulatory bodies focusing on the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). ICP-MS systems must incorporate appropriate electronic records management with audit trails, user access controls, and data backup procedures that comply with 21 CFR Part 11 or EU Annex 11 requirements.
Ongoing performance verification represents another regulatory expectation, requiring pharmaceutical manufacturers to implement regular proficiency testing, participate in interlaboratory comparison programs, and conduct periodic revalidation of their ICP-MS methods. This ensures continued reliability of analytical results and demonstrates commitment to maintaining compliance with evolving regulatory standards.
Change control procedures must be established to manage modifications to validated ICP-MS methods, including changes to equipment, software, reagents, or analytical parameters. Any significant change requires appropriate validation and regulatory notification depending on the nature and impact of the modification.
Cost-Benefit Analysis of ICP-MS Implementation
Implementing ICP-MS technology for pharmaceutical quality control requires substantial initial investment balanced against long-term operational benefits. The capital expenditure typically ranges from $250,000 to $500,000 for a standard ICP-MS system, with additional costs for specialized pharmaceutical-grade equipment modifications. Laboratory infrastructure adaptations, including clean room facilities and specialized gas handling systems, may add $50,000-$100,000 to implementation costs.
Training expenses represent another significant investment, requiring specialized personnel development programs costing approximately $10,000-$20,000 per analyst. Ongoing operational costs include high-purity gases ($5,000-$10,000 annually), replacement parts ($15,000-$25,000 annually), and maintenance contracts ($20,000-$30,000 annually).
Against these expenses, pharmaceutical manufacturers must weigh considerable benefits. Enhanced detection capabilities allow identification of trace contaminants at parts-per-trillion levels, significantly exceeding traditional methods like atomic absorption spectroscopy. This superior sensitivity translates to improved product safety profiles and reduced risk of regulatory non-compliance.
Efficiency gains represent substantial cost savings, with ICP-MS reducing analysis time by 60-70% compared to conventional methods. Multi-element analysis capabilities further enhance throughput, allowing simultaneous detection of numerous elements in a single analytical run. These operational improvements typically yield labor cost reductions of $50,000-$100,000 annually for medium-sized pharmaceutical operations.
Regulatory compliance benefits provide additional value, as ICP-MS meets or exceeds requirements specified in USP <232>, <233>, ICH Q3D, and FDA guidelines for elemental impurity testing. This compliance advantage reduces regulatory submission delays and minimizes the risk of costly product recalls.
Return on investment calculations indicate most pharmaceutical manufacturers achieve break-even within 2-3 years of implementation. Long-term financial modeling demonstrates 5-year ROI ranging from 150-300%, depending on facility size and testing volume. These calculations typically exclude less quantifiable benefits such as enhanced brand reputation and reduced liability exposure from improved quality control processes.
For smaller pharmaceutical operations, shared resource models or contract testing services may provide more economically viable alternatives to full implementation, with per-sample costs ranging from $100-$300 depending on analytical requirements and volume commitments.
Training expenses represent another significant investment, requiring specialized personnel development programs costing approximately $10,000-$20,000 per analyst. Ongoing operational costs include high-purity gases ($5,000-$10,000 annually), replacement parts ($15,000-$25,000 annually), and maintenance contracts ($20,000-$30,000 annually).
Against these expenses, pharmaceutical manufacturers must weigh considerable benefits. Enhanced detection capabilities allow identification of trace contaminants at parts-per-trillion levels, significantly exceeding traditional methods like atomic absorption spectroscopy. This superior sensitivity translates to improved product safety profiles and reduced risk of regulatory non-compliance.
Efficiency gains represent substantial cost savings, with ICP-MS reducing analysis time by 60-70% compared to conventional methods. Multi-element analysis capabilities further enhance throughput, allowing simultaneous detection of numerous elements in a single analytical run. These operational improvements typically yield labor cost reductions of $50,000-$100,000 annually for medium-sized pharmaceutical operations.
Regulatory compliance benefits provide additional value, as ICP-MS meets or exceeds requirements specified in USP <232>, <233>, ICH Q3D, and FDA guidelines for elemental impurity testing. This compliance advantage reduces regulatory submission delays and minimizes the risk of costly product recalls.
Return on investment calculations indicate most pharmaceutical manufacturers achieve break-even within 2-3 years of implementation. Long-term financial modeling demonstrates 5-year ROI ranging from 150-300%, depending on facility size and testing volume. These calculations typically exclude less quantifiable benefits such as enhanced brand reputation and reduced liability exposure from improved quality control processes.
For smaller pharmaceutical operations, shared resource models or contract testing services may provide more economically viable alternatives to full implementation, with per-sample costs ranging from $100-$300 depending on analytical requirements and volume commitments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!