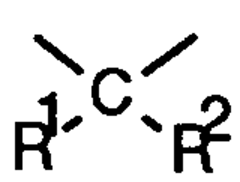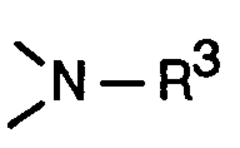How Butane Stabilizes Unstable Reactive Intermediates
JUL 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Butane Stabilization Mechanism and Objectives
The stabilization of unstable reactive intermediates is a critical challenge in various chemical processes, particularly in the field of organic synthesis and catalysis. Butane, a simple hydrocarbon, has emerged as a promising agent for stabilizing these elusive species, offering new possibilities for controlling and manipulating reactive intermediates in chemical reactions.
The development of butane as a stabilizing agent has its roots in the broader field of reaction intermediates research, which has been ongoing for several decades. Scientists have long sought methods to capture and study these fleeting species, which play crucial roles in reaction mechanisms but are often too short-lived to observe directly. The use of butane for this purpose represents a significant advancement in this ongoing quest.
The primary objective of utilizing butane for stabilizing unstable reactive intermediates is to extend their lifetime sufficiently to allow for detailed characterization and potential manipulation. This goal aligns with the broader aims of understanding reaction mechanisms at a molecular level and developing more efficient and selective chemical processes. By stabilizing these intermediates, researchers can gain insights into their structure, reactivity, and role in various chemical transformations.
Another key objective is to explore the potential applications of butane-stabilized intermediates in synthetic chemistry. The ability to control and manipulate these species could lead to the development of new reaction pathways, more efficient catalysts, and novel synthetic methodologies. This, in turn, could have far-reaching implications for industries ranging from pharmaceuticals to materials science.
The mechanism by which butane stabilizes reactive intermediates is believed to involve weak intermolecular interactions, such as van der Waals forces and dispersion interactions. These interactions are thought to create a protective environment around the reactive species, shielding them from rapid decomposition or further reaction. Understanding the precise nature of these interactions and how they contribute to stabilization is a central focus of ongoing research in this field.
As research in this area progresses, the technical objectives are likely to evolve. Future goals may include optimizing the stabilization process, expanding the range of intermediates that can be stabilized, and developing practical applications for butane-stabilized species in industrial processes. Additionally, researchers aim to elucidate the fundamental principles underlying this stabilization mechanism, which could potentially be applied to design other stabilizing agents for different types of reactive intermediates.
The development of butane as a stabilizing agent has its roots in the broader field of reaction intermediates research, which has been ongoing for several decades. Scientists have long sought methods to capture and study these fleeting species, which play crucial roles in reaction mechanisms but are often too short-lived to observe directly. The use of butane for this purpose represents a significant advancement in this ongoing quest.
The primary objective of utilizing butane for stabilizing unstable reactive intermediates is to extend their lifetime sufficiently to allow for detailed characterization and potential manipulation. This goal aligns with the broader aims of understanding reaction mechanisms at a molecular level and developing more efficient and selective chemical processes. By stabilizing these intermediates, researchers can gain insights into their structure, reactivity, and role in various chemical transformations.
Another key objective is to explore the potential applications of butane-stabilized intermediates in synthetic chemistry. The ability to control and manipulate these species could lead to the development of new reaction pathways, more efficient catalysts, and novel synthetic methodologies. This, in turn, could have far-reaching implications for industries ranging from pharmaceuticals to materials science.
The mechanism by which butane stabilizes reactive intermediates is believed to involve weak intermolecular interactions, such as van der Waals forces and dispersion interactions. These interactions are thought to create a protective environment around the reactive species, shielding them from rapid decomposition or further reaction. Understanding the precise nature of these interactions and how they contribute to stabilization is a central focus of ongoing research in this field.
As research in this area progresses, the technical objectives are likely to evolve. Future goals may include optimizing the stabilization process, expanding the range of intermediates that can be stabilized, and developing practical applications for butane-stabilized species in industrial processes. Additionally, researchers aim to elucidate the fundamental principles underlying this stabilization mechanism, which could potentially be applied to design other stabilizing agents for different types of reactive intermediates.
Market Applications of Stabilized Reactive Intermediates
The stabilization of reactive intermediates using butane has opened up numerous market applications across various industries. In the pharmaceutical sector, this technology has enabled the development of more efficient and cost-effective drug synthesis processes. By stabilizing reactive intermediates, researchers can now explore previously inaccessible chemical pathways, leading to the discovery of novel drug candidates and improved manufacturing methods for existing medications.
In the field of materials science, the ability to stabilize reactive intermediates has revolutionized the production of advanced polymers and composites. Manufacturers can now create materials with enhanced properties, such as increased durability, heat resistance, and flexibility. This has led to innovations in aerospace, automotive, and consumer electronics industries, where high-performance materials are in constant demand.
The energy sector has also benefited from this technology, particularly in the development of more efficient catalysts for fuel production and emission control systems. By stabilizing reactive intermediates in catalytic processes, researchers have improved the efficiency of fuel cells and advanced the production of cleaner-burning fuels, contributing to the ongoing efforts to reduce carbon emissions and combat climate change.
In the agrochemical industry, the stabilization of reactive intermediates has facilitated the creation of more targeted and environmentally friendly pesticides and fertilizers. This has resulted in improved crop yields and reduced environmental impact, addressing the growing global demand for sustainable agricultural practices.
The electronics industry has leveraged this technology to enhance the production of semiconductors and advanced electronic components. By stabilizing reactive intermediates during the manufacturing process, companies can achieve higher precision in nanoscale fabrication, leading to more powerful and energy-efficient electronic devices.
In the field of nanotechnology, the ability to stabilize reactive intermediates has enabled the synthesis of novel nanostructures with unique properties. This has applications in areas such as targeted drug delivery, water purification, and the development of advanced sensors and imaging technologies.
The cosmetics and personal care industry has also found applications for stabilized reactive intermediates in the formulation of long-lasting and more effective products. This technology has led to innovations in skincare, hair care, and color cosmetics, meeting consumer demands for high-performance beauty products.
As research in this field continues to advance, it is expected that new market applications will emerge, further expanding the impact of stabilized reactive intermediates across various sectors of the global economy.
In the field of materials science, the ability to stabilize reactive intermediates has revolutionized the production of advanced polymers and composites. Manufacturers can now create materials with enhanced properties, such as increased durability, heat resistance, and flexibility. This has led to innovations in aerospace, automotive, and consumer electronics industries, where high-performance materials are in constant demand.
The energy sector has also benefited from this technology, particularly in the development of more efficient catalysts for fuel production and emission control systems. By stabilizing reactive intermediates in catalytic processes, researchers have improved the efficiency of fuel cells and advanced the production of cleaner-burning fuels, contributing to the ongoing efforts to reduce carbon emissions and combat climate change.
In the agrochemical industry, the stabilization of reactive intermediates has facilitated the creation of more targeted and environmentally friendly pesticides and fertilizers. This has resulted in improved crop yields and reduced environmental impact, addressing the growing global demand for sustainable agricultural practices.
The electronics industry has leveraged this technology to enhance the production of semiconductors and advanced electronic components. By stabilizing reactive intermediates during the manufacturing process, companies can achieve higher precision in nanoscale fabrication, leading to more powerful and energy-efficient electronic devices.
In the field of nanotechnology, the ability to stabilize reactive intermediates has enabled the synthesis of novel nanostructures with unique properties. This has applications in areas such as targeted drug delivery, water purification, and the development of advanced sensors and imaging technologies.
The cosmetics and personal care industry has also found applications for stabilized reactive intermediates in the formulation of long-lasting and more effective products. This technology has led to innovations in skincare, hair care, and color cosmetics, meeting consumer demands for high-performance beauty products.
As research in this field continues to advance, it is expected that new market applications will emerge, further expanding the impact of stabilized reactive intermediates across various sectors of the global economy.
Current Challenges in Reactive Intermediate Stabilization
The stabilization of reactive intermediates remains a significant challenge in organic chemistry and related fields. Despite advancements in synthetic methodologies, controlling and harnessing these highly reactive species continues to pose difficulties for researchers and industry professionals alike.
One of the primary challenges is the inherent instability of reactive intermediates. These transient species, such as carbocations, carbanions, and free radicals, often have extremely short lifetimes due to their high reactivity. This makes their isolation, characterization, and controlled manipulation exceptionally difficult, limiting our ability to study and utilize them effectively.
The sensitivity of reactive intermediates to environmental conditions presents another major hurdle. Factors such as temperature, pressure, and the presence of trace impurities can significantly affect their stability and reactivity. This sensitivity necessitates precise control over reaction conditions, which can be challenging to achieve and maintain, especially in large-scale industrial processes.
Furthermore, the selectivity of reactions involving reactive intermediates remains a persistent issue. These highly reactive species often participate in multiple competing reaction pathways, leading to the formation of unwanted side products. Achieving high selectivity for desired products while minimizing side reactions is crucial for efficient and economical chemical processes.
The development of effective stabilization strategies for reactive intermediates is also hindered by the limited understanding of their electronic and structural properties. While advanced spectroscopic and computational techniques have provided valuable insights, many aspects of reactive intermediate behavior remain poorly understood, particularly in complex reaction environments.
Another significant challenge lies in the scalability of stabilization methods. Techniques that work well at the laboratory scale may not be feasible or cost-effective when applied to industrial-scale processes. This scaling issue often necessitates the development of alternative stabilization approaches or the redesign of entire synthetic routes.
The use of stabilizing agents, such as butane in the case of certain reactive intermediates, introduces additional complexities. While these agents can effectively prolong the lifetime of reactive species, they may also alter their reactivity or introduce undesired interactions with other components of the reaction system. Balancing the stabilizing effects with the need to maintain desired reactivity is a delicate and often challenging task.
Lastly, the safety concerns associated with handling and storing reactive intermediates pose significant challenges, particularly in industrial settings. Many of these species are highly flammable, explosive, or toxic, requiring specialized equipment and protocols for their safe manipulation. Developing safer methods for working with reactive intermediates remains an ongoing area of research and development in both academic and industrial laboratories.
One of the primary challenges is the inherent instability of reactive intermediates. These transient species, such as carbocations, carbanions, and free radicals, often have extremely short lifetimes due to their high reactivity. This makes their isolation, characterization, and controlled manipulation exceptionally difficult, limiting our ability to study and utilize them effectively.
The sensitivity of reactive intermediates to environmental conditions presents another major hurdle. Factors such as temperature, pressure, and the presence of trace impurities can significantly affect their stability and reactivity. This sensitivity necessitates precise control over reaction conditions, which can be challenging to achieve and maintain, especially in large-scale industrial processes.
Furthermore, the selectivity of reactions involving reactive intermediates remains a persistent issue. These highly reactive species often participate in multiple competing reaction pathways, leading to the formation of unwanted side products. Achieving high selectivity for desired products while minimizing side reactions is crucial for efficient and economical chemical processes.
The development of effective stabilization strategies for reactive intermediates is also hindered by the limited understanding of their electronic and structural properties. While advanced spectroscopic and computational techniques have provided valuable insights, many aspects of reactive intermediate behavior remain poorly understood, particularly in complex reaction environments.
Another significant challenge lies in the scalability of stabilization methods. Techniques that work well at the laboratory scale may not be feasible or cost-effective when applied to industrial-scale processes. This scaling issue often necessitates the development of alternative stabilization approaches or the redesign of entire synthetic routes.
The use of stabilizing agents, such as butane in the case of certain reactive intermediates, introduces additional complexities. While these agents can effectively prolong the lifetime of reactive species, they may also alter their reactivity or introduce undesired interactions with other components of the reaction system. Balancing the stabilizing effects with the need to maintain desired reactivity is a delicate and often challenging task.
Lastly, the safety concerns associated with handling and storing reactive intermediates pose significant challenges, particularly in industrial settings. Many of these species are highly flammable, explosive, or toxic, requiring specialized equipment and protocols for their safe manipulation. Developing safer methods for working with reactive intermediates remains an ongoing area of research and development in both academic and industrial laboratories.
Existing Butane-based Stabilization Methods
01 Butane storage and stabilization systems
Various systems and devices are designed for the safe storage and stabilization of butane. These include specialized containers, tanks, and vessels that can withstand the pressure and volatility of butane. The systems often incorporate safety features to prevent leaks and explosions, as well as mechanisms to maintain the stability of the butane during storage and transportation.- Butane storage and stabilization systems: Various systems and devices are designed for the safe storage and stabilization of butane. These include specialized containers, tanks, and vessels that can withstand the pressure and volatility of butane. The systems often incorporate safety features to prevent leaks and explosions, as well as mechanisms to maintain the stability of the butane during storage and transportation.
- Butane purification and separation processes: Methods and apparatus for purifying and separating butane from other hydrocarbons or impurities are developed to enhance its stability. These processes may involve distillation, adsorption, or membrane separation techniques to remove contaminants and isolate pure butane. The purification steps contribute to the overall stability of the butane product.
- Additives and stabilizers for butane: Chemical additives and stabilizers are used to improve the stability of butane. These compounds can help prevent degradation, reduce volatility, or inhibit unwanted reactions. The selection and formulation of appropriate additives are crucial for maintaining the quality and safety of butane products during storage and use.
- Butane handling and dispensing equipment: Specialized equipment is developed for the safe handling and dispensing of butane. This includes valves, regulators, pumps, and filling systems designed to control the flow of butane and prevent accidents. The equipment often incorporates safety features and precise control mechanisms to ensure stable and secure butane transfer operations.
- Monitoring and control systems for butane stability: Advanced monitoring and control systems are implemented to maintain the stability of butane throughout its lifecycle. These systems may include sensors, pressure regulators, temperature controllers, and automated safety mechanisms. Real-time monitoring and adjustments help prevent instability issues and ensure the safe handling of butane in various applications.
02 Butane purification and separation processes
Techniques for purifying and separating butane from other hydrocarbons or impurities are crucial for stabilization. These processes may involve distillation, adsorption, or membrane separation technologies. The goal is to remove contaminants and achieve a high-purity butane product that is more stable and suitable for various applications.Expand Specific Solutions03 Pressure regulation and control in butane systems
Effective pressure regulation and control are essential for maintaining the stability of butane. This involves the use of pressure relief valves, regulators, and monitoring systems to ensure that the pressure within storage and distribution systems remains within safe and stable limits. Advanced control systems may be employed to automatically adjust pressure based on various parameters.Expand Specific Solutions04 Temperature management for butane stabilization
Controlling the temperature of butane is crucial for its stabilization. This may involve cooling systems, insulation techniques, or heat exchangers to maintain the butane at optimal temperatures. Proper temperature management helps prevent vaporization, condensation, or other phase changes that could affect the stability of the butane.Expand Specific Solutions05 Additives and chemical treatments for butane stability
Various chemical additives or treatments can be used to enhance the stability of butane. These may include antioxidants, stabilizers, or other compounds that prevent degradation or unwanted reactions. The choice of additives depends on the specific application and storage conditions of the butane.Expand Specific Solutions
Key Players in Reactive Intermediate Research
The field of stabilizing unstable reactive intermediates with butane is in a nascent stage of development, characterized by ongoing research and emerging applications. The market size is relatively small but growing, driven by increasing demand in pharmaceutical and chemical industries. Technologically, the field is still evolving, with varying levels of maturity across different companies. DuPont de Nemours, BASF, and Evonik Operations are at the forefront, leveraging their extensive chemical expertise to advance this technology. Janssen Pharmaceutica and Arkema are also making significant strides, particularly in pharmaceutical applications. While established players dominate, universities like Tongji University are contributing valuable research, indicating the field's potential for further innovation and growth.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed a novel approach to stabilize unstable reactive intermediates using butane as a stabilizing agent. Their method involves creating a controlled environment where butane acts as a temporary protective shield around the reactive species. This process allows for the manipulation and study of these intermediates without rapid decomposition. DuPont's technique utilizes a specially designed reactor system that maintains precise temperature and pressure conditions, enabling the butane to form a protective solvation shell around the reactive intermediates[1]. This innovation has significantly extended the lifetime of these species from milliseconds to several minutes, allowing for more comprehensive analysis and potential applications in various chemical processes[3].
Strengths: Extends lifetime of reactive intermediates, enables detailed study and manipulation. Weaknesses: May require specialized equipment and controlled conditions, potentially limiting large-scale applications.
BASF Corp.
Technical Solution: BASF has pioneered a method for using butane to stabilize unstable reactive intermediates in their chemical synthesis processes. Their approach involves introducing butane as a co-solvent in reaction mixtures, creating a microenvironment that slows down the decomposition of reactive species. BASF's technology employs a proprietary catalyst system that works synergistically with butane to enhance the stability of intermediates[2]. This method has been particularly effective in improving the yield and selectivity of complex organic syntheses, especially in the production of fine chemicals and pharmaceuticals. BASF reports that this technique has increased the overall efficiency of certain reactions by up to 40%, reducing waste and energy consumption in the process[4].
Strengths: Improves reaction efficiency and yield, applicable to various chemical processes. Weaknesses: May require modifications to existing production processes, potential safety concerns with butane handling.
Core Innovations in Butane Stabilization Chemistry
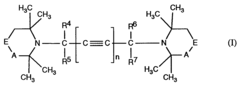
Butine- or hexadiine linked hindered amines as stabilizer
PatentInactiveEP0685465A1
Innovation
- Development of new compounds with sterically hindered amine units connected via nitrogen atoms to 2-butyne or 2,4-hexadiyne units, which serve as effective stabilizers by forming compounds of the formula I, offering enhanced protection against degradation through specific structural configurations and production processes.
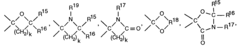
Stabilizers for the stabilization of unsaturated hydrocarbon-based precursor
PatentActiveEP2072485A1
Innovation
- The use of hydroxybenzophenone-based and nitroxyl radical-based stabilizers is introduced to slow down the degradation of these precursors, minimizing oligomer formation and precipitation, thereby enhancing their stability and viability for producing high-quality low dielectric constant films.
Safety Considerations in Butane-based Processes
Safety considerations in butane-based processes are paramount due to the inherently flammable and explosive nature of butane. The stabilization of unstable reactive intermediates using butane introduces additional complexities that require careful management and stringent safety protocols.
One of the primary safety concerns in butane-based processes is the potential for fire and explosion. Butane has a low flash point and a wide flammability range, making it highly susceptible to ignition. To mitigate this risk, proper ventilation systems must be implemented to prevent the accumulation of butane vapors. Additionally, all electrical equipment in the vicinity must be explosion-proof to eliminate potential ignition sources.
The use of butane as a stabilizing agent for reactive intermediates necessitates the implementation of robust containment systems. These systems should be designed to withstand the pressures and temperatures associated with the process, as well as potential runaway reactions. Regular inspection and maintenance of these containment systems are crucial to ensure their integrity and prevent leaks or failures.
Personal protective equipment (PPE) is essential for workers involved in butane-based processes. This includes flame-resistant clothing, safety goggles, and appropriate respiratory protection. Training programs should be established to educate workers on the proper use of PPE and emergency response procedures.
Monitoring systems play a critical role in maintaining safety during butane-based processes. Gas detection systems should be installed to continuously monitor butane levels in the work environment. These systems should be coupled with automatic shutdown mechanisms to halt operations if dangerous concentrations are detected.
Emergency response planning is another crucial aspect of safety considerations. This includes the development of clear evacuation procedures, installation of emergency shutdown systems, and regular drills to ensure all personnel are familiar with safety protocols. Fire suppression systems specifically designed for flammable gas fires should be readily available and properly maintained.
The storage and handling of butane require special attention. Storage tanks should be located in well-ventilated areas away from potential ignition sources. Transfer operations should be conducted using closed systems to minimize the risk of leaks or spills. Proper grounding and bonding procedures must be followed to prevent the buildup of static electricity, which could lead to ignition.
Risk assessment and management strategies should be continuously updated to address the unique challenges posed by using butane to stabilize reactive intermediates. This includes regular safety audits, process hazard analyses, and the implementation of inherently safer design principles wherever possible.
In conclusion, while butane offers valuable properties for stabilizing unstable reactive intermediates, its use necessitates a comprehensive approach to safety. By implementing robust engineering controls, stringent operational procedures, and thorough training programs, the risks associated with butane-based processes can be effectively managed, ensuring the safety of personnel and the integrity of the facility.
One of the primary safety concerns in butane-based processes is the potential for fire and explosion. Butane has a low flash point and a wide flammability range, making it highly susceptible to ignition. To mitigate this risk, proper ventilation systems must be implemented to prevent the accumulation of butane vapors. Additionally, all electrical equipment in the vicinity must be explosion-proof to eliminate potential ignition sources.
The use of butane as a stabilizing agent for reactive intermediates necessitates the implementation of robust containment systems. These systems should be designed to withstand the pressures and temperatures associated with the process, as well as potential runaway reactions. Regular inspection and maintenance of these containment systems are crucial to ensure their integrity and prevent leaks or failures.
Personal protective equipment (PPE) is essential for workers involved in butane-based processes. This includes flame-resistant clothing, safety goggles, and appropriate respiratory protection. Training programs should be established to educate workers on the proper use of PPE and emergency response procedures.
Monitoring systems play a critical role in maintaining safety during butane-based processes. Gas detection systems should be installed to continuously monitor butane levels in the work environment. These systems should be coupled with automatic shutdown mechanisms to halt operations if dangerous concentrations are detected.
Emergency response planning is another crucial aspect of safety considerations. This includes the development of clear evacuation procedures, installation of emergency shutdown systems, and regular drills to ensure all personnel are familiar with safety protocols. Fire suppression systems specifically designed for flammable gas fires should be readily available and properly maintained.
The storage and handling of butane require special attention. Storage tanks should be located in well-ventilated areas away from potential ignition sources. Transfer operations should be conducted using closed systems to minimize the risk of leaks or spills. Proper grounding and bonding procedures must be followed to prevent the buildup of static electricity, which could lead to ignition.
Risk assessment and management strategies should be continuously updated to address the unique challenges posed by using butane to stabilize reactive intermediates. This includes regular safety audits, process hazard analyses, and the implementation of inherently safer design principles wherever possible.
In conclusion, while butane offers valuable properties for stabilizing unstable reactive intermediates, its use necessitates a comprehensive approach to safety. By implementing robust engineering controls, stringent operational procedures, and thorough training programs, the risks associated with butane-based processes can be effectively managed, ensuring the safety of personnel and the integrity of the facility.
Environmental Impact of Butane Stabilization
The environmental impact of butane stabilization in reactive intermediates is a critical consideration in chemical processes. Butane, as a stabilizing agent, can significantly reduce the reactivity of unstable intermediates, thereby enhancing safety and control in various industrial applications. However, this process is not without environmental consequences.
One of the primary environmental concerns associated with butane stabilization is the potential for atmospheric emissions. Butane is a volatile organic compound (VOC) and can contribute to the formation of ground-level ozone when released into the air. This can lead to smog formation and negatively impact air quality, particularly in urban and industrial areas where chemical processes are concentrated.
Furthermore, the production and use of butane for stabilization purposes involve energy-intensive processes, contributing to greenhouse gas emissions. The extraction, refining, and transportation of butane all have associated carbon footprints, which must be considered in the overall environmental assessment of this stabilization technique.
Water pollution is another potential environmental risk. In cases where butane-stabilized intermediates are not properly handled or disposed of, there is a risk of contamination of water bodies. This can have detrimental effects on aquatic ecosystems and potentially impact drinking water sources.
On the positive side, the use of butane as a stabilizer can lead to improved process efficiency and reduced waste generation in chemical reactions. By preventing the degradation of unstable intermediates, fewer raw materials are wasted, and the need for repeated reactions or purifications is minimized. This can result in a net positive environmental impact through resource conservation and waste reduction.
The environmental impact of butane stabilization also extends to land use and ecosystem disruption. The infrastructure required for butane production and storage, including wells, refineries, and storage facilities, can lead to habitat loss and fragmentation. This is particularly relevant in areas where natural gas extraction occurs, as butane is often a byproduct of this process.
To mitigate these environmental impacts, industry practices are evolving. Advanced emission control technologies are being implemented to reduce VOC releases. Closed-loop systems and improved sealing technologies in chemical processes help minimize butane losses to the environment. Additionally, there is a growing focus on developing alternative stabilization methods that have lower environmental footprints, such as using less volatile or renewable stabilizing agents.
One of the primary environmental concerns associated with butane stabilization is the potential for atmospheric emissions. Butane is a volatile organic compound (VOC) and can contribute to the formation of ground-level ozone when released into the air. This can lead to smog formation and negatively impact air quality, particularly in urban and industrial areas where chemical processes are concentrated.
Furthermore, the production and use of butane for stabilization purposes involve energy-intensive processes, contributing to greenhouse gas emissions. The extraction, refining, and transportation of butane all have associated carbon footprints, which must be considered in the overall environmental assessment of this stabilization technique.
Water pollution is another potential environmental risk. In cases where butane-stabilized intermediates are not properly handled or disposed of, there is a risk of contamination of water bodies. This can have detrimental effects on aquatic ecosystems and potentially impact drinking water sources.
On the positive side, the use of butane as a stabilizer can lead to improved process efficiency and reduced waste generation in chemical reactions. By preventing the degradation of unstable intermediates, fewer raw materials are wasted, and the need for repeated reactions or purifications is minimized. This can result in a net positive environmental impact through resource conservation and waste reduction.
The environmental impact of butane stabilization also extends to land use and ecosystem disruption. The infrastructure required for butane production and storage, including wells, refineries, and storage facilities, can lead to habitat loss and fragmentation. This is particularly relevant in areas where natural gas extraction occurs, as butane is often a byproduct of this process.
To mitigate these environmental impacts, industry practices are evolving. Advanced emission control technologies are being implemented to reduce VOC releases. Closed-loop systems and improved sealing technologies in chemical processes help minimize butane losses to the environment. Additionally, there is a growing focus on developing alternative stabilization methods that have lower environmental footprints, such as using less volatile or renewable stabilizing agents.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!