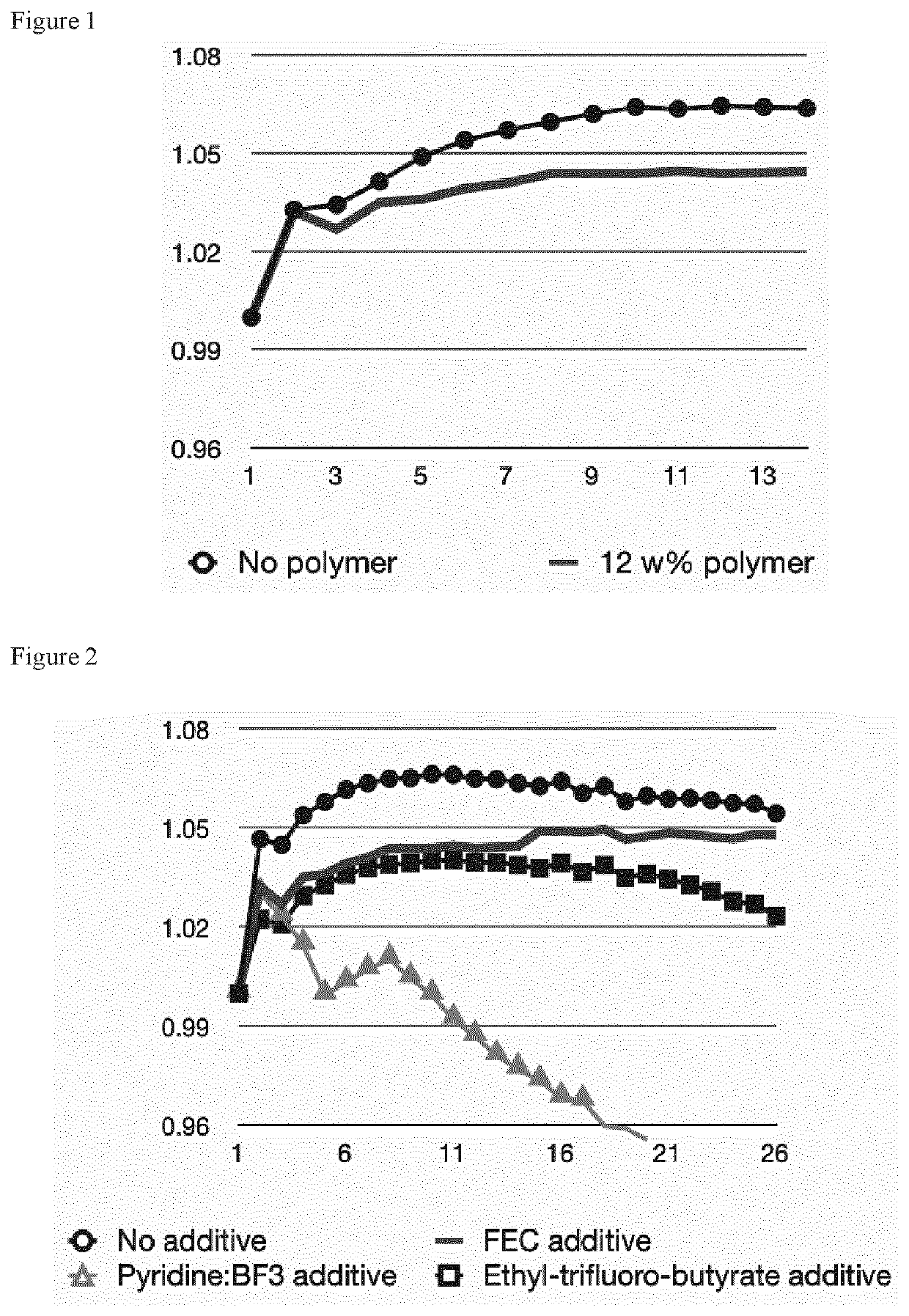
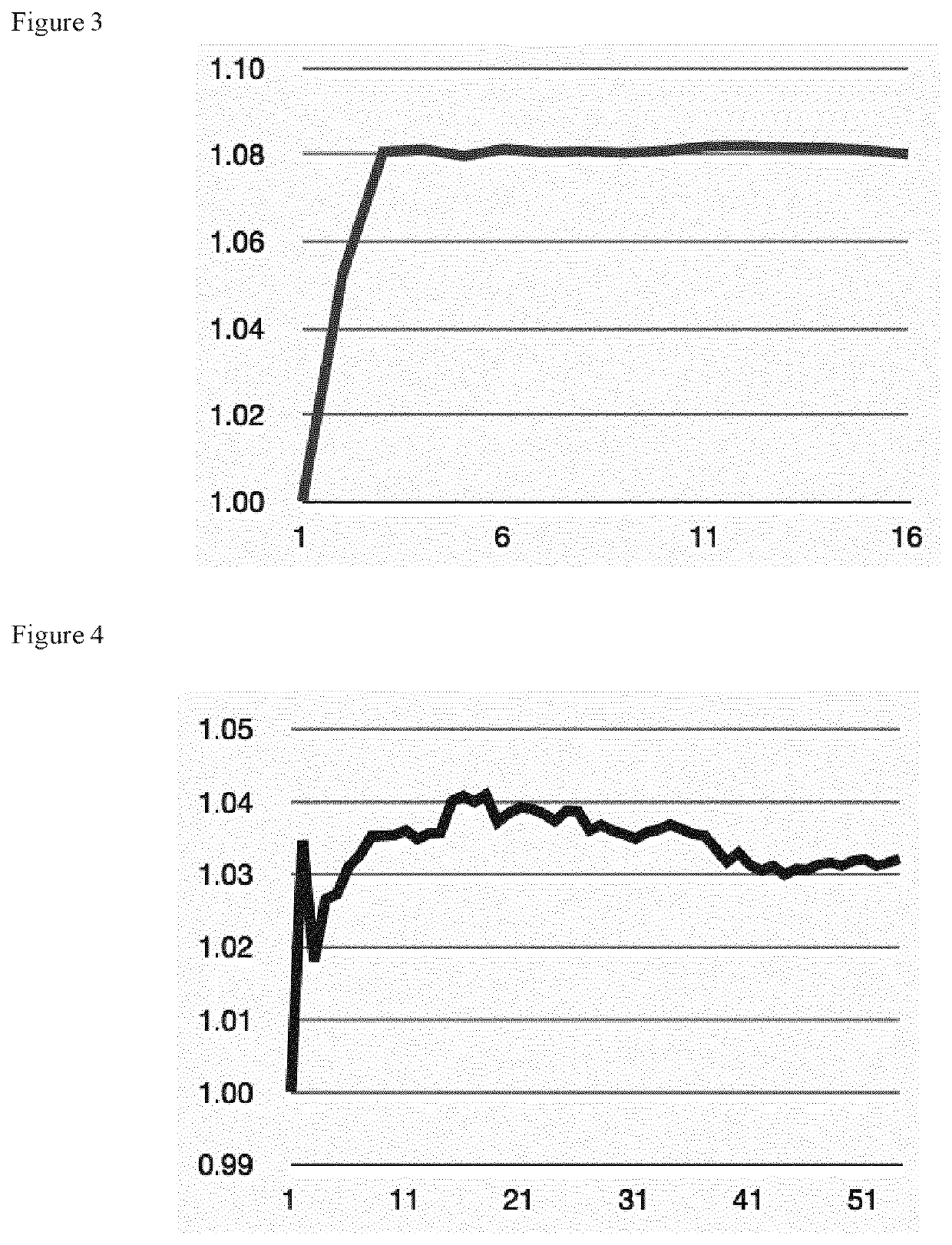
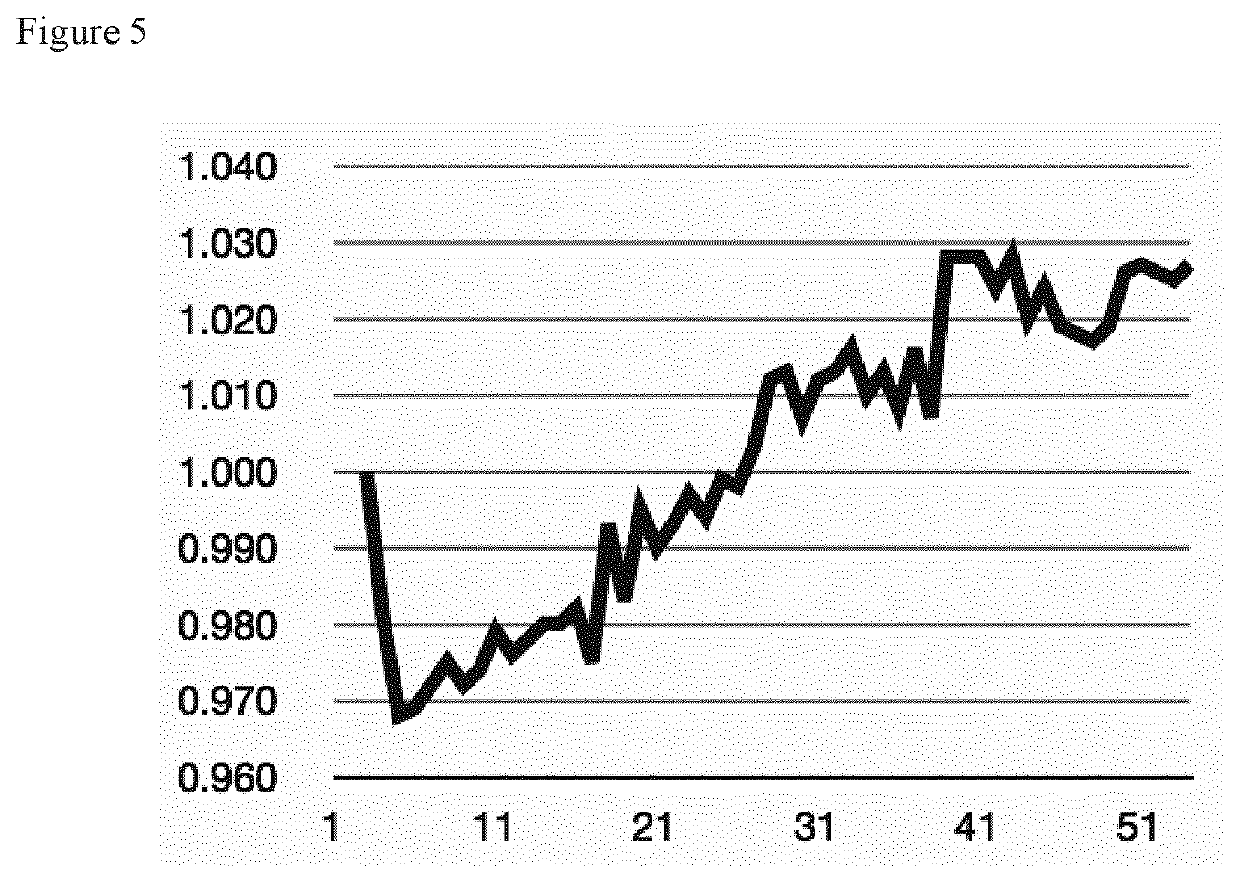
Innovations in Electrolyte Formulations for Enhanced Electrolytic Cell Functionality
AUG 1, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Electrolyte Evolution
The evolution of electrolyte formulations has been a critical factor in enhancing the functionality and performance of electrolytic cells. Over the past decades, researchers and engineers have made significant strides in developing innovative electrolyte compositions that address various challenges in electrochemical systems.
In the early stages of electrolyte development, simple aqueous solutions of acids, bases, or salts were predominantly used. These basic formulations provided the necessary ionic conductivity but often suffered from limitations such as narrow electrochemical stability windows and corrosion issues. As the demand for more efficient and durable electrolytic cells grew, the focus shifted towards tailoring electrolyte compositions to specific applications.
The introduction of organic solvents marked a significant milestone in electrolyte evolution. These solvents, such as propylene carbonate and ethylene carbonate, enabled the expansion of the electrochemical stability window and improved the solubility of electrolyte salts. This advancement paved the way for the development of high-energy-density batteries and more efficient electrochemical processes.
Subsequent research led to the development of ionic liquids as electrolytes. These room-temperature molten salts offered unique properties, including high thermal stability, negligible vapor pressure, and wide electrochemical windows. Their customizable nature allowed for fine-tuning of electrolyte properties to meet specific application requirements.
The advent of solid electrolytes represented another paradigm shift in electrolyte technology. These materials, including ceramic and polymer electrolytes, addressed safety concerns associated with liquid electrolytes and opened up possibilities for new cell designs and form factors. Solid electrolytes have shown particular promise in the development of all-solid-state batteries and fuel cells.
Recent years have seen a surge in the development of composite and hybrid electrolytes. These formulations combine different types of electrolytes to leverage their respective advantages while mitigating individual limitations. For instance, gel polymer electrolytes incorporate liquid electrolytes into polymer matrices, offering a balance between the high conductivity of liquids and the mechanical stability of solids.
Nanotechnology has also played a crucial role in electrolyte evolution. The incorporation of nanoparticles and nanostructured materials into electrolyte formulations has led to enhanced ionic conductivity, improved interfacial properties, and better overall cell performance. This approach has been particularly effective in addressing issues related to dendrite formation in battery systems.
Looking ahead, the future of electrolyte evolution is likely to focus on sustainable and environmentally friendly formulations. Bio-derived electrolytes and water-in-salt electrolytes are emerging as promising candidates for next-generation energy storage and conversion devices. Additionally, the integration of artificial intelligence and machine learning in electrolyte design is expected to accelerate the discovery of novel formulations with optimized properties for specific applications.
In the early stages of electrolyte development, simple aqueous solutions of acids, bases, or salts were predominantly used. These basic formulations provided the necessary ionic conductivity but often suffered from limitations such as narrow electrochemical stability windows and corrosion issues. As the demand for more efficient and durable electrolytic cells grew, the focus shifted towards tailoring electrolyte compositions to specific applications.
The introduction of organic solvents marked a significant milestone in electrolyte evolution. These solvents, such as propylene carbonate and ethylene carbonate, enabled the expansion of the electrochemical stability window and improved the solubility of electrolyte salts. This advancement paved the way for the development of high-energy-density batteries and more efficient electrochemical processes.
Subsequent research led to the development of ionic liquids as electrolytes. These room-temperature molten salts offered unique properties, including high thermal stability, negligible vapor pressure, and wide electrochemical windows. Their customizable nature allowed for fine-tuning of electrolyte properties to meet specific application requirements.
The advent of solid electrolytes represented another paradigm shift in electrolyte technology. These materials, including ceramic and polymer electrolytes, addressed safety concerns associated with liquid electrolytes and opened up possibilities for new cell designs and form factors. Solid electrolytes have shown particular promise in the development of all-solid-state batteries and fuel cells.
Recent years have seen a surge in the development of composite and hybrid electrolytes. These formulations combine different types of electrolytes to leverage their respective advantages while mitigating individual limitations. For instance, gel polymer electrolytes incorporate liquid electrolytes into polymer matrices, offering a balance between the high conductivity of liquids and the mechanical stability of solids.
Nanotechnology has also played a crucial role in electrolyte evolution. The incorporation of nanoparticles and nanostructured materials into electrolyte formulations has led to enhanced ionic conductivity, improved interfacial properties, and better overall cell performance. This approach has been particularly effective in addressing issues related to dendrite formation in battery systems.
Looking ahead, the future of electrolyte evolution is likely to focus on sustainable and environmentally friendly formulations. Bio-derived electrolytes and water-in-salt electrolytes are emerging as promising candidates for next-generation energy storage and conversion devices. Additionally, the integration of artificial intelligence and machine learning in electrolyte design is expected to accelerate the discovery of novel formulations with optimized properties for specific applications.
Market Demand Analysis
The market demand for innovations in electrolyte formulations for enhanced electrolytic cell functionality has been steadily growing, driven by the increasing need for more efficient and sustainable energy storage solutions. This demand spans across various industries, including renewable energy, electric vehicles, consumer electronics, and industrial applications.
In the renewable energy sector, the push for grid-scale energy storage has created a significant market for advanced electrolyte formulations. As intermittent energy sources like solar and wind power become more prevalent, the need for reliable and high-capacity energy storage systems has intensified. Improved electrolytes can enhance the performance and longevity of large-scale batteries, making them more economically viable for grid applications.
The electric vehicle (EV) industry represents another major driver of market demand for innovative electrolyte formulations. With global EV sales projected to grow exponentially in the coming years, there is a pressing need for batteries with higher energy density, faster charging capabilities, and longer lifespans. Advanced electrolytes play a crucial role in achieving these performance improvements, making them a key focus area for automotive manufacturers and battery suppliers.
Consumer electronics continue to be a significant market for electrolyte innovations. As devices become more powerful and energy-intensive, there is a growing demand for batteries that can provide longer usage times without increasing in size or weight. Improved electrolyte formulations can contribute to higher energy density and better cycle life in portable electronic devices, meeting consumer expectations for longer-lasting and faster-charging products.
In industrial applications, the demand for enhanced electrolytic cell functionality is driven by the need for more efficient and cost-effective processes. Industries such as metal refining, chlor-alkali production, and water treatment rely heavily on electrolytic processes. Innovations in electrolyte formulations can lead to improved productivity, reduced energy consumption, and lower operational costs in these sectors.
The market for advanced electrolytes is also influenced by environmental concerns and regulatory pressures. There is a growing emphasis on developing safer, more environmentally friendly electrolyte formulations that can reduce the environmental impact of battery production and disposal. This trend is particularly evident in regions with strict environmental regulations, creating opportunities for innovations in green electrolyte technologies.
Overall, the market demand for innovations in electrolyte formulations is robust and diverse, spanning multiple industries and applications. As technology continues to advance and sustainability becomes increasingly important, the demand for enhanced electrolytic cell functionality is expected to grow further, driving ongoing research and development in this field.
In the renewable energy sector, the push for grid-scale energy storage has created a significant market for advanced electrolyte formulations. As intermittent energy sources like solar and wind power become more prevalent, the need for reliable and high-capacity energy storage systems has intensified. Improved electrolytes can enhance the performance and longevity of large-scale batteries, making them more economically viable for grid applications.
The electric vehicle (EV) industry represents another major driver of market demand for innovative electrolyte formulations. With global EV sales projected to grow exponentially in the coming years, there is a pressing need for batteries with higher energy density, faster charging capabilities, and longer lifespans. Advanced electrolytes play a crucial role in achieving these performance improvements, making them a key focus area for automotive manufacturers and battery suppliers.
Consumer electronics continue to be a significant market for electrolyte innovations. As devices become more powerful and energy-intensive, there is a growing demand for batteries that can provide longer usage times without increasing in size or weight. Improved electrolyte formulations can contribute to higher energy density and better cycle life in portable electronic devices, meeting consumer expectations for longer-lasting and faster-charging products.
In industrial applications, the demand for enhanced electrolytic cell functionality is driven by the need for more efficient and cost-effective processes. Industries such as metal refining, chlor-alkali production, and water treatment rely heavily on electrolytic processes. Innovations in electrolyte formulations can lead to improved productivity, reduced energy consumption, and lower operational costs in these sectors.
The market for advanced electrolytes is also influenced by environmental concerns and regulatory pressures. There is a growing emphasis on developing safer, more environmentally friendly electrolyte formulations that can reduce the environmental impact of battery production and disposal. This trend is particularly evident in regions with strict environmental regulations, creating opportunities for innovations in green electrolyte technologies.
Overall, the market demand for innovations in electrolyte formulations is robust and diverse, spanning multiple industries and applications. As technology continues to advance and sustainability becomes increasingly important, the demand for enhanced electrolytic cell functionality is expected to grow further, driving ongoing research and development in this field.
Technical Challenges
The development of innovative electrolyte formulations for enhanced electrolytic cell functionality faces several significant technical challenges. One of the primary obstacles is achieving a balance between ionic conductivity and electrochemical stability. As researchers strive to improve cell performance, they must contend with the trade-off between high conductivity and the potential for unwanted side reactions that can degrade the electrolyte or electrode materials.
Another major challenge lies in the development of electrolytes that can operate effectively across a wide temperature range. Many current formulations suffer from reduced performance or even solidification at low temperatures, while high temperatures can lead to accelerated degradation and safety concerns. Creating electrolytes that maintain optimal functionality in diverse environmental conditions remains a critical area of research.
The quest for improved safety in electrolytic cells presents yet another hurdle. Traditional liquid electrolytes often pose risks due to their flammability and potential for leakage. While solid-state electrolytes offer promising safety advantages, they typically struggle with lower ionic conductivity and poor interfacial contact with electrodes, limiting their practical application.
Compatibility between the electrolyte and electrode materials is a persistent challenge in electrolytic cell design. Unwanted reactions at the electrode-electrolyte interface can lead to the formation of resistive layers, reducing cell efficiency and lifespan. Developing electrolyte formulations that form stable and beneficial interfaces with both cathode and anode materials is crucial for long-term cell performance.
The environmental impact and sustainability of electrolyte materials present additional technical challenges. As the demand for electrolytic cells grows, particularly in energy storage applications, there is an increasing need for electrolytes composed of abundant, non-toxic, and easily recyclable materials. However, finding formulations that meet these criteria while maintaining high performance standards remains difficult.
Scalability and cost-effectiveness of new electrolyte formulations also pose significant challenges. Many promising electrolyte innovations developed in laboratory settings face obstacles when transitioning to large-scale production. Researchers must consider not only the performance characteristics but also the feasibility of mass production and the overall economic viability of new formulations.
Lastly, the development of electrolytes for specific applications, such as high-voltage batteries or specialized industrial processes, presents unique challenges. These applications often require tailored electrolyte properties that may not be achievable with conventional formulations, necessitating innovative approaches and potentially new classes of electrolyte materials.
Another major challenge lies in the development of electrolytes that can operate effectively across a wide temperature range. Many current formulations suffer from reduced performance or even solidification at low temperatures, while high temperatures can lead to accelerated degradation and safety concerns. Creating electrolytes that maintain optimal functionality in diverse environmental conditions remains a critical area of research.
The quest for improved safety in electrolytic cells presents yet another hurdle. Traditional liquid electrolytes often pose risks due to their flammability and potential for leakage. While solid-state electrolytes offer promising safety advantages, they typically struggle with lower ionic conductivity and poor interfacial contact with electrodes, limiting their practical application.
Compatibility between the electrolyte and electrode materials is a persistent challenge in electrolytic cell design. Unwanted reactions at the electrode-electrolyte interface can lead to the formation of resistive layers, reducing cell efficiency and lifespan. Developing electrolyte formulations that form stable and beneficial interfaces with both cathode and anode materials is crucial for long-term cell performance.
The environmental impact and sustainability of electrolyte materials present additional technical challenges. As the demand for electrolytic cells grows, particularly in energy storage applications, there is an increasing need for electrolytes composed of abundant, non-toxic, and easily recyclable materials. However, finding formulations that meet these criteria while maintaining high performance standards remains difficult.
Scalability and cost-effectiveness of new electrolyte formulations also pose significant challenges. Many promising electrolyte innovations developed in laboratory settings face obstacles when transitioning to large-scale production. Researchers must consider not only the performance characteristics but also the feasibility of mass production and the overall economic viability of new formulations.
Lastly, the development of electrolytes for specific applications, such as high-voltage batteries or specialized industrial processes, presents unique challenges. These applications often require tailored electrolyte properties that may not be achievable with conventional formulations, necessitating innovative approaches and potentially new classes of electrolyte materials.
Current Formulations
01 Electrolyte composition for lithium-ion batteries
Formulations of electrolytes for lithium-ion batteries focus on improving battery performance, safety, and longevity. These compositions often include specific combinations of solvents, lithium salts, and additives to enhance conductivity, stability, and electrode interface properties.- Electrolyte composition for improved battery performance: Formulations of electrolytes designed to enhance battery performance, including improved conductivity, stability, and cycle life. These compositions may include specific combinations of solvents, salts, and additives tailored to optimize electrochemical properties and safety features in various types of batteries.
- Functional additives in electrolyte formulations: Incorporation of functional additives into electrolyte formulations to address specific performance issues or enhance certain properties. These additives may include film-forming agents, stabilizers, or compounds that mitigate unwanted side reactions, thereby improving the overall functionality and longevity of the electrolyte system.
- Novel electrolyte systems for advanced energy storage: Development of innovative electrolyte systems for next-generation energy storage devices, such as solid-state batteries or supercapacitors. These formulations may involve new materials, unique solvent blends, or hybrid electrolyte concepts to achieve superior energy density, faster charging capabilities, or enhanced safety features.
- Electrolyte formulations for specific applications: Tailored electrolyte formulations designed for specific applications or operating conditions, such as high-temperature environments, aerospace use, or medical devices. These specialized formulations may prioritize certain properties like thermal stability, low volatility, or biocompatibility to meet the unique requirements of their intended use.
- Eco-friendly and sustainable electrolyte compositions: Development of environmentally friendly and sustainable electrolyte formulations, focusing on biodegradable components, reduced toxicity, or the use of renewable resources. These green electrolyte solutions aim to minimize environmental impact while maintaining or improving the functional performance of energy storage systems.
02 Polymer electrolyte formulations
Polymer-based electrolyte formulations are developed to improve the mechanical and thermal stability of electrolytes. These formulations often incorporate various polymers and additives to create solid or gel-like electrolytes with enhanced safety features and flexibility for use in different battery designs.Expand Specific Solutions03 Electrolyte additives for performance enhancement
Specific additives are incorporated into electrolyte formulations to enhance various aspects of battery performance. These additives can improve factors such as ionic conductivity, solid electrolyte interphase (SEI) formation, cycle life, and high-temperature stability of the electrolyte system.Expand Specific Solutions04 Electrolyte formulations for extreme temperature applications
Specialized electrolyte formulations are developed for batteries operating in extreme temperature conditions. These formulations focus on maintaining electrolyte stability, preventing freezing or decomposition, and ensuring consistent battery performance across a wide temperature range.Expand Specific Solutions05 Eco-friendly and sustainable electrolyte formulations
Research is conducted on developing environmentally friendly and sustainable electrolyte formulations. These efforts include using bio-derived solvents, reducing toxic components, and improving the recyclability of battery materials while maintaining or enhancing electrolyte functionality.Expand Specific Solutions
Key Industry Players
The electrolyte formulation innovation landscape for enhanced electrolytic cell functionality is in a dynamic growth phase, with significant market potential driven by increasing demand for advanced energy storage solutions. The market is characterized by intense competition among established chemical companies like Merck, BASF, and DuPont, alongside specialized battery technology firms such as Wildcat Discovery Technologies and PolyPlus Battery. Research institutions and universities also play a crucial role in advancing the field. The technology is rapidly evolving, with companies focusing on developing novel electrolyte compositions to improve battery performance, safety, and longevity. While some formulations are reaching commercial maturity, ongoing research indicates substantial room for further innovation and market expansion.
Merck Patent GmbH
Technical Solution: Merck Patent GmbH has developed innovative electrolyte formulations for enhanced electrolytic cell functionality. Their approach focuses on using novel ionic liquids and additives to improve the performance and stability of electrolytes. The company has introduced a new class of fluorinated ether-based electrolytes that demonstrate superior electrochemical stability and ionic conductivity[1]. These electrolytes incorporate carefully selected salt combinations and functional additives to create a stable solid electrolyte interphase (SEI) layer, which is crucial for long-term battery performance[2]. Merck's formulations also address the challenge of high-voltage operation, enabling the use of high-energy cathode materials while maintaining electrolyte stability[3].
Strengths: High electrochemical stability, improved ionic conductivity, and enhanced SEI formation. Weaknesses: Potential high cost of specialized materials and complexity in large-scale production.
Wildcat Discovery Technologies, Inc.
Technical Solution: Wildcat Discovery Technologies has pioneered a high-throughput screening approach to electrolyte formulation development. Their proprietary platform allows for rapid testing of thousands of electrolyte combinations, accelerating the discovery of optimal formulations[4]. The company has developed electrolytes specifically tailored for next-generation battery chemistries, including silicon anodes and high-nickel cathodes. Their formulations often include multi-component additive packages that work synergistically to enhance cell performance and longevity[5]. Wildcat's electrolytes have demonstrated significant improvements in cycle life, fast-charging capabilities, and low-temperature performance compared to conventional formulations[6].
Strengths: Rapid discovery of novel formulations, tailored solutions for advanced battery chemistries. Weaknesses: Reliance on proprietary screening technology may limit broader industry adoption.
Breakthrough Patents
Improved rechargeable batteries and production thereof
PatentPendingUS20220359914A1
Innovation
- A carbonate:nitrile type solvent mixture-based electrolyte formulation using lithium-difluoro(oxalato)borate (LiDFOB) salt, combined with polymer additives like poly(methyl vinyl ether-alt-maleic anhydride) and fluoro-ethylenecarbonate, enhances stability, safety, and ionic conductivity, allowing for extended temperature ranges and stable cycling of advanced battery electrodes.
Electrolyte composition
PatentPendingGB2625308A
Innovation
- An electrolyte composition featuring ethyl acetate as the solvent component and lithium salts at concentrations between 1.0 and 3.0 mol-dm^-3, which enhances ionic conductivity and reduces internal resistance, and a hybrid electrolyte composition that combines this with traditional electrolytes to improve rate capability.
Environmental Impact
The environmental impact of innovations in electrolyte formulations for enhanced electrolytic cell functionality is a critical consideration in the development and implementation of these technologies. As electrolytic cells play a significant role in various industrial processes, including energy storage and chemical production, the environmental implications of their operation and the materials used in their construction are of paramount importance.
One of the primary environmental benefits of improved electrolyte formulations is the potential for increased energy efficiency in electrolytic processes. By enhancing the functionality of electrolytic cells, these innovations can lead to reduced energy consumption, which in turn results in lower greenhouse gas emissions associated with power generation. This improvement in energy efficiency aligns with global efforts to mitigate climate change and reduce the carbon footprint of industrial operations.
Furthermore, advanced electrolyte formulations can contribute to the longevity and durability of electrolytic cells. This extended lifespan reduces the frequency of cell replacement and maintenance, thereby minimizing waste generation and the environmental impact associated with manufacturing and disposing of cell components. The reduction in material consumption and waste production represents a significant step towards more sustainable industrial practices.
Another important aspect of environmental impact is the potential for these innovations to enable the use of more environmentally friendly materials in electrolyte formulations. By developing electrolytes that are less toxic, more biodegradable, or derived from renewable sources, the overall environmental footprint of electrolytic processes can be substantially reduced. This shift towards greener materials aligns with the principles of green chemistry and supports the transition to more sustainable industrial practices.
The improved performance of electrolytic cells resulting from innovative electrolyte formulations can also lead to more efficient resource utilization. For instance, in water treatment applications, enhanced electrolytic processes can result in more effective pollutant removal, contributing to improved water quality and reduced environmental contamination. Similarly, in metal recovery processes, more efficient electrolysis can lead to higher recovery rates, reducing the need for primary resource extraction and its associated environmental impacts.
However, it is crucial to consider potential negative environmental impacts as well. The production of new electrolyte formulations may involve the use of rare or environmentally sensitive materials, which could pose challenges in terms of resource depletion and ecosystem disruption. Additionally, the disposal or recycling of spent electrolytes must be carefully managed to prevent environmental contamination and ensure proper handling of potentially hazardous substances.
In conclusion, while innovations in electrolyte formulations for enhanced electrolytic cell functionality offer significant potential for positive environmental impact through improved efficiency and sustainability, a comprehensive life cycle assessment is necessary to fully understand and mitigate any potential negative consequences. Ongoing research and development in this field should prioritize not only performance enhancements but also environmental considerations to ensure a holistic approach to technological advancement.
One of the primary environmental benefits of improved electrolyte formulations is the potential for increased energy efficiency in electrolytic processes. By enhancing the functionality of electrolytic cells, these innovations can lead to reduced energy consumption, which in turn results in lower greenhouse gas emissions associated with power generation. This improvement in energy efficiency aligns with global efforts to mitigate climate change and reduce the carbon footprint of industrial operations.
Furthermore, advanced electrolyte formulations can contribute to the longevity and durability of electrolytic cells. This extended lifespan reduces the frequency of cell replacement and maintenance, thereby minimizing waste generation and the environmental impact associated with manufacturing and disposing of cell components. The reduction in material consumption and waste production represents a significant step towards more sustainable industrial practices.
Another important aspect of environmental impact is the potential for these innovations to enable the use of more environmentally friendly materials in electrolyte formulations. By developing electrolytes that are less toxic, more biodegradable, or derived from renewable sources, the overall environmental footprint of electrolytic processes can be substantially reduced. This shift towards greener materials aligns with the principles of green chemistry and supports the transition to more sustainable industrial practices.
The improved performance of electrolytic cells resulting from innovative electrolyte formulations can also lead to more efficient resource utilization. For instance, in water treatment applications, enhanced electrolytic processes can result in more effective pollutant removal, contributing to improved water quality and reduced environmental contamination. Similarly, in metal recovery processes, more efficient electrolysis can lead to higher recovery rates, reducing the need for primary resource extraction and its associated environmental impacts.
However, it is crucial to consider potential negative environmental impacts as well. The production of new electrolyte formulations may involve the use of rare or environmentally sensitive materials, which could pose challenges in terms of resource depletion and ecosystem disruption. Additionally, the disposal or recycling of spent electrolytes must be carefully managed to prevent environmental contamination and ensure proper handling of potentially hazardous substances.
In conclusion, while innovations in electrolyte formulations for enhanced electrolytic cell functionality offer significant potential for positive environmental impact through improved efficiency and sustainability, a comprehensive life cycle assessment is necessary to fully understand and mitigate any potential negative consequences. Ongoing research and development in this field should prioritize not only performance enhancements but also environmental considerations to ensure a holistic approach to technological advancement.
Safety Regulations
Safety regulations play a crucial role in the development and implementation of innovative electrolyte formulations for enhanced electrolytic cell functionality. As the field progresses, regulatory bodies have established comprehensive guidelines to ensure the safe handling, storage, and use of electrolytes in various applications.
One of the primary concerns addressed by safety regulations is the potential for chemical reactions and hazardous emissions. Electrolytes often contain corrosive or toxic substances, necessitating strict protocols for their containment and disposal. Regulatory frameworks mandate the use of appropriate personal protective equipment (PPE) for workers handling these materials, as well as the implementation of proper ventilation systems in laboratories and production facilities.
The transportation of electrolytes is another area heavily regulated by safety guidelines. Specific packaging requirements and labeling standards have been established to minimize the risk of accidents during transit. These regulations often vary depending on the chemical composition of the electrolyte and its potential reactivity with other substances.
Environmental protection is a key focus of safety regulations in this field. Stringent measures are in place to prevent the release of electrolytes into ecosystems, with particular emphasis on aquatic environments. Companies are required to implement robust waste management systems and adhere to strict disposal protocols to mitigate environmental impact.
Fire safety is a critical aspect of electrolyte-related regulations, especially in the context of battery technologies. Guidelines specify the need for fire suppression systems, thermal management protocols, and emergency response procedures in facilities where electrolytes are used or stored. These measures are designed to prevent and mitigate potential fire hazards associated with certain electrolyte formulations.
Regulatory bodies also mandate regular safety audits and inspections of facilities involved in electrolyte research and production. These assessments ensure ongoing compliance with safety standards and provide opportunities for continuous improvement in safety practices.
As new electrolyte formulations emerge, safety regulations evolve to address novel risks and challenges. Regulatory agencies work closely with industry experts and researchers to develop updated guidelines that reflect the latest scientific understanding of electrolyte properties and potential hazards.
The implementation of these safety regulations has a significant impact on the innovation process. While they may introduce additional complexity and cost to research and development efforts, they also drive the creation of safer, more sustainable electrolyte formulations. This regulatory framework ultimately contributes to the long-term viability and public acceptance of advanced electrolytic cell technologies across various industries.
One of the primary concerns addressed by safety regulations is the potential for chemical reactions and hazardous emissions. Electrolytes often contain corrosive or toxic substances, necessitating strict protocols for their containment and disposal. Regulatory frameworks mandate the use of appropriate personal protective equipment (PPE) for workers handling these materials, as well as the implementation of proper ventilation systems in laboratories and production facilities.
The transportation of electrolytes is another area heavily regulated by safety guidelines. Specific packaging requirements and labeling standards have been established to minimize the risk of accidents during transit. These regulations often vary depending on the chemical composition of the electrolyte and its potential reactivity with other substances.
Environmental protection is a key focus of safety regulations in this field. Stringent measures are in place to prevent the release of electrolytes into ecosystems, with particular emphasis on aquatic environments. Companies are required to implement robust waste management systems and adhere to strict disposal protocols to mitigate environmental impact.
Fire safety is a critical aspect of electrolyte-related regulations, especially in the context of battery technologies. Guidelines specify the need for fire suppression systems, thermal management protocols, and emergency response procedures in facilities where electrolytes are used or stored. These measures are designed to prevent and mitigate potential fire hazards associated with certain electrolyte formulations.
Regulatory bodies also mandate regular safety audits and inspections of facilities involved in electrolyte research and production. These assessments ensure ongoing compliance with safety standards and provide opportunities for continuous improvement in safety practices.
As new electrolyte formulations emerge, safety regulations evolve to address novel risks and challenges. Regulatory agencies work closely with industry experts and researchers to develop updated guidelines that reflect the latest scientific understanding of electrolyte properties and potential hazards.
The implementation of these safety regulations has a significant impact on the innovation process. While they may introduce additional complexity and cost to research and development efforts, they also drive the creation of safer, more sustainable electrolyte formulations. This regulatory framework ultimately contributes to the long-term viability and public acceptance of advanced electrolytic cell technologies across various industries.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!