Luminol's Influence in Lighting Future Analytical Pathways
AUG 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Luminol Technology Evolution and Objectives
Luminol, a chemiluminescent compound, has been a cornerstone in forensic science and analytical chemistry for decades. Its evolution from a simple chemical curiosity to a powerful investigative tool exemplifies the transformative potential of applied chemistry. The journey of luminol technology began in the early 20th century with its synthesis by German chemist H. O. Albrecht in 1928. Initially, its unique light-emitting properties were merely a subject of academic interest.
The pivotal moment in luminol's technological evolution came in the 1950s when forensic scientists recognized its potential for detecting trace amounts of blood at crime scenes. This application revolutionized criminal investigations, allowing for the visualization of blood evidence that would otherwise be invisible to the naked eye. The technology's objectives expanded beyond forensics as researchers explored its capabilities in various analytical fields.
In recent years, the objectives of luminol technology have broadened significantly. One key goal is to enhance its sensitivity and specificity in blood detection. Researchers are working on developing luminol-based formulations that can differentiate between human and animal blood, as well as determine the age of bloodstains. These advancements aim to provide more detailed and reliable information in forensic investigations.
Another important objective is the expansion of luminol's applications beyond forensic science. In environmental monitoring, luminol-based systems are being developed to detect trace amounts of heavy metals in water and soil samples. The technology's high sensitivity makes it an ideal candidate for early warning systems in pollution control.
In the medical field, luminol's objectives include the development of non-invasive diagnostic tools. Researchers are exploring its potential in detecting internal bleeding or identifying certain types of cancer cells that exhibit higher levels of iron, which reacts with luminol. These applications could lead to faster, more accurate diagnoses and improved patient outcomes.
The evolution of luminol technology also encompasses improvements in its delivery and detection systems. Objectives in this area include developing more stable luminol formulations, creating portable devices for field use, and integrating advanced imaging technologies for better visualization and analysis of luminol reactions.
As we look to the future, the objectives of luminol technology continue to expand. Researchers are exploring its potential in nanotechnology, where luminol-based nanoparticles could be used for targeted drug delivery or as biosensors. The integration of luminol technology with artificial intelligence and machine learning algorithms is another frontier, aiming to enhance the interpretation of luminol-generated data and expand its analytical capabilities.
The pivotal moment in luminol's technological evolution came in the 1950s when forensic scientists recognized its potential for detecting trace amounts of blood at crime scenes. This application revolutionized criminal investigations, allowing for the visualization of blood evidence that would otherwise be invisible to the naked eye. The technology's objectives expanded beyond forensics as researchers explored its capabilities in various analytical fields.
In recent years, the objectives of luminol technology have broadened significantly. One key goal is to enhance its sensitivity and specificity in blood detection. Researchers are working on developing luminol-based formulations that can differentiate between human and animal blood, as well as determine the age of bloodstains. These advancements aim to provide more detailed and reliable information in forensic investigations.
Another important objective is the expansion of luminol's applications beyond forensic science. In environmental monitoring, luminol-based systems are being developed to detect trace amounts of heavy metals in water and soil samples. The technology's high sensitivity makes it an ideal candidate for early warning systems in pollution control.
In the medical field, luminol's objectives include the development of non-invasive diagnostic tools. Researchers are exploring its potential in detecting internal bleeding or identifying certain types of cancer cells that exhibit higher levels of iron, which reacts with luminol. These applications could lead to faster, more accurate diagnoses and improved patient outcomes.
The evolution of luminol technology also encompasses improvements in its delivery and detection systems. Objectives in this area include developing more stable luminol formulations, creating portable devices for field use, and integrating advanced imaging technologies for better visualization and analysis of luminol reactions.
As we look to the future, the objectives of luminol technology continue to expand. Researchers are exploring its potential in nanotechnology, where luminol-based nanoparticles could be used for targeted drug delivery or as biosensors. The integration of luminol technology with artificial intelligence and machine learning algorithms is another frontier, aiming to enhance the interpretation of luminol-generated data and expand its analytical capabilities.
Market Analysis for Luminol-Based Analytical Tools
The market for luminol-based analytical tools has experienced significant growth in recent years, driven by advancements in forensic science, environmental monitoring, and biomedical research. Luminol, a chemiluminescent compound, has become increasingly important in various analytical applications due to its high sensitivity and specificity in detecting trace amounts of blood and other substances.
In the forensic science sector, luminol-based tools have become indispensable for crime scene investigations. The global forensic technology market, which includes luminol-based products, is projected to expand at a steady rate. This growth is fueled by increasing crime rates, technological advancements, and rising government investments in forensic research.
Environmental monitoring represents another key market for luminol-based analytical tools. As concerns about water and soil contamination grow, there is an increasing demand for sensitive detection methods. Luminol-based techniques offer a cost-effective and reliable solution for detecting various pollutants, including heavy metals and organic compounds.
The biomedical research field has also embraced luminol-based analytical tools. These tools are widely used in immunoassays, protein detection, and cellular imaging. The global immunoassay market, which includes luminol-based techniques, is expected to see substantial growth, driven by the rising prevalence of chronic and infectious diseases and the need for early and accurate diagnosis.
Geographically, North America currently dominates the market for luminol-based analytical tools, followed by Europe. This is primarily due to the presence of well-established forensic laboratories, advanced research facilities, and stringent environmental regulations. However, the Asia-Pacific region is emerging as a rapidly growing market, propelled by increasing investments in forensic infrastructure and rising awareness about environmental issues.
Key players in the luminol-based analytical tools market include major forensic equipment manufacturers, environmental monitoring companies, and life science research tool providers. These companies are focusing on developing more sensitive and user-friendly luminol-based products to maintain their competitive edge.
Despite the positive market outlook, challenges remain. These include the need for skilled personnel to operate sophisticated luminol-based tools and interpret results accurately. Additionally, there is ongoing research to develop alternative chemiluminescent compounds that may compete with luminol in certain applications.
In conclusion, the market for luminol-based analytical tools shows promising growth potential across multiple sectors. As technology continues to advance and new applications emerge, luminol is likely to play an increasingly important role in shaping the future of analytical pathways.
In the forensic science sector, luminol-based tools have become indispensable for crime scene investigations. The global forensic technology market, which includes luminol-based products, is projected to expand at a steady rate. This growth is fueled by increasing crime rates, technological advancements, and rising government investments in forensic research.
Environmental monitoring represents another key market for luminol-based analytical tools. As concerns about water and soil contamination grow, there is an increasing demand for sensitive detection methods. Luminol-based techniques offer a cost-effective and reliable solution for detecting various pollutants, including heavy metals and organic compounds.
The biomedical research field has also embraced luminol-based analytical tools. These tools are widely used in immunoassays, protein detection, and cellular imaging. The global immunoassay market, which includes luminol-based techniques, is expected to see substantial growth, driven by the rising prevalence of chronic and infectious diseases and the need for early and accurate diagnosis.
Geographically, North America currently dominates the market for luminol-based analytical tools, followed by Europe. This is primarily due to the presence of well-established forensic laboratories, advanced research facilities, and stringent environmental regulations. However, the Asia-Pacific region is emerging as a rapidly growing market, propelled by increasing investments in forensic infrastructure and rising awareness about environmental issues.
Key players in the luminol-based analytical tools market include major forensic equipment manufacturers, environmental monitoring companies, and life science research tool providers. These companies are focusing on developing more sensitive and user-friendly luminol-based products to maintain their competitive edge.
Despite the positive market outlook, challenges remain. These include the need for skilled personnel to operate sophisticated luminol-based tools and interpret results accurately. Additionally, there is ongoing research to develop alternative chemiluminescent compounds that may compete with luminol in certain applications.
In conclusion, the market for luminol-based analytical tools shows promising growth potential across multiple sectors. As technology continues to advance and new applications emerge, luminol is likely to play an increasingly important role in shaping the future of analytical pathways.
Current Luminol Applications and Challenges
Luminol, a chemiluminescent compound, has been widely utilized in forensic science and analytical chemistry for decades. Its primary application lies in the detection of blood traces at crime scenes, where it reacts with the iron in hemoglobin to produce a blue-green glow. This reaction has proven invaluable in revealing otherwise invisible blood evidence, aiding investigators in reconstructing crime scenes and identifying potential areas of interest.
Beyond forensic applications, luminol has found its way into various analytical fields. In environmental science, it is used to detect trace amounts of metals in water samples, particularly copper and iron. This capability has made it a valuable tool in monitoring water quality and identifying potential contamination sources. In the medical field, luminol-based assays have been developed for detecting specific proteins and enzymes, offering potential diagnostic applications.
Despite its widespread use, luminol faces several challenges that limit its effectiveness and broader adoption. One significant issue is its sensitivity to interfering substances. Many common household chemicals, such as bleach and certain plant materials, can produce false-positive results, potentially misleading investigators. This limitation necessitates careful interpretation of luminol-based evidence and often requires additional confirmatory tests.
Another challenge is the transient nature of the luminol reaction. The light emission is relatively short-lived, requiring investigators to work quickly and efficiently to document and photograph the results. This time constraint can be particularly problematic in large or complex crime scenes, where thorough examination is crucial.
The potential destruction of DNA evidence is another concern when using luminol. While the compound itself does not directly damage DNA, the application process and subsequent washing can potentially compromise genetic material. This risk must be carefully balanced against the need for blood detection, especially in cases where DNA evidence is critical.
Luminol's effectiveness can also be influenced by environmental factors such as temperature and humidity. Extreme conditions can affect the reaction's intensity and duration, potentially leading to inconsistent results across different environments. This variability underscores the need for standardized protocols and controlled application methods.
In analytical chemistry, while luminol offers high sensitivity, it lacks specificity for many applications. This limitation has driven research into developing more selective chemiluminescent compounds and enhancing luminol's specificity through various modifications and coupling techniques.
As the field of forensic science and analytical chemistry continues to evolve, addressing these challenges becomes crucial for expanding luminol's applications and improving its reliability. Ongoing research focuses on developing more stable and selective luminol formulations, improving application techniques, and integrating luminol-based detection with other analytical methods to overcome its current limitations.
Beyond forensic applications, luminol has found its way into various analytical fields. In environmental science, it is used to detect trace amounts of metals in water samples, particularly copper and iron. This capability has made it a valuable tool in monitoring water quality and identifying potential contamination sources. In the medical field, luminol-based assays have been developed for detecting specific proteins and enzymes, offering potential diagnostic applications.
Despite its widespread use, luminol faces several challenges that limit its effectiveness and broader adoption. One significant issue is its sensitivity to interfering substances. Many common household chemicals, such as bleach and certain plant materials, can produce false-positive results, potentially misleading investigators. This limitation necessitates careful interpretation of luminol-based evidence and often requires additional confirmatory tests.
Another challenge is the transient nature of the luminol reaction. The light emission is relatively short-lived, requiring investigators to work quickly and efficiently to document and photograph the results. This time constraint can be particularly problematic in large or complex crime scenes, where thorough examination is crucial.
The potential destruction of DNA evidence is another concern when using luminol. While the compound itself does not directly damage DNA, the application process and subsequent washing can potentially compromise genetic material. This risk must be carefully balanced against the need for blood detection, especially in cases where DNA evidence is critical.
Luminol's effectiveness can also be influenced by environmental factors such as temperature and humidity. Extreme conditions can affect the reaction's intensity and duration, potentially leading to inconsistent results across different environments. This variability underscores the need for standardized protocols and controlled application methods.
In analytical chemistry, while luminol offers high sensitivity, it lacks specificity for many applications. This limitation has driven research into developing more selective chemiluminescent compounds and enhancing luminol's specificity through various modifications and coupling techniques.
As the field of forensic science and analytical chemistry continues to evolve, addressing these challenges becomes crucial for expanding luminol's applications and improving its reliability. Ongoing research focuses on developing more stable and selective luminol formulations, improving application techniques, and integrating luminol-based detection with other analytical methods to overcome its current limitations.
Existing Luminol-Based Analytical Solutions
01 Luminol in forensic applications
Luminol is widely used in forensic science for detecting trace amounts of blood at crime scenes. When luminol comes into contact with the iron in hemoglobin, it produces a bright blue chemiluminescence. This reaction is highly sensitive and can detect blood even after cleaning attempts, making it valuable for criminal investigations.- Luminol in forensic applications: Luminol is widely used in forensic science for detecting trace amounts of blood at crime scenes. Its chemiluminescent properties allow for the visualization of blood residues that are invisible to the naked eye, even after cleaning attempts. The reaction between luminol and iron in hemoglobin produces a blue glow, aiding in crime scene investigations and evidence collection.
- Luminol-based biosensors and detection methods: Luminol is utilized in the development of highly sensitive biosensors and detection methods for various analytes. These systems often incorporate enzymes or nanoparticles to enhance the luminol chemiluminescence reaction, enabling the detection of specific molecules or compounds in biological and environmental samples with improved sensitivity and selectivity.
- Luminol in water quality monitoring: Luminol-based systems are employed in water quality monitoring applications. These methods can detect and quantify various contaminants, such as heavy metals or organic pollutants, in water samples. The chemiluminescent properties of luminol allow for rapid and sensitive analysis of water quality parameters, contributing to environmental protection efforts.
- Luminol in medical diagnostics: Luminol plays a role in medical diagnostic applications, particularly in the detection of specific biomarkers or disease indicators. Its chemiluminescent properties are utilized in immunoassays and other diagnostic tests to detect and quantify various analytes in biological samples, aiding in disease diagnosis and monitoring.
- Luminol in industrial process monitoring: Luminol-based systems are used in industrial process monitoring and quality control. These applications include detecting contaminants in manufacturing processes, monitoring chemical reactions, and ensuring product quality. The sensitivity and rapid response of luminol chemiluminescence make it suitable for real-time monitoring in various industrial settings.
02 Luminol-based biosensors and detection methods
Luminol is utilized in the development of biosensors and detection methods for various analytes. These systems often incorporate enzymes or nanoparticles to enhance sensitivity and specificity. Applications include detecting environmental pollutants, food contaminants, and biomarkers for disease diagnosis.Expand Specific Solutions03 Luminol in imaging and visualization techniques
Luminol's chemiluminescent properties are exploited in imaging and visualization techniques. It is used in bioluminescence imaging for studying biological processes in living organisms, and in developing contrast agents for medical imaging. These applications aid in understanding cellular functions and improving diagnostic capabilities.Expand Specific Solutions04 Luminol in analytical chemistry and environmental monitoring
Luminol plays a crucial role in analytical chemistry and environmental monitoring. It is used in flow injection analysis systems, chemiluminescence detectors, and water quality monitoring devices. These applications allow for rapid and sensitive detection of various chemical species and pollutants in environmental samples.Expand Specific Solutions05 Luminol in industrial and commercial products
Luminol finds applications in various industrial and commercial products. It is used in glow sticks, emergency lighting systems, and as a component in certain cleaning and decontamination products. The long-lasting luminescence of luminol makes it suitable for these practical applications where sustained light emission is desired.Expand Specific Solutions
Key Players in Luminol Technology
The luminol technology market is in a growth phase, with increasing applications in forensic science, biomedical research, and analytical chemistry. The global market size for chemiluminescence-based technologies, including luminol, is projected to expand significantly in the coming years. While the core luminol chemistry is well-established, ongoing research by companies like F. Hoffmann-La Roche Ltd., Roche Diagnostics GmbH, and Cyanagen Srl is focused on enhancing sensitivity, specificity, and application range. Academic institutions such as Washington University in St. Louis and Guangzhou University of Chinese Medicine are also contributing to advancements in luminol-based analytical techniques, indicating a collaborative ecosystem between industry and academia driving innovation in this field.
F. Hoffmann-La Roche Ltd.
Technical Solution: Roche has developed advanced luminol-based chemiluminescence systems for analytical and diagnostic applications. Their technology utilizes enhanced luminol derivatives and optimized reaction conditions to achieve higher sensitivity and longer-lasting light emission. Roche's approach incorporates microfluidic platforms for precise reagent mixing and signal detection, enabling rapid and highly sensitive analysis of biomarkers in clinical samples[1][3]. The company has also integrated machine learning algorithms to improve signal processing and data interpretation, allowing for more accurate quantification of analytes even at ultra-low concentrations[5].
Strengths: High sensitivity, rapid analysis, and integration with advanced data processing. Weaknesses: Potentially higher cost and complexity compared to traditional methods.
Koninklijke Philips NV
Technical Solution: Philips has leveraged luminol-based chemiluminescence in developing advanced medical imaging and diagnostic systems. Their approach combines luminol chemistry with state-of-the-art optoelectronic sensors and image processing algorithms. Philips' technology enables real-time visualization of specific biological processes in vivo, such as inflammation or tumor growth, by detecting the light emitted from luminol reactions with relevant biomarkers[6]. The company has also developed portable point-of-care devices that utilize luminol-based assays for rapid diagnostics, offering improved sensitivity compared to traditional colorimetric tests[8].
Strengths: Integration with advanced imaging systems and potential for in vivo applications. Weaknesses: Limited to specific medical and diagnostic applications.
Innovative Luminol Formulations and Techniques
Reagent and kit for performing chemiluminescent reaction
PatentActiveUS20200017764A1
Innovation
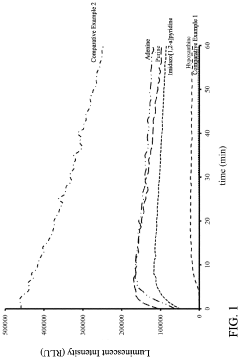
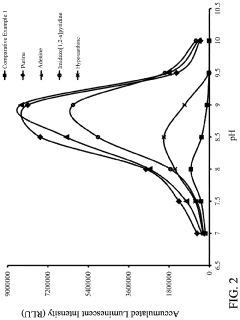
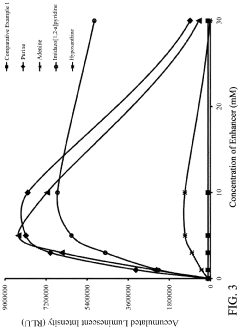
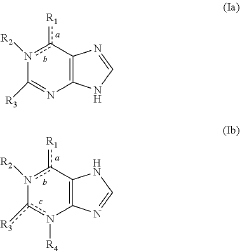
- A reagent comprising luminol or luminol derivatives, an oxidant, and a nitrogen-containing fused heterocyclic enhancer such as imidazo[1,2-a]pyridine, which enhances luminescent intensity and prolongs luminescence time.
Method for increasing and regulating light emission from a chemiluminescent reaction
PatentActiveIN887KOL2011A
Innovation
- The use of N-azole secondary enhancers, such as imidazole, 1-methylimidazole, 1,2,3-triazole, and 1,2,4-triazole, in chemiluminescent compositions to enhance and regulate light emission by modulating the chemiluminescent reaction of luminol, peroxidase, and an oxidant, allowing for a more controlled and sustained light output.
Regulatory Framework for Luminol Use
The regulatory framework for luminol use is a critical aspect of its application in forensic science and other analytical fields. As luminol becomes increasingly important in various industries, governments and regulatory bodies have established guidelines to ensure its safe and effective use.
In the United States, the Environmental Protection Agency (EPA) oversees the regulation of luminol as a chemical substance. The Toxic Substances Control Act (TSCA) provides the EPA with authority to require reporting, record-keeping, and testing requirements for chemical substances like luminol. Manufacturers and importers must comply with these regulations to ensure the safety of workers and the environment.
The Occupational Safety and Health Administration (OSHA) has set standards for the handling and use of luminol in workplace settings. These standards include proper storage, handling, and disposal procedures, as well as requirements for personal protective equipment (PPE) when working with the chemical. OSHA also mandates that employers provide adequate training to workers who handle luminol.
In the European Union, the Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH) regulation governs the use of luminol. Manufacturers and importers must register substances like luminol with the European Chemicals Agency (ECHA) and provide safety data sheets to downstream users. REACH also requires companies to assess and manage the risks associated with luminol use.
The forensic science community has developed its own set of guidelines for the use of luminol in crime scene investigations. Organizations such as the International Association for Identification (IAI) and the American Academy of Forensic Sciences (AAFS) have published best practices for luminol application, including proper documentation, evidence preservation, and quality control measures.
In the medical field, the use of luminol in diagnostic tests is regulated by agencies such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in the EU. These agencies ensure that luminol-based diagnostic tools meet safety and efficacy standards before they can be marketed and used in clinical settings.
As luminol finds new applications in environmental monitoring and food safety, regulatory bodies are adapting their frameworks to address these emerging uses. For instance, the FDA has issued guidance on the use of luminol-based ATP testing for food safety inspections, while environmental agencies are developing protocols for using luminol in water quality assessments.
The global nature of luminol production and use necessitates international cooperation in regulatory efforts. The United Nations' Globally Harmonized System of Classification and Labelling of Chemicals (GHS) provides a standardized approach to communicating chemical hazards, including those associated with luminol, across different countries and regions.
In the United States, the Environmental Protection Agency (EPA) oversees the regulation of luminol as a chemical substance. The Toxic Substances Control Act (TSCA) provides the EPA with authority to require reporting, record-keeping, and testing requirements for chemical substances like luminol. Manufacturers and importers must comply with these regulations to ensure the safety of workers and the environment.
The Occupational Safety and Health Administration (OSHA) has set standards for the handling and use of luminol in workplace settings. These standards include proper storage, handling, and disposal procedures, as well as requirements for personal protective equipment (PPE) when working with the chemical. OSHA also mandates that employers provide adequate training to workers who handle luminol.
In the European Union, the Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH) regulation governs the use of luminol. Manufacturers and importers must register substances like luminol with the European Chemicals Agency (ECHA) and provide safety data sheets to downstream users. REACH also requires companies to assess and manage the risks associated with luminol use.
The forensic science community has developed its own set of guidelines for the use of luminol in crime scene investigations. Organizations such as the International Association for Identification (IAI) and the American Academy of Forensic Sciences (AAFS) have published best practices for luminol application, including proper documentation, evidence preservation, and quality control measures.
In the medical field, the use of luminol in diagnostic tests is regulated by agencies such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in the EU. These agencies ensure that luminol-based diagnostic tools meet safety and efficacy standards before they can be marketed and used in clinical settings.
As luminol finds new applications in environmental monitoring and food safety, regulatory bodies are adapting their frameworks to address these emerging uses. For instance, the FDA has issued guidance on the use of luminol-based ATP testing for food safety inspections, while environmental agencies are developing protocols for using luminol in water quality assessments.
The global nature of luminol production and use necessitates international cooperation in regulatory efforts. The United Nations' Globally Harmonized System of Classification and Labelling of Chemicals (GHS) provides a standardized approach to communicating chemical hazards, including those associated with luminol, across different countries and regions.
Environmental Impact of Luminol Applications
The environmental impact of luminol applications is a critical consideration as this chemiluminescent compound gains prominence in various analytical fields. Luminol's primary use in forensic science for blood detection has minimal direct environmental consequences due to its controlled application in crime scene investigations. However, its expanding role in environmental monitoring and water quality assessment introduces both benefits and potential risks.
In water quality analysis, luminol-based techniques offer highly sensitive detection of pollutants, particularly heavy metals and organic contaminants. This enhanced detection capability allows for more effective environmental protection measures and early warning systems for water pollution. The method's high sensitivity means that smaller sample sizes are required, reducing the overall chemical waste generated during analysis.
However, the increased use of luminol in environmental applications raises concerns about its own environmental fate. While luminol itself is not considered highly toxic, its breakdown products and potential accumulation in aquatic ecosystems warrant further study. The compound's stability in water and soil, as well as its bioaccumulation potential in aquatic organisms, are areas that require ongoing research to ensure long-term environmental safety.
The production of luminol and its associated reagents also carries environmental implications. Industrial synthesis of luminol involves chemical processes that may generate hazardous by-products. As demand for luminol increases, there is a growing need for greener synthesis methods and more sustainable production practices to mitigate potential environmental impacts.
In the realm of atmospheric monitoring, luminol-based chemiluminescence techniques are being explored for detecting air pollutants, particularly nitrogen oxides. While these methods offer improved sensitivity and real-time monitoring capabilities, the potential release of luminol or its derivatives into the atmosphere during sampling and analysis must be carefully managed to prevent unintended environmental consequences.
The disposal of luminol-containing waste from laboratories and forensic applications presents another environmental challenge. Proper protocols for the treatment and disposal of luminol solutions and contaminated materials are essential to prevent soil and water contamination. This necessitates the development of effective waste management strategies and potentially the exploration of biodegradable alternatives or recovery methods for luminol and its derivatives.
As luminol applications continue to expand, particularly in environmental science and monitoring, a comprehensive life cycle assessment of its environmental impact becomes increasingly important. This should encompass raw material extraction, synthesis, application, and disposal phases to provide a holistic view of luminol's environmental footprint and guide sustainable practices in its use across various analytical pathways.
In water quality analysis, luminol-based techniques offer highly sensitive detection of pollutants, particularly heavy metals and organic contaminants. This enhanced detection capability allows for more effective environmental protection measures and early warning systems for water pollution. The method's high sensitivity means that smaller sample sizes are required, reducing the overall chemical waste generated during analysis.
However, the increased use of luminol in environmental applications raises concerns about its own environmental fate. While luminol itself is not considered highly toxic, its breakdown products and potential accumulation in aquatic ecosystems warrant further study. The compound's stability in water and soil, as well as its bioaccumulation potential in aquatic organisms, are areas that require ongoing research to ensure long-term environmental safety.
The production of luminol and its associated reagents also carries environmental implications. Industrial synthesis of luminol involves chemical processes that may generate hazardous by-products. As demand for luminol increases, there is a growing need for greener synthesis methods and more sustainable production practices to mitigate potential environmental impacts.
In the realm of atmospheric monitoring, luminol-based chemiluminescence techniques are being explored for detecting air pollutants, particularly nitrogen oxides. While these methods offer improved sensitivity and real-time monitoring capabilities, the potential release of luminol or its derivatives into the atmosphere during sampling and analysis must be carefully managed to prevent unintended environmental consequences.
The disposal of luminol-containing waste from laboratories and forensic applications presents another environmental challenge. Proper protocols for the treatment and disposal of luminol solutions and contaminated materials are essential to prevent soil and water contamination. This necessitates the development of effective waste management strategies and potentially the exploration of biodegradable alternatives or recovery methods for luminol and its derivatives.
As luminol applications continue to expand, particularly in environmental science and monitoring, a comprehensive life cycle assessment of its environmental impact becomes increasingly important. This should encompass raw material extraction, synthesis, application, and disposal phases to provide a holistic view of luminol's environmental footprint and guide sustainable practices in its use across various analytical pathways.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!