Model Predictive Control For Bioprocess Engineering
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Bioprocess MPC Background and Objectives
Model Predictive Control (MPC) has emerged as a powerful advanced control strategy in bioprocess engineering over the past three decades. Initially developed for petroleum and chemical industries in the 1970s, MPC has gradually found its way into bioprocess applications due to its ability to handle complex, nonlinear, and time-varying systems with multiple inputs and outputs. The evolution of this technology has been accelerated by advancements in computational capabilities, allowing for real-time implementation of increasingly sophisticated control algorithms.
Bioprocesses present unique challenges for control systems due to their inherent complexity, including cellular metabolism variations, substrate limitations, product inhibition, and the presence of living organisms that exhibit non-linear behaviors. Traditional control methods such as PID controllers often fail to provide optimal performance in such dynamic environments, creating a significant need for more advanced control strategies like MPC.
The fundamental principle of MPC in bioprocess engineering involves using a process model to predict future system behavior, optimizing control actions over a prediction horizon, and implementing only the first control move while continuously updating predictions as new measurements become available. This receding horizon approach allows for adaptive control that can respond to the changing dynamics of biological systems.
Recent technological developments have further expanded MPC capabilities in bioprocesses, including the integration of artificial intelligence and machine learning techniques for improved model development and adaptation. The emergence of digital twins and Industry 4.0 concepts has also created new opportunities for implementing MPC in bioprocess control systems with enhanced monitoring and predictive capabilities.
The primary objectives of implementing MPC in bioprocess engineering include maximizing product yield and quality, minimizing process variability, reducing energy consumption, and ensuring consistent operation despite disturbances and uncertainties. Additionally, MPC aims to facilitate the transition from batch to continuous bioprocessing, which represents a significant trend in the biopharmaceutical industry for improving efficiency and reducing costs.
From a regulatory perspective, the implementation of MPC aligns with Quality by Design (QbD) principles advocated by regulatory agencies, as it provides a systematic approach to process understanding and control. The ability of MPC to maintain critical process parameters within their design space makes it particularly valuable for regulated bioprocesses such as pharmaceutical manufacturing.
Looking forward, the continued evolution of MPC in bioprocess engineering is expected to focus on developing more accurate and adaptable process models, improving computational efficiency for real-time applications, and enhancing robustness against model uncertainties and process disturbances. These advancements will be crucial for addressing the increasing complexity of modern bioprocesses and meeting the growing demands for process efficiency and product quality.
Bioprocesses present unique challenges for control systems due to their inherent complexity, including cellular metabolism variations, substrate limitations, product inhibition, and the presence of living organisms that exhibit non-linear behaviors. Traditional control methods such as PID controllers often fail to provide optimal performance in such dynamic environments, creating a significant need for more advanced control strategies like MPC.
The fundamental principle of MPC in bioprocess engineering involves using a process model to predict future system behavior, optimizing control actions over a prediction horizon, and implementing only the first control move while continuously updating predictions as new measurements become available. This receding horizon approach allows for adaptive control that can respond to the changing dynamics of biological systems.
Recent technological developments have further expanded MPC capabilities in bioprocesses, including the integration of artificial intelligence and machine learning techniques for improved model development and adaptation. The emergence of digital twins and Industry 4.0 concepts has also created new opportunities for implementing MPC in bioprocess control systems with enhanced monitoring and predictive capabilities.
The primary objectives of implementing MPC in bioprocess engineering include maximizing product yield and quality, minimizing process variability, reducing energy consumption, and ensuring consistent operation despite disturbances and uncertainties. Additionally, MPC aims to facilitate the transition from batch to continuous bioprocessing, which represents a significant trend in the biopharmaceutical industry for improving efficiency and reducing costs.
From a regulatory perspective, the implementation of MPC aligns with Quality by Design (QbD) principles advocated by regulatory agencies, as it provides a systematic approach to process understanding and control. The ability of MPC to maintain critical process parameters within their design space makes it particularly valuable for regulated bioprocesses such as pharmaceutical manufacturing.
Looking forward, the continued evolution of MPC in bioprocess engineering is expected to focus on developing more accurate and adaptable process models, improving computational efficiency for real-time applications, and enhancing robustness against model uncertainties and process disturbances. These advancements will be crucial for addressing the increasing complexity of modern bioprocesses and meeting the growing demands for process efficiency and product quality.
Market Analysis for MPC in Bioprocessing
The global market for Model Predictive Control (MPC) in bioprocessing is experiencing significant growth, driven by increasing demand for biopharmaceuticals and the need for more efficient manufacturing processes. Current market valuations indicate that the bioprocess automation market, of which MPC is a growing segment, reached approximately $4.2 billion in 2022 and is projected to grow at a CAGR of 8.5% through 2028.
Biopharmaceutical manufacturing represents the largest application segment for MPC technologies, accounting for nearly 40% of the market share. This dominance is attributed to the stringent regulatory requirements and the high value of biopharmaceutical products, which justify investments in advanced control systems. The industrial enzyme production sector follows as the second-largest application area, representing about 25% of the market.
Geographically, North America leads the MPC bioprocessing market with approximately 35% market share, followed by Europe at 30% and Asia-Pacific at 25%. The Asia-Pacific region, particularly China and India, is expected to witness the fastest growth due to expanding biopharmaceutical manufacturing capabilities and increasing adoption of automation technologies.
Key market drivers include the rising complexity of bioprocesses, increasing regulatory pressures for consistent product quality, and the industry-wide push toward continuous manufacturing. The COVID-19 pandemic has accelerated market growth by highlighting the need for flexible and robust manufacturing processes that can rapidly adapt to changing demands.
Customer segments for MPC in bioprocessing include large pharmaceutical companies (40% of the market), contract manufacturing organizations (30%), academic and research institutions (15%), and small to medium biotech companies (15%). Large pharmaceutical companies typically seek enterprise-wide solutions, while smaller organizations often prefer modular implementations that can be scaled over time.
Market barriers include high initial implementation costs, technical complexity requiring specialized expertise, and integration challenges with existing systems. The return on investment period for MPC implementations in bioprocessing typically ranges from 12 to 24 months, depending on the scale and complexity of operations.
Future market trends indicate a shift toward cloud-based MPC solutions, integration with artificial intelligence for enhanced predictive capabilities, and the development of industry-specific MPC packages tailored to particular bioprocessing applications. The market is also witnessing increased collaboration between technology providers and bioprocess equipment manufacturers to offer integrated solutions.
Biopharmaceutical manufacturing represents the largest application segment for MPC technologies, accounting for nearly 40% of the market share. This dominance is attributed to the stringent regulatory requirements and the high value of biopharmaceutical products, which justify investments in advanced control systems. The industrial enzyme production sector follows as the second-largest application area, representing about 25% of the market.
Geographically, North America leads the MPC bioprocessing market with approximately 35% market share, followed by Europe at 30% and Asia-Pacific at 25%. The Asia-Pacific region, particularly China and India, is expected to witness the fastest growth due to expanding biopharmaceutical manufacturing capabilities and increasing adoption of automation technologies.
Key market drivers include the rising complexity of bioprocesses, increasing regulatory pressures for consistent product quality, and the industry-wide push toward continuous manufacturing. The COVID-19 pandemic has accelerated market growth by highlighting the need for flexible and robust manufacturing processes that can rapidly adapt to changing demands.
Customer segments for MPC in bioprocessing include large pharmaceutical companies (40% of the market), contract manufacturing organizations (30%), academic and research institutions (15%), and small to medium biotech companies (15%). Large pharmaceutical companies typically seek enterprise-wide solutions, while smaller organizations often prefer modular implementations that can be scaled over time.
Market barriers include high initial implementation costs, technical complexity requiring specialized expertise, and integration challenges with existing systems. The return on investment period for MPC implementations in bioprocessing typically ranges from 12 to 24 months, depending on the scale and complexity of operations.
Future market trends indicate a shift toward cloud-based MPC solutions, integration with artificial intelligence for enhanced predictive capabilities, and the development of industry-specific MPC packages tailored to particular bioprocessing applications. The market is also witnessing increased collaboration between technology providers and bioprocess equipment manufacturers to offer integrated solutions.
Current State and Challenges of Bioprocess Control
Bioprocess control has evolved significantly over the past decades, transitioning from simple feedback control systems to sophisticated model-based approaches. Currently, the implementation of Model Predictive Control (MPC) in bioprocess engineering represents a significant advancement, yet remains limited in industrial applications compared to other sectors like petrochemical or automotive industries. This disparity stems from the inherent complexity and nonlinearity of biological systems, which present unique challenges for control engineers.
The state-of-the-art in bioprocess control predominantly features hybrid approaches that combine first-principles models with data-driven techniques. These systems typically incorporate real-time monitoring through Process Analytical Technology (PAT) tools, enabling continuous adjustment of process parameters. However, the integration of these technologies with MPC frameworks remains fragmented across the industry, with varying levels of sophistication and implementation success.
A significant challenge in current bioprocess control is the development of accurate and robust dynamic models that can capture the complex behavior of biological systems. Traditional mechanistic models often struggle with parameter uncertainty and process variability, while purely data-driven approaches may lack interpretability and extrapolation capabilities. This modeling challenge directly impacts the performance of MPC strategies, particularly in handling disturbances and maintaining optimal operating conditions.
Real-time monitoring presents another critical challenge, as many key bioprocess parameters cannot be measured directly or require offline analysis with significant time delays. This limitation creates difficulties in state estimation and feedback control, necessitating the development of soft sensors and inferential measurement techniques. The integration of these estimation methods with MPC frameworks remains an active area of research with considerable implementation hurdles.
Computational complexity and solver efficiency constitute additional barriers to widespread MPC adoption in bioprocess engineering. The nonlinear nature of bioprocesses often requires computationally intensive optimization algorithms that may not be suitable for real-time applications, particularly in systems with fast dynamics or multiple control objectives.
Regulatory considerations further complicate the implementation landscape, as any advanced control strategy in biopharmaceutical manufacturing must comply with stringent validation requirements and quality standards. This regulatory burden often discourages innovation and experimentation with novel control approaches, leading many manufacturers to rely on well-established but potentially suboptimal control strategies.
Despite these challenges, recent advances in computational capabilities, machine learning techniques, and sensor technologies are gradually enabling more sophisticated MPC implementations in bioprocess engineering, suggesting a promising trajectory for future developments in this field.
The state-of-the-art in bioprocess control predominantly features hybrid approaches that combine first-principles models with data-driven techniques. These systems typically incorporate real-time monitoring through Process Analytical Technology (PAT) tools, enabling continuous adjustment of process parameters. However, the integration of these technologies with MPC frameworks remains fragmented across the industry, with varying levels of sophistication and implementation success.
A significant challenge in current bioprocess control is the development of accurate and robust dynamic models that can capture the complex behavior of biological systems. Traditional mechanistic models often struggle with parameter uncertainty and process variability, while purely data-driven approaches may lack interpretability and extrapolation capabilities. This modeling challenge directly impacts the performance of MPC strategies, particularly in handling disturbances and maintaining optimal operating conditions.
Real-time monitoring presents another critical challenge, as many key bioprocess parameters cannot be measured directly or require offline analysis with significant time delays. This limitation creates difficulties in state estimation and feedback control, necessitating the development of soft sensors and inferential measurement techniques. The integration of these estimation methods with MPC frameworks remains an active area of research with considerable implementation hurdles.
Computational complexity and solver efficiency constitute additional barriers to widespread MPC adoption in bioprocess engineering. The nonlinear nature of bioprocesses often requires computationally intensive optimization algorithms that may not be suitable for real-time applications, particularly in systems with fast dynamics or multiple control objectives.
Regulatory considerations further complicate the implementation landscape, as any advanced control strategy in biopharmaceutical manufacturing must comply with stringent validation requirements and quality standards. This regulatory burden often discourages innovation and experimentation with novel control approaches, leading many manufacturers to rely on well-established but potentially suboptimal control strategies.
Despite these challenges, recent advances in computational capabilities, machine learning techniques, and sensor technologies are gradually enabling more sophisticated MPC implementations in bioprocess engineering, suggesting a promising trajectory for future developments in this field.
Current MPC Implementation Approaches for Bioprocesses
01 Industrial process control applications
Model Predictive Control (MPC) is widely applied in industrial processes to optimize operations and improve efficiency. These systems use mathematical models to predict future behavior of processes and calculate optimal control actions. MPC algorithms can handle complex constraints and multiple variables simultaneously, making them particularly valuable in manufacturing, chemical processing, and energy production where precise control is critical for product quality and operational safety.- Industrial process control applications: Model Predictive Control (MPC) is widely applied in industrial processes to optimize operations and improve efficiency. These systems use mathematical models to predict future behavior of processes and calculate optimal control actions. MPC algorithms can handle complex constraints and multiple variables simultaneously, making them suitable for manufacturing, chemical processing, and energy production systems where precise control is critical for product quality and operational safety.
- Advanced automotive control systems: Model Predictive Control is increasingly implemented in automotive applications for enhanced vehicle performance and safety. These systems predict vehicle behavior under various conditions and optimize control actions for engine management, transmission control, and autonomous driving features. MPC algorithms enable vehicles to adapt to changing road conditions, optimize fuel efficiency, and improve handling characteristics while maintaining safety constraints and driver comfort.
- Energy management and optimization: Model Predictive Control provides sophisticated solutions for energy management systems by optimizing resource allocation and consumption patterns. These controllers predict energy demand, manage storage systems, and balance generation sources to minimize costs and environmental impact. MPC algorithms are particularly valuable in renewable energy integration, building climate control, and grid management where they can anticipate fluctuations and optimize operations across multiple time horizons.
- Machine learning enhanced MPC: Integration of machine learning techniques with Model Predictive Control creates more adaptive and robust control systems. These hybrid approaches use data-driven methods to improve model accuracy, handle uncertainties, and optimize controller performance. Machine learning algorithms can identify patterns in system behavior, adapt to changing conditions, and enhance the predictive capabilities of traditional MPC frameworks, resulting in more efficient and resilient control systems.
- Distributed and networked MPC architectures: Distributed Model Predictive Control architectures enable coordination of multiple subsystems across networked environments. These approaches divide complex control problems into manageable components while maintaining overall system optimization. Distributed MPC is particularly valuable in large-scale applications such as smart grids, water distribution networks, and interconnected manufacturing systems where centralized control would be computationally prohibitive or communication-constrained.
02 Advanced automotive control systems
Model Predictive Control is increasingly implemented in automotive applications for enhanced vehicle performance and safety. These systems predict vehicle behavior under various conditions and optimize control actions accordingly. Applications include engine management, transmission control, adaptive cruise control, and autonomous driving features. MPC algorithms help vehicles maintain optimal performance while adapting to changing road conditions and driver inputs, resulting in improved fuel efficiency and driving safety.Expand Specific Solutions03 Energy management and optimization
Model Predictive Control provides sophisticated solutions for energy management systems by optimizing resource allocation and consumption patterns. These controllers predict energy demand and supply fluctuations to minimize costs and environmental impact. Applications include smart grid management, building climate control, renewable energy integration, and power plant operation. MPC algorithms balance multiple objectives such as energy efficiency, cost reduction, and emission control while maintaining system stability and reliability.Expand Specific Solutions04 Machine learning integration with MPC
The integration of machine learning techniques with Model Predictive Control represents a significant advancement in control system technology. These hybrid approaches use data-driven methods to improve model accuracy and adaptability. Neural networks and other AI techniques can enhance MPC performance by continuously refining predictive models based on operational data. This combination enables more robust control in complex, nonlinear systems and environments with significant uncertainties or changing dynamics.Expand Specific Solutions05 Distributed and networked MPC systems
Distributed Model Predictive Control architectures enable coordination among multiple interconnected subsystems while maintaining computational efficiency. These approaches divide large-scale control problems into smaller, manageable components that communicate and collaborate. Applications include large industrial complexes, smart city infrastructure, and multi-agent robotic systems. Distributed MPC provides scalability and resilience against single-point failures while optimizing overall system performance through coordinated local decision-making.Expand Specific Solutions
Leading Companies in Bioprocess Control Systems
Model Predictive Control (MPC) for bioprocess engineering is currently in a growth phase, with the market expanding as biopharmaceutical manufacturing becomes increasingly sophisticated. The global market size is estimated to be growing at 8-10% annually, driven by demand for optimized bioprocessing operations. Technologically, MPC implementation in bioprocesses is maturing but still evolving, with varying levels of adoption across companies. Industry leaders like Rockwell Automation, Sartorius Stedim Data Analytics, and Emerson Process Management offer established control solutions, while biopharmaceutical companies such as Amgen, Biogen, and Lonza are implementing these technologies in production environments. Academic institutions including Katholieke Universiteit Leuven and Colorado State University are advancing fundamental research, while newer entrants like Form Bio and Lynceus are introducing AI-enhanced predictive control solutions for bioprocessing applications.
Fisher-Rosemount Systems, Inc.
Technical Solution: Fisher-Rosemount Systems has developed DeltaV Predict, an advanced Model Predictive Control solution adapted specifically for bioprocess engineering applications. Their approach leverages the company's extensive experience in process control across multiple industries, now tailored to address the unique challenges of biological systems. The DeltaV Predict platform utilizes a multi-model framework that can simultaneously handle the different phases of bioprocessing from inoculation through production and harvest. Their implementation features state estimation algorithms that can infer critical biological parameters that cannot be measured directly, such as specific growth rates and metabolic flux distributions. The system employs a dynamic constraint handling mechanism that adapts control boundaries based on the current state of the bioprocess, allowing for tighter control during critical phases while providing flexibility during less sensitive periods. Fisher-Rosemount's solution includes robust disturbance rejection capabilities specifically designed to handle the inherent variability in raw materials and biological responses common in bioprocessing[5]. Their MPC framework integrates seamlessly with their broader automation platform, providing a unified interface for operators and facilitating implementation across existing bioprocess facilities. The system has been successfully deployed across various bioprocess applications including vaccine production, enzyme manufacturing, and therapeutic protein expression.
Strengths: Exceptional integration with existing automation infrastructure reduces implementation barriers. Their industrial-scale control expertise translates to robust solutions that can handle real-world manufacturing variability. Weaknesses: The general-purpose control platform may require significant customization for specialized bioprocesses, and the system may not capture all biological complexities without extensive model development.
Sartorius Stedim Data Analytics AB
Technical Solution: Sartorius Stedim Data Analytics AB has developed a comprehensive Model Predictive Control (MPC) solution specifically for bioprocess engineering called SIMCA-online. This platform integrates multivariate data analysis with advanced predictive control algorithms to optimize bioprocess performance in real-time. Their approach utilizes both mechanistic and data-driven models to capture complex biological interactions and process dynamics. The system continuously monitors critical process parameters (CPPs) and predicts critical quality attributes (CQAs) several steps ahead, allowing for proactive control adjustments. Sartorius has implemented a hierarchical control structure where MPC algorithms operate at different time scales - from rapid adjustments of basic parameters to longer-term trajectory optimization for entire bioprocesses. Their solution includes adaptive model updating capabilities that account for biological variability and process drift over time, ensuring sustained performance even as conditions change[1][2]. The platform also features a digital twin component that enables offline scenario testing and process optimization before implementation in actual production environments.
Strengths: Specialized expertise in bioprocess applications with proven implementation across multiple biopharmaceutical processes. Their integration of multivariate data analysis with MPC provides superior handling of complex biological systems. Weaknesses: The system requires significant upfront modeling effort and process knowledge to implement effectively, potentially limiting accessibility for smaller biotech operations without extensive data science capabilities.
Key Algorithms and Models for Bioprocess MPC
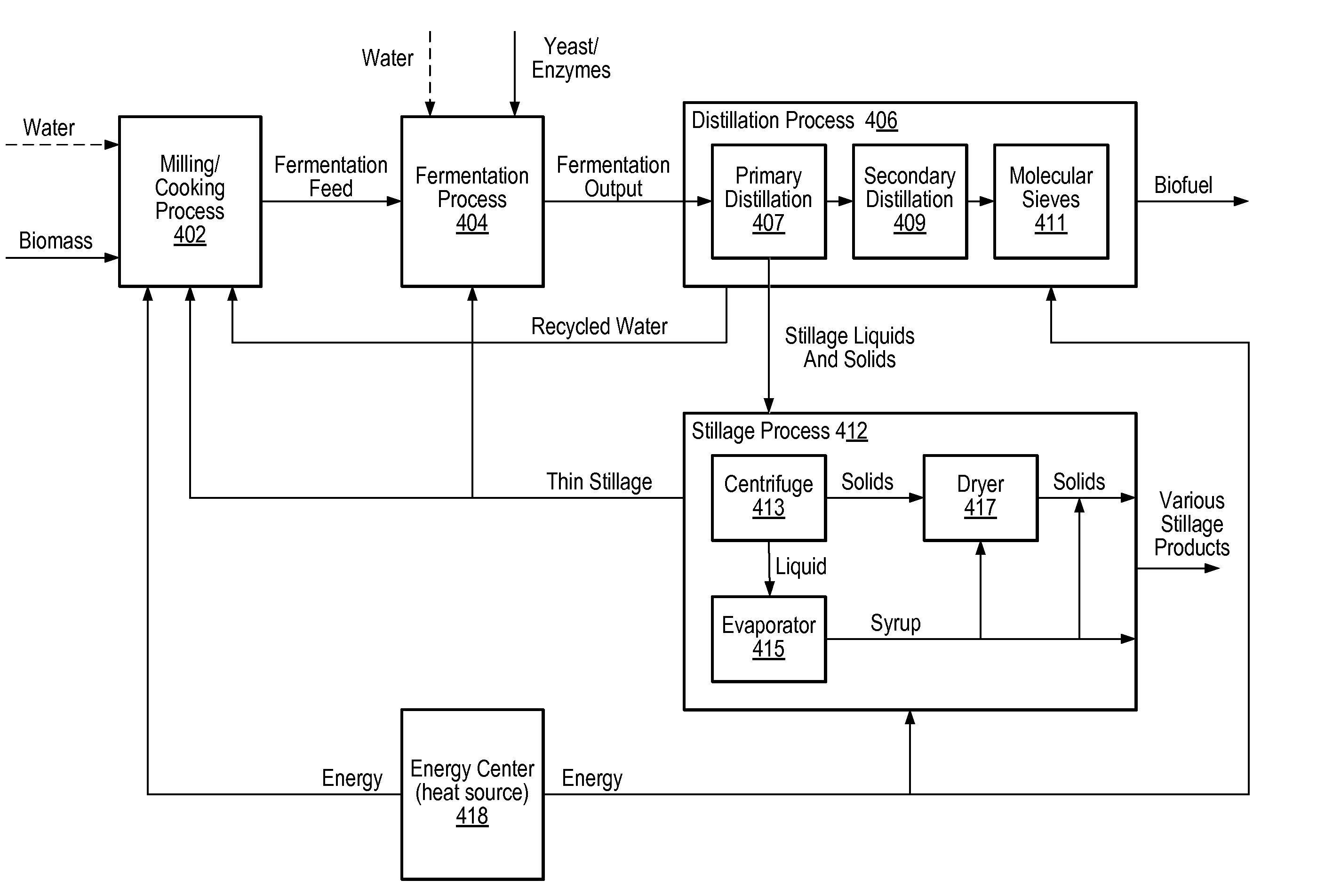
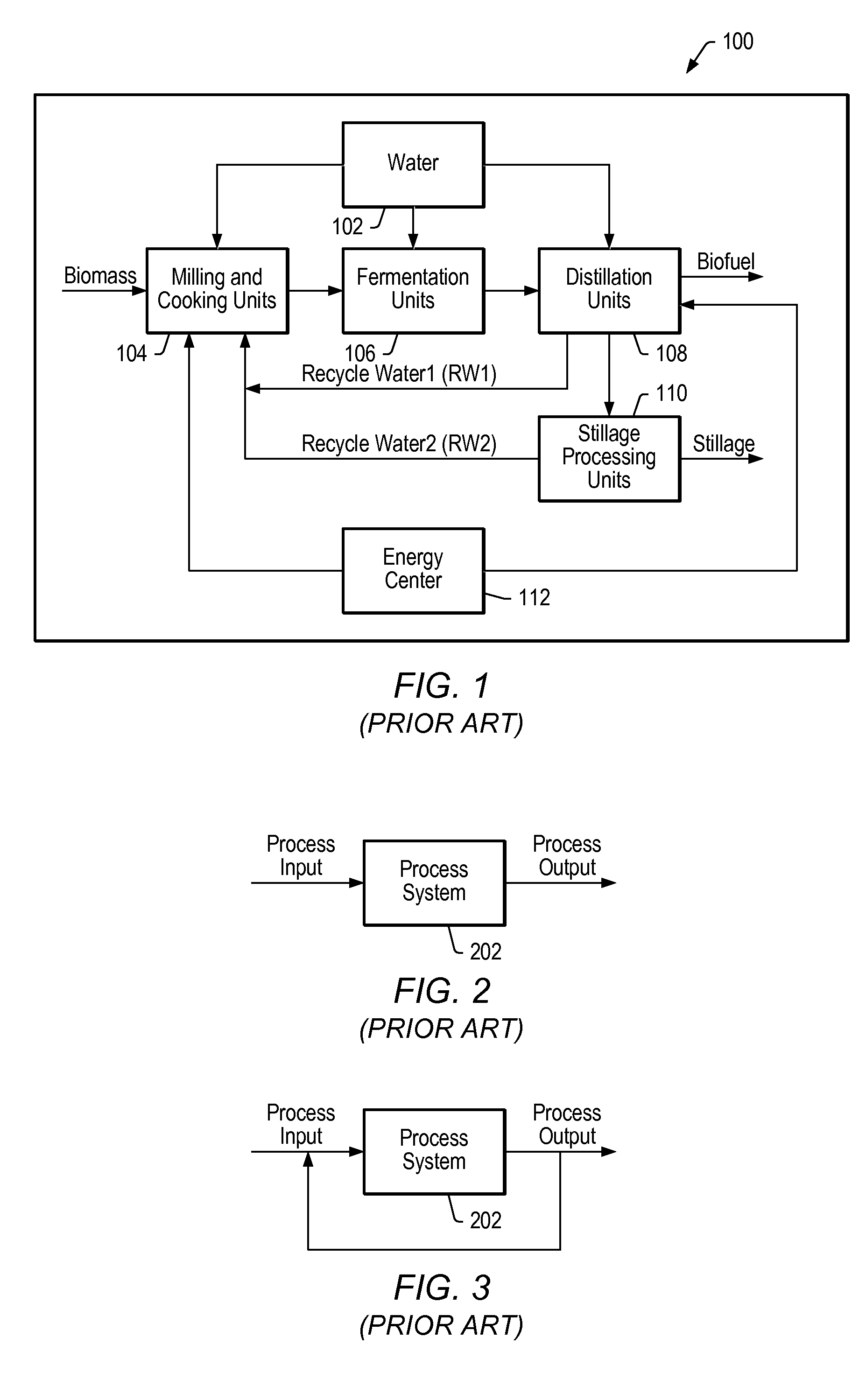
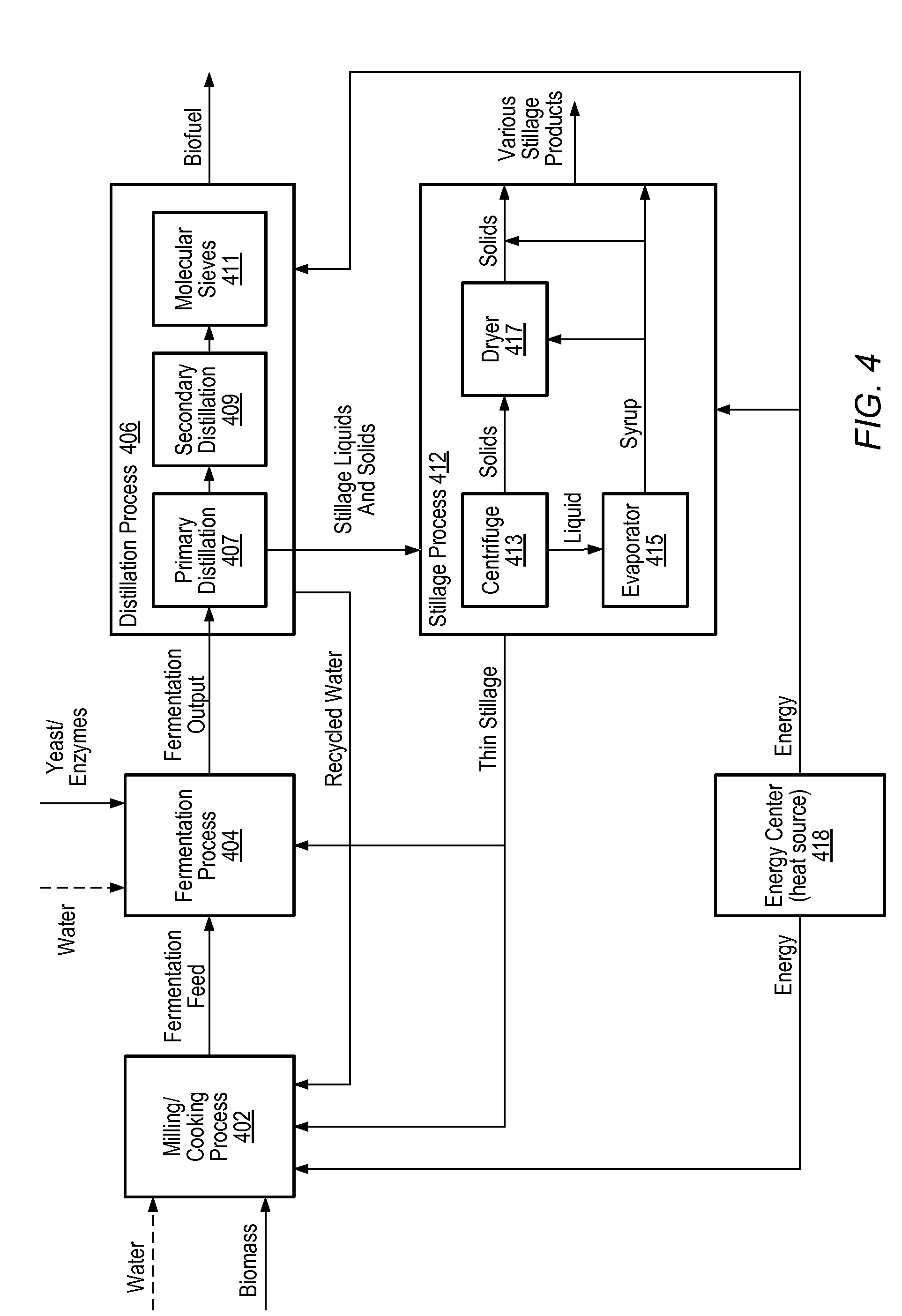
Model predictive control of a fermentation feed in biofuel production
PatentActiveUS20080104003A1
Innovation
- A dynamic multivariate predictive model-based controller is implemented to stabilize water/biomass balance in the fermentation feed by adjusting flow rates of water and biomass, incorporating target values for manipulated variables such as mill speed, pump speeds, and enzyme flow rates, while considering equipment constraints and temperature control.
Hybrid predictive modeling for control of cell culture
PatentWO2023167802A1
Innovation
- A hybrid predictive modeling approach combining first-principle and data-driven models, using linear regressors or neural networks to predict metabolite concentrations and cell culture attributes, while first-principle models like mass balance predict glucose concentration, optimizing control actions through model predictive control.
Regulatory Compliance for Bioprocess Control Systems
Regulatory compliance represents a critical dimension in the implementation of Model Predictive Control (MPC) systems for bioprocess engineering. The biopharmaceutical industry operates under stringent regulatory frameworks established by authorities such as the FDA, EMA, and ICH. These regulations ensure product quality, patient safety, and process consistency. For MPC implementation in bioprocesses, compliance with 21 CFR Part 11 (Electronic Records and Electronic Signatures) is particularly significant as these systems rely heavily on digital data collection, processing, and storage.
The validation of MPC systems in bioprocess applications must adhere to GAMP 5 (Good Automated Manufacturing Practice) guidelines, which provide a risk-based approach to computerized system compliance. This includes comprehensive documentation of system requirements, design specifications, risk assessments, and testing protocols. The qualification process typically follows the IQ/OQ/PQ (Installation Qualification/Operational Qualification/Performance Qualification) methodology to ensure that control systems perform as intended within the regulated environment.
Process Analytical Technology (PAT) initiatives, encouraged by regulatory bodies, align perfectly with MPC implementation in bioprocesses. PAT frameworks promote real-time monitoring and control of critical quality attributes, which MPC systems can effectively manage through their predictive capabilities. Companies implementing MPC must demonstrate how these advanced control strategies contribute to consistent product quality and process understanding, key requirements under Quality by Design (QbD) principles.
Change management represents another significant regulatory consideration. Any modification to an MPC system post-validation requires careful assessment through established change control procedures. This includes evaluating the potential impact on product quality, process performance, and regulatory compliance. Depending on the significance of changes, additional validation activities and regulatory notifications may be necessary.
Data integrity within MPC systems must comply with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). This ensures that all data used for model development, validation, and ongoing control decisions meets regulatory expectations. Audit trails must capture all system activities, particularly those affecting critical process parameters or quality attributes.
International harmonization efforts are gradually standardizing regulatory approaches to advanced process control technologies like MPC. The ICH Q8, Q9, Q10, and Q12 guidelines collectively provide a framework for implementing innovative control strategies within a compliant quality management system. Organizations must navigate these evolving regulatory landscapes while demonstrating that their MPC implementations maintain or enhance product quality and process robustness.
The validation of MPC systems in bioprocess applications must adhere to GAMP 5 (Good Automated Manufacturing Practice) guidelines, which provide a risk-based approach to computerized system compliance. This includes comprehensive documentation of system requirements, design specifications, risk assessments, and testing protocols. The qualification process typically follows the IQ/OQ/PQ (Installation Qualification/Operational Qualification/Performance Qualification) methodology to ensure that control systems perform as intended within the regulated environment.
Process Analytical Technology (PAT) initiatives, encouraged by regulatory bodies, align perfectly with MPC implementation in bioprocesses. PAT frameworks promote real-time monitoring and control of critical quality attributes, which MPC systems can effectively manage through their predictive capabilities. Companies implementing MPC must demonstrate how these advanced control strategies contribute to consistent product quality and process understanding, key requirements under Quality by Design (QbD) principles.
Change management represents another significant regulatory consideration. Any modification to an MPC system post-validation requires careful assessment through established change control procedures. This includes evaluating the potential impact on product quality, process performance, and regulatory compliance. Depending on the significance of changes, additional validation activities and regulatory notifications may be necessary.
Data integrity within MPC systems must comply with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). This ensures that all data used for model development, validation, and ongoing control decisions meets regulatory expectations. Audit trails must capture all system activities, particularly those affecting critical process parameters or quality attributes.
International harmonization efforts are gradually standardizing regulatory approaches to advanced process control technologies like MPC. The ICH Q8, Q9, Q10, and Q12 guidelines collectively provide a framework for implementing innovative control strategies within a compliant quality management system. Organizations must navigate these evolving regulatory landscapes while demonstrating that their MPC implementations maintain or enhance product quality and process robustness.
Economic Impact and ROI Analysis of MPC Implementation
The implementation of Model Predictive Control (MPC) in bioprocess engineering represents a significant capital investment that requires thorough economic justification. Initial implementation costs typically range from $50,000 to $500,000 depending on process complexity, encompassing software licensing, hardware upgrades, sensor integration, and specialized engineering expertise. However, these investments have demonstrated compelling returns across multiple bioprocess applications.
Operational cost reductions constitute the primary economic benefit, with MPC implementations consistently achieving 10-15% reductions in energy consumption through optimized heating, cooling, and mixing operations. Raw material utilization improvements of 5-8% have been documented in commercial biopharmaceutical processes, particularly in media component optimization and feeding strategies for cell cultures. These efficiencies translate directly to lower production costs per batch.
Quality improvements represent another significant economic advantage. By maintaining critical process parameters within tighter control bands, MPC implementations have reduced batch-to-batch variability by up to 40% in industrial fermentation processes. This enhanced consistency directly impacts product quality attributes, reducing rejection rates by 15-25% in documented case studies. For high-value biopharmaceuticals, even marginal improvements in product quality can translate to millions in recovered revenue.
Productivity enhancements further bolster the economic case for MPC adoption. Throughput increases of 8-12% have been achieved through cycle time optimization and reduced downtime between batches. In continuous bioprocessing applications, MPC has enabled stable operation at higher cell densities, increasing volumetric productivity by up to 20% in perfusion bioreactors.
Return on investment timelines vary by application but typically range from 12-24 months for standard bioprocesses. Particularly compelling ROI cases emerge in high-value product manufacturing, where even minor yield improvements justify implementation costs. A comprehensive analysis of 15 industrial bioprocess MPC implementations revealed average ROI of 200-300% over a five-year period, with some applications in vaccine manufacturing achieving ROI exceeding 500%.
Risk mitigation benefits, while more difficult to quantify, provide additional economic value through reduced regulatory compliance issues and fewer production deviations. The predictive capabilities of MPC allow for proactive intervention before process excursions occur, potentially avoiding costly batch failures that can exceed $1 million per incident in commercial biopharmaceutical production.
Operational cost reductions constitute the primary economic benefit, with MPC implementations consistently achieving 10-15% reductions in energy consumption through optimized heating, cooling, and mixing operations. Raw material utilization improvements of 5-8% have been documented in commercial biopharmaceutical processes, particularly in media component optimization and feeding strategies for cell cultures. These efficiencies translate directly to lower production costs per batch.
Quality improvements represent another significant economic advantage. By maintaining critical process parameters within tighter control bands, MPC implementations have reduced batch-to-batch variability by up to 40% in industrial fermentation processes. This enhanced consistency directly impacts product quality attributes, reducing rejection rates by 15-25% in documented case studies. For high-value biopharmaceuticals, even marginal improvements in product quality can translate to millions in recovered revenue.
Productivity enhancements further bolster the economic case for MPC adoption. Throughput increases of 8-12% have been achieved through cycle time optimization and reduced downtime between batches. In continuous bioprocessing applications, MPC has enabled stable operation at higher cell densities, increasing volumetric productivity by up to 20% in perfusion bioreactors.
Return on investment timelines vary by application but typically range from 12-24 months for standard bioprocesses. Particularly compelling ROI cases emerge in high-value product manufacturing, where even minor yield improvements justify implementation costs. A comprehensive analysis of 15 industrial bioprocess MPC implementations revealed average ROI of 200-300% over a five-year period, with some applications in vaccine manufacturing achieving ROI exceeding 500%.
Risk mitigation benefits, while more difficult to quantify, provide additional economic value through reduced regulatory compliance issues and fewer production deviations. The predictive capabilities of MPC allow for proactive intervention before process excursions occur, potentially avoiding costly batch failures that can exceed $1 million per incident in commercial biopharmaceutical production.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!