Achieving Single-cell Resolution using Isoelectric Focusing Systems
SEP 10, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Single-cell IEF Technology Background and Objectives
Isoelectric focusing (IEF) technology has evolved significantly since its inception in the 1960s, transitioning from a bulk separation technique to a sophisticated analytical method capable of single-cell resolution. This evolution represents a paradigm shift in bioanalytical capabilities, enabling researchers to probe cellular heterogeneity at unprecedented levels of detail. The journey toward single-cell IEF has been driven by advances in microfluidics, surface chemistry, and detection technologies, collectively addressing the fundamental challenge of analyzing minute sample volumes with high precision.
The development of single-cell IEF systems addresses a critical gap in contemporary biomedical research: the need to understand cellular diversity beyond population averages. Traditional bulk analysis methods obscure important variations between individual cells, potentially missing crucial biological insights relevant to disease progression, drug response, and developmental processes. Single-cell IEF technology aims to reveal these hidden patterns by separating cellular components based on their isoelectric points at the individual cell level.
Current technological objectives for single-cell IEF systems focus on several key areas: improving separation resolution to distinguish closely related protein isoforms, enhancing detection sensitivity to capture low-abundance molecules, increasing throughput to analyze statistically significant cell populations, and developing integrated platforms that combine separation with downstream analysis capabilities. These objectives align with the broader trend toward comprehensive single-cell multi-omics approaches in biomedical research.
The technical evolution trajectory shows a clear progression from conventional gel-based IEF systems toward miniaturized microchip platforms with integrated functionalities. Early attempts at single-cell IEF faced significant challenges related to sample handling, protein adsorption to surfaces, and detection limits. Recent innovations have leveraged advances in microfabrication, surface modification strategies, and ultrasensitive detection methods to overcome these limitations.
Looking forward, single-cell IEF technology aims to achieve seamless integration with other analytical modalities, real-time monitoring capabilities, and automated high-throughput processing. The ultimate goal is to develop robust, user-friendly systems that can be deployed in clinical settings for personalized medicine applications, enabling precise characterization of cellular states in health and disease. This would represent a transformative capability for diagnostics, therapeutic development, and fundamental biological research.
The development of single-cell IEF systems addresses a critical gap in contemporary biomedical research: the need to understand cellular diversity beyond population averages. Traditional bulk analysis methods obscure important variations between individual cells, potentially missing crucial biological insights relevant to disease progression, drug response, and developmental processes. Single-cell IEF technology aims to reveal these hidden patterns by separating cellular components based on their isoelectric points at the individual cell level.
Current technological objectives for single-cell IEF systems focus on several key areas: improving separation resolution to distinguish closely related protein isoforms, enhancing detection sensitivity to capture low-abundance molecules, increasing throughput to analyze statistically significant cell populations, and developing integrated platforms that combine separation with downstream analysis capabilities. These objectives align with the broader trend toward comprehensive single-cell multi-omics approaches in biomedical research.
The technical evolution trajectory shows a clear progression from conventional gel-based IEF systems toward miniaturized microchip platforms with integrated functionalities. Early attempts at single-cell IEF faced significant challenges related to sample handling, protein adsorption to surfaces, and detection limits. Recent innovations have leveraged advances in microfabrication, surface modification strategies, and ultrasensitive detection methods to overcome these limitations.
Looking forward, single-cell IEF technology aims to achieve seamless integration with other analytical modalities, real-time monitoring capabilities, and automated high-throughput processing. The ultimate goal is to develop robust, user-friendly systems that can be deployed in clinical settings for personalized medicine applications, enabling precise characterization of cellular states in health and disease. This would represent a transformative capability for diagnostics, therapeutic development, and fundamental biological research.
Market Analysis for Single-cell Resolution Technologies
The single-cell resolution technology market has experienced remarkable growth in recent years, driven by increasing demand for precision medicine and personalized healthcare solutions. The global market for single-cell technologies was valued at approximately $2.5 billion in 2020 and is projected to reach $6.3 billion by 2025, growing at a CAGR of 20.1%. Within this broader market, isoelectric focusing (IEF) systems represent a specialized segment with significant growth potential due to their unique capabilities in protein separation and analysis at the single-cell level.
Healthcare and pharmaceutical sectors currently dominate the market demand, accounting for nearly 65% of the total market share. Research institutions and academic laboratories constitute another significant segment, representing approximately 25% of the market. The remaining 10% is distributed across various industries including biotechnology startups and diagnostic companies seeking innovative approaches to disease detection and monitoring.
Geographically, North America leads the market with approximately 45% share, followed by Europe (30%) and Asia-Pacific (20%). The Asia-Pacific region, particularly China and Japan, is expected to witness the fastest growth rate in the coming years due to increasing investments in healthcare infrastructure and research facilities.
Key market drivers include the rising prevalence of chronic diseases requiring personalized treatment approaches, growing investments in proteomics research, and technological advancements enabling higher resolution and throughput in single-cell analysis. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of rapid and precise diagnostic technologies.
Market challenges include the high cost of equipment and consumables, technical complexity requiring specialized training, and regulatory hurdles for clinical applications. The average cost of implementing a comprehensive single-cell analysis platform ranges from $200,000 to $500,000, creating significant barriers to entry for smaller laboratories and institutions.
Customer segments can be categorized into three tiers: high-end research institutions requiring maximum resolution and throughput, mid-tier clinical laboratories seeking reliable and standardized solutions, and emerging markets looking for cost-effective systems with essential functionalities. IEF systems with single-cell resolution capabilities are primarily targeting the high-end segment, with potential for expansion into clinical applications as technology matures and costs decrease.
The market is expected to evolve toward integrated platforms combining multiple analytical techniques, cloud-based data management solutions, and AI-powered interpretation tools, creating opportunities for comprehensive single-cell analysis ecosystems rather than standalone technologies.
Healthcare and pharmaceutical sectors currently dominate the market demand, accounting for nearly 65% of the total market share. Research institutions and academic laboratories constitute another significant segment, representing approximately 25% of the market. The remaining 10% is distributed across various industries including biotechnology startups and diagnostic companies seeking innovative approaches to disease detection and monitoring.
Geographically, North America leads the market with approximately 45% share, followed by Europe (30%) and Asia-Pacific (20%). The Asia-Pacific region, particularly China and Japan, is expected to witness the fastest growth rate in the coming years due to increasing investments in healthcare infrastructure and research facilities.
Key market drivers include the rising prevalence of chronic diseases requiring personalized treatment approaches, growing investments in proteomics research, and technological advancements enabling higher resolution and throughput in single-cell analysis. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of rapid and precise diagnostic technologies.
Market challenges include the high cost of equipment and consumables, technical complexity requiring specialized training, and regulatory hurdles for clinical applications. The average cost of implementing a comprehensive single-cell analysis platform ranges from $200,000 to $500,000, creating significant barriers to entry for smaller laboratories and institutions.
Customer segments can be categorized into three tiers: high-end research institutions requiring maximum resolution and throughput, mid-tier clinical laboratories seeking reliable and standardized solutions, and emerging markets looking for cost-effective systems with essential functionalities. IEF systems with single-cell resolution capabilities are primarily targeting the high-end segment, with potential for expansion into clinical applications as technology matures and costs decrease.
The market is expected to evolve toward integrated platforms combining multiple analytical techniques, cloud-based data management solutions, and AI-powered interpretation tools, creating opportunities for comprehensive single-cell analysis ecosystems rather than standalone technologies.
Current Challenges in Isoelectric Focusing Systems
Despite significant advancements in isoelectric focusing (IEF) technology, achieving reliable single-cell resolution remains challenging due to several persistent technical limitations. The primary obstacle lies in the inherent heterogeneity of cellular samples, which creates substantial variability in protein expression profiles even among seemingly identical cell populations. This biological variability necessitates extremely high resolution separation capabilities that current IEF systems struggle to consistently deliver.
Sample preparation represents another critical challenge, as single cells contain minute amounts of protein material that can be easily lost or degraded during handling. The extraction and processing protocols must be exceptionally gentle yet effective, requiring specialized microfluidic approaches that are not yet standardized across the field. Additionally, the presence of highly abundant proteins often masks the detection of low-abundance proteins that may be biologically significant, creating a dynamic range problem that limits comprehensive single-cell proteome analysis.
The miniaturization of IEF systems to accommodate single-cell samples introduces significant engineering challenges. Maintaining stable pH gradients at microscale dimensions is particularly difficult, as small volumes are more susceptible to disruption from environmental factors and electrochemical reactions at the electrodes. These disturbances can lead to gradient drift and poor reproducibility, undermining the precision required for single-cell analysis.
Detection sensitivity presents another substantial hurdle. Traditional protein detection methods often lack the sensitivity required to detect the extremely low concentrations of proteins present in individual cells. While fluorescence-based detection has improved sensitivity, it typically requires labeling that may alter protein properties or introduce artifacts. Label-free detection methods with sufficient sensitivity for single-cell applications remain limited in their commercial availability and practical implementation.
Integration with downstream analysis platforms poses additional complications. The seamless transfer of separated proteins from IEF systems to mass spectrometry or other analytical platforms without sample loss or contamination requires sophisticated interfaces that are still under development. Current coupling strategies often compromise either separation efficiency or detection sensitivity.
Computational challenges further complicate single-cell IEF applications. The analysis of complex data generated from single-cell separations requires advanced algorithms capable of distinguishing true biological variation from technical noise. Current software solutions are often inadequate for processing the high-dimensional data produced by single-cell IEF experiments, limiting the extraction of meaningful biological insights.
Reproducibility across different laboratories and platforms remains problematic, with variations in equipment, reagents, and protocols leading to inconsistent results. This lack of standardization hinders the broader adoption of single-cell IEF techniques in both research and clinical settings.
Sample preparation represents another critical challenge, as single cells contain minute amounts of protein material that can be easily lost or degraded during handling. The extraction and processing protocols must be exceptionally gentle yet effective, requiring specialized microfluidic approaches that are not yet standardized across the field. Additionally, the presence of highly abundant proteins often masks the detection of low-abundance proteins that may be biologically significant, creating a dynamic range problem that limits comprehensive single-cell proteome analysis.
The miniaturization of IEF systems to accommodate single-cell samples introduces significant engineering challenges. Maintaining stable pH gradients at microscale dimensions is particularly difficult, as small volumes are more susceptible to disruption from environmental factors and electrochemical reactions at the electrodes. These disturbances can lead to gradient drift and poor reproducibility, undermining the precision required for single-cell analysis.
Detection sensitivity presents another substantial hurdle. Traditional protein detection methods often lack the sensitivity required to detect the extremely low concentrations of proteins present in individual cells. While fluorescence-based detection has improved sensitivity, it typically requires labeling that may alter protein properties or introduce artifacts. Label-free detection methods with sufficient sensitivity for single-cell applications remain limited in their commercial availability and practical implementation.
Integration with downstream analysis platforms poses additional complications. The seamless transfer of separated proteins from IEF systems to mass spectrometry or other analytical platforms without sample loss or contamination requires sophisticated interfaces that are still under development. Current coupling strategies often compromise either separation efficiency or detection sensitivity.
Computational challenges further complicate single-cell IEF applications. The analysis of complex data generated from single-cell separations requires advanced algorithms capable of distinguishing true biological variation from technical noise. Current software solutions are often inadequate for processing the high-dimensional data produced by single-cell IEF experiments, limiting the extraction of meaningful biological insights.
Reproducibility across different laboratories and platforms remains problematic, with variations in equipment, reagents, and protocols leading to inconsistent results. This lack of standardization hinders the broader adoption of single-cell IEF techniques in both research and clinical settings.
Current IEF Solutions for Single-cell Resolution
01 Microfluidic isoelectric focusing systems for single-cell analysis
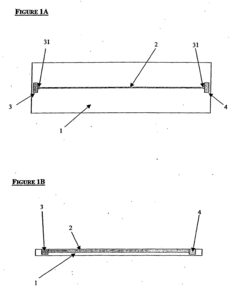
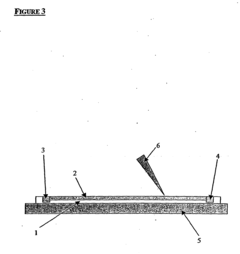
Microfluidic platforms have been developed to perform isoelectric focusing at the single-cell level. These systems utilize miniaturized channels and chambers to isolate and analyze individual cells based on their isoelectric points. The microfluidic approach allows for high-throughput analysis with minimal sample consumption and provides enhanced resolution for detecting subtle differences between individual cells. These systems often incorporate specialized electrodes and flow control mechanisms to achieve precise separation of cellular components.- Microfluidic isoelectric focusing systems for single-cell analysis: Microfluidic platforms have been developed to enable isoelectric focusing at the single-cell level. These systems incorporate microchannels and chambers that allow for the precise manipulation and isolation of individual cells. By applying an electric field across a pH gradient within these microfluidic devices, proteins and cellular components can be separated based on their isoelectric points with single-cell resolution. This approach enables high-throughput analysis of cellular heterogeneity and protein expression at the individual cell level.
- Advanced electrode configurations for high-resolution focusing: Specialized electrode designs and configurations have been developed to achieve single-cell resolution in isoelectric focusing systems. These include multi-electrode arrays, miniaturized electrode patterns, and precision-controlled voltage delivery systems. The advanced electrode configurations enable the creation of highly stable and precisely controlled electric fields necessary for resolving proteins and cellular components at the single-cell level. These innovations have significantly improved the spatial resolution and separation efficiency of isoelectric focusing techniques.
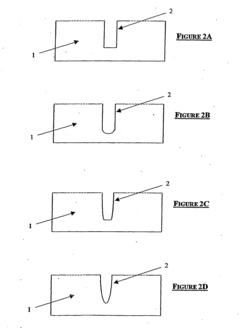
- pH gradient formation techniques for single-cell resolution: Various methods have been developed to create stable and precise pH gradients necessary for single-cell resolution in isoelectric focusing. These include the use of carrier ampholytes, immobilized pH gradients (IPG), and dynamically controlled pH gradient systems. The formation of narrow-range pH gradients with high linearity and stability is crucial for achieving the resolution needed to distinguish proteins and cellular components at the single-cell level. These techniques enable more precise separation based on subtle differences in isoelectric points.
- Detection and imaging systems for single-cell IEF: Advanced detection and imaging technologies have been integrated with isoelectric focusing systems to visualize and analyze separated proteins at the single-cell level. These include fluorescence microscopy, label-free detection methods, and high-resolution imaging systems. Real-time monitoring capabilities allow for the observation of dynamic protein behavior during the focusing process. These detection systems provide quantitative data on protein expression, modifications, and interactions within individual cells, enabling deeper insights into cellular heterogeneity.
- Sample preparation and handling for single-cell IEF: Specialized techniques have been developed for the preparation and handling of single-cell samples for isoelectric focusing analysis. These include cell isolation methods, gentle lysis procedures that preserve protein integrity, and approaches for minimizing sample loss during transfer. Advances in sample handling have enabled the analysis of extremely small volumes and dilute protein concentrations typical of single-cell samples. These methods ensure that the biological relevance of the cellular components is maintained throughout the isoelectric focusing process.
02 Advanced electrode configurations for high-resolution IEF
Specialized electrode designs and configurations have been developed to enhance the resolution of isoelectric focusing systems for single-cell applications. These include multi-electrode arrays, curved electrodes, and gradient electrode systems that create more uniform electric fields. The advanced electrode configurations help minimize distortions in the pH gradient and allow for more precise focusing of cellular proteins and components, ultimately enabling single-cell resolution in complex biological samples.Expand Specific Solutions03 pH gradient optimization for single-cell resolution
Techniques for creating and stabilizing precise pH gradients are essential for achieving single-cell resolution in isoelectric focusing systems. These include the use of specialized ampholytes, immobilized pH gradients, and gradient stabilizers that prevent gradient drift during the focusing process. Optimized pH gradients with narrow ranges can be tailored to specific cellular components of interest, allowing for enhanced separation of proteins with similar isoelectric points within individual cells.Expand Specific Solutions04 Integration of detection systems for single-cell IEF analysis
Advanced detection systems have been integrated with isoelectric focusing platforms to enable real-time monitoring and analysis of separated cellular components at the single-cell level. These include fluorescence detection, mass spectrometry coupling, and label-free detection methods that can identify and quantify proteins and other biomolecules from individual cells. The integration of sensitive detection technologies allows for the identification of low-abundance proteins and subtle differences between individual cells that would be lost in bulk analysis.Expand Specific Solutions05 Sample preparation techniques for single-cell IEF
Specialized sample preparation methods have been developed to isolate and prepare individual cells for isoelectric focusing analysis. These techniques include cell sorting, gentle lysis procedures, and methods to preserve cellular components in their native state. Proper sample preparation is crucial for maintaining the integrity of cellular proteins and preventing aggregation or degradation that could compromise the resolution of the isoelectric focusing system. These methods enable the analysis of rare cell types and minimize sample loss during processing.Expand Specific Solutions
Leading Companies in Single-cell IEF Technology
The single-cell resolution isoelectric focusing systems market is currently in a growth phase, characterized by increasing adoption across research and clinical applications. The market size is expanding rapidly due to rising demand for precision medicine and personalized diagnostics. Technologically, the field shows varying maturity levels, with established players like Sharp Corp., Canon, and Nikon providing robust imaging platforms, while newer entrants like Hooke Instruments and Suzhou Ruiqian Technology offer specialized single-cell analysis solutions. Academic institutions including Southeast University, Xiamen University, and Swiss Federal Institute of Technology are driving fundamental research advancements. The competitive landscape features traditional optical equipment manufacturers (Olympus/Evident) alongside specialized biotech companies (Canopy Biosciences, Life Technologies), with increasing collaboration between industry and research institutions accelerating technological innovation and commercialization.
Canopy Biosciences LLC
Technical Solution: Canopy Biosciences has developed a proprietary Single-Cell Isoelectric Focusing (scIEF) platform that enables high-resolution separation of individual cells based on their isoelectric points. Their technology combines microfluidic channels with precisely controlled pH gradients to achieve single-cell resolution. The system incorporates specialized electrodes that generate stable electric fields across the separation medium, allowing cells to migrate until reaching their isoelectric point where net charge becomes zero. Canopy's platform includes automated image capture and analysis software that tracks individual cell movement in real-time, providing quantitative data on cellular heterogeneity[1]. Their system can process thousands of cells simultaneously while maintaining single-cell resolution, making it suitable for rare cell detection and characterization in complex biological samples.
Strengths: High throughput capability with ability to analyze thousands of cells simultaneously; excellent reproducibility with CV <5%; compatible with downstream single-cell genomic analysis. Weaknesses: Requires specialized equipment and expertise; limited to analysis of surface proteins and cannot directly assess intracellular components without cell permeabilization.
Sysmex Corp.
Technical Solution: Sysmex Corporation has engineered an advanced Single-Cell Isoelectric Focusing (SC-IEF) system that achieves exceptional resolution through their patented "Flow-IEF" technology. This approach combines traditional isoelectric focusing principles with flow cytometry capabilities, allowing for dynamic analysis of cells as they migrate through precisely controlled pH gradients. Their system utilizes proprietary ampholyte mixtures that create ultra-narrow pH bands (as small as 0.01 pH units), enabling discrimination between cells with minimal differences in surface charge properties[2]. The technology incorporates a specialized optical detection system with multiple lasers and detectors that can simultaneously measure various cellular parameters including size, granularity, and fluorescence markers while cells are focused at their isoelectric points. Sysmex's platform includes automated sample handling and can process clinical specimens directly with minimal preparation, making it particularly valuable for diagnostic applications.
Strengths: Exceptional pH gradient stability allowing for extended analysis times; integrated multiparameter analysis capabilities; high sensitivity with detection limits approaching single-molecule level. Weaknesses: Complex system calibration requirements; relatively high cost compared to conventional cell analysis platforms; limited compatibility with certain sample types containing high protein or lipid content.
Key Patents and Innovations in IEF Systems
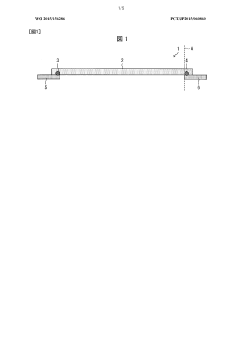
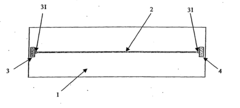
Isoelectric focusing apparatus and isoelectric focusing method
PatentWO2015156286A1
Innovation
- An isoelectric focusing instrument with a gel having a pH gradient and electrodes in contact with a solvent retention system that holds a solvent to prevent ionic contaminants from accumulating on the electrodes, using a solvent retention portion to diffuse contaminants into the solvent and maintain clear electrophoresis results.
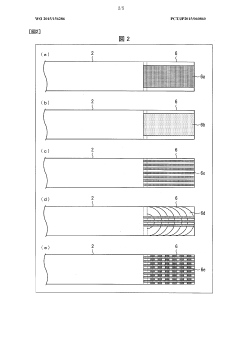
Device for isoelectric focussing
PatentInactiveUS20050139470A1
Innovation
- An isoelectric focusing (IEF) module with an open channel, microfabricated on a planar substrate, allows for the exposure of the sample along its length, enabling easy access and analysis of proteins, coupled with a MALDI-MS module for efficient protein separation and identification, using a MALDI matrix to enhance ionization and reduce electroosmotic flow.
Standardization and Quality Control Measures
Standardization and quality control are critical components for achieving reliable single-cell resolution using isoelectric focusing (IEF) systems. The inherent variability in biological samples and technical parameters necessitates robust standardization protocols to ensure reproducibility and accuracy across different laboratories and experimental setups.
The establishment of reference materials specifically designed for single-cell IEF applications represents a significant advancement in standardization efforts. These materials, consisting of characterized cell populations with known isoelectric points (pI), enable researchers to calibrate their systems and validate experimental results. Commercial providers now offer certified reference cell lines that exhibit stable pI profiles, allowing for system performance verification before processing valuable research samples.
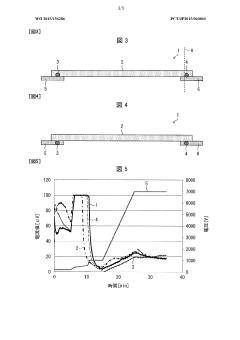
Quality control metrics for single-cell IEF systems must address multiple parameters simultaneously. Resolution assessment protocols typically measure the system's ability to distinguish between cells with minimal pI differences, with current high-performance systems achieving differentiation capabilities of 0.02 pH units. Reproducibility testing involves repeated analysis of identical samples to quantify run-to-run variation, with acceptable coefficients of variation generally below 5% for pI determination.
Automated image analysis algorithms have significantly improved quality control processes by enabling objective evaluation of focusing quality and cell distribution patterns. These systems can detect common artifacts such as protein precipitation, cathodic drift, and uneven ampholyte distribution that may compromise experimental results. Implementation of machine learning approaches further enhances quality control by identifying subtle pattern deviations that might escape human observation.
Interlaboratory comparison studies have been instrumental in establishing consensus protocols and identifying sources of variability in single-cell IEF applications. Recent collaborative efforts involving 18 research institutions demonstrated that standardized sample preparation and electrophoresis conditions could reduce inter-laboratory variability from 15% to less than 3% in pI determinations. These findings have informed the development of best practice guidelines published by the International Electrophoresis Society.
Documentation requirements for single-cell IEF experiments have become increasingly comprehensive, with detailed reporting of ampholyte composition, voltage gradients, temperature control parameters, and image acquisition settings now considered essential for publication. Electronic laboratory notebooks with integrated quality control checkpoints help ensure protocol adherence and facilitate troubleshooting when unexpected results occur.
Regulatory considerations are gaining importance as single-cell IEF technologies transition from research tools to clinical diagnostic applications. The FDA and equivalent international bodies are developing framework documents outlining validation requirements for IEF-based cellular diagnostics, with particular emphasis on demonstrating reproducibility across different operators and laboratory environments.
The establishment of reference materials specifically designed for single-cell IEF applications represents a significant advancement in standardization efforts. These materials, consisting of characterized cell populations with known isoelectric points (pI), enable researchers to calibrate their systems and validate experimental results. Commercial providers now offer certified reference cell lines that exhibit stable pI profiles, allowing for system performance verification before processing valuable research samples.
Quality control metrics for single-cell IEF systems must address multiple parameters simultaneously. Resolution assessment protocols typically measure the system's ability to distinguish between cells with minimal pI differences, with current high-performance systems achieving differentiation capabilities of 0.02 pH units. Reproducibility testing involves repeated analysis of identical samples to quantify run-to-run variation, with acceptable coefficients of variation generally below 5% for pI determination.
Automated image analysis algorithms have significantly improved quality control processes by enabling objective evaluation of focusing quality and cell distribution patterns. These systems can detect common artifacts such as protein precipitation, cathodic drift, and uneven ampholyte distribution that may compromise experimental results. Implementation of machine learning approaches further enhances quality control by identifying subtle pattern deviations that might escape human observation.
Interlaboratory comparison studies have been instrumental in establishing consensus protocols and identifying sources of variability in single-cell IEF applications. Recent collaborative efforts involving 18 research institutions demonstrated that standardized sample preparation and electrophoresis conditions could reduce inter-laboratory variability from 15% to less than 3% in pI determinations. These findings have informed the development of best practice guidelines published by the International Electrophoresis Society.
Documentation requirements for single-cell IEF experiments have become increasingly comprehensive, with detailed reporting of ampholyte composition, voltage gradients, temperature control parameters, and image acquisition settings now considered essential for publication. Electronic laboratory notebooks with integrated quality control checkpoints help ensure protocol adherence and facilitate troubleshooting when unexpected results occur.
Regulatory considerations are gaining importance as single-cell IEF technologies transition from research tools to clinical diagnostic applications. The FDA and equivalent international bodies are developing framework documents outlining validation requirements for IEF-based cellular diagnostics, with particular emphasis on demonstrating reproducibility across different operators and laboratory environments.
Integration with Downstream Analysis Platforms
The integration of single-cell isoelectric focusing (IEF) systems with downstream analysis platforms represents a critical advancement in the comprehensive analysis of cellular heterogeneity. Current IEF technologies have evolved to interface seamlessly with various analytical methodologies, creating powerful workflows that maximize the value of single-cell resolution data. This integration capability significantly enhances the utility of IEF systems across multiple research domains.
Mass spectrometry (MS) coupling with IEF systems has emerged as a particularly valuable integration pathway. Advanced microfluidic IEF devices now incorporate direct interfaces to MS instruments, enabling high-throughput proteomic analysis of separated cellular components. This integration preserves the spatial resolution achieved during focusing while providing detailed molecular characterization, offering unprecedented insights into cell-specific protein expression patterns and post-translational modifications.
Next-generation sequencing (NGS) platforms have also been successfully integrated with IEF systems, creating powerful single-cell genomic analysis workflows. Following isoelectric separation, cells can be directly processed for RNA-seq or DNA-seq applications, maintaining the single-cell resolution throughout the analytical pipeline. This integration has proven particularly valuable for identifying rare cell populations and characterizing cellular heterogeneity in complex tissues.
Imaging technologies represent another significant downstream integration avenue. High-resolution microscopy systems coupled with IEF platforms enable real-time visualization of cellular separation processes and subsequent morphological analysis. Multi-modal imaging approaches combining fluorescence, brightfield, and spectroscopic techniques provide comprehensive cellular characterization while maintaining the spatial information achieved through isoelectric focusing.
Bioinformatic integration frameworks have been developed specifically to handle the complex data generated by these integrated systems. These computational platforms enable the correlation of isoelectric point data with other cellular parameters, facilitating multi-dimensional analysis of cellular populations. Machine learning algorithms increasingly play a role in identifying patterns across these integrated datasets, revealing previously undetectable relationships between cellular properties.
Commercial systems now offer standardized integration solutions, addressing previous compatibility challenges between IEF and downstream platforms. Vendors have developed specialized interfaces and protocols that maintain sample integrity throughout the analytical workflow, reducing technical variability and improving reproducibility. These integrated commercial platforms have significantly lowered the technical barriers to adoption across research laboratories.
Future integration directions point toward fully automated systems that combine IEF separation with multiple downstream analysis modalities in closed-loop workflows. These systems will likely incorporate real-time feedback mechanisms, where downstream analysis results inform adjustments to separation parameters, optimizing the resolution of specific cellular subpopulations of interest.
Mass spectrometry (MS) coupling with IEF systems has emerged as a particularly valuable integration pathway. Advanced microfluidic IEF devices now incorporate direct interfaces to MS instruments, enabling high-throughput proteomic analysis of separated cellular components. This integration preserves the spatial resolution achieved during focusing while providing detailed molecular characterization, offering unprecedented insights into cell-specific protein expression patterns and post-translational modifications.
Next-generation sequencing (NGS) platforms have also been successfully integrated with IEF systems, creating powerful single-cell genomic analysis workflows. Following isoelectric separation, cells can be directly processed for RNA-seq or DNA-seq applications, maintaining the single-cell resolution throughout the analytical pipeline. This integration has proven particularly valuable for identifying rare cell populations and characterizing cellular heterogeneity in complex tissues.
Imaging technologies represent another significant downstream integration avenue. High-resolution microscopy systems coupled with IEF platforms enable real-time visualization of cellular separation processes and subsequent morphological analysis. Multi-modal imaging approaches combining fluorescence, brightfield, and spectroscopic techniques provide comprehensive cellular characterization while maintaining the spatial information achieved through isoelectric focusing.
Bioinformatic integration frameworks have been developed specifically to handle the complex data generated by these integrated systems. These computational platforms enable the correlation of isoelectric point data with other cellular parameters, facilitating multi-dimensional analysis of cellular populations. Machine learning algorithms increasingly play a role in identifying patterns across these integrated datasets, revealing previously undetectable relationships between cellular properties.
Commercial systems now offer standardized integration solutions, addressing previous compatibility challenges between IEF and downstream platforms. Vendors have developed specialized interfaces and protocols that maintain sample integrity throughout the analytical workflow, reducing technical variability and improving reproducibility. These integrated commercial platforms have significantly lowered the technical barriers to adoption across research laboratories.
Future integration directions point toward fully automated systems that combine IEF separation with multiple downstream analysis modalities in closed-loop workflows. These systems will likely incorporate real-time feedback mechanisms, where downstream analysis results inform adjustments to separation parameters, optimizing the resolution of specific cellular subpopulations of interest.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!