Evaluating Technical Challenges in Isoelectric Focusing Applications
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
IEF Technology Evolution and Objectives
Isoelectric focusing (IEF) emerged in the 1960s as a revolutionary technique for protein separation based on their isoelectric points. This analytical method has evolved significantly over the decades, transitioning from conventional gel-based systems to more sophisticated capillary and microchip platforms. The fundamental principle—separating amphoteric molecules in a pH gradient until they reach a position where their net charge is zero—has remained unchanged, but the implementation technologies have undergone substantial refinement.
The evolution of IEF technology can be traced through several key developmental phases. Initially, carrier ampholytes were used to establish pH gradients in polyacrylamide gels. The 1980s saw the introduction of immobilized pH gradients (IPGs), which dramatically improved reproducibility and resolution. By the 1990s, capillary isoelectric focusing (cIEF) emerged, offering advantages in automation, speed, and sample conservation. The early 2000s witnessed the integration of IEF with mass spectrometry, creating powerful hybrid analytical platforms for proteomics research.
Recent technological advancements have focused on miniaturization and integration. Microfluidic IEF systems have been developed, allowing for analysis of extremely small sample volumes with enhanced efficiency. Additionally, digital microfluidics has enabled more precise control over the separation process, while also facilitating integration with downstream analytical techniques.
The primary objectives of current IEF technology development are multifaceted. First, there is a push toward higher resolution separation to distinguish proteins with minimal differences in isoelectric points, particularly important for identifying post-translational modifications. Second, researchers aim to improve detection sensitivity to identify low-abundance proteins in complex biological samples. Third, there is significant interest in developing more robust and reproducible methods suitable for clinical diagnostics and quality control applications.
Another critical objective is the reduction of analysis time without compromising separation quality. Traditional IEF methods often require hours to achieve optimal separation, which limits throughput in research and clinical settings. Innovations in electric field application, buffer systems, and surface chemistry are being explored to accelerate the focusing process.
Automation and integration represent additional key goals in IEF technology development. The ideal systems would seamlessly combine sample preparation, separation, detection, and data analysis in a single platform, minimizing manual intervention and potential sources of error. This integration would make IEF more accessible to non-specialist laboratories and facilitate its adoption in routine analytical workflows.
The evolution of IEF technology can be traced through several key developmental phases. Initially, carrier ampholytes were used to establish pH gradients in polyacrylamide gels. The 1980s saw the introduction of immobilized pH gradients (IPGs), which dramatically improved reproducibility and resolution. By the 1990s, capillary isoelectric focusing (cIEF) emerged, offering advantages in automation, speed, and sample conservation. The early 2000s witnessed the integration of IEF with mass spectrometry, creating powerful hybrid analytical platforms for proteomics research.
Recent technological advancements have focused on miniaturization and integration. Microfluidic IEF systems have been developed, allowing for analysis of extremely small sample volumes with enhanced efficiency. Additionally, digital microfluidics has enabled more precise control over the separation process, while also facilitating integration with downstream analytical techniques.
The primary objectives of current IEF technology development are multifaceted. First, there is a push toward higher resolution separation to distinguish proteins with minimal differences in isoelectric points, particularly important for identifying post-translational modifications. Second, researchers aim to improve detection sensitivity to identify low-abundance proteins in complex biological samples. Third, there is significant interest in developing more robust and reproducible methods suitable for clinical diagnostics and quality control applications.
Another critical objective is the reduction of analysis time without compromising separation quality. Traditional IEF methods often require hours to achieve optimal separation, which limits throughput in research and clinical settings. Innovations in electric field application, buffer systems, and surface chemistry are being explored to accelerate the focusing process.
Automation and integration represent additional key goals in IEF technology development. The ideal systems would seamlessly combine sample preparation, separation, detection, and data analysis in a single platform, minimizing manual intervention and potential sources of error. This integration would make IEF more accessible to non-specialist laboratories and facilitate its adoption in routine analytical workflows.
Market Analysis for IEF Applications
The global market for Isoelectric Focusing (IEF) applications has shown consistent growth over the past decade, primarily driven by expanding applications in proteomics research, pharmaceutical development, and clinical diagnostics. Current market valuations place the IEF technology sector at approximately 1.2 billion USD, with projected annual growth rates of 5-7% through 2028, according to recent industry analyses.
Biopharmaceutical research and development represents the largest market segment, accounting for nearly 40% of total IEF applications. This dominance stems from the critical role IEF plays in protein characterization, quality control, and formulation development processes. Academic research institutions constitute the second-largest market segment at 30%, followed by clinical diagnostic laboratories at 20%.
Geographically, North America leads the market with approximately 45% share, benefiting from substantial research funding and a concentrated presence of biopharmaceutical companies. Europe follows at 30%, with particularly strong adoption in Germany, Switzerland, and the UK. The Asia-Pacific region, while currently representing only 20% of the market, demonstrates the highest growth potential with annual expansion rates exceeding 9%, primarily driven by increasing research investments in China, Japan, and South Korea.
Market demand analysis reveals several key trends shaping the IEF landscape. First, there is growing demand for integrated systems that combine IEF with other separation techniques, particularly mass spectrometry, enabling more comprehensive protein analysis workflows. Second, miniaturization and automation of IEF systems are increasingly sought after, as laboratories seek higher throughput capabilities with reduced sample volumes.
Customer segmentation shows distinct requirements across different user groups. Research laboratories prioritize resolution and reproducibility, while clinical settings emphasize throughput, ease of use, and regulatory compliance. Industrial users, particularly in biopharmaceutical manufacturing, focus on scalability, validation capabilities, and integration with existing quality control systems.
Competitive analysis indicates a moderately fragmented market with approximately 15 significant players. Market leaders include Bio-Rad, Thermo Fisher Scientific, and Agilent Technologies, collectively holding about 65% market share. Several specialized companies focusing exclusively on electrophoresis technologies maintain competitive positions through technical innovation and specialized applications.
Future market growth will likely be driven by emerging applications in personalized medicine, biomarker discovery, and biotherapeutic development. The increasing adoption of multi-omics approaches in life sciences research presents significant opportunities for advanced IEF technologies that can interface with complementary analytical platforms.
Biopharmaceutical research and development represents the largest market segment, accounting for nearly 40% of total IEF applications. This dominance stems from the critical role IEF plays in protein characterization, quality control, and formulation development processes. Academic research institutions constitute the second-largest market segment at 30%, followed by clinical diagnostic laboratories at 20%.
Geographically, North America leads the market with approximately 45% share, benefiting from substantial research funding and a concentrated presence of biopharmaceutical companies. Europe follows at 30%, with particularly strong adoption in Germany, Switzerland, and the UK. The Asia-Pacific region, while currently representing only 20% of the market, demonstrates the highest growth potential with annual expansion rates exceeding 9%, primarily driven by increasing research investments in China, Japan, and South Korea.
Market demand analysis reveals several key trends shaping the IEF landscape. First, there is growing demand for integrated systems that combine IEF with other separation techniques, particularly mass spectrometry, enabling more comprehensive protein analysis workflows. Second, miniaturization and automation of IEF systems are increasingly sought after, as laboratories seek higher throughput capabilities with reduced sample volumes.
Customer segmentation shows distinct requirements across different user groups. Research laboratories prioritize resolution and reproducibility, while clinical settings emphasize throughput, ease of use, and regulatory compliance. Industrial users, particularly in biopharmaceutical manufacturing, focus on scalability, validation capabilities, and integration with existing quality control systems.
Competitive analysis indicates a moderately fragmented market with approximately 15 significant players. Market leaders include Bio-Rad, Thermo Fisher Scientific, and Agilent Technologies, collectively holding about 65% market share. Several specialized companies focusing exclusively on electrophoresis technologies maintain competitive positions through technical innovation and specialized applications.
Future market growth will likely be driven by emerging applications in personalized medicine, biomarker discovery, and biotherapeutic development. The increasing adoption of multi-omics approaches in life sciences research presents significant opportunities for advanced IEF technologies that can interface with complementary analytical platforms.
Current IEF Limitations and Technical Barriers
Despite significant advancements in isoelectric focusing (IEF) technology, several persistent technical barriers continue to limit its broader application and effectiveness. One of the most challenging limitations is the phenomenon of protein precipitation at their isoelectric points (pI), particularly for proteins with extreme pI values. This precipitation occurs because proteins exhibit minimum solubility at their pI, leading to aggregation and loss of sample during analysis, which significantly compromises the reliability of results.
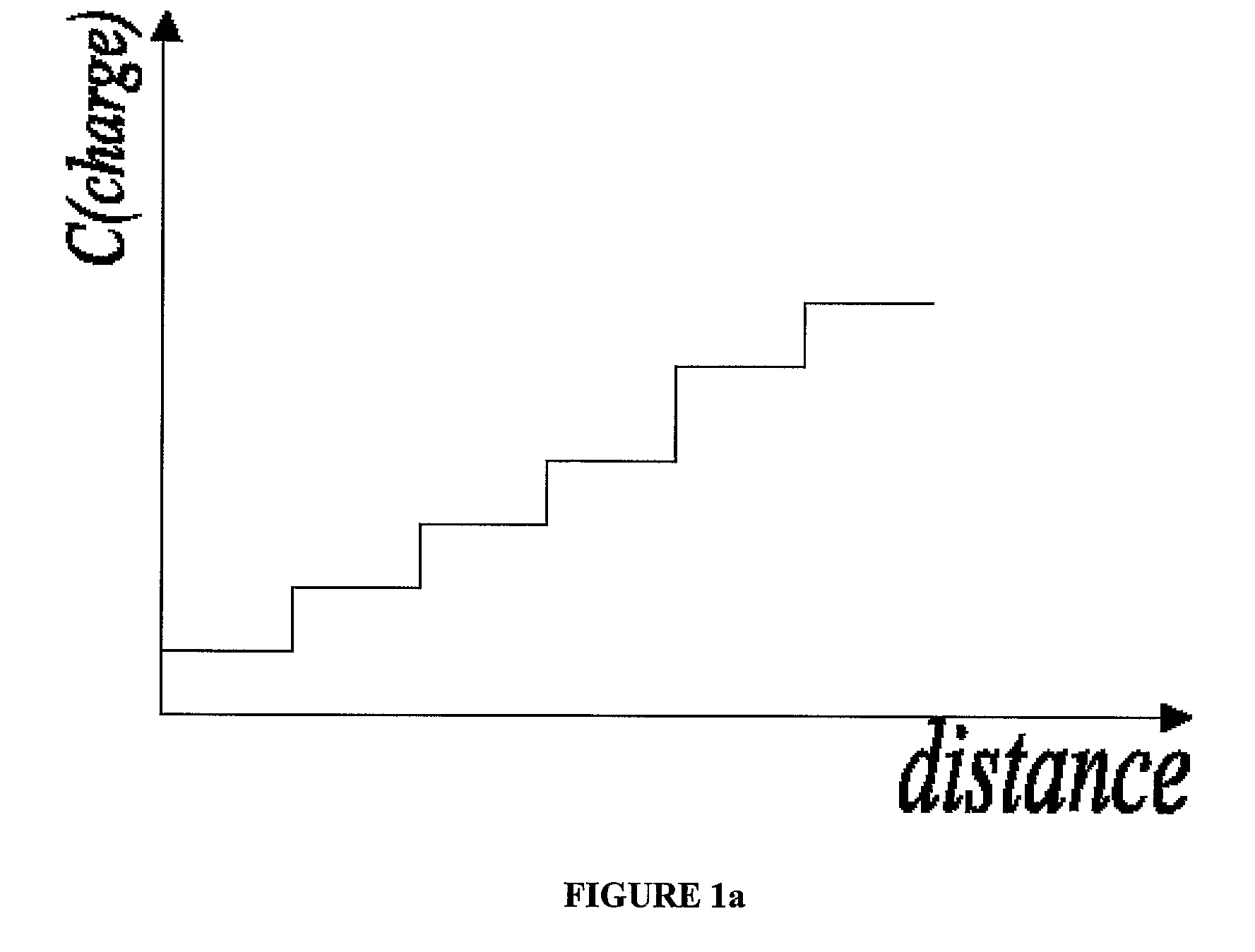
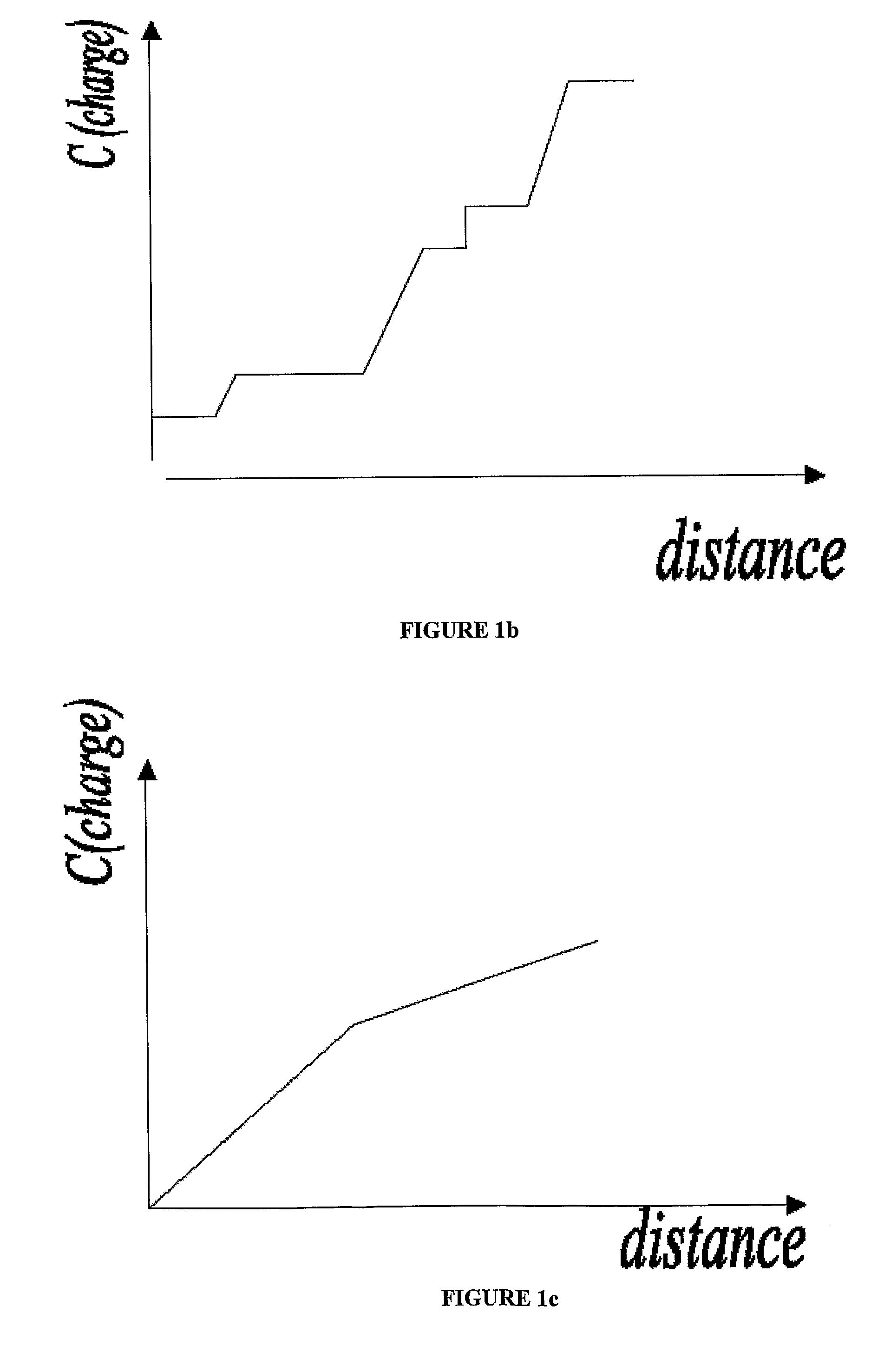
Carrier ampholyte-based IEF systems face inherent limitations in resolution capability, typically achieving pH gradient stability only between pH 3-10, with resolution declining dramatically at the extremes of this range. This restricts the analysis of proteins with very acidic or basic properties, creating blind spots in proteomic studies. Additionally, the batch-to-batch variability of carrier ampholytes introduces reproducibility challenges that undermine the consistency required for standardized analytical procedures.
The establishment and maintenance of stable pH gradients represent another significant technical hurdle. Gradient drift, particularly in extended separations, leads to unpredictable protein migration and compromised resolution. This instability is exacerbated by electroendosmosis effects and the cathodic drift phenomenon, where the pH gradient gradually shifts toward the cathode during the separation process, distorting the final protein distribution pattern.
Sample loading capacity constraints severely limit the applicability of IEF for preparative purposes. Current systems can typically handle only microgram quantities of protein, whereas many downstream applications require substantially larger amounts. This scaling limitation has prevented IEF from becoming a mainstream preparative technique despite its exceptional resolving power.
Detection sensitivity issues also plague IEF applications, particularly when dealing with low-abundance proteins. The conventional staining methods used post-separation often lack the sensitivity required to visualize proteins present in trace amounts, creating a significant blind spot in comprehensive proteomic analyses. This limitation is particularly problematic in biomarker discovery and clinical diagnostics, where proteins of interest may be present at extremely low concentrations.
The integration of IEF with downstream analytical techniques presents additional technical challenges. While coupling with mass spectrometry has shown promise, issues related to ampholyte interference, salt contamination, and recovery efficiency continue to hinder seamless workflow integration. These interface problems often necessitate additional sample processing steps that can lead to sample loss and increased analytical variability.
Carrier ampholyte-based IEF systems face inherent limitations in resolution capability, typically achieving pH gradient stability only between pH 3-10, with resolution declining dramatically at the extremes of this range. This restricts the analysis of proteins with very acidic or basic properties, creating blind spots in proteomic studies. Additionally, the batch-to-batch variability of carrier ampholytes introduces reproducibility challenges that undermine the consistency required for standardized analytical procedures.
The establishment and maintenance of stable pH gradients represent another significant technical hurdle. Gradient drift, particularly in extended separations, leads to unpredictable protein migration and compromised resolution. This instability is exacerbated by electroendosmosis effects and the cathodic drift phenomenon, where the pH gradient gradually shifts toward the cathode during the separation process, distorting the final protein distribution pattern.
Sample loading capacity constraints severely limit the applicability of IEF for preparative purposes. Current systems can typically handle only microgram quantities of protein, whereas many downstream applications require substantially larger amounts. This scaling limitation has prevented IEF from becoming a mainstream preparative technique despite its exceptional resolving power.
Detection sensitivity issues also plague IEF applications, particularly when dealing with low-abundance proteins. The conventional staining methods used post-separation often lack the sensitivity required to visualize proteins present in trace amounts, creating a significant blind spot in comprehensive proteomic analyses. This limitation is particularly problematic in biomarker discovery and clinical diagnostics, where proteins of interest may be present at extremely low concentrations.
The integration of IEF with downstream analytical techniques presents additional technical challenges. While coupling with mass spectrometry has shown promise, issues related to ampholyte interference, salt contamination, and recovery efficiency continue to hinder seamless workflow integration. These interface problems often necessitate additional sample processing steps that can lead to sample loss and increased analytical variability.
Contemporary IEF Implementation Approaches
01 pH gradient stability and control
One of the major technical challenges in isoelectric focusing is maintaining a stable pH gradient throughout the separation process. The pH gradient can drift or collapse over time due to various factors such as electrolysis, temperature fluctuations, and carrier ampholyte mobility. Innovations in this area include improved buffer systems, specialized ampholytes, and temperature control mechanisms that help maintain gradient stability for more reproducible separations.- pH gradient stability and control: One of the major technical challenges in isoelectric focusing is maintaining a stable pH gradient throughout the separation process. The pH gradient can drift over time due to various factors such as electrolysis, temperature fluctuations, and carrier ampholyte migration. This instability can lead to poor reproducibility and resolution of protein separation. Advanced techniques for stabilizing pH gradients include the use of immobilized pH gradients (IPGs), specialized buffer systems, and temperature control mechanisms to ensure consistent and reliable separation of proteins based on their isoelectric points.
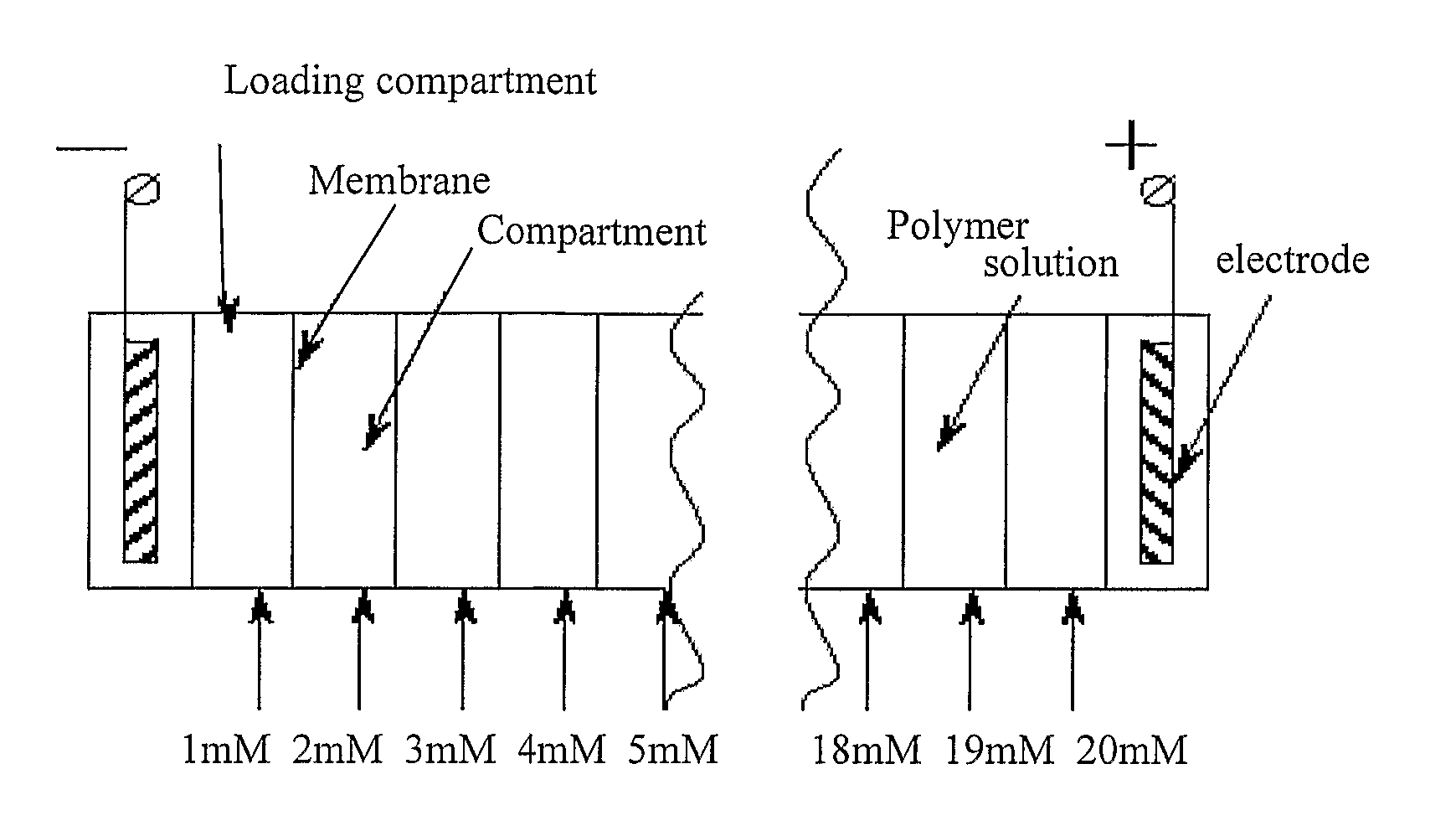
- Sample loading and protein solubility issues: Proper sample preparation and loading are critical challenges in isoelectric focusing. Proteins may precipitate at their isoelectric points due to minimal electrostatic repulsion, leading to poor resolution and recovery. Additionally, hydrophobic proteins and membrane proteins are particularly difficult to analyze using conventional isoelectric focusing techniques. Innovations in sample preparation methods include the development of specialized detergents, chaotropic agents, and loading techniques to improve protein solubility and prevent precipitation during the focusing process.
- Equipment and apparatus design challenges: The design of isoelectric focusing equipment presents several technical challenges. These include issues with heat dissipation, electrode design, and the need for specialized materials that can withstand the electrical fields applied during separation. Modern isoelectric focusing systems incorporate advanced cooling systems, precise power supplies, and innovative electrode configurations to overcome these challenges. Additionally, miniaturization and integration with other analytical techniques require novel engineering approaches to maintain separation efficiency while reducing sample and reagent consumption.
- Detection and quantification methods: Detecting and quantifying separated proteins after isoelectric focusing presents significant technical challenges. Traditional staining methods may have limited sensitivity or dynamic range, while more advanced detection techniques require specialized equipment and expertise. Innovations in this area include the development of fluorescent dyes with improved sensitivity, label-free detection methods, and integration with mass spectrometry for protein identification. Digital imaging and analysis software have also been developed to improve the accuracy of protein quantification following isoelectric focusing separation.
- Data analysis and automation challenges: Processing and interpreting the complex data generated by isoelectric focusing experiments presents significant challenges. This includes issues with reproducibility, standardization of results, and integration with other proteomics data. Advanced software solutions have been developed to automate data analysis, improve pattern recognition, and facilitate comparison between experiments. Additionally, machine learning approaches are being applied to enhance the interpretation of isoelectric focusing results and identify subtle patterns that may be missed by conventional analysis methods.
02 Sample loading and resolution optimization
Achieving optimal resolution in isoelectric focusing requires careful consideration of sample loading techniques. Challenges include preventing protein precipitation at their isoelectric points, managing sample overloading that can distort the pH gradient, and optimizing focusing time to prevent diffusion after separation. Advanced sample application methods and gradient optimization techniques have been developed to improve resolution and reproducibility of protein separation.Expand Specific Solutions03 Miniaturization and microfluidic integration
The miniaturization of isoelectric focusing systems presents unique technical challenges related to scaling effects, surface interactions, and heat dissipation. Microfluidic isoelectric focusing requires specialized channel designs, surface treatments to prevent protein adsorption, and precise control of electroosmotic flow. These systems aim to reduce sample volume requirements while maintaining or improving separation efficiency for proteomic applications.Expand Specific Solutions04 Detection and imaging technologies
Detecting and visualizing separated proteins after isoelectric focusing presents significant technical challenges. Traditional staining methods may interfere with downstream analysis, while label-free detection often lacks sensitivity. Advanced imaging technologies including fluorescence detection, mass spectrometry coupling, and digital imaging systems have been developed to improve detection limits and enable quantitative analysis of separated proteins without compromising sample integrity.Expand Specific Solutions05 Automation and data processing
Automation of isoelectric focusing procedures faces challenges in reproducibility, standardization, and data analysis. Automated systems must precisely control electric fields, temperature, and timing while handling multiple samples. Sophisticated software solutions are needed for pattern recognition, peak identification, and comparative analysis across multiple gels or capillaries. These systems aim to reduce operator variability and increase throughput for clinical and research applications.Expand Specific Solutions
Leading Companies and Research Institutions in IEF
The isoelectric focusing (IEF) technology market is currently in a growth phase, with increasing applications in proteomics research and biopharmaceutical development. The global market size is estimated to reach approximately $1.2 billion by 2025, driven by advancements in protein separation techniques. Technology maturity varies across applications, with established players like Bio-Rad Laboratories and Life Technologies leading with comprehensive IEF solutions. Emerging competitors include ProteinSimple and PARC, who are developing next-generation microfluidic IEF platforms. Academic institutions such as MIT, Texas A&M, and EPFL are advancing fundamental research, while companies like Hamamatsu Photonics and Sharp Corp are contributing imaging technologies critical for detection systems. The competitive landscape is characterized by increasing integration of IEF with other analytical techniques to enhance resolution and throughput.
Life Technologies Corp.
Technical Solution: Life Technologies has developed innovative solutions for isoelectric focusing challenges through their integrated proteomics platforms. Their technology incorporates specialized ampholyte mixtures that create highly linear and stable pH gradients, crucial for reproducible protein separation. The company has engineered novel gel matrices with optimized pore structures that minimize electroendosmosis while enhancing protein mobility during focusing. Their IEF systems feature advanced electrode designs that ensure uniform electric field distribution across the separation medium, reducing edge effects that commonly distort protein migration patterns. Life Technologies has implemented sophisticated temperature regulation systems that maintain thermal stability throughout the focusing process, preventing protein denaturation and improving reproducibility. Their platforms also incorporate proprietary sample preparation protocols that effectively remove contaminants known to interfere with IEF performance, such as salts and detergents, without significant protein loss.
Strengths: Comprehensive workflow integration from sample preparation through analysis, excellent compatibility with downstream mass spectrometry applications, and robust performance with diverse sample types. Weaknesses: Complex operational protocols requiring technical expertise, higher consumable costs compared to traditional electrophoresis, and limited throughput for high-volume clinical applications.
Bio-Rad Laboratories, Inc.
Technical Solution: Bio-Rad has developed advanced isoelectric focusing (IEF) systems that utilize immobilized pH gradient (IPG) strips for high-resolution protein separation. Their technology incorporates proprietary buffer systems and gel formulations that enhance protein solubility and minimize protein aggregation during the focusing process. Bio-Rad's IEF platforms feature precise temperature control mechanisms that maintain consistent conditions throughout the separation process, reducing the thermal gradients that can distort protein migration patterns. Their systems also implement advanced power supply algorithms that automatically adjust voltage and current parameters during focusing to prevent sample burning while maximizing resolution. Bio-Rad has integrated digital imaging capabilities that allow for real-time monitoring of the focusing process and immediate analysis of separated proteins without additional staining steps.
Strengths: Superior resolution for complex protein mixtures, excellent reproducibility across runs, and comprehensive software integration for automated analysis. Weaknesses: Higher cost compared to traditional electrophoresis systems, requires specialized training for optimal results, and limited throughput for high-volume applications.
Critical Patents and Innovations in IEF Technology
An apparatus for protein separation using capillary isoelectric focusing-hollow fiber flow field flow fractionation and method thereof
PatentInactiveEP1987053A1
Innovation
- A capillary isoelectric focusing-hollow fiber flow field flow fractionation apparatus that separates proteins based on isoelectric point and molecular weight in a two-dimensional, non-gel, and liquid phase manner, using a combination of capillary isoelectric focusing and hollow fiber flow field flow fractionation units to automatically remove ampholytes and prevent protein denaturation.
Electrophoretic Separation of Analytes by Molecular Mass
PatentInactiveUS20080314751A1
Innovation
- A novel method using a matrix modified with a charged separation agent, where analytes form a charged complex and are separated based on their total charge, allowing for separation by molecular weight in a low-friction medium with a charge density gradient, eliminating the need for gradient gels and improving mass determination accuracy.
Regulatory Compliance for IEF Applications
Isoelectric focusing (IEF) applications are subject to stringent regulatory frameworks that vary significantly across different regions and industries. In the United States, the FDA regulates IEF technologies used in clinical diagnostics through the Clinical Laboratory Improvement Amendments (CLIA) and medical device regulations. These frameworks establish quality standards for laboratory testing and validation protocols that must be rigorously followed to ensure accurate and reliable results.
The European Union implements the In Vitro Diagnostic Regulation (IVDR) and Medical Device Regulation (MDR), which have introduced more comprehensive requirements for performance evaluation, technical documentation, and post-market surveillance of IEF technologies. Manufacturers must demonstrate compliance with these regulations through extensive validation studies and risk management procedures before their products can enter the European market.
For pharmaceutical applications, IEF technologies must adhere to Good Manufacturing Practice (GMP) guidelines, which ensure consistent quality and purity of pharmaceutical products. These guidelines mandate detailed documentation of procedures, validation of analytical methods, and regular calibration of equipment used in IEF processes.
Environmental considerations also play a crucial role in regulatory compliance for IEF applications. The disposal of ampholytes, carrier ampholytes, and other chemicals used in IEF processes must comply with local waste management regulations. Many jurisdictions have implemented specific protocols for the handling and disposal of these potentially hazardous materials.
Data integrity and security requirements have become increasingly important in regulatory frameworks governing IEF applications. This is particularly relevant for automated IEF systems that generate and store digital data. Compliance with regulations such as 21 CFR Part 11 in the United States requires implementation of electronic records management systems with appropriate access controls, audit trails, and data backup procedures.
International harmonization efforts, such as those led by the International Conference on Harmonisation (ICH), aim to standardize regulatory requirements across different regions. However, significant variations still exist, creating challenges for global deployment of IEF technologies. Companies must navigate these complex regulatory landscapes by developing comprehensive compliance strategies tailored to each target market.
Emerging technologies in IEF applications, such as microfluidic platforms and integrated analytical systems, present novel regulatory challenges. Regulatory agencies are continuously updating their frameworks to address these innovations, requiring manufacturers to maintain vigilant monitoring of evolving compliance requirements.
The European Union implements the In Vitro Diagnostic Regulation (IVDR) and Medical Device Regulation (MDR), which have introduced more comprehensive requirements for performance evaluation, technical documentation, and post-market surveillance of IEF technologies. Manufacturers must demonstrate compliance with these regulations through extensive validation studies and risk management procedures before their products can enter the European market.
For pharmaceutical applications, IEF technologies must adhere to Good Manufacturing Practice (GMP) guidelines, which ensure consistent quality and purity of pharmaceutical products. These guidelines mandate detailed documentation of procedures, validation of analytical methods, and regular calibration of equipment used in IEF processes.
Environmental considerations also play a crucial role in regulatory compliance for IEF applications. The disposal of ampholytes, carrier ampholytes, and other chemicals used in IEF processes must comply with local waste management regulations. Many jurisdictions have implemented specific protocols for the handling and disposal of these potentially hazardous materials.
Data integrity and security requirements have become increasingly important in regulatory frameworks governing IEF applications. This is particularly relevant for automated IEF systems that generate and store digital data. Compliance with regulations such as 21 CFR Part 11 in the United States requires implementation of electronic records management systems with appropriate access controls, audit trails, and data backup procedures.
International harmonization efforts, such as those led by the International Conference on Harmonisation (ICH), aim to standardize regulatory requirements across different regions. However, significant variations still exist, creating challenges for global deployment of IEF technologies. Companies must navigate these complex regulatory landscapes by developing comprehensive compliance strategies tailored to each target market.
Emerging technologies in IEF applications, such as microfluidic platforms and integrated analytical systems, present novel regulatory challenges. Regulatory agencies are continuously updating their frameworks to address these innovations, requiring manufacturers to maintain vigilant monitoring of evolving compliance requirements.
Emerging Materials for IEF Performance Improvement
Recent advancements in materials science have opened new frontiers for improving isoelectric focusing (IEF) performance. Novel carrier ampholytes with enhanced buffering capacity and solubility are emerging as promising alternatives to traditional ampholytes. These next-generation materials feature more precise pH gradients and improved stability during extended separation runs, addressing the long-standing challenge of pH drift in conventional IEF applications.
Immobilized pH gradient (IPG) materials are undergoing significant innovation with the development of acrylamide derivatives that offer superior mechanical strength and chemical resistance. These materials maintain gradient integrity even under high voltage conditions, enabling higher resolution separations while minimizing the distortion effects commonly observed with traditional IPG strips.
Surface-modified capillaries represent another breakthrough area, with new coatings that substantially reduce electroosmotic flow and protein adsorption. Hydrophilic polymers and zwitterionic materials applied to capillary walls have demonstrated remarkable improvements in separation efficiency and reproducibility, particularly for hydrophobic proteins that typically present challenges in conventional IEF systems.
Nanomaterials are increasingly being incorporated into IEF platforms, with carbon nanotubes, graphene, and metal-organic frameworks showing particular promise. These materials enhance electrical conductivity while providing increased surface area for protein interaction, resulting in faster separation times and improved detection sensitivity. Preliminary studies indicate up to 40% reduction in analysis time when using graphene-enhanced separation media.
Smart hydrogels that respond to electrical fields represent a cutting-edge development for IEF applications. These materials can dynamically adjust their properties during separation, potentially enabling adaptive focusing that responds to sample complexity in real-time. Early prototypes have demonstrated the ability to maintain resolution across wider pH ranges than conventional materials.
Biodegradable and environmentally friendly materials are gaining attention as sustainable alternatives for IEF applications. Plant-derived polymers and biocompatible synthetic materials offer comparable performance to traditional petroleum-based products while reducing environmental impact and improving biocompatibility for downstream biological applications.
Hybrid materials combining inorganic components with organic polymers are showing exceptional promise for specialized IEF applications. These composites leverage the mechanical stability of inorganic substrates with the chemical versatility of organic components, creating platforms with tunable properties that can be optimized for specific protein classes or separation challenges.
Immobilized pH gradient (IPG) materials are undergoing significant innovation with the development of acrylamide derivatives that offer superior mechanical strength and chemical resistance. These materials maintain gradient integrity even under high voltage conditions, enabling higher resolution separations while minimizing the distortion effects commonly observed with traditional IPG strips.
Surface-modified capillaries represent another breakthrough area, with new coatings that substantially reduce electroosmotic flow and protein adsorption. Hydrophilic polymers and zwitterionic materials applied to capillary walls have demonstrated remarkable improvements in separation efficiency and reproducibility, particularly for hydrophobic proteins that typically present challenges in conventional IEF systems.
Nanomaterials are increasingly being incorporated into IEF platforms, with carbon nanotubes, graphene, and metal-organic frameworks showing particular promise. These materials enhance electrical conductivity while providing increased surface area for protein interaction, resulting in faster separation times and improved detection sensitivity. Preliminary studies indicate up to 40% reduction in analysis time when using graphene-enhanced separation media.
Smart hydrogels that respond to electrical fields represent a cutting-edge development for IEF applications. These materials can dynamically adjust their properties during separation, potentially enabling adaptive focusing that responds to sample complexity in real-time. Early prototypes have demonstrated the ability to maintain resolution across wider pH ranges than conventional materials.
Biodegradable and environmentally friendly materials are gaining attention as sustainable alternatives for IEF applications. Plant-derived polymers and biocompatible synthetic materials offer comparable performance to traditional petroleum-based products while reducing environmental impact and improving biocompatibility for downstream biological applications.
Hybrid materials combining inorganic components with organic polymers are showing exceptional promise for specialized IEF applications. These composites leverage the mechanical stability of inorganic substrates with the chemical versatility of organic components, creating platforms with tunable properties that can be optimized for specific protein classes or separation challenges.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!