Optimization Modalities for Isoelectric Focusing Protocol Consistency
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
IEF Technology Background and Objectives
Isoelectric focusing (IEF) emerged in the 1960s as a groundbreaking electrophoretic technique for separating amphoteric molecules, particularly proteins, based on their isoelectric points (pI). The technique was pioneered by Harry Svensson and subsequently refined by researchers like Olof Vesterberg and Righetti, who established its theoretical foundations and practical applications. Over decades, IEF has evolved from conventional gel-based systems to more sophisticated capillary and microchip formats, significantly enhancing resolution and automation capabilities.
The technology's evolution has been marked by several key milestones, including the development of immobilized pH gradients (IPGs) in the 1980s, which addressed reproducibility issues inherent in carrier ampholyte-generated gradients. The integration of IEF with mass spectrometry and the advent of multi-dimensional protein separation techniques like 2D-PAGE have further expanded its analytical power and application scope.
Current technological trends in IEF focus on miniaturization, increased throughput, and enhanced reproducibility. The shift toward microfluidic platforms and lab-on-a-chip devices represents a significant advancement, allowing for reduced sample volumes, faster analysis times, and potential for point-of-care applications. Additionally, computational modeling of electric field distributions and protein behavior within pH gradients has enabled more precise protocol optimization.
Despite these advancements, IEF faces persistent challenges in achieving consistent results across laboratories and platforms. Protocol variability, environmental fluctuations, and differences in equipment specifications contribute to inconsistencies that hamper standardization efforts. These challenges are particularly pronounced in clinical and industrial settings where reproducibility is paramount.
The primary objective of optimizing IEF protocol consistency is to establish robust, transferable methodologies that yield reliable and comparable results regardless of operator, equipment, or laboratory environment. This entails developing standardized procedures, reference materials, and quality control metrics that can be universally applied.
Secondary objectives include enhancing the technique's sensitivity for detecting low-abundance proteins, improving resolution for closely spaced isoelectric points, and reducing analysis time without compromising separation quality. Additionally, there is growing interest in developing environmentally sustainable IEF protocols that minimize the use of toxic reagents and reduce waste generation.
The ultimate goal is to transform IEF from a highly specialized technique requiring considerable expertise into a more accessible, user-friendly analytical tool that can be reliably implemented across diverse research, clinical, and industrial applications. This transformation would significantly expand the technique's utility in proteomics, diagnostics, and quality control processes.
The technology's evolution has been marked by several key milestones, including the development of immobilized pH gradients (IPGs) in the 1980s, which addressed reproducibility issues inherent in carrier ampholyte-generated gradients. The integration of IEF with mass spectrometry and the advent of multi-dimensional protein separation techniques like 2D-PAGE have further expanded its analytical power and application scope.
Current technological trends in IEF focus on miniaturization, increased throughput, and enhanced reproducibility. The shift toward microfluidic platforms and lab-on-a-chip devices represents a significant advancement, allowing for reduced sample volumes, faster analysis times, and potential for point-of-care applications. Additionally, computational modeling of electric field distributions and protein behavior within pH gradients has enabled more precise protocol optimization.
Despite these advancements, IEF faces persistent challenges in achieving consistent results across laboratories and platforms. Protocol variability, environmental fluctuations, and differences in equipment specifications contribute to inconsistencies that hamper standardization efforts. These challenges are particularly pronounced in clinical and industrial settings where reproducibility is paramount.
The primary objective of optimizing IEF protocol consistency is to establish robust, transferable methodologies that yield reliable and comparable results regardless of operator, equipment, or laboratory environment. This entails developing standardized procedures, reference materials, and quality control metrics that can be universally applied.
Secondary objectives include enhancing the technique's sensitivity for detecting low-abundance proteins, improving resolution for closely spaced isoelectric points, and reducing analysis time without compromising separation quality. Additionally, there is growing interest in developing environmentally sustainable IEF protocols that minimize the use of toxic reagents and reduce waste generation.
The ultimate goal is to transform IEF from a highly specialized technique requiring considerable expertise into a more accessible, user-friendly analytical tool that can be reliably implemented across diverse research, clinical, and industrial applications. This transformation would significantly expand the technique's utility in proteomics, diagnostics, and quality control processes.
Market Analysis for Consistent IEF Applications
The global market for isoelectric focusing (IEF) applications continues to expand significantly, driven by increasing demand for high-resolution protein separation techniques across multiple industries. The current market size for IEF equipment and consumables is estimated at $1.2 billion, with a compound annual growth rate of 6.8% projected through 2028. This growth trajectory reflects the critical importance of consistent and reliable IEF protocols in various research and industrial applications.
Pharmaceutical and biotechnology sectors represent the largest market segments, collectively accounting for approximately 65% of total IEF application demand. These industries rely heavily on consistent IEF protocols for protein characterization, quality control in biopharmaceutical production, and development of novel biologics. The increasing pipeline of protein-based therapeutics has created substantial demand for reliable IEF methodologies that can deliver reproducible results across different laboratory settings.
Academic and research institutions constitute the second-largest market segment at 22%, where IEF techniques are extensively utilized in proteomics research, biomarker discovery, and fundamental protein science. These institutions are particularly sensitive to protocol consistency issues, as reproducibility challenges can significantly impact research outcomes and publication potential.
Clinical diagnostics represents an emerging growth sector for IEF applications, currently at 8% of the market but expanding rapidly at 9.3% annually. The integration of IEF techniques into diagnostic workflows for protein-based disease markers has created new demand for standardized protocols that can deliver consistent results in clinical settings where regulatory compliance is paramount.
Geographically, North America leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (24%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth rate at 8.5% annually, driven by expanding biotechnology sectors and increasing research funding.
Customer surveys indicate that protocol inconsistency remains a significant pain point, with 78% of users reporting challenges in achieving reproducible results across different batches or laboratory settings. This has created a distinct market opportunity for optimized IEF solutions that can deliver consistent performance. Approximately 62% of respondents indicated willingness to pay premium prices for IEF systems and reagents that demonstrably improve protocol consistency.
The market analysis reveals growing demand for integrated IEF systems that combine hardware, software, and standardized reagents to minimize variability. This trend is particularly pronounced in regulated environments such as pharmaceutical manufacturing and clinical applications, where protocol consistency directly impacts compliance requirements and operational efficiency.
Pharmaceutical and biotechnology sectors represent the largest market segments, collectively accounting for approximately 65% of total IEF application demand. These industries rely heavily on consistent IEF protocols for protein characterization, quality control in biopharmaceutical production, and development of novel biologics. The increasing pipeline of protein-based therapeutics has created substantial demand for reliable IEF methodologies that can deliver reproducible results across different laboratory settings.
Academic and research institutions constitute the second-largest market segment at 22%, where IEF techniques are extensively utilized in proteomics research, biomarker discovery, and fundamental protein science. These institutions are particularly sensitive to protocol consistency issues, as reproducibility challenges can significantly impact research outcomes and publication potential.
Clinical diagnostics represents an emerging growth sector for IEF applications, currently at 8% of the market but expanding rapidly at 9.3% annually. The integration of IEF techniques into diagnostic workflows for protein-based disease markers has created new demand for standardized protocols that can deliver consistent results in clinical settings where regulatory compliance is paramount.
Geographically, North America leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (24%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth rate at 8.5% annually, driven by expanding biotechnology sectors and increasing research funding.
Customer surveys indicate that protocol inconsistency remains a significant pain point, with 78% of users reporting challenges in achieving reproducible results across different batches or laboratory settings. This has created a distinct market opportunity for optimized IEF solutions that can deliver consistent performance. Approximately 62% of respondents indicated willingness to pay premium prices for IEF systems and reagents that demonstrably improve protocol consistency.
The market analysis reveals growing demand for integrated IEF systems that combine hardware, software, and standardized reagents to minimize variability. This trend is particularly pronounced in regulated environments such as pharmaceutical manufacturing and clinical applications, where protocol consistency directly impacts compliance requirements and operational efficiency.
Current Challenges in IEF Protocol Standardization
Despite significant advancements in isoelectric focusing (IEF) technology, the field continues to face substantial challenges in protocol standardization. The inherent complexity of IEF procedures creates considerable variability in experimental outcomes, hampering reproducibility across different laboratories and research settings. This inconsistency represents a critical bottleneck in the broader adoption of IEF as a reliable analytical technique in both research and industrial applications.
One of the primary challenges stems from the sensitivity of IEF to environmental conditions. Temperature fluctuations, even within small ranges, can significantly alter protein mobility and focusing patterns. Current temperature control systems vary widely in precision and methodology, leading to inconsistent results when protocols are transferred between different laboratory environments.
pH gradient establishment and stability present another major hurdle. The formation of stable, reproducible pH gradients is fundamental to IEF success, yet current methodologies struggle with gradient drift and cathodic mobilization. Commercial ampholyte compositions often exhibit batch-to-batch variations, further complicating standardization efforts. The lack of universal calibration standards for pH gradients exacerbates this issue.
Sample preparation protocols represent a significant source of variability. Differences in sample solubilization methods, buffer compositions, and additive concentrations can dramatically alter protein behavior during focusing. The absence of standardized guidelines for sample preparation tailored to specific protein classes contributes to inconsistent results across different research groups.
Instrumentation diversity further compounds standardization challenges. The wide array of IEF platforms—ranging from conventional gel-based systems to capillary and microfluidic formats—each introduce unique variables that affect protocol transferability. Variations in electrode design, chamber geometry, and power supply characteristics create additional layers of complexity when attempting to standardize protocols.
Data analysis and interpretation methodologies lack uniformity across the field. Different image acquisition systems, analysis software, and quantification approaches lead to divergent interpretations of similar experimental results. The absence of standardized reporting formats makes it difficult to compare findings between studies or establish reliable reference points.
Regulatory considerations also impede standardization efforts, particularly in clinical and pharmaceutical applications. Different regulatory bodies maintain varying requirements for validation and quality control, necessitating protocol modifications that further complicate standardization across international boundaries.
The economic implications of these challenges are substantial. The cost and time associated with protocol optimization and validation discourage smaller laboratories from adopting IEF technology, limiting its broader implementation and slowing technological advancement in the field.
One of the primary challenges stems from the sensitivity of IEF to environmental conditions. Temperature fluctuations, even within small ranges, can significantly alter protein mobility and focusing patterns. Current temperature control systems vary widely in precision and methodology, leading to inconsistent results when protocols are transferred between different laboratory environments.
pH gradient establishment and stability present another major hurdle. The formation of stable, reproducible pH gradients is fundamental to IEF success, yet current methodologies struggle with gradient drift and cathodic mobilization. Commercial ampholyte compositions often exhibit batch-to-batch variations, further complicating standardization efforts. The lack of universal calibration standards for pH gradients exacerbates this issue.
Sample preparation protocols represent a significant source of variability. Differences in sample solubilization methods, buffer compositions, and additive concentrations can dramatically alter protein behavior during focusing. The absence of standardized guidelines for sample preparation tailored to specific protein classes contributes to inconsistent results across different research groups.
Instrumentation diversity further compounds standardization challenges. The wide array of IEF platforms—ranging from conventional gel-based systems to capillary and microfluidic formats—each introduce unique variables that affect protocol transferability. Variations in electrode design, chamber geometry, and power supply characteristics create additional layers of complexity when attempting to standardize protocols.
Data analysis and interpretation methodologies lack uniformity across the field. Different image acquisition systems, analysis software, and quantification approaches lead to divergent interpretations of similar experimental results. The absence of standardized reporting formats makes it difficult to compare findings between studies or establish reliable reference points.
Regulatory considerations also impede standardization efforts, particularly in clinical and pharmaceutical applications. Different regulatory bodies maintain varying requirements for validation and quality control, necessitating protocol modifications that further complicate standardization across international boundaries.
The economic implications of these challenges are substantial. The cost and time associated with protocol optimization and validation discourage smaller laboratories from adopting IEF technology, limiting its broader implementation and slowing technological advancement in the field.
Contemporary Approaches to IEF Protocol Optimization
01 Optimization of buffer systems for consistent isoelectric focusing
Buffer systems play a crucial role in maintaining consistent isoelectric focusing protocols. The composition, concentration, and pH range of buffer systems directly impact the resolution and reproducibility of protein separation. Ampholytes and other buffer components must be carefully selected to create stable pH gradients that allow proteins to migrate to their isoelectric points with high precision. Optimized buffer formulations help minimize drift and ensure consistent results across multiple runs.- Buffer composition and pH gradient optimization: The composition of buffer solutions and optimization of pH gradients are critical for consistent isoelectric focusing protocols. Proper selection of ampholytes, carrier ampholytes, and buffer systems helps establish stable pH gradients across the gel or capillary. Optimizing buffer concentrations and pH ranges ensures reproducible separation of proteins based on their isoelectric points, leading to more consistent and reliable results in isoelectric focusing experiments.
- Gel preparation and polymerization techniques: Consistent gel preparation and polymerization techniques are essential for reproducible isoelectric focusing. This includes standardized methods for preparing polyacrylamide gels with uniform thickness and pore size, controlling polymerization conditions, and incorporating ampholytes into the gel matrix. Proper gel preparation ensures uniform electric field distribution and consistent protein migration, contributing to protocol consistency and reproducible separation of proteins based on their isoelectric points.
- Temperature control and cooling systems: Temperature control is crucial for maintaining consistency in isoelectric focusing protocols. Effective cooling systems help dissipate heat generated during electrophoresis, preventing temperature gradients that can distort protein migration patterns. Maintaining uniform temperature across the separation medium ensures that protein mobility is determined solely by isoelectric points rather than thermal effects, leading to more reproducible and reliable results in isoelectric focusing experiments.
- Sample preparation and loading techniques: Standardized sample preparation and loading techniques are essential for consistent isoelectric focusing results. This includes methods for protein extraction, purification, and denaturation, as well as techniques for uniform sample application to the gel or capillary. Controlling sample volume, concentration, and composition helps ensure reproducible protein migration patterns and prevents artifacts that can affect the consistency of isoelectric focusing protocols.
- Automated systems and quality control measures: Automated isoelectric focusing systems and standardized quality control measures improve protocol consistency. These include computerized control of electrophoresis parameters, automated gel imaging and analysis, and the use of reference standards for system validation. Implementing quality control procedures, such as regular calibration and performance verification, ensures consistent and reliable results across multiple experiments and between different laboratories performing isoelectric focusing.
02 Temperature control methods for reproducible isoelectric focusing
Temperature management is critical for consistent isoelectric focusing results. Fluctuations in temperature can alter protein mobility, affect pH gradients, and lead to band distortion. Advanced temperature control systems that maintain uniform conditions throughout the separation process help ensure reproducibility. These systems may include cooling plates, thermostatic controls, and heat dissipation mechanisms that prevent Joule heating effects that would otherwise compromise focusing precision and protocol consistency.Expand Specific Solutions03 Standardized sample preparation techniques for isoelectric focusing
Consistent sample preparation is fundamental to achieving reproducible isoelectric focusing results. This includes standardized protocols for protein extraction, purification, and loading. Techniques such as desalting, denaturation control, and precise sample application methods ensure that proteins enter the focusing system under uniform conditions. Proper sample handling minimizes interference from contaminants and ensures that observed differences in migration patterns reflect actual protein properties rather than preparation artifacts.Expand Specific Solutions04 Gel formulation and polymerization standardization
The physical properties of separation media significantly impact isoelectric focusing consistency. Standardized procedures for gel preparation, including precise control of acrylamide concentration, crosslinking density, and polymerization conditions, ensure uniform pore size distribution and mechanical stability. Consistent gel formulations provide reproducible environments for protein migration and focusing. Advanced manufacturing techniques help minimize batch-to-batch variations that would otherwise lead to inconsistent separation patterns.Expand Specific Solutions05 Automated systems and quality control for isoelectric focusing
Automation technologies have significantly improved the consistency of isoelectric focusing protocols by reducing human error and standardizing operational parameters. Integrated systems that control voltage application, timing, and detection provide highly reproducible conditions across experiments. Additionally, implementation of quality control measures, including the use of standard markers, calibration proteins, and validation procedures, ensures protocol reliability. These systems often incorporate data analysis tools that help identify and correct for any remaining variability.Expand Specific Solutions
Leading Manufacturers and Research Institutions in IEF
The isoelectric focusing protocol consistency optimization market is in a growth phase, characterized by increasing demand for reliable protein separation techniques in proteomics research and biopharmaceutical development. The global market size is expanding steadily, driven by advancements in precision medicine and biomarker discovery. Technologically, this field shows moderate maturity with ongoing innovation. Key players include specialized companies like Intabio LLC and ProteoSys AG focusing exclusively on separation technologies, alongside diversified corporations such as Huawei Technologies and NEC Corporation that leverage their analytical capabilities. Academic institutions including Southeast University and Nanjing University contribute significant research advancements. The competitive landscape features collaboration between industry and academia to address reproducibility challenges, with emerging players from China increasingly gaining market share through cost-effective solutions.
Intabio LLC
Technical Solution: Intabio has developed a proprietary iCIEF-MS (imaged capillary isoelectric focusing-mass spectrometry) platform that addresses optimization challenges in isoelectric focusing protocols. Their technology integrates microfluidic cartridges with automated sample preparation and analysis systems to ensure consistent protein separation. The system employs real-time pH gradient monitoring and control mechanisms that automatically adjust buffer compositions and electric field parameters during the focusing process. This approach minimizes run-to-run variability by implementing closed-loop feedback systems that detect deviations in pH gradient formation and make compensatory adjustments. Additionally, Intabio's platform incorporates machine learning algorithms that analyze historical run data to predict optimal parameters for specific sample types, further enhancing protocol consistency across different laboratories and instrument setups.
Strengths: Integration of real-time monitoring with automated adjustments significantly reduces human error and improves reproducibility. The machine learning component enables continuous improvement of protocols based on accumulated data. Weaknesses: The proprietary nature of the system creates dependency on Intabio's consumables and software ecosystem, potentially increasing operational costs for laboratories.
ProteoSys AG
Technical Solution: ProteoSys AG has pioneered advanced isoelectric focusing (IEF) optimization through their CIEF-Stabilizer™ technology platform. This system addresses protocol consistency challenges by implementing temperature-controlled focusing chambers with precision regulation to within ±0.1°C, eliminating one of the primary sources of run-to-run variability. Their approach includes specialized carrier ampholyte formulations that resist degradation during extended focusing periods, maintaining stable pH gradients for up to 24 hours. ProteoSys has also developed proprietary electrode designs that minimize electrolysis effects at the buffer interfaces, preventing pH drift during extended separations. The company's software suite provides automated protocol optimization based on sample characteristics, adjusting voltage ramping profiles, focusing times, and mobilization parameters to achieve consistent results across different sample types and concentrations. This comprehensive approach has been validated across multiple laboratories, demonstrating significant improvements in peak resolution and positional reproducibility compared to conventional IEF methods.
Strengths: The temperature-controlled system with specialized ampholytes provides exceptional stability for extended runs, making it ideal for complex protein mixtures requiring high resolution. The automated protocol optimization reduces the expertise barrier for achieving consistent results. Weaknesses: The system requires specialized equipment and consumables, potentially limiting accessibility for smaller laboratories with budget constraints.
Critical Patents and Innovations in IEF Consistency
Method for analysis of proteins by solution isoelectric focusing
PatentInactiveEP1564550B1
Innovation
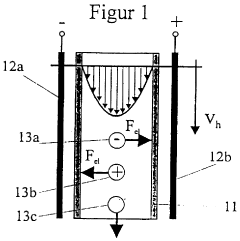
- A novel small-scale solution isofocusing method using a chamber with multiple compartments separated by pH gradient gel membranes, allowing for reproducible fractionation of charged molecules into well-defined pools, suitable for complex eukaryotic proteome samples with over 10,000 protein components.
Process and device for isoelectric particle separation
PatentWO1998013689A1
Innovation
- The method involves controlling the pH of a guide liquid to separate particles based on their charge, using an external electric field to move charged particles to a collecting device where they are temporarily fixed, allowing for efficient separation and collection without additional additives, and enabling continuous processing.
Quality Control Metrics for IEF Performance Evaluation
Establishing robust quality control metrics is essential for evaluating the performance and consistency of Isoelectric Focusing (IEF) protocols. These metrics provide quantitative frameworks to assess whether an IEF procedure meets predefined standards and delivers reproducible results across multiple runs.
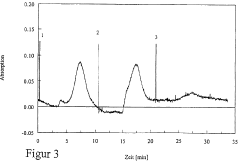
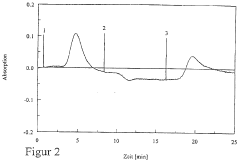
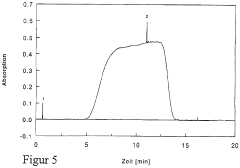
Peak resolution represents a primary quality indicator in IEF performance evaluation. This metric quantifies the system's ability to separate proteins with minimal pI differences, typically measured by calculating the distance between adjacent protein bands divided by their average peak width. Higher resolution values indicate superior separation capability, with industry standards typically requiring resolution values above 0.8 for analytical applications and above 1.2 for preparative purposes.
Reproducibility metrics track the consistency of results across multiple IEF runs under identical conditions. These include Coefficient of Variation (CV) for band positions (target: <2%), intensity measurements (acceptable range: 5-10%), and migration time stability (target: <3%). Systematic monitoring of these parameters enables identification of protocol drift and equipment degradation before they compromise experimental outcomes.
Signal-to-noise ratio (SNR) serves as another critical quality parameter, measuring the ratio between protein band intensity and background noise. Effective IEF protocols should consistently achieve SNR values exceeding 10:1, with high-sensitivity applications requiring 20:1 or greater. Regular SNR monitoring helps detect contamination issues, reagent degradation, or electrode deterioration.
pH gradient stability metrics evaluate the linearity and consistency of the established pH gradient. Deviations from linearity should not exceed ±0.2 pH units across the separation range, with gradient drift remaining below 0.1 pH units per hour. Modern IEF systems incorporate internal pH markers to continuously monitor gradient integrity throughout the separation process.
Focusing time optimization metrics balance separation quality against operational efficiency. Complete focusing is typically confirmed when current decreases to 10-15% of initial values and remains stable for at least 30 minutes. Excessive focusing times (>8 hours) may indicate suboptimal buffer compositions or sample overloading.
Implementation of standardized quality control charts tracking these metrics over time enables laboratories to establish statistical process control for their IEF protocols. These charts facilitate early detection of systematic errors, equipment failures, or reagent inconsistencies before they impact experimental outcomes, ultimately ensuring reliable protein characterization and consistent downstream applications.
Peak resolution represents a primary quality indicator in IEF performance evaluation. This metric quantifies the system's ability to separate proteins with minimal pI differences, typically measured by calculating the distance between adjacent protein bands divided by their average peak width. Higher resolution values indicate superior separation capability, with industry standards typically requiring resolution values above 0.8 for analytical applications and above 1.2 for preparative purposes.
Reproducibility metrics track the consistency of results across multiple IEF runs under identical conditions. These include Coefficient of Variation (CV) for band positions (target: <2%), intensity measurements (acceptable range: 5-10%), and migration time stability (target: <3%). Systematic monitoring of these parameters enables identification of protocol drift and equipment degradation before they compromise experimental outcomes.
Signal-to-noise ratio (SNR) serves as another critical quality parameter, measuring the ratio between protein band intensity and background noise. Effective IEF protocols should consistently achieve SNR values exceeding 10:1, with high-sensitivity applications requiring 20:1 or greater. Regular SNR monitoring helps detect contamination issues, reagent degradation, or electrode deterioration.
pH gradient stability metrics evaluate the linearity and consistency of the established pH gradient. Deviations from linearity should not exceed ±0.2 pH units across the separation range, with gradient drift remaining below 0.1 pH units per hour. Modern IEF systems incorporate internal pH markers to continuously monitor gradient integrity throughout the separation process.
Focusing time optimization metrics balance separation quality against operational efficiency. Complete focusing is typically confirmed when current decreases to 10-15% of initial values and remains stable for at least 30 minutes. Excessive focusing times (>8 hours) may indicate suboptimal buffer compositions or sample overloading.
Implementation of standardized quality control charts tracking these metrics over time enables laboratories to establish statistical process control for their IEF protocols. These charts facilitate early detection of systematic errors, equipment failures, or reagent inconsistencies before they impact experimental outcomes, ultimately ensuring reliable protein characterization and consistent downstream applications.
Regulatory Compliance for IEF in Clinical Applications
Regulatory compliance for Isoelectric Focusing (IEF) in clinical applications represents a critical consideration for laboratories and manufacturers implementing this analytical technique. The FDA, EMA, and other global regulatory bodies have established stringent frameworks governing the use of IEF in diagnostic and therapeutic contexts, particularly when results directly impact patient care decisions.
Clinical IEF applications must adhere to Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP) standards, which mandate comprehensive documentation of protocol validation, equipment calibration, and quality control measures. These requirements ensure reproducibility and reliability of results across different laboratory settings and timeframes.
The Clinical Laboratory Improvement Amendments (CLIA) in the United States specifically addresses IEF when used for clinical diagnostics, requiring laboratories to demonstrate proficiency testing and maintain detailed records of protocol consistency. Similarly, the EU's In Vitro Diagnostic Regulation (IVDR) imposes additional verification requirements for IEF-based diagnostic methods, emphasizing the need for standardized protocols.
Protocol consistency in IEF faces particular regulatory scrutiny due to its sensitivity to minor procedural variations. Regulatory bodies typically require extensive validation studies demonstrating robustness across different operators, reagent lots, and environmental conditions. This validation must include statistical analysis of reproducibility parameters and establishment of acceptance criteria for quality control samples.
Risk management frameworks must be implemented for IEF clinical applications, identifying potential failure modes that could compromise protocol consistency. These frameworks should include mitigation strategies and contingency plans for addressing deviations, with clear documentation of corrective and preventive actions (CAPA) when inconsistencies arise.
Regulatory compliance also extends to reagent and equipment qualification. IEF reagents used in clinical settings often require certification as medical devices or diagnostic components, necessitating additional quality control measures and stability testing. Equipment validation protocols must demonstrate that instruments consistently maintain critical parameters such as temperature gradients and electrical field strength.
Data integrity requirements present another regulatory dimension, with agencies increasingly focusing on electronic record compliance with 21 CFR Part 11 or equivalent standards. This includes audit trails for any modifications to IEF protocols, secure data storage, and validated analysis software that maintains the integrity of results from raw data through final reporting.
Clinical IEF applications must adhere to Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP) standards, which mandate comprehensive documentation of protocol validation, equipment calibration, and quality control measures. These requirements ensure reproducibility and reliability of results across different laboratory settings and timeframes.
The Clinical Laboratory Improvement Amendments (CLIA) in the United States specifically addresses IEF when used for clinical diagnostics, requiring laboratories to demonstrate proficiency testing and maintain detailed records of protocol consistency. Similarly, the EU's In Vitro Diagnostic Regulation (IVDR) imposes additional verification requirements for IEF-based diagnostic methods, emphasizing the need for standardized protocols.
Protocol consistency in IEF faces particular regulatory scrutiny due to its sensitivity to minor procedural variations. Regulatory bodies typically require extensive validation studies demonstrating robustness across different operators, reagent lots, and environmental conditions. This validation must include statistical analysis of reproducibility parameters and establishment of acceptance criteria for quality control samples.
Risk management frameworks must be implemented for IEF clinical applications, identifying potential failure modes that could compromise protocol consistency. These frameworks should include mitigation strategies and contingency plans for addressing deviations, with clear documentation of corrective and preventive actions (CAPA) when inconsistencies arise.
Regulatory compliance also extends to reagent and equipment qualification. IEF reagents used in clinical settings often require certification as medical devices or diagnostic components, necessitating additional quality control measures and stability testing. Equipment validation protocols must demonstrate that instruments consistently maintain critical parameters such as temperature gradients and electrical field strength.
Data integrity requirements present another regulatory dimension, with agencies increasingly focusing on electronic record compliance with 21 CFR Part 11 or equivalent standards. This includes audit trails for any modifications to IEF protocols, secure data storage, and validated analysis software that maintains the integrity of results from raw data through final reporting.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!