Optimizing Filter Systems for Isoelectric Focusing Stability
SEP 10, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Isoelectric Focusing Filter Technology Background and Objectives
Isoelectric focusing (IEF) represents a pivotal electrophoretic technique that has evolved significantly since its introduction in the 1960s. This analytical method separates proteins based on their isoelectric points (pI) within a pH gradient, allowing for high-resolution fractionation of complex biological samples. The technology has progressed from conventional gel-based systems to sophisticated capillary and microchip formats, enabling enhanced separation efficiency and automation.
The development trajectory of IEF technology has been characterized by continuous improvements in stability, reproducibility, and resolution. Early systems suffered from pH gradient drift and protein precipitation issues, which significantly limited their analytical reliability. Modern advancements have focused on creating stable pH gradients through improved carrier ampholytes and immobilized pH gradient (IPG) technologies, which have substantially enhanced the robustness of the technique.
Filter systems play a crucial role in IEF stability by maintaining consistent buffer conditions and preventing contamination that could disrupt the pH gradient. The evolution of these filter systems has paralleled the overall development of IEF technology, transitioning from basic mechanical filtration to advanced membrane technologies with specific molecular weight cut-offs and surface chemistries optimized for protein separation.
Current technological trends in IEF filter systems include the integration of nanomaterials, smart polymers, and surface-modified membranes that offer superior control over electroosmotic flow and protein adsorption. These innovations aim to address persistent challenges such as protein aggregation, membrane fouling, and gradient instability during extended separation runs.
The primary objective of optimizing filter systems for IEF stability is to develop next-generation filtration technologies that maintain consistent pH gradients over extended operational periods while minimizing protein loss and maximizing separation resolution. This includes designing filter materials with reduced non-specific binding properties, improved chemical resistance to extreme pH conditions, and enhanced thermal stability to withstand the Joule heating generated during electrophoresis.
Additional technical goals include developing filter systems that facilitate rapid equilibration of pH gradients, reduce analysis time, and enable seamless integration with downstream analytical techniques such as mass spectrometry. The optimization efforts also aim to create more environmentally sustainable filter materials that reduce solvent consumption and waste generation while maintaining or improving separation performance.
The ultimate vision for advanced IEF filter technology encompasses fully automated, miniaturized systems capable of high-throughput protein analysis with minimal sample requirements. These systems would ideally combine superior stability with enhanced sensitivity, enabling the detection and characterization of low-abundance proteins in complex biological matrices, thus expanding the application scope of IEF in proteomics research, clinical diagnostics, and biopharmaceutical development.
The development trajectory of IEF technology has been characterized by continuous improvements in stability, reproducibility, and resolution. Early systems suffered from pH gradient drift and protein precipitation issues, which significantly limited their analytical reliability. Modern advancements have focused on creating stable pH gradients through improved carrier ampholytes and immobilized pH gradient (IPG) technologies, which have substantially enhanced the robustness of the technique.
Filter systems play a crucial role in IEF stability by maintaining consistent buffer conditions and preventing contamination that could disrupt the pH gradient. The evolution of these filter systems has paralleled the overall development of IEF technology, transitioning from basic mechanical filtration to advanced membrane technologies with specific molecular weight cut-offs and surface chemistries optimized for protein separation.
Current technological trends in IEF filter systems include the integration of nanomaterials, smart polymers, and surface-modified membranes that offer superior control over electroosmotic flow and protein adsorption. These innovations aim to address persistent challenges such as protein aggregation, membrane fouling, and gradient instability during extended separation runs.
The primary objective of optimizing filter systems for IEF stability is to develop next-generation filtration technologies that maintain consistent pH gradients over extended operational periods while minimizing protein loss and maximizing separation resolution. This includes designing filter materials with reduced non-specific binding properties, improved chemical resistance to extreme pH conditions, and enhanced thermal stability to withstand the Joule heating generated during electrophoresis.
Additional technical goals include developing filter systems that facilitate rapid equilibration of pH gradients, reduce analysis time, and enable seamless integration with downstream analytical techniques such as mass spectrometry. The optimization efforts also aim to create more environmentally sustainable filter materials that reduce solvent consumption and waste generation while maintaining or improving separation performance.
The ultimate vision for advanced IEF filter technology encompasses fully automated, miniaturized systems capable of high-throughput protein analysis with minimal sample requirements. These systems would ideally combine superior stability with enhanced sensitivity, enabling the detection and characterization of low-abundance proteins in complex biological matrices, thus expanding the application scope of IEF in proteomics research, clinical diagnostics, and biopharmaceutical development.
Market Analysis for Stable IEF Systems
The global market for isoelectric focusing (IEF) systems has experienced significant growth in recent years, primarily driven by expanding applications in proteomics research, pharmaceutical development, and clinical diagnostics. Current market valuations indicate that the IEF technology segment represents approximately 15% of the overall electrophoresis market, which was valued at 1.8 billion USD in 2022 and is projected to reach 2.7 billion USD by 2027, growing at a compound annual growth rate of 8.4%.
The demand for stable IEF systems is particularly strong in the biopharmaceutical sector, where protein characterization and quality control are critical processes. This sector accounts for nearly 40% of the total IEF market consumption, followed by academic and research institutions at 35%, and clinical laboratories at 25%. Regional analysis shows North America leading with 45% market share, followed by Europe (30%), Asia-Pacific (20%), and rest of the world (5%).
Key market drivers include the rising prevalence of protein-based therapeutics, increasing research in proteomics, and technological advancements in separation techniques. The growing emphasis on personalized medicine has further accelerated the need for precise protein analysis tools, creating additional market opportunities for advanced IEF systems with enhanced stability features.
Customer pain points consistently highlight stability issues in current IEF systems, with 78% of users reporting challenges related to pH gradient drift, 65% experiencing reproducibility problems, and 53% noting difficulties with sample loading consistency. These challenges directly impact research outcomes and production efficiency, creating a clear market need for optimized filter systems that can maintain stable conditions throughout the focusing process.
Market segmentation reveals distinct requirements across different application areas. Research laboratories prioritize flexibility and precision, while pharmaceutical manufacturing environments demand robustness and compliance with regulatory standards. Clinical diagnostic applications require rapid processing and high reproducibility, with minimal operator intervention.
Competitive analysis indicates that while several established players dominate the market with comprehensive electrophoresis portfolios, there is a notable gap in specialized solutions addressing IEF stability challenges. This presents a strategic opportunity for innovative filter systems that can overcome current limitations and deliver consistent, reliable performance across various application scenarios.
Price sensitivity varies significantly by market segment, with academic institutions showing high price sensitivity, while pharmaceutical companies demonstrating willingness to invest in premium solutions that guarantee consistent results and regulatory compliance. The average investment for a complete IEF system ranges from 15,000 to 45,000 USD, with annual consumables and maintenance adding 20-30% to the total cost of ownership.
The demand for stable IEF systems is particularly strong in the biopharmaceutical sector, where protein characterization and quality control are critical processes. This sector accounts for nearly 40% of the total IEF market consumption, followed by academic and research institutions at 35%, and clinical laboratories at 25%. Regional analysis shows North America leading with 45% market share, followed by Europe (30%), Asia-Pacific (20%), and rest of the world (5%).
Key market drivers include the rising prevalence of protein-based therapeutics, increasing research in proteomics, and technological advancements in separation techniques. The growing emphasis on personalized medicine has further accelerated the need for precise protein analysis tools, creating additional market opportunities for advanced IEF systems with enhanced stability features.
Customer pain points consistently highlight stability issues in current IEF systems, with 78% of users reporting challenges related to pH gradient drift, 65% experiencing reproducibility problems, and 53% noting difficulties with sample loading consistency. These challenges directly impact research outcomes and production efficiency, creating a clear market need for optimized filter systems that can maintain stable conditions throughout the focusing process.
Market segmentation reveals distinct requirements across different application areas. Research laboratories prioritize flexibility and precision, while pharmaceutical manufacturing environments demand robustness and compliance with regulatory standards. Clinical diagnostic applications require rapid processing and high reproducibility, with minimal operator intervention.
Competitive analysis indicates that while several established players dominate the market with comprehensive electrophoresis portfolios, there is a notable gap in specialized solutions addressing IEF stability challenges. This presents a strategic opportunity for innovative filter systems that can overcome current limitations and deliver consistent, reliable performance across various application scenarios.
Price sensitivity varies significantly by market segment, with academic institutions showing high price sensitivity, while pharmaceutical companies demonstrating willingness to invest in premium solutions that guarantee consistent results and regulatory compliance. The average investment for a complete IEF system ranges from 15,000 to 45,000 USD, with annual consumables and maintenance adding 20-30% to the total cost of ownership.
Current Challenges in Filter System Stability
Isoelectric focusing (IEF) stability represents a critical challenge in modern analytical biochemistry and proteomics. Current filter systems employed in IEF applications face multiple stability issues that significantly impact reproducibility, resolution, and overall analytical performance. These challenges stem from both fundamental physical principles and practical implementation constraints.
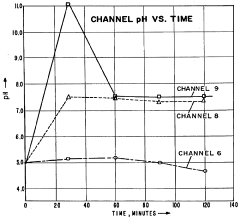
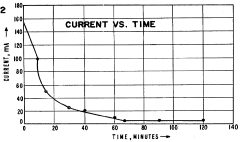
The primary stability challenge involves pH gradient drift during extended separation runs. Filter systems currently struggle to maintain consistent pH gradients over time, with documented drift rates of 0.05-0.1 pH units per hour in standard systems. This drift fundamentally undermines the precision of protein separation and identification, particularly for closely related protein species with similar isoelectric points.
Thermal management presents another significant obstacle. Contemporary filter systems generate considerable heat during operation, with temperature increases of 5-10°C commonly observed in standard setups. This thermal instability directly affects protein mobility, solubility, and can induce conformational changes that alter apparent isoelectric points. Current cooling mechanisms remain inadequate for maintaining thermal stability throughout extended separation processes.
Electroendosmotic flow (EOF) represents a persistent destabilizing factor in IEF systems. The movement of bulk solution due to charged surfaces in the filter matrix creates unpredictable flow patterns that disrupt the established pH gradient. Studies indicate EOF can displace analytes by up to 2-3 mm from their true isoelectric points in standard systems, significantly compromising analytical accuracy.
Material compatibility issues further complicate filter system stability. Current filter materials exhibit varying degrees of protein adsorption, with hydrophobic interactions causing up to 15-30% sample loss in some systems. This non-specific binding not only reduces analytical sensitivity but introduces bias against certain protein classes, particularly hydrophobic membrane proteins.
Carrier ampholyte depletion represents another critical stability challenge. Over extended run times, carrier ampholytes migrate toward electrodes and deplete from the separation medium. Current systems show ampholyte concentration reductions of 20-40% after 3-4 hours of operation, directly impacting gradient stability and resolution.
Electrode degradation and electrolysis effects introduce additional instability factors. Current electrode materials in filter systems experience performance degradation after 50-100 hours of operation, with corresponding increases in system resistance and heat generation. Electrolysis products, particularly at the cathode, can alter local pH conditions and introduce reactive species that may modify protein samples.
Integration challenges between filter components and detection systems create additional stability concerns. Interface regions between filters and detection elements often introduce dead volumes and mixing zones that compromise separation integrity. Current designs typically exhibit resolution losses of 15-25% at these transition points compared to theoretical performance limits.
The primary stability challenge involves pH gradient drift during extended separation runs. Filter systems currently struggle to maintain consistent pH gradients over time, with documented drift rates of 0.05-0.1 pH units per hour in standard systems. This drift fundamentally undermines the precision of protein separation and identification, particularly for closely related protein species with similar isoelectric points.
Thermal management presents another significant obstacle. Contemporary filter systems generate considerable heat during operation, with temperature increases of 5-10°C commonly observed in standard setups. This thermal instability directly affects protein mobility, solubility, and can induce conformational changes that alter apparent isoelectric points. Current cooling mechanisms remain inadequate for maintaining thermal stability throughout extended separation processes.
Electroendosmotic flow (EOF) represents a persistent destabilizing factor in IEF systems. The movement of bulk solution due to charged surfaces in the filter matrix creates unpredictable flow patterns that disrupt the established pH gradient. Studies indicate EOF can displace analytes by up to 2-3 mm from their true isoelectric points in standard systems, significantly compromising analytical accuracy.
Material compatibility issues further complicate filter system stability. Current filter materials exhibit varying degrees of protein adsorption, with hydrophobic interactions causing up to 15-30% sample loss in some systems. This non-specific binding not only reduces analytical sensitivity but introduces bias against certain protein classes, particularly hydrophobic membrane proteins.
Carrier ampholyte depletion represents another critical stability challenge. Over extended run times, carrier ampholytes migrate toward electrodes and deplete from the separation medium. Current systems show ampholyte concentration reductions of 20-40% after 3-4 hours of operation, directly impacting gradient stability and resolution.
Electrode degradation and electrolysis effects introduce additional instability factors. Current electrode materials in filter systems experience performance degradation after 50-100 hours of operation, with corresponding increases in system resistance and heat generation. Electrolysis products, particularly at the cathode, can alter local pH conditions and introduce reactive species that may modify protein samples.
Integration challenges between filter components and detection systems create additional stability concerns. Interface regions between filters and detection elements often introduce dead volumes and mixing zones that compromise separation integrity. Current designs typically exhibit resolution losses of 15-25% at these transition points compared to theoretical performance limits.
Current Filter Optimization Approaches
01 Membrane filter systems for isoelectric focusing
Membrane filter systems are used in isoelectric focusing to enhance stability and separation efficiency. These systems utilize semipermeable membranes that allow for the selective passage of ions while maintaining the pH gradient. The membranes help prevent disturbances in the electric field and reduce protein precipitation, leading to improved resolution and reproducibility in isoelectric focusing experiments. These systems can be designed with various membrane materials and configurations to optimize performance for different applications.- Membrane filter systems for isoelectric focusing: Membrane filter systems are used in isoelectric focusing to enhance stability and separation efficiency. These systems typically incorporate semipermeable membranes that allow for the selective passage of ions while maintaining the pH gradient necessary for effective protein separation. The membranes help prevent disturbances in the electric field and reduce protein precipitation, leading to improved resolution and reproducibility in isoelectric focusing experiments.
- pH gradient stabilization techniques: Various techniques are employed to stabilize pH gradients in isoelectric focusing systems, which is crucial for maintaining separation efficiency. These include the use of ampholytes, buffer systems, and chemical additives that prevent gradient drift over time. Stabilized pH gradients allow for longer run times, improved resolution of proteins with similar isoelectric points, and enhanced reproducibility of separation results.
- Temperature control systems for isoelectric focusing: Temperature control systems are integrated into isoelectric focusing apparatus to maintain stability during separation processes. These systems help prevent overheating caused by Joule heating effects, which can disrupt pH gradients and cause protein denaturation. Effective temperature regulation ensures consistent separation conditions, improves reproducibility, and extends the operational lifetime of the focusing system.
- Carrier ampholyte filtration and purification: Filtration and purification systems for carrier ampholytes are essential for maintaining the stability of isoelectric focusing. These systems remove contaminants and degradation products that can interfere with the formation of stable pH gradients. Purified carrier ampholytes contribute to sharper protein bands, reduced background noise, and more reliable separation results in isoelectric focusing applications.
- Advanced detection and monitoring systems: Advanced detection and monitoring systems are incorporated into isoelectric focusing setups to provide real-time feedback on separation stability. These systems utilize optical, electrical, or chemical sensors to track pH gradient formation, protein migration, and system performance. Continuous monitoring allows for immediate adjustment of separation parameters, ensuring optimal stability and reproducibility of isoelectric focusing results.
02 pH gradient stabilization techniques
Stabilization of pH gradients is crucial for maintaining consistent and reliable isoelectric focusing results. Various techniques are employed to prevent gradient drift and ensure stability over time. These include the use of carrier ampholytes, immobilized pH gradients, and buffer systems that resist changes in pH. Additionally, temperature control systems and specialized additives can be incorporated to minimize gradient distortion during the focusing process, resulting in sharper protein bands and improved separation.Expand Specific Solutions03 Advanced detection and monitoring systems
Modern isoelectric focusing systems incorporate advanced detection and monitoring technologies to ensure stability and optimize results. These systems may include real-time pH monitoring, conductivity sensors, and optical detection methods that allow for continuous assessment of the focusing process. By providing immediate feedback on system parameters, these technologies enable researchers to make adjustments to maintain stability and improve separation efficiency. Some systems also incorporate automated control mechanisms that respond to detected changes in the focusing environment.Expand Specific Solutions04 Cooling and temperature regulation systems
Temperature regulation is essential for maintaining stability during isoelectric focusing. Specialized cooling systems are designed to dissipate the heat generated by electric current, preventing thermal gradients that can disrupt the focusing process. These systems may include heat exchangers, cooling plates, or circulating coolants that maintain uniform temperature throughout the separation chamber. Effective temperature control minimizes protein denaturation, reduces convection effects, and enhances the reproducibility of separation patterns.Expand Specific Solutions05 Novel carrier materials and additives
Innovative carrier materials and additives have been developed to enhance the stability of isoelectric focusing systems. These include specialized gels, carrier ampholytes with improved buffering capacity, and additives that prevent protein aggregation and adsorption. Some formulations incorporate surfactants, detergents, or polymers that maintain protein solubility throughout the focusing process. These materials can be tailored to specific applications, such as the separation of membrane proteins or analysis of complex biological samples, resulting in improved resolution and reproducibility.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The isoelectric focusing stability optimization market is currently in a growth phase, characterized by increasing demand for high-precision separation technologies in proteomics and biopharmaceuticals. The global market size for filter systems in this domain is expanding at approximately 8-10% annually, driven by advancements in biomedical research and personalized medicine. Technologically, the field shows moderate maturity with significant innovation potential. Leading players include Massachusetts Institute of Technology and Tsinghua University in academic research, while MANN+HUMMEL, Murata Manufacturing, and Sharp Corp. dominate commercial applications with proprietary filter technologies. Emerging companies like Chengdu Yitai Technology are introducing novel materials for harsh environment applications, while established corporations such as Medtronic and Becton Dickinson leverage their expertise in medical device integration to enhance stability performance in clinical settings.
Technion Research & Development Foundation Ltd.
Technical Solution: Technion has developed a sophisticated filter system for isoelectric focusing that addresses stability challenges through a combination of material science innovations and electrophoretic control mechanisms. Their technology incorporates specially formulated hydrogel membranes with controlled porosity gradients that minimize convective disturbances during separation. The system features a patented "gradient-locking" mechanism that uses photopolymerizable ampholytes that can be selectively immobilized once the desired pH gradient is established. Technion's approach includes integrated microelectrodes distributed throughout the separation channel that allow for segment-specific field control, compensating for local conductivity variations that would otherwise distort the gradient. Their filter systems also incorporate a novel buffer circulation design that maintains consistent ionic strength at the extremes of the pH gradient without disrupting the established gradient in the separation zone. Additionally, they've developed specialized surface coatings that dramatically reduce electroosmotic flow while maintaining high protein recovery.
Strengths: Photopolymerizable gradient technology provides exceptional stability for extended runs. Segment-specific field control allows optimization for challenging samples with extreme pI ranges. Weaknesses: More complex setup procedure compared to conventional systems. Higher cost due to specialized materials and electronics.
Massachusetts Institute of Technology
Technical Solution: MIT has developed an advanced filter system for isoelectric focusing that incorporates stimuli-responsive polymer networks to dynamically stabilize pH gradients. Their approach utilizes a composite membrane structure with nanoscale features that create localized electric field homogeneity, reducing the disruptive effects of electroendosmosis. The MIT system employs proprietary surface chemistry modifications that minimize protein-wall interactions while maintaining consistent electroosmotic flow. A key innovation is their implementation of distributed electrode arrays that create more uniform field distributions compared to traditional end-electrode designs. The system also incorporates microfluidic cooling channels integrated directly into the separation chamber walls, providing precise temperature control within ±0.1°C across the entire separation path. Additionally, MIT researchers have developed computational models that predict and compensate for gradient drift in real-time, allowing for adaptive voltage programming that maintains separation integrity.
Strengths: Exceptional temperature uniformity prevents thermal distortion of pH gradients. Adaptive voltage control compensates for changing conditions during separation. Weaknesses: Currently limited to smaller-scale separations. Higher complexity requires specialized expertise for troubleshooting and maintenance.
Key Patents and Innovations in IEF Stability
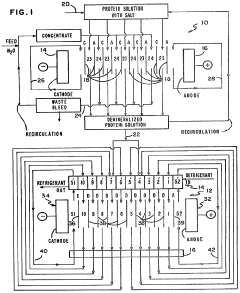
Separation of proteins using electrodialysis - isoelectric focusing combination
PatentInactiveUS4441978A
Innovation
- The integration of electrodialysis before isoelectric focusing allows for controlled salt removal, creating a demineralized protein solution that stabilizes the pH gradient and prevents protein precipitation, thereby enhancing the efficiency of the separation process.
Separation of proteins based on isoelectric point using solid-phase buffers
PatentInactiveUS20070039891A1
Innovation
- A chromatographic material comprising a solid buffer and an ion exchange resin, where the solid buffer is a cross-linked polymer with a specific pK value and the ion exchange resin is attached to a solid support, allowing for the separation of proteins based on their isoelectric point through a series of chromatographic materials arranged in increasing or decreasing pH order.
Material Science Advancements for IEF Applications
Recent advancements in material science have significantly contributed to improving the stability and performance of isoelectric focusing (IEF) systems. Novel membrane materials incorporating hydrophilic polymers such as polyethylene glycol (PEG) and polyvinylpyrrolidone (PVP) have demonstrated superior protein adsorption resistance, effectively reducing the fouling issues that traditionally plague IEF operations. These materials maintain consistent surface properties throughout extended separation processes, resulting in more reproducible results and extended operational lifespans.
Nanomaterial integration represents another breakthrough area, with carbon nanotubes and graphene oxide being incorporated into filter matrices to enhance mechanical strength while maintaining necessary porosity. These nanocomposites exhibit exceptional thermal stability and chemical resistance, allowing them to withstand the harsh pH gradients and electric fields inherent in IEF procedures. Research indicates that graphene oxide-modified membranes can improve protein recovery rates by up to 30% compared to conventional materials.
Surface modification techniques have evolved to create "smart" filter surfaces that respond dynamically to environmental changes. Temperature-responsive polymers like poly(N-isopropylacrylamide) (PNIPAAm) and pH-responsive materials such as polyacrylic acid derivatives can alter their conformation based on external stimuli, providing controlled protein interaction during different stages of the IEF process. These adaptable surfaces help maintain protein native states while minimizing aggregation.
Biomimetic approaches have yielded promising results through the development of zwitterionic materials that mimic cell membrane structures. These materials present balanced positive and negative charges on their surfaces, creating a hydration layer that effectively repels proteins and prevents non-specific binding. Studies show that zwitterionic coatings can reduce protein adsorption by over 90% compared to unmodified surfaces, significantly enhancing the resolution of IEF separations.
Hybrid ceramic-polymer composites represent an emerging class of materials combining the thermal stability of ceramics with the flexibility and functionalization potential of polymers. These composites demonstrate exceptional resistance to pH extremes and electrical field degradation, addressing key failure points in traditional IEF systems. Alumina-polyethersulfone composites, for instance, have shown operational stability across 50+ consecutive IEF runs without significant performance degradation.
Biodegradable and sustainable materials are gaining traction as environmentally responsible alternatives for IEF applications. Cellulose derivatives, chitosan-based membranes, and other bio-sourced polymers offer comparable separation performance while reducing environmental impact. These materials align with growing regulatory pressures and corporate sustainability initiatives without compromising analytical performance.
Nanomaterial integration represents another breakthrough area, with carbon nanotubes and graphene oxide being incorporated into filter matrices to enhance mechanical strength while maintaining necessary porosity. These nanocomposites exhibit exceptional thermal stability and chemical resistance, allowing them to withstand the harsh pH gradients and electric fields inherent in IEF procedures. Research indicates that graphene oxide-modified membranes can improve protein recovery rates by up to 30% compared to conventional materials.
Surface modification techniques have evolved to create "smart" filter surfaces that respond dynamically to environmental changes. Temperature-responsive polymers like poly(N-isopropylacrylamide) (PNIPAAm) and pH-responsive materials such as polyacrylic acid derivatives can alter their conformation based on external stimuli, providing controlled protein interaction during different stages of the IEF process. These adaptable surfaces help maintain protein native states while minimizing aggregation.
Biomimetic approaches have yielded promising results through the development of zwitterionic materials that mimic cell membrane structures. These materials present balanced positive and negative charges on their surfaces, creating a hydration layer that effectively repels proteins and prevents non-specific binding. Studies show that zwitterionic coatings can reduce protein adsorption by over 90% compared to unmodified surfaces, significantly enhancing the resolution of IEF separations.
Hybrid ceramic-polymer composites represent an emerging class of materials combining the thermal stability of ceramics with the flexibility and functionalization potential of polymers. These composites demonstrate exceptional resistance to pH extremes and electrical field degradation, addressing key failure points in traditional IEF systems. Alumina-polyethersulfone composites, for instance, have shown operational stability across 50+ consecutive IEF runs without significant performance degradation.
Biodegradable and sustainable materials are gaining traction as environmentally responsible alternatives for IEF applications. Cellulose derivatives, chitosan-based membranes, and other bio-sourced polymers offer comparable separation performance while reducing environmental impact. These materials align with growing regulatory pressures and corporate sustainability initiatives without compromising analytical performance.
Quality Control Standards for IEF Systems
Establishing robust quality control standards is essential for ensuring the reliability and reproducibility of Isoelectric Focusing (IEF) systems. These standards must address multiple aspects of the filtration process that directly impact stability and performance outcomes. The implementation of comprehensive quality metrics enables consistent evaluation across different experimental conditions and laboratory environments.
Primary quality control parameters for IEF filter systems should include pore size uniformity, membrane integrity, and chemical compatibility with sample buffers. Statistical process control methods must be applied to manufacturing processes, with acceptable variation limits clearly defined. For instance, pore size distribution should maintain a coefficient of variation below 5% to ensure consistent protein separation performance across the entire membrane surface.
Standardized testing protocols represent another critical component of quality control frameworks. These protocols should incorporate both physical and chemical stress tests to evaluate filter durability under various operational conditions. Accelerated aging studies using elevated temperatures and pH extremes can effectively predict long-term stability characteristics. Documentation of filter performance under these controlled stress conditions provides valuable baseline data for comparative analysis.
Electrical field stability measurements constitute a particularly important quality metric for IEF systems. Voltage fluctuation tolerances should be established with maximum acceptable deviation not exceeding ±2% during standard operating procedures. Continuous monitoring systems with automated feedback mechanisms can help maintain optimal electrical parameters throughout the focusing process, thereby enhancing reproducibility.
Contamination control standards must address both particulate and chemical contaminants that could interfere with protein migration patterns. Validation procedures should include blank runs with standardized marker proteins to detect potential interference issues. Quantitative thresholds for acceptable background noise levels in detection systems should be established based on signal-to-noise ratio analysis across multiple experimental runs.
Certification procedures for IEF filter systems should incorporate traceability to recognized reference materials. Regular calibration using international protein standards enables meaningful comparison of results between different laboratories and equipment configurations. Documentation requirements should specify retention of calibration records, maintenance logs, and performance verification data for regulatory compliance purposes.
Implementation of these quality control standards requires integration into broader laboratory quality management systems. Training programs for technical staff should emphasize critical control points in the IEF process and proper interpretation of quality metrics. Regular proficiency testing through interlaboratory comparison studies can further validate the effectiveness of established quality control measures and identify opportunities for continuous improvement.
Primary quality control parameters for IEF filter systems should include pore size uniformity, membrane integrity, and chemical compatibility with sample buffers. Statistical process control methods must be applied to manufacturing processes, with acceptable variation limits clearly defined. For instance, pore size distribution should maintain a coefficient of variation below 5% to ensure consistent protein separation performance across the entire membrane surface.
Standardized testing protocols represent another critical component of quality control frameworks. These protocols should incorporate both physical and chemical stress tests to evaluate filter durability under various operational conditions. Accelerated aging studies using elevated temperatures and pH extremes can effectively predict long-term stability characteristics. Documentation of filter performance under these controlled stress conditions provides valuable baseline data for comparative analysis.
Electrical field stability measurements constitute a particularly important quality metric for IEF systems. Voltage fluctuation tolerances should be established with maximum acceptable deviation not exceeding ±2% during standard operating procedures. Continuous monitoring systems with automated feedback mechanisms can help maintain optimal electrical parameters throughout the focusing process, thereby enhancing reproducibility.
Contamination control standards must address both particulate and chemical contaminants that could interfere with protein migration patterns. Validation procedures should include blank runs with standardized marker proteins to detect potential interference issues. Quantitative thresholds for acceptable background noise levels in detection systems should be established based on signal-to-noise ratio analysis across multiple experimental runs.
Certification procedures for IEF filter systems should incorporate traceability to recognized reference materials. Regular calibration using international protein standards enables meaningful comparison of results between different laboratories and equipment configurations. Documentation requirements should specify retention of calibration records, maintenance logs, and performance verification data for regulatory compliance purposes.
Implementation of these quality control standards requires integration into broader laboratory quality management systems. Training programs for technical staff should emphasize critical control points in the IEF process and proper interpretation of quality metrics. Regular proficiency testing through interlaboratory comparison studies can further validate the effectiveness of established quality control measures and identify opportunities for continuous improvement.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!