Optimizing Sample Application Techniques in Isoelectric Focusing
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
IEF Technology Background and Objectives
Isoelectric focusing (IEF) emerged in the 1960s as a groundbreaking electrophoretic technique for separating amphoteric molecules, particularly proteins, based on their isoelectric points (pI). The technique was pioneered by Harry Svensson and subsequently refined by researchers like Righetti and Bjellqvist, who introduced immobilized pH gradients (IPGs) in the 1980s, significantly enhancing reproducibility and resolution.
The evolution of IEF technology has been characterized by continuous improvements in sample application methodologies, which directly impact separation efficiency, resolution, and reproducibility. Traditional sample application techniques involved direct loading onto gel surfaces or within sample cups, often resulting in protein precipitation, streaking, and poor resolution due to salt interference and uneven sample distribution.
Current technological trends in IEF sample application are moving toward automated, precise, and miniaturized systems. The integration of microfluidic platforms, robotic sample handlers, and advanced liquid handling technologies has enabled more consistent and reproducible sample application. Additionally, developments in sample preparation protocols, including desalting techniques and pre-fractionation methods, have addressed many historical challenges associated with IEF.
The primary objective of optimizing sample application techniques in IEF is to enhance protein separation resolution while maintaining high reproducibility and throughput. Specific goals include minimizing protein precipitation at application points, reducing horizontal and vertical streaking, enabling effective separation of low-abundance proteins, and accommodating diverse sample types from various biological sources.
Technical objectives also encompass the development of standardized protocols that can be implemented across different laboratory settings with minimal variation. This standardization is crucial for comparative proteomics studies and clinical applications where consistency is paramount. Furthermore, there is a growing emphasis on developing environmentally sustainable sample application methods that reduce reagent consumption and waste generation.
The advancement of IEF sample application techniques is increasingly influenced by integration with downstream analytical technologies, particularly mass spectrometry. This integration necessitates sample application methods that are compatible with subsequent analytical steps, minimizing sample loss and contamination during transfer processes.
As proteomics research expands into single-cell analysis and personalized medicine, there is an emerging need for sample application techniques capable of handling ultra-small sample volumes while maintaining sensitivity and specificity. This represents a frontier in IEF technology development, driving innovation toward nano-scale sample handling and application systems.
The evolution of IEF technology has been characterized by continuous improvements in sample application methodologies, which directly impact separation efficiency, resolution, and reproducibility. Traditional sample application techniques involved direct loading onto gel surfaces or within sample cups, often resulting in protein precipitation, streaking, and poor resolution due to salt interference and uneven sample distribution.
Current technological trends in IEF sample application are moving toward automated, precise, and miniaturized systems. The integration of microfluidic platforms, robotic sample handlers, and advanced liquid handling technologies has enabled more consistent and reproducible sample application. Additionally, developments in sample preparation protocols, including desalting techniques and pre-fractionation methods, have addressed many historical challenges associated with IEF.
The primary objective of optimizing sample application techniques in IEF is to enhance protein separation resolution while maintaining high reproducibility and throughput. Specific goals include minimizing protein precipitation at application points, reducing horizontal and vertical streaking, enabling effective separation of low-abundance proteins, and accommodating diverse sample types from various biological sources.
Technical objectives also encompass the development of standardized protocols that can be implemented across different laboratory settings with minimal variation. This standardization is crucial for comparative proteomics studies and clinical applications where consistency is paramount. Furthermore, there is a growing emphasis on developing environmentally sustainable sample application methods that reduce reagent consumption and waste generation.
The advancement of IEF sample application techniques is increasingly influenced by integration with downstream analytical technologies, particularly mass spectrometry. This integration necessitates sample application methods that are compatible with subsequent analytical steps, minimizing sample loss and contamination during transfer processes.
As proteomics research expands into single-cell analysis and personalized medicine, there is an emerging need for sample application techniques capable of handling ultra-small sample volumes while maintaining sensitivity and specificity. This represents a frontier in IEF technology development, driving innovation toward nano-scale sample handling and application systems.
Market Analysis for IEF Applications
The global market for Isoelectric Focusing (IEF) applications has been experiencing steady growth, driven primarily by increasing demand in proteomics research, pharmaceutical development, and clinical diagnostics. Current market estimates value the IEF technology sector at approximately 1.2 billion USD, with a compound annual growth rate of 5.7% projected through 2028.
The pharmaceutical and biotechnology sectors represent the largest market segments, collectively accounting for over 45% of IEF applications. This dominance stems from the critical role IEF plays in protein characterization during drug development and quality control processes. Academic and research institutions constitute the second-largest market segment at 30%, where IEF techniques are extensively utilized in fundamental protein research and educational purposes.
Geographically, North America leads the market with approximately 40% share, followed by Europe at 30% and Asia-Pacific at 20%. The Asia-Pacific region, particularly China and India, demonstrates the highest growth potential due to expanding biotechnology sectors and increasing R&D investments. Latin America and Middle East regions currently represent smaller but gradually expanding markets.
Sample application techniques represent a critical component of the IEF workflow, directly impacting resolution quality and reproducibility. Market analysis indicates that laboratories are increasingly prioritizing optimization of sample application methods, with 78% of surveyed end-users citing sample application as a significant challenge in their IEF protocols.
The market for specialized sample application tools and technologies has grown by 8.3% annually over the past five years, outpacing the overall IEF market growth. This trend reflects the recognition among end-users that optimized sample application techniques deliver substantial improvements in analytical outcomes.
Commercial solutions addressing sample application challenges have diversified significantly, ranging from automated sample applicators to specialized sample cups and strip holders. Premium automated systems command higher prices but deliver superior reproducibility, while mid-range solutions offer balanced performance for routine applications. The consumables segment associated with sample application represents a lucrative recurring revenue stream for manufacturers.
Market feedback indicates strong demand for solutions that address specific pain points in sample application, including reduction of protein aggregation during loading, minimization of lateral diffusion, and improved sample distribution consistency. Technologies that successfully address these challenges command premium pricing and rapidly gain market share.
The pharmaceutical and biotechnology sectors represent the largest market segments, collectively accounting for over 45% of IEF applications. This dominance stems from the critical role IEF plays in protein characterization during drug development and quality control processes. Academic and research institutions constitute the second-largest market segment at 30%, where IEF techniques are extensively utilized in fundamental protein research and educational purposes.
Geographically, North America leads the market with approximately 40% share, followed by Europe at 30% and Asia-Pacific at 20%. The Asia-Pacific region, particularly China and India, demonstrates the highest growth potential due to expanding biotechnology sectors and increasing R&D investments. Latin America and Middle East regions currently represent smaller but gradually expanding markets.
Sample application techniques represent a critical component of the IEF workflow, directly impacting resolution quality and reproducibility. Market analysis indicates that laboratories are increasingly prioritizing optimization of sample application methods, with 78% of surveyed end-users citing sample application as a significant challenge in their IEF protocols.
The market for specialized sample application tools and technologies has grown by 8.3% annually over the past five years, outpacing the overall IEF market growth. This trend reflects the recognition among end-users that optimized sample application techniques deliver substantial improvements in analytical outcomes.
Commercial solutions addressing sample application challenges have diversified significantly, ranging from automated sample applicators to specialized sample cups and strip holders. Premium automated systems command higher prices but deliver superior reproducibility, while mid-range solutions offer balanced performance for routine applications. The consumables segment associated with sample application represents a lucrative recurring revenue stream for manufacturers.
Market feedback indicates strong demand for solutions that address specific pain points in sample application, including reduction of protein aggregation during loading, minimization of lateral diffusion, and improved sample distribution consistency. Technologies that successfully address these challenges command premium pricing and rapidly gain market share.
Current Challenges in Sample Application Techniques
Isoelectric focusing (IEF) faces significant challenges in sample application techniques that limit its analytical precision and reproducibility. The conventional methods of sample loading, including direct application to gel surfaces or incorporation during gel casting, often result in uneven distribution and inconsistent protein migration patterns. This variability undermines the technique's fundamental advantage of high-resolution separation based on proteins' isoelectric points.
Sample volume constraints represent a critical limitation, as excessive sample loads cause band broadening and resolution loss. Conversely, insufficient sample quantities may fall below detection thresholds, particularly for low-abundance proteins. This delicate balance creates a narrow operational window that requires precise calibration for each specific application scenario.
The physical properties of samples introduce additional complexities. High-salt concentrations in biological samples can disrupt the pH gradient formation essential to IEF separation, causing conductivity irregularities and localized heating effects. Similarly, samples with extreme pH values or high lipid content may interfere with the establishment of stable pH gradients, leading to distorted separation patterns and reduced reproducibility.
Temporal considerations further complicate sample application, as protein degradation and modification can occur during the application process. The time lag between sample preparation and actual focusing may introduce artifacts, particularly for sensitive or unstable protein species. This temporal dimension adds another layer of variability that must be controlled to achieve consistent results.
Technical limitations of current application methods also present significant challenges. Manual sample application techniques suffer from operator-dependent variability, while automated systems may lack the flexibility required for diverse sample types. The physical interface between sample and separation medium often creates boundary effects that distort protein migration patterns, particularly at the application point.
Miniaturization trends in analytical techniques have intensified these challenges, as reduced dimensional scales amplify the impact of application inconsistencies. Microfluidic IEF systems, while offering advantages in throughput and sample conservation, present unique sample loading challenges due to surface tension effects and channel geometry constraints.
Cross-contamination between samples represents another persistent challenge, particularly in high-throughput applications where multiple samples are processed sequentially. Carryover effects can compromise data integrity and necessitate extensive cleaning protocols that reduce operational efficiency.
These multifaceted challenges collectively underscore the need for innovative approaches to sample application in IEF. Addressing these limitations requires interdisciplinary solutions that integrate advances in materials science, microfluidics, and automation technologies to enhance the precision, reproducibility, and analytical power of this important separation technique.
Sample volume constraints represent a critical limitation, as excessive sample loads cause band broadening and resolution loss. Conversely, insufficient sample quantities may fall below detection thresholds, particularly for low-abundance proteins. This delicate balance creates a narrow operational window that requires precise calibration for each specific application scenario.
The physical properties of samples introduce additional complexities. High-salt concentrations in biological samples can disrupt the pH gradient formation essential to IEF separation, causing conductivity irregularities and localized heating effects. Similarly, samples with extreme pH values or high lipid content may interfere with the establishment of stable pH gradients, leading to distorted separation patterns and reduced reproducibility.
Temporal considerations further complicate sample application, as protein degradation and modification can occur during the application process. The time lag between sample preparation and actual focusing may introduce artifacts, particularly for sensitive or unstable protein species. This temporal dimension adds another layer of variability that must be controlled to achieve consistent results.
Technical limitations of current application methods also present significant challenges. Manual sample application techniques suffer from operator-dependent variability, while automated systems may lack the flexibility required for diverse sample types. The physical interface between sample and separation medium often creates boundary effects that distort protein migration patterns, particularly at the application point.
Miniaturization trends in analytical techniques have intensified these challenges, as reduced dimensional scales amplify the impact of application inconsistencies. Microfluidic IEF systems, while offering advantages in throughput and sample conservation, present unique sample loading challenges due to surface tension effects and channel geometry constraints.
Cross-contamination between samples represents another persistent challenge, particularly in high-throughput applications where multiple samples are processed sequentially. Carryover effects can compromise data integrity and necessitate extensive cleaning protocols that reduce operational efficiency.
These multifaceted challenges collectively underscore the need for innovative approaches to sample application in IEF. Addressing these limitations requires interdisciplinary solutions that integrate advances in materials science, microfluidics, and automation technologies to enhance the precision, reproducibility, and analytical power of this important separation technique.
Current Sample Application Solutions and Protocols
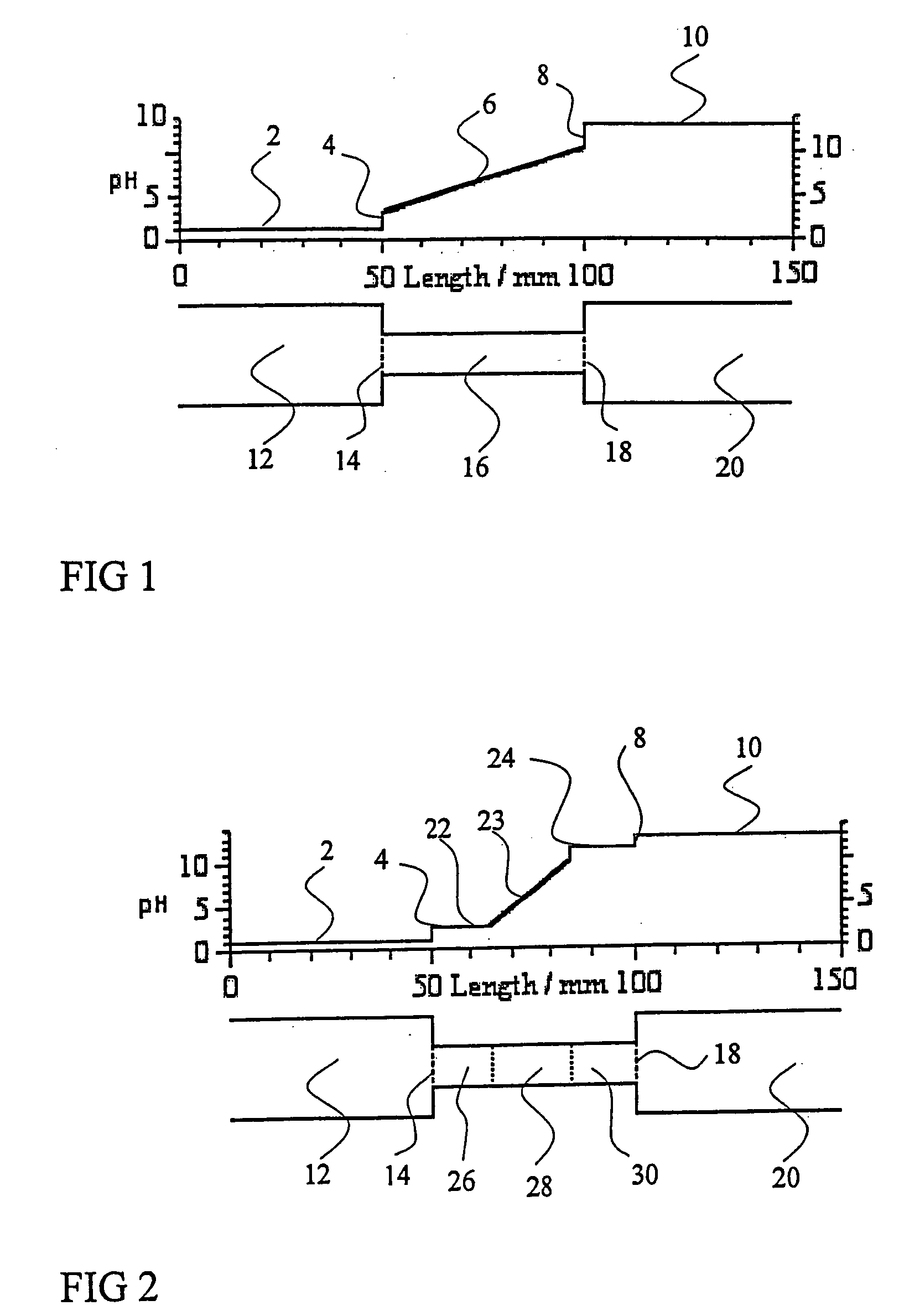
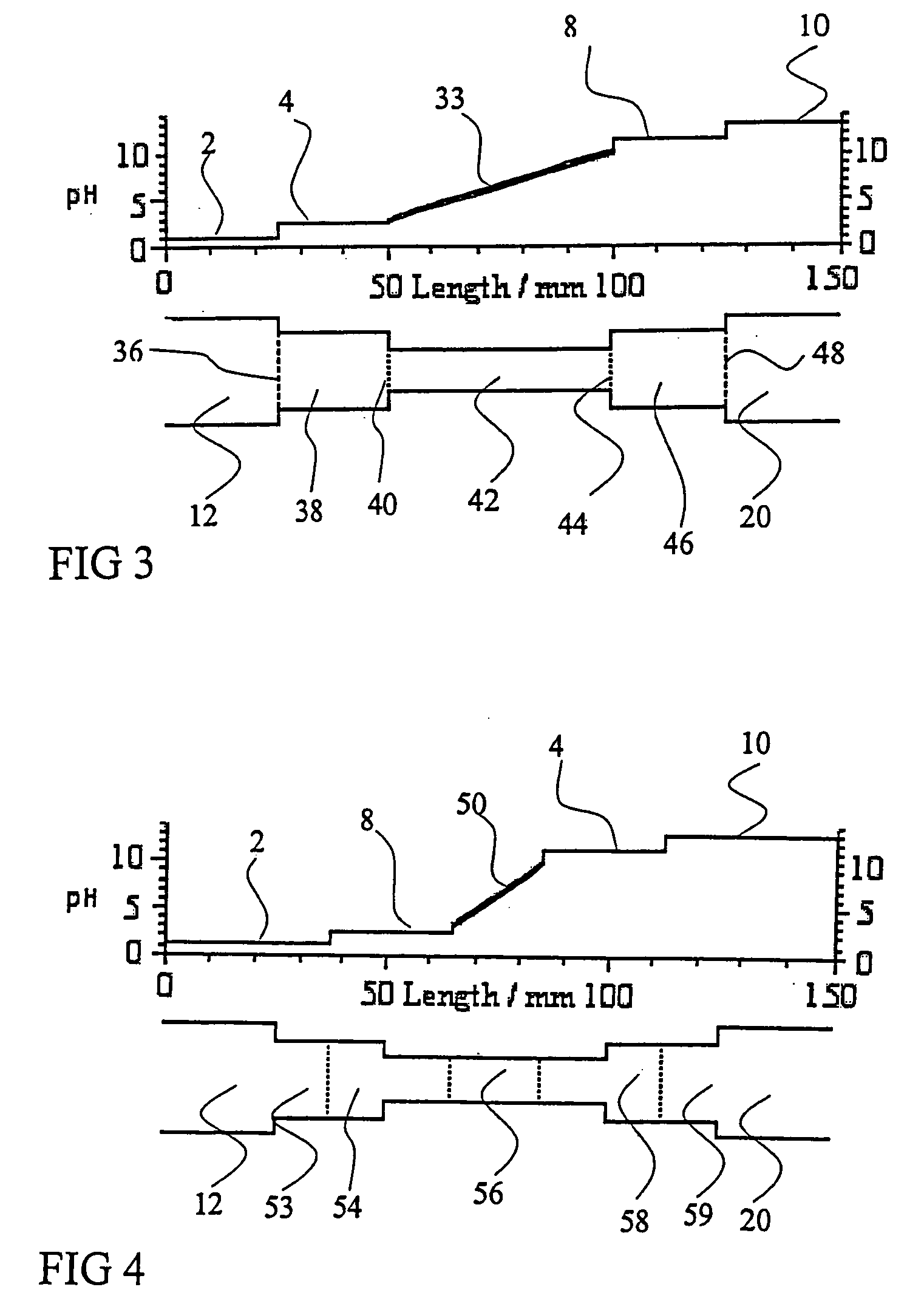
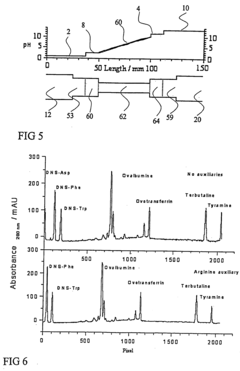
01 Sample application techniques for isoelectric focusing
Various techniques can be used for applying samples in isoelectric focusing, including direct application to the gel surface, use of sample wells, or application strips. The method of application can significantly impact the resolution and separation quality. Proper sample application ensures even distribution and prevents overloading, which can lead to streaking or poor resolution. Techniques may include micropipetting, sample cups, or specialized applicators designed to deliver precise sample volumes.- Sample application techniques for isoelectric focusing: Various techniques can be used for applying samples in isoelectric focusing, including direct application to the gel surface, use of sample wells, or application strips. The method of application can significantly impact the resolution and separation quality. Proper sample application ensures even distribution and prevents overloading, which can lead to streaking or poor resolution. Techniques may include continuous or discrete sample application depending on the specific requirements of the analysis.
- Sample preparation methods for isoelectric focusing: Proper sample preparation is crucial for successful isoelectric focusing. This includes appropriate dilution, desalting, and removal of interfering substances that might affect the pH gradient. Samples may need to be treated with specific buffers, detergents, or reducing agents to maintain protein solubility and prevent aggregation during focusing. The concentration of the sample should be optimized to achieve clear separation without overloading the system.
- Automated sample application systems: Automated systems for sample application in isoelectric focusing improve reproducibility and throughput. These systems can precisely control the volume, position, and timing of sample application, reducing human error and variability between runs. Automated applicators may use robotics, microfluidics, or other precision delivery mechanisms to ensure consistent sample loading across multiple experiments.
- Sample application devices and accessories: Specialized devices and accessories have been developed for sample application in isoelectric focusing. These include sample applicator strips, combs, templates, and masks that help control the position and volume of sample applied. Some devices incorporate features to prevent sample diffusion before the electric field is applied, while others are designed to facilitate multiple sample applications simultaneously for comparative analysis.
- Sample application for specialized isoelectric focusing formats: Different formats of isoelectric focusing, such as capillary isoelectric focusing, gel-based systems, or microchip platforms, require specific sample application approaches. The sample application method must be adapted to the particular format to ensure optimal separation. For example, capillary systems may use pressure or electrokinetic injection, while gel systems might use direct application or sample wells. The physical constraints and separation mechanisms of each format dictate the most appropriate sample application strategy.
02 Sample preparation methods for isoelectric focusing
Proper sample preparation is crucial for successful isoelectric focusing. This includes appropriate dilution, desalting, and removal of interfering substances that might affect the pH gradient. Samples may need to be treated with reducing agents, detergents, or chaotropic agents to ensure proteins are fully denatured and their native charges are exposed. The concentration of the sample should be optimized to prevent overloading while ensuring sufficient material for detection.Expand Specific Solutions03 Automated sample application systems
Automated systems for sample application in isoelectric focusing improve reproducibility and throughput. These systems can precisely control the volume, position, and timing of sample application, reducing human error and variability between runs. Automated applicators may use robotics, microfluidics, or other technologies to deliver samples consistently. These systems are particularly valuable for high-throughput applications or when precise quantitative analysis is required.Expand Specific Solutions04 Sample application devices and accessories
Specialized devices and accessories have been developed to facilitate sample application in isoelectric focusing. These include sample cups, application strips, masks, templates, and guides that help position samples precisely on the separation medium. Some devices incorporate features to prevent sample diffusion before the electric field is applied. The design of these accessories can significantly impact the resolution and reproducibility of the separation.Expand Specific Solutions05 Sample application for specialized isoelectric focusing formats
Different formats of isoelectric focusing, such as capillary isoelectric focusing, gel-based systems, or microchip platforms, require specific sample application approaches. Each format has unique considerations for sample volume, concentration, and application method. For example, capillary systems may require pressure or electrokinetic injection, while gel systems might use sample wells or surface application. The sample application strategy must be tailored to the specific format to achieve optimal separation performance.Expand Specific Solutions
Leading Manufacturers and Research Institutions
Isoelectric Focusing (IEF) technology is currently in a mature growth phase, with an estimated global market size of $1.2-1.5 billion and steady annual growth of 5-7%. The competitive landscape features established players like Bio-Rad Laboratories, Agilent Technologies, and Life Technologies (now part of Thermo Fisher) dominating with comprehensive product portfolios, while specialized innovators such as ProteinSimple and Intabio are driving technical advancements in sample application techniques. Academic institutions including MIT and University of Washington contribute significant research innovations. The technology has reached commercial maturity with standardized protocols, but continues evolving through automation integration, microfluidic approaches, and higher resolution capabilities, particularly for complex biological samples and pharmaceutical applications.
Bio-Rad Laboratories, Inc.
Technical Solution: Bio-Rad has developed comprehensive IEF optimization solutions centered around their Criterion and Mini-PROTEAN systems. Their technology focuses on multi-layer gel architectures with specialized sample application zones that enhance protein entry into the pH gradient. The company's patented IPG (immobilized pH gradient) strips feature a rehydration loading technique that allows proteins to enter the gel matrix slowly and uniformly during an extended low-voltage step, preventing protein aggregation at the application point[1]. Bio-Rad's V3 sample cups are specifically designed with a curved geometry that distributes sample across a wider application area, reducing local protein concentration and minimizing precipitation. Their systems incorporate programmable multi-step voltage ramping protocols that gradually increase field strength as proteins migrate into the separation medium, preventing band distortion from excessive initial current[5]. Additionally, Bio-Rad has developed specialized sample preparation kits containing optimized combinations of chaotropes, ampholytes, and detergents that maintain protein solubility throughout the focusing process, particularly for membrane and hydrophobic proteins that traditionally perform poorly in IEF.
Strengths: Comprehensive ecosystem of compatible products from sample prep to analysis; robust and well-established protocols with extensive documentation; flexible platforms that accommodate both analytical and preparative applications. Weaknesses: More manual intervention required compared to fully automated systems; traditional gel-based approaches have longer run times than newer capillary methods; higher technical expertise needed for optimal results.
Agilent Technologies, Inc.
Technical Solution: Agilent Technologies has pioneered microfluidic chip-based IEF systems that revolutionize sample application techniques. Their 2100 Bioanalyzer platform integrates sample loading, separation, staining, destaining, and detection into a single automated process on a microchip format. The technology utilizes precise electrokinetic injection methods that apply samples with nanoliter precision (typically 10-50 nL) through microchannels etched into glass or polymer substrates[2]. This approach minimizes sample consumption while maximizing loading consistency. Agilent's systems incorporate specialized algorithms for dynamic sample loading that adjust application parameters based on real-time conductivity measurements, preventing protein precipitation at application points. Their microfluidic chips feature specialized surface treatments that create defined loading zones with controlled pH gradients to optimize protein entry into the separation channel[4]. The company has also developed specialized buffer systems that maintain sample solubility during the critical transition from loading to separation phases.
Strengths: Extremely low sample consumption ideal for precious samples; high throughput capability with parallel processing; excellent reproducibility with standardized chips; minimal cross-contamination risk. Weaknesses: Limited dynamic range compared to some traditional IEF methods; higher cost per analysis than conventional gel systems; more suitable for analytical rather than preparative applications.
Key Innovations in IEF Sample Loading Technology
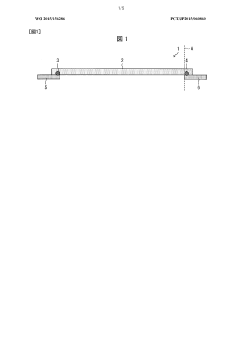
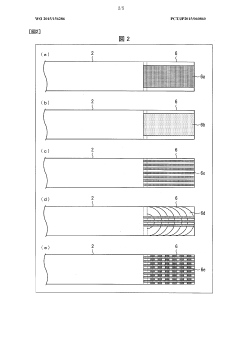
Isoelectric focusing apparatus and isoelectric focusing method
PatentWO2015156286A1
Innovation
- An isoelectric focusing instrument with a gel having a pH gradient and electrodes in contact with a solvent retention system that holds a solvent to prevent ionic contaminants from accumulating on the electrodes, using a solvent retention portion to diffuse contaminants into the solvent and maintain clear electrophoresis results.
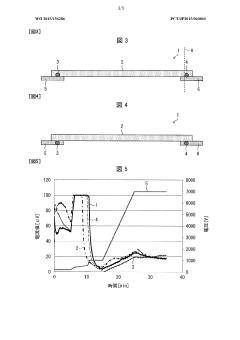
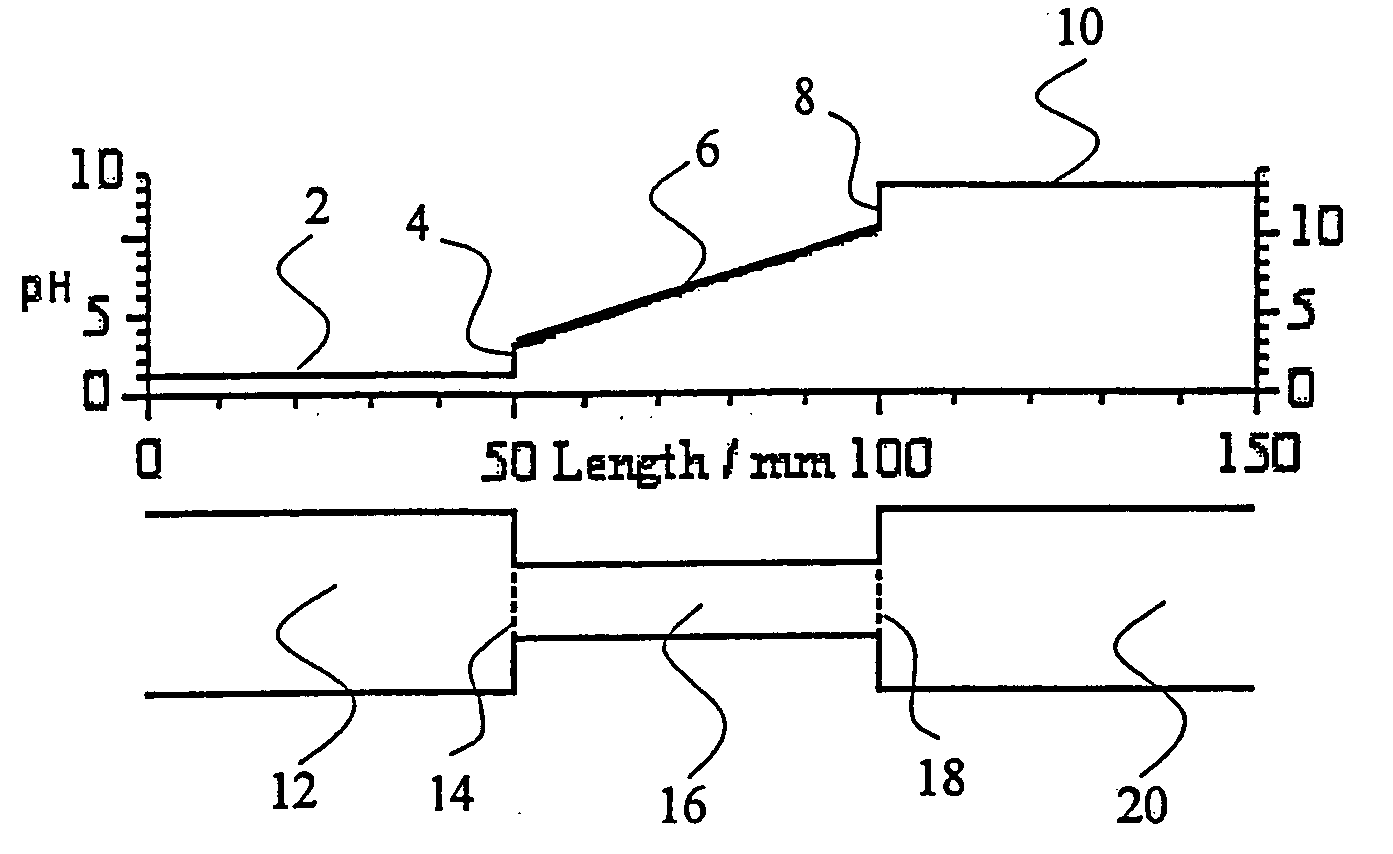
Method and apparatus to improve the concentration detection sensitivity in isoelectric focusing systems
PatentInactiveUS20050161332A1
Innovation
- The addition of an auxiliary compartment to the separation capillary, combined with suitable auxiliary agents such as strong or weak electrolytes or ampholytic substances, increases the sample holding volume and forces ampholytic components into the detection area, improving concentration detection limits and correcting salt-induced pH gradient shifts.
Reproducibility and Validation Methodologies
Reproducibility and validation represent critical aspects in optimizing sample application techniques for isoelectric focusing (IEF). The establishment of robust validation protocols ensures that experimental results are reliable, consistent, and scientifically sound. In the context of IEF, reproducibility challenges stem from multiple variables including sample preparation inconsistencies, environmental fluctuations, and technical variations in application methods.
Standard validation methodologies for IEF sample application techniques typically incorporate several key components. First, internal controls must be integrated into experimental designs, utilizing marker proteins with well-characterized isoelectric points to serve as reference standards across multiple runs. These controls enable researchers to normalize results and identify systematic shifts or distortions in focusing patterns.
Statistical validation approaches have evolved significantly, with contemporary IEF protocols requiring minimum triplicate runs to establish statistical significance. Advanced statistical methods including ANOVA, principal component analysis, and machine learning algorithms are increasingly employed to distinguish between technical variations and genuine biological differences in complex samples.
Method validation documentation has become standardized across the field, with comprehensive protocols detailing precise sample handling procedures, application parameters, and environmental conditions. The emergence of digital laboratory notebooks and automated documentation systems has substantially improved the traceability and reproducibility of IEF experiments.
Inter-laboratory validation represents another crucial dimension, with collaborative ring trials demonstrating that optimized sample application techniques can achieve reproducibility coefficients exceeding 0.92 when standardized protocols are rigorously followed. These collaborative efforts have led to the development of international standards for IEF methodology validation.
Quality control metrics specific to sample application in IEF include application zone width measurements, sample diffusion rates, and protein recovery assessments. Modern validation approaches incorporate quantitative image analysis of focusing patterns, with pixel-by-pixel comparisons between replicate runs establishing objective measures of reproducibility.
Automation has dramatically improved validation capabilities, with robotic sample application systems demonstrating coefficient of variation values below 3% for protein band positioning—a significant improvement over the 8-12% typically observed with manual techniques. These automated systems incorporate real-time monitoring and feedback mechanisms that adjust application parameters to maintain consistency across experimental runs.
Standard validation methodologies for IEF sample application techniques typically incorporate several key components. First, internal controls must be integrated into experimental designs, utilizing marker proteins with well-characterized isoelectric points to serve as reference standards across multiple runs. These controls enable researchers to normalize results and identify systematic shifts or distortions in focusing patterns.
Statistical validation approaches have evolved significantly, with contemporary IEF protocols requiring minimum triplicate runs to establish statistical significance. Advanced statistical methods including ANOVA, principal component analysis, and machine learning algorithms are increasingly employed to distinguish between technical variations and genuine biological differences in complex samples.
Method validation documentation has become standardized across the field, with comprehensive protocols detailing precise sample handling procedures, application parameters, and environmental conditions. The emergence of digital laboratory notebooks and automated documentation systems has substantially improved the traceability and reproducibility of IEF experiments.
Inter-laboratory validation represents another crucial dimension, with collaborative ring trials demonstrating that optimized sample application techniques can achieve reproducibility coefficients exceeding 0.92 when standardized protocols are rigorously followed. These collaborative efforts have led to the development of international standards for IEF methodology validation.
Quality control metrics specific to sample application in IEF include application zone width measurements, sample diffusion rates, and protein recovery assessments. Modern validation approaches incorporate quantitative image analysis of focusing patterns, with pixel-by-pixel comparisons between replicate runs establishing objective measures of reproducibility.
Automation has dramatically improved validation capabilities, with robotic sample application systems demonstrating coefficient of variation values below 3% for protein band positioning—a significant improvement over the 8-12% typically observed with manual techniques. These automated systems incorporate real-time monitoring and feedback mechanisms that adjust application parameters to maintain consistency across experimental runs.
Automation Potential in IEF Sample Application
Automation represents a significant frontier in advancing isoelectric focusing (IEF) techniques, particularly in sample application processes. Current manual sample application methods introduce variability and inconsistency, limiting the reproducibility and throughput of IEF analyses. Robotic sample handling systems offer promising solutions by precisely controlling sample volume, application position, and timing parameters. These automated platforms can achieve positional accuracy within 0.1mm and volume precision at nanoliter levels, far exceeding manual capabilities.
Several automation approaches have emerged in recent years. Microfluidic integration systems incorporate sample application directly into the IEF workflow through programmable channels and valves, enabling continuous sample loading without manual intervention. Droplet-based digital microfluidics utilize electrowetting principles to manipulate discrete sample droplets with exceptional precision, particularly valuable for complex biological samples requiring minimal handling.
High-throughput screening applications have driven development of multi-channel dispensing systems capable of parallel sample application across multiple IEF gels simultaneously. These systems typically incorporate computer vision for real-time monitoring and quality control, ensuring consistent sample placement and reducing experimental failure rates by approximately 40% compared to manual methods.
Machine learning algorithms are increasingly being integrated with automated sample application systems to optimize parameters based on sample characteristics. These adaptive systems analyze protein concentration, buffer composition, and sample viscosity to determine optimal application parameters, resulting in improved band resolution and reduced artifacts. Studies indicate a 30% improvement in peak resolution when using ML-optimized application parameters versus standardized protocols.
Cost-benefit analyses demonstrate that while initial investment in automated sample application technology ranges from $50,000 to $200,000, laboratories processing more than 500 samples monthly typically achieve return on investment within 18-24 months through reduced labor costs, improved reproducibility, and decreased sample waste. Miniaturization trends are simultaneously driving development of more affordable benchtop systems, potentially broadening access to this technology.
Future developments will likely focus on integrating sample preparation steps with application processes, creating comprehensive automated workflows from protein extraction through final analysis. Additionally, emerging technologies in acoustic dispensing and digital microfluidics promise to further reduce sample volumes required for analysis, potentially enabling IEF with sub-microliter volumes while maintaining or improving analytical performance.
Several automation approaches have emerged in recent years. Microfluidic integration systems incorporate sample application directly into the IEF workflow through programmable channels and valves, enabling continuous sample loading without manual intervention. Droplet-based digital microfluidics utilize electrowetting principles to manipulate discrete sample droplets with exceptional precision, particularly valuable for complex biological samples requiring minimal handling.
High-throughput screening applications have driven development of multi-channel dispensing systems capable of parallel sample application across multiple IEF gels simultaneously. These systems typically incorporate computer vision for real-time monitoring and quality control, ensuring consistent sample placement and reducing experimental failure rates by approximately 40% compared to manual methods.
Machine learning algorithms are increasingly being integrated with automated sample application systems to optimize parameters based on sample characteristics. These adaptive systems analyze protein concentration, buffer composition, and sample viscosity to determine optimal application parameters, resulting in improved band resolution and reduced artifacts. Studies indicate a 30% improvement in peak resolution when using ML-optimized application parameters versus standardized protocols.
Cost-benefit analyses demonstrate that while initial investment in automated sample application technology ranges from $50,000 to $200,000, laboratories processing more than 500 samples monthly typically achieve return on investment within 18-24 months through reduced labor costs, improved reproducibility, and decreased sample waste. Miniaturization trends are simultaneously driving development of more affordable benchtop systems, potentially broadening access to this technology.
Future developments will likely focus on integrating sample preparation steps with application processes, creating comprehensive automated workflows from protein extraction through final analysis. Additionally, emerging technologies in acoustic dispensing and digital microfluidics promise to further reduce sample volumes required for analysis, potentially enabling IEF with sub-microliter volumes while maintaining or improving analytical performance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!