Exploit Protonation Dynamics in Isoelectric Focusing Scalability
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Protonation Dynamics in IEF: Background and Objectives
Isoelectric focusing (IEF) represents one of the most powerful analytical techniques in protein separation science, with its development tracing back to the 1960s when Svensson and Vesterberg pioneered the concept of creating pH gradients for protein separation based on their isoelectric points. The fundamental principle leverages the amphoteric nature of proteins, which carry different net charges depending on the surrounding pH environment, ultimately reaching a neutral state at their specific isoelectric point (pI).
The evolution of IEF technology has progressed through several significant phases, from carrier ampholyte-based systems to immobilized pH gradient (IPG) gels, which dramatically improved reproducibility and resolution. Recent advancements have focused on miniaturization and integration with other analytical platforms, particularly mass spectrometry, enhancing the technique's analytical power and application scope.
Protonation dynamics—the process by which proteins gain or lose protons in response to environmental pH changes—forms the theoretical cornerstone of IEF. This phenomenon involves complex interactions between amino acid side chains, solvent molecules, and buffer components, creating a multifaceted system that determines separation efficiency and resolution. Understanding these dynamics at a molecular level has become increasingly crucial as applications demand higher resolution and throughput.
The scalability of IEF represents a significant technical challenge that has limited its broader industrial application. While laboratory-scale IEF delivers exceptional resolution, scaling to preparative or industrial levels introduces complications in heat dissipation, pH gradient stability, and protein precipitation. These challenges directly relate to protonation dynamics, as scaling affects the kinetics and equilibrium of protonation-deprotonation events.
This technical research aims to explore innovative approaches to exploit protonation dynamics specifically to overcome scalability limitations in IEF. The primary objectives include: developing mathematical models that accurately predict protonation behavior in scaled-up systems; investigating novel buffer systems that maintain stable pH gradients under high-voltage, high-throughput conditions; and designing electrode configurations that minimize disruptive electrochemical reactions while maximizing separation efficiency.
Additionally, we seek to understand how protein concentration affects protonation kinetics in crowded environments typical of industrial-scale separations, and to develop strategies for controlling Joule heating—a major limiting factor in IEF scalability—through manipulation of ionic strength and conductivity parameters that directly influence protonation dynamics.
By addressing these objectives, we aim to bridge the gap between analytical-scale IEF and industrial applications, potentially revolutionizing protein purification processes in biopharmaceutical manufacturing, enzyme production, and other biotechnology sectors where high-resolution, high-throughput protein separation represents a critical bottleneck.
The evolution of IEF technology has progressed through several significant phases, from carrier ampholyte-based systems to immobilized pH gradient (IPG) gels, which dramatically improved reproducibility and resolution. Recent advancements have focused on miniaturization and integration with other analytical platforms, particularly mass spectrometry, enhancing the technique's analytical power and application scope.
Protonation dynamics—the process by which proteins gain or lose protons in response to environmental pH changes—forms the theoretical cornerstone of IEF. This phenomenon involves complex interactions between amino acid side chains, solvent molecules, and buffer components, creating a multifaceted system that determines separation efficiency and resolution. Understanding these dynamics at a molecular level has become increasingly crucial as applications demand higher resolution and throughput.
The scalability of IEF represents a significant technical challenge that has limited its broader industrial application. While laboratory-scale IEF delivers exceptional resolution, scaling to preparative or industrial levels introduces complications in heat dissipation, pH gradient stability, and protein precipitation. These challenges directly relate to protonation dynamics, as scaling affects the kinetics and equilibrium of protonation-deprotonation events.
This technical research aims to explore innovative approaches to exploit protonation dynamics specifically to overcome scalability limitations in IEF. The primary objectives include: developing mathematical models that accurately predict protonation behavior in scaled-up systems; investigating novel buffer systems that maintain stable pH gradients under high-voltage, high-throughput conditions; and designing electrode configurations that minimize disruptive electrochemical reactions while maximizing separation efficiency.
Additionally, we seek to understand how protein concentration affects protonation kinetics in crowded environments typical of industrial-scale separations, and to develop strategies for controlling Joule heating—a major limiting factor in IEF scalability—through manipulation of ionic strength and conductivity parameters that directly influence protonation dynamics.
By addressing these objectives, we aim to bridge the gap between analytical-scale IEF and industrial applications, potentially revolutionizing protein purification processes in biopharmaceutical manufacturing, enzyme production, and other biotechnology sectors where high-resolution, high-throughput protein separation represents a critical bottleneck.
Market Analysis for Scalable IEF Technologies
The global market for Isoelectric Focusing (IEF) technologies is experiencing significant growth, driven by expanding applications in proteomics, pharmaceutical development, and biomarker discovery. Current market valuations place the analytical separation technologies sector at approximately $4.9 billion, with IEF technologies representing a growing segment estimated at $650 million in 2023, projected to reach $1.2 billion by 2028 at a CAGR of 13.5%.
The demand for scalable IEF technologies is particularly strong in biopharmaceutical manufacturing, where the need for efficient protein separation and purification continues to increase with the rise of biologics and biosimilars. The biologics market, valued at $366 billion in 2022, is expected to grow at 12% annually, creating substantial pull for advanced separation technologies that can operate at industrial scales.
Regional analysis reveals North America dominates the market with approximately 42% share, followed by Europe (28%) and Asia-Pacific (22%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth rate at 16.7% annually, attributed to expanding biopharmaceutical manufacturing capabilities and increasing R&D investments.
Key market segments for scalable IEF technologies include academic research (31%), pharmaceutical and biotechnology companies (45%), clinical diagnostics (18%), and other industrial applications (6%). The pharmaceutical segment shows the strongest growth potential due to increasing adoption of precision medicine approaches and personalized therapeutics.
Customer needs analysis indicates growing demand for IEF systems that can maintain resolution while scaling up, with particular emphasis on reproducibility, automation compatibility, and reduced sample preparation requirements. Survey data from end-users highlights that 78% of respondents identify scalability limitations as a significant barrier to broader IEF adoption in industrial settings.
Market barriers include high initial investment costs, technical complexity requiring specialized expertise, and competition from alternative separation technologies. However, the unique advantages of IEF in achieving high-resolution separation of amphoteric molecules position it favorably against competing technologies like chromatography for specific applications.
Emerging market opportunities exist in continuous bioprocessing, where scalable IEF could enable real-time monitoring and separation, potentially reducing manufacturing costs by 25-30%. Additionally, the growing field of proteomics research, expanding at 14% annually, represents a significant opportunity for advanced IEF technologies that can exploit protonation dynamics for improved resolution and throughput.
The demand for scalable IEF technologies is particularly strong in biopharmaceutical manufacturing, where the need for efficient protein separation and purification continues to increase with the rise of biologics and biosimilars. The biologics market, valued at $366 billion in 2022, is expected to grow at 12% annually, creating substantial pull for advanced separation technologies that can operate at industrial scales.
Regional analysis reveals North America dominates the market with approximately 42% share, followed by Europe (28%) and Asia-Pacific (22%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth rate at 16.7% annually, attributed to expanding biopharmaceutical manufacturing capabilities and increasing R&D investments.
Key market segments for scalable IEF technologies include academic research (31%), pharmaceutical and biotechnology companies (45%), clinical diagnostics (18%), and other industrial applications (6%). The pharmaceutical segment shows the strongest growth potential due to increasing adoption of precision medicine approaches and personalized therapeutics.
Customer needs analysis indicates growing demand for IEF systems that can maintain resolution while scaling up, with particular emphasis on reproducibility, automation compatibility, and reduced sample preparation requirements. Survey data from end-users highlights that 78% of respondents identify scalability limitations as a significant barrier to broader IEF adoption in industrial settings.
Market barriers include high initial investment costs, technical complexity requiring specialized expertise, and competition from alternative separation technologies. However, the unique advantages of IEF in achieving high-resolution separation of amphoteric molecules position it favorably against competing technologies like chromatography for specific applications.
Emerging market opportunities exist in continuous bioprocessing, where scalable IEF could enable real-time monitoring and separation, potentially reducing manufacturing costs by 25-30%. Additionally, the growing field of proteomics research, expanding at 14% annually, represents a significant opportunity for advanced IEF technologies that can exploit protonation dynamics for improved resolution and throughput.
Current Limitations in IEF Scalability
Despite significant advancements in isoelectric focusing (IEF) technology, several critical limitations continue to impede its scalability for industrial applications. The fundamental challenge lies in maintaining stable pH gradients during extended separation processes, particularly when scaling up from analytical to preparative dimensions. As separation chambers increase in size, heat dissipation becomes increasingly problematic, leading to temperature gradients that distort the pH environment and compromise resolution.
Carrier ampholyte-based systems, while effective at laboratory scale, demonstrate significant drawbacks in larger formats. The limited buffering capacity of conventional carrier ampholytes results in pH drift over time, especially when processing high-protein loads typical in industrial applications. This instability directly impacts reproducibility and separation efficiency, creating a significant barrier to consistent large-scale operations.
Electrode reactions present another substantial challenge, generating proton and hydroxide ions that progressively disrupt established pH gradients. In scaled-up systems, these electrochemical effects become magnified, causing more pronounced gradient decay. Current mitigation strategies, such as membrane barriers and specialized electrode solutions, add complexity and cost while providing only partial solutions to this fundamental issue.
Power management represents a critical limitation in scaled IEF systems. Higher voltages required for larger separation distances increase Joule heating effects exponentially. Contemporary cooling systems struggle to maintain uniform temperature profiles across expanded separation chambers, resulting in thermal gradients that directly impact protein mobility and focusing behavior through altered protonation dynamics.
Sample loading capacity constraints further limit industrial applications. As separation chambers increase in size, the relationship between sample volume, protein concentration, and focusing efficiency becomes increasingly non-linear. Protein-protein interactions and localized concentration effects create zones of altered conductivity and pH, disrupting the theoretical separation model that functions effectively at analytical scale.
Instrumentation limitations also present significant barriers to scalability. Current detection and monitoring systems lack the spatial resolution and response time necessary to track dynamic pH changes across large-format separations. This technological gap prevents real-time process optimization and quality control essential for industrial implementation.
The economic considerations of scaled IEF systems present additional challenges. The exponential increase in carrier ampholyte consumption with increased separation volume creates prohibitive operational costs. Furthermore, the extended processing times required for large-scale separations reduce throughput and increase exposure to gradient instability, directly impacting production economics.
Carrier ampholyte-based systems, while effective at laboratory scale, demonstrate significant drawbacks in larger formats. The limited buffering capacity of conventional carrier ampholytes results in pH drift over time, especially when processing high-protein loads typical in industrial applications. This instability directly impacts reproducibility and separation efficiency, creating a significant barrier to consistent large-scale operations.
Electrode reactions present another substantial challenge, generating proton and hydroxide ions that progressively disrupt established pH gradients. In scaled-up systems, these electrochemical effects become magnified, causing more pronounced gradient decay. Current mitigation strategies, such as membrane barriers and specialized electrode solutions, add complexity and cost while providing only partial solutions to this fundamental issue.
Power management represents a critical limitation in scaled IEF systems. Higher voltages required for larger separation distances increase Joule heating effects exponentially. Contemporary cooling systems struggle to maintain uniform temperature profiles across expanded separation chambers, resulting in thermal gradients that directly impact protein mobility and focusing behavior through altered protonation dynamics.
Sample loading capacity constraints further limit industrial applications. As separation chambers increase in size, the relationship between sample volume, protein concentration, and focusing efficiency becomes increasingly non-linear. Protein-protein interactions and localized concentration effects create zones of altered conductivity and pH, disrupting the theoretical separation model that functions effectively at analytical scale.
Instrumentation limitations also present significant barriers to scalability. Current detection and monitoring systems lack the spatial resolution and response time necessary to track dynamic pH changes across large-format separations. This technological gap prevents real-time process optimization and quality control essential for industrial implementation.
The economic considerations of scaled IEF systems present additional challenges. The exponential increase in carrier ampholyte consumption with increased separation volume creates prohibitive operational costs. Furthermore, the extended processing times required for large-scale separations reduce throughput and increase exposure to gradient instability, directly impacting production economics.
Current Approaches to Exploit Protonation Dynamics
01 Principles and mechanisms of isoelectric focusing
Isoelectric focusing (IEF) is a technique that separates proteins based on their isoelectric points (pI). The process involves creating a pH gradient in a gel or solution, where proteins migrate until they reach the pH that matches their pI, at which point they become neutrally charged and stop moving. The protonation dynamics in this process are critical as proteins gain or lose protons depending on the surrounding pH, affecting their net charge and mobility in the electric field.- Principles of isoelectric focusing and pH gradients: Isoelectric focusing (IEF) is an electrophoretic technique that separates proteins based on their isoelectric points (pI) in a pH gradient. When proteins are placed in a pH gradient under an electric field, they migrate until reaching a position where their net charge is zero (their pI). The protonation dynamics of amino acid residues determine the protein's charge state at different pH values, affecting migration behavior. Stable pH gradients are crucial for effective separation and can be established using carrier ampholytes or immobilized pH gradient (IPG) strips.
- Advanced detection and monitoring of protonation states: Modern IEF systems incorporate advanced detection methods to monitor protonation dynamics during separation. These include real-time pH measurements, fluorescence detection, and conductivity monitoring that can track the changing protonation states of molecules as they migrate through pH gradients. These detection systems allow for precise determination of isoelectric points and provide insights into the conformational changes that occur as proteins gain or lose protons in response to their microenvironment.
- Microfluidic and miniaturized IEF systems: Miniaturized and microfluidic IEF platforms enable analysis of protonation dynamics with minimal sample volumes and enhanced resolution. These systems utilize microfabricated channels or chambers where pH gradients are established, allowing for rapid equilibration and separation. The confined environment in these platforms provides better control over local pH conditions and electric fields, resulting in improved observation of protonation/deprotonation events during protein focusing. Some systems incorporate integrated sensors for real-time monitoring of the separation process.
- Computational modeling of protonation dynamics: Computational approaches are increasingly used to model and predict protonation dynamics in isoelectric focusing. These models simulate the behavior of amphoteric molecules in pH gradients by calculating protonation/deprotonation rates of ionizable groups under varying conditions. Machine learning algorithms can analyze experimental IEF data to predict protein migration patterns and isoelectric points. These computational tools help optimize separation conditions and provide insights into the molecular mechanisms underlying the focusing process.
- Novel carrier ampholytes and buffer systems: Innovative carrier ampholytes and buffer systems have been developed to enhance control over protonation dynamics during IEF. These include synthetic ampholytes with precisely defined pKa values, temperature-responsive buffer components, and additives that stabilize pH gradients against cathodic drift. Some formulations incorporate molecules that can modulate local protonation environments or prevent protein aggregation at their isoelectric points. These advances allow for more precise manipulation of the separation environment and improved resolution of closely spaced protein species.
02 Advanced pH gradient formation techniques
Various methods have been developed to create stable and reproducible pH gradients for isoelectric focusing. These include carrier ampholytes, immobilized pH gradients (IPG), and specialized buffer systems. The dynamics of protonation and deprotonation of these gradient-forming components are essential for maintaining a stable pH environment throughout the separation process, allowing for precise protein focusing and improved resolution.Expand Specific Solutions03 Instrumentation and detection systems for monitoring protonation states
Advanced instrumentation has been developed to monitor and analyze the protonation dynamics during isoelectric focusing. These systems include specialized electrodes, optical sensors, and imaging technologies that can detect changes in protein charge states, pH fluctuations, and focusing patterns in real-time. Such monitoring capabilities allow for better control of the separation process and provide insights into the behavior of proteins under different conditions.Expand Specific Solutions04 Applications in protein characterization and analysis
Isoelectric focusing with controlled protonation dynamics has numerous applications in protein characterization and analysis. It can be used to determine protein isoelectric points, analyze post-translational modifications, identify protein variants, and separate complex protein mixtures. The technique is particularly valuable in proteomics, biomarker discovery, and quality control of biopharmaceuticals, where understanding protein charge properties is crucial.Expand Specific Solutions05 Integration with other analytical techniques
Isoelectric focusing is often integrated with other analytical techniques to enhance separation power and information content. Two-dimensional electrophoresis combines IEF with SDS-PAGE, while coupling with mass spectrometry allows for detailed structural analysis. Microfluidic platforms enable miniaturized IEF with precise control over protonation dynamics. These integrated approaches provide comprehensive characterization of proteins by leveraging the charge-based separation of IEF with complementary analytical methods.Expand Specific Solutions
Leading Organizations in IEF Technology Development
The isoelectric focusing (IEF) scalability landscape is currently in a growth phase, with the market expanding as analytical proteomics applications increase across pharmaceutical and biotechnology sectors. The technology is approaching maturity with established players like Bio-Rad Laboratories and Thermo Fisher Scientific leading commercial development, while academic institutions such as Technion Research Foundation and South China University of Technology drive fundamental research innovations. Life Technologies and Becton Dickinson contribute significant advancements in instrumentation and consumables. The market is characterized by a blend of specialized biotechnology firms (ProteoSys AG, ReLign) and larger diversified corporations (Koninklijke Philips), with competition focused on improving resolution, throughput, and automation capabilities for complex protein separation applications.
Life Technologies Corp.
Technical Solution: Life Technologies has developed the PureProteome™ IEF system that addresses scalability challenges in isoelectric focusing through innovative management of protonation dynamics. Their technology employs a combination of specialized immobilized pH gradient (IPG) strips with enhanced buffer capacity and proprietary electrode designs that minimize pH drift during extended separations. The system features adaptive power control algorithms that respond to conductivity changes in real-time, preventing localized overheating that can disrupt protonation equilibria. Life Technologies has also pioneered multi-dimensional separation workflows that integrate their IEF technology with orthogonal separation methods, enabling processing of complex samples at preparative scales. Their approach incorporates specialized cooling systems that maintain uniform temperature distribution across the separation medium, ensuring consistent protonation/deprotonation kinetics even with increased sample loads[5][6].
Strengths: Excellent protein recovery rates even at preparative scales; high compatibility with downstream mass spectrometry applications; user-friendly interface requiring minimal specialized training. Weaknesses: Limited flexibility for customizing separation parameters; higher consumable costs compared to traditional methods; requires significant bench space for the complete system setup.
Bio-Rad Laboratories, Inc.
Technical Solution: Bio-Rad has developed advanced isoelectric focusing (IEF) systems that address scalability challenges through innovative protonation dynamics management. Their technology utilizes specialized ampholyte mixtures with optimized buffering capacities to maintain stable pH gradients during extended separations. Bio-Rad's approach incorporates proprietary electrode designs that minimize electrochemical reactions interfering with protein focusing while enabling higher sample loads. Their PROTEAN i12 IEF system employs computer-controlled voltage gradients that adapt to changing conductivity conditions during focusing, preventing protein precipitation at isoelectric points. Additionally, Bio-Rad has pioneered temperature control systems that counteract Joule heating effects, allowing for consistent protonation/deprotonation kinetics across larger separation distances[1][3].
Strengths: Superior resolution of complex protein mixtures with high reproducibility; scalable to both analytical and preparative applications; compatible with downstream mass spectrometry analysis. Weaknesses: Higher cost compared to conventional electrophoresis systems; requires specialized training for optimal operation; limited throughput for very high-volume applications.
Key Patents and Research in IEF Scalability
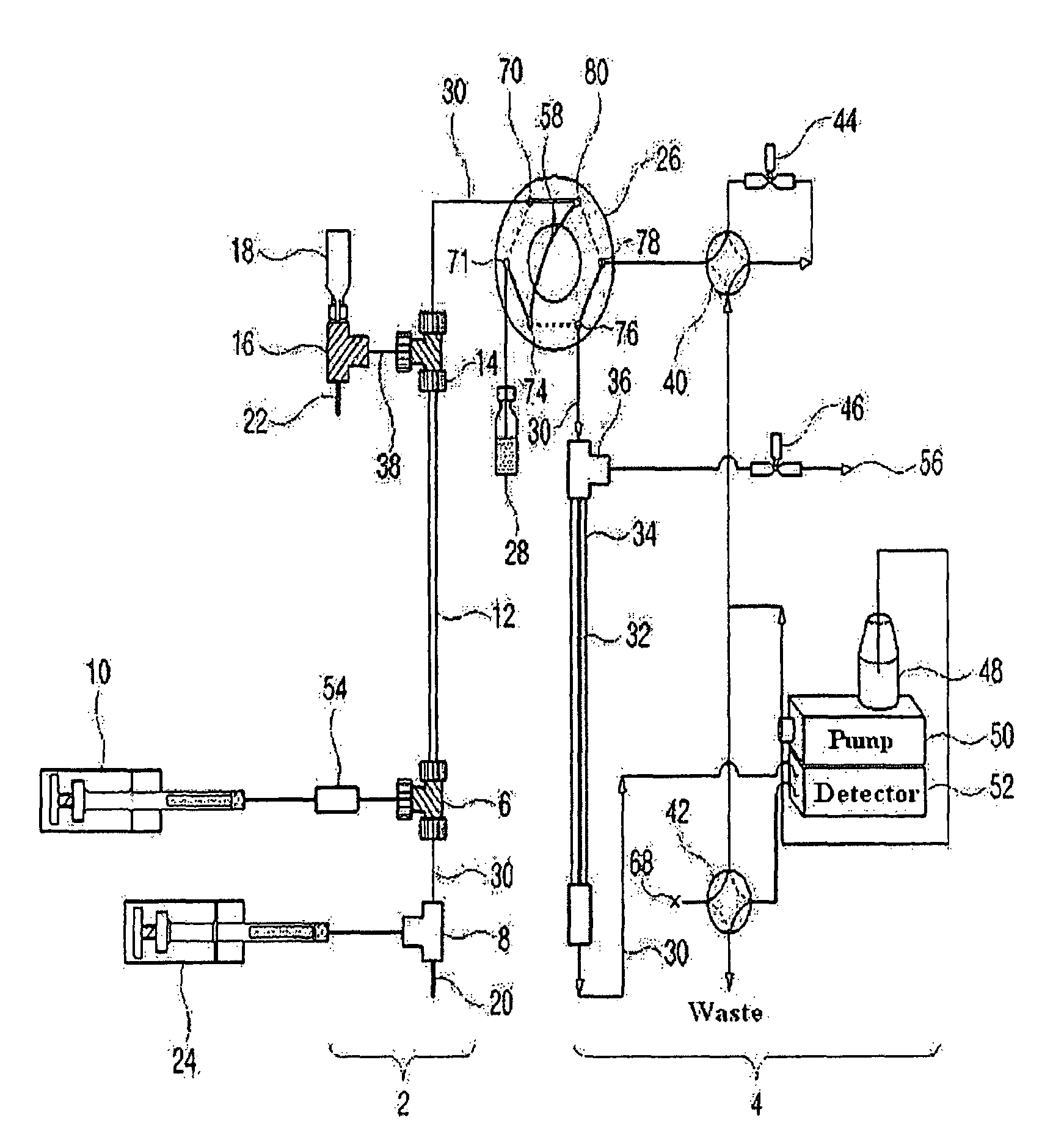
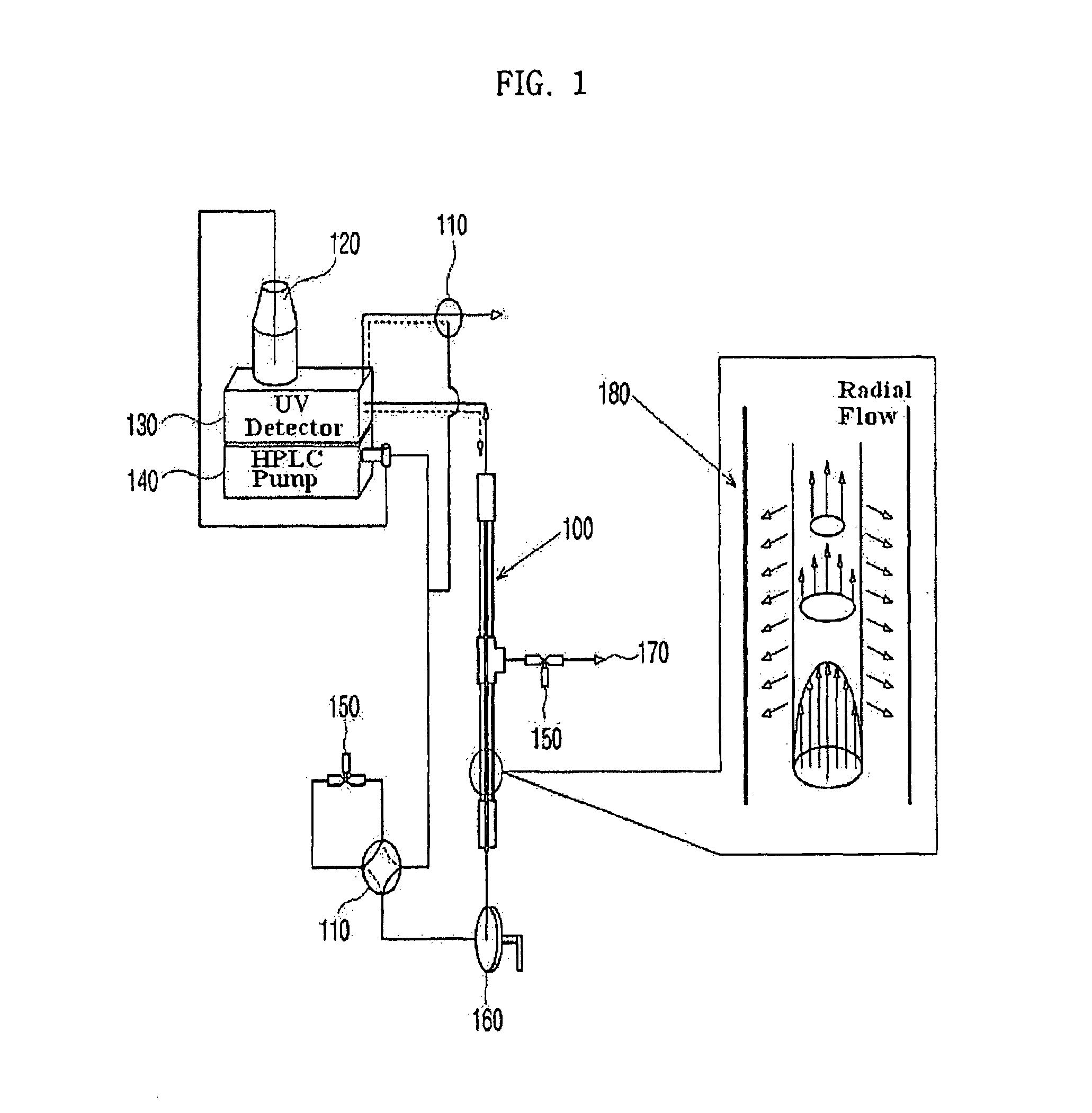
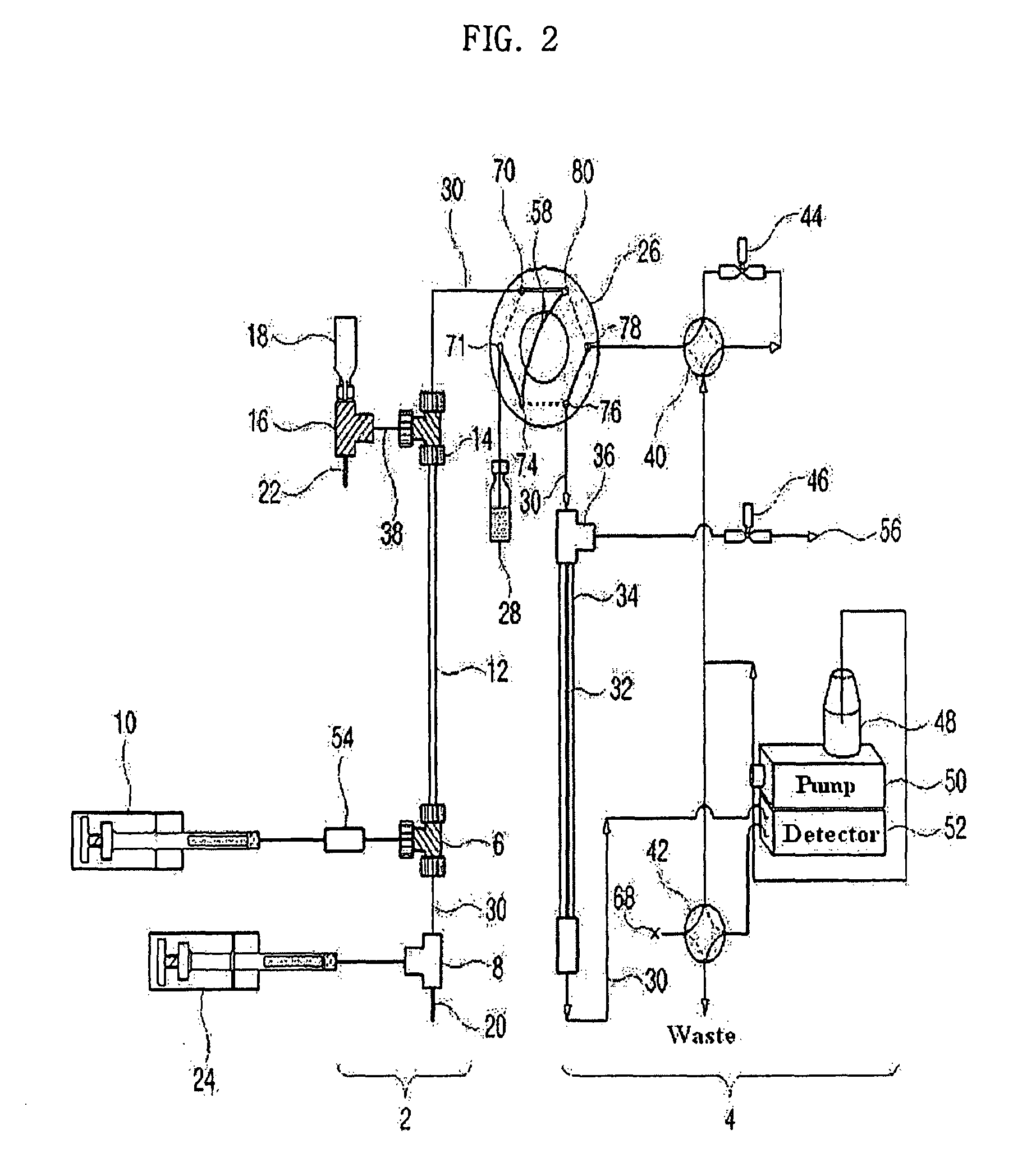
Apparatus for protein separation using capillary isoelectric focusing-hollow fiber flow field flow fractionation and method thereof
PatentInactiveUS8585884B2
Innovation
- A capillary isoelectric focusing-hollow fiber flow field flow fractionation apparatus that separates proteins based on isoelectric point (pI) and molecular weight in a two-dimensional, non-gel, and liquid phase manner, using a capillary isoelectric focusing unit connected to a hollow fiber flow field flow fractionation unit, allowing for automatic removal of ampholytes and avoiding protein denaturation.
Detection of genetic sequences using PNA probes and isotachophoresis
PatentActiveUS20170198339A1
Innovation
- The use of a peptide nucleic acid (PNA) molecule with an anti-sense sequence, combined with an Isotachophoresis (ITP) system, where the PNA hybridizes with nucleic acid molecules in a specific solution and focuses at the ITP interface based on mobility differences between leading and trailing electrolytes, allowing for sequence-specific separation and detection.
Regulatory Considerations for Scaled IEF Applications
The regulatory landscape for scaled isoelectric focusing (IEF) applications presents significant considerations that must be addressed when exploiting protonation dynamics at industrial scales. Regulatory bodies such as the FDA, EMA, and other international agencies have established frameworks that govern the implementation of separation technologies in various sectors, particularly in pharmaceutical and biomedical applications.
Quality control standards for scaled IEF processes require validation protocols that demonstrate reproducibility and robustness across batches. This includes establishing acceptable ranges for pH gradient stability, ampholyte distribution, and resolution parameters. The FDA's Process Analytical Technology (PAT) initiative specifically encourages implementation of real-time monitoring systems for critical process parameters, which becomes increasingly complex when scaling up protonation-dependent separations.
Environmental regulations present another layer of compliance requirements for industrial IEF applications. The disposal of ampholytes, carrier electrolytes, and other chemicals used in large-scale operations must adhere to local and international waste management guidelines. Organizations like the EPA in the United States and the European Chemicals Agency impose strict limitations on the discharge of these materials, necessitating effective waste treatment strategies.
Product safety and efficacy documentation requirements differ significantly between diagnostic, therapeutic, and research applications of scaled IEF technology. For diagnostic applications, regulatory bodies require extensive validation of the correlation between separation profiles and clinical outcomes. Therapeutic applications face even more stringent requirements, including demonstration of batch-to-batch consistency in separation profiles and impurity removal capabilities.
Good Manufacturing Practice (GMP) compliance presents unique challenges for scaled IEF processes due to the sensitivity of protonation dynamics to environmental conditions. Temperature control, electrolyte composition, and equipment calibration must be meticulously documented and validated. The International Conference on Harmonisation (ICH) guidelines provide specific recommendations for validation of analytical procedures that must be adapted for large-scale IEF operations.
Regulatory pathways for novel IEF technologies exploiting advanced protonation dynamics often require specialized approaches. The FDA's Emerging Technology Program and the EMA's Innovation Task Force offer frameworks for early engagement with regulators when implementing novel separation methodologies at scale. These pathways can significantly reduce regulatory uncertainty but require comprehensive characterization of the fundamental protonation mechanisms underlying the separation process.
Quality control standards for scaled IEF processes require validation protocols that demonstrate reproducibility and robustness across batches. This includes establishing acceptable ranges for pH gradient stability, ampholyte distribution, and resolution parameters. The FDA's Process Analytical Technology (PAT) initiative specifically encourages implementation of real-time monitoring systems for critical process parameters, which becomes increasingly complex when scaling up protonation-dependent separations.
Environmental regulations present another layer of compliance requirements for industrial IEF applications. The disposal of ampholytes, carrier electrolytes, and other chemicals used in large-scale operations must adhere to local and international waste management guidelines. Organizations like the EPA in the United States and the European Chemicals Agency impose strict limitations on the discharge of these materials, necessitating effective waste treatment strategies.
Product safety and efficacy documentation requirements differ significantly between diagnostic, therapeutic, and research applications of scaled IEF technology. For diagnostic applications, regulatory bodies require extensive validation of the correlation between separation profiles and clinical outcomes. Therapeutic applications face even more stringent requirements, including demonstration of batch-to-batch consistency in separation profiles and impurity removal capabilities.
Good Manufacturing Practice (GMP) compliance presents unique challenges for scaled IEF processes due to the sensitivity of protonation dynamics to environmental conditions. Temperature control, electrolyte composition, and equipment calibration must be meticulously documented and validated. The International Conference on Harmonisation (ICH) guidelines provide specific recommendations for validation of analytical procedures that must be adapted for large-scale IEF operations.
Regulatory pathways for novel IEF technologies exploiting advanced protonation dynamics often require specialized approaches. The FDA's Emerging Technology Program and the EMA's Innovation Task Force offer frameworks for early engagement with regulators when implementing novel separation methodologies at scale. These pathways can significantly reduce regulatory uncertainty but require comprehensive characterization of the fundamental protonation mechanisms underlying the separation process.
Environmental Impact of Large-Scale IEF Processes
The scaling up of Isoelectric Focusing (IEF) processes to industrial levels introduces significant environmental considerations that must be addressed for sustainable implementation. Large-scale IEF operations consume substantial amounts of carrier ampholytes and buffer solutions, which can contribute to chemical waste streams if not properly managed. These chemicals, particularly ampholytes containing nitrogen compounds, may lead to increased biochemical oxygen demand in wastewater and potential eutrophication of aquatic ecosystems when discharged without adequate treatment.
Energy consumption represents another critical environmental factor in scaled IEF processes. The application of electric fields across large separation chambers requires considerable power input, especially during extended separation runs. This energy demand translates to increased carbon footprints unless renewable energy sources are integrated into facility operations. Modern IEF equipment manufacturers have begun implementing energy recovery systems that can recapture up to 30% of applied power, though further efficiency improvements remain necessary.
Water usage in large-scale IEF presents additional environmental challenges. The process requires ultrapure water for buffer preparation and system cleaning, with industrial implementations potentially consuming thousands of liters daily. Water recycling technologies specifically designed for electrophoretic processes have emerged, capable of recovering approximately 70-85% of process water through specialized filtration and purification steps.
The environmental impact of cooling systems necessary for heat dissipation in scaled IEF operations must also be considered. Traditional water-based cooling systems contribute to thermal pollution when discharged into natural water bodies. Alternative approaches utilizing closed-loop cooling with heat exchangers have demonstrated reduced environmental impact while maintaining separation efficiency.
Recent advances in green chemistry approaches to IEF have focused on developing biodegradable ampholytes and environmentally friendly buffer systems. These innovations leverage protonation dynamics to achieve comparable separation performance while reducing environmental persistence of process chemicals. Several research groups have reported success with plant-derived ampholytes that maintain separation efficiency while demonstrating improved biodegradability profiles.
Regulatory frameworks governing large-scale IEF implementations vary globally, with the European Union's REACH regulations and the United States EPA guidelines providing the most comprehensive environmental standards. Companies deploying industrial IEF processes must navigate these requirements through careful waste management planning and environmental impact assessments prior to facility construction.
Energy consumption represents another critical environmental factor in scaled IEF processes. The application of electric fields across large separation chambers requires considerable power input, especially during extended separation runs. This energy demand translates to increased carbon footprints unless renewable energy sources are integrated into facility operations. Modern IEF equipment manufacturers have begun implementing energy recovery systems that can recapture up to 30% of applied power, though further efficiency improvements remain necessary.
Water usage in large-scale IEF presents additional environmental challenges. The process requires ultrapure water for buffer preparation and system cleaning, with industrial implementations potentially consuming thousands of liters daily. Water recycling technologies specifically designed for electrophoretic processes have emerged, capable of recovering approximately 70-85% of process water through specialized filtration and purification steps.
The environmental impact of cooling systems necessary for heat dissipation in scaled IEF operations must also be considered. Traditional water-based cooling systems contribute to thermal pollution when discharged into natural water bodies. Alternative approaches utilizing closed-loop cooling with heat exchangers have demonstrated reduced environmental impact while maintaining separation efficiency.
Recent advances in green chemistry approaches to IEF have focused on developing biodegradable ampholytes and environmentally friendly buffer systems. These innovations leverage protonation dynamics to achieve comparable separation performance while reducing environmental persistence of process chemicals. Several research groups have reported success with plant-derived ampholytes that maintain separation efficiency while demonstrating improved biodegradability profiles.
Regulatory frameworks governing large-scale IEF implementations vary globally, with the European Union's REACH regulations and the United States EPA guidelines providing the most comprehensive environmental standards. Companies deploying industrial IEF processes must navigate these requirements through careful waste management planning and environmental impact assessments prior to facility construction.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!