Electride Surface Defect Engineering To Boost Selectivity
AUG 28, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Electride Defect Engineering Background and Objectives
Electride materials represent a unique class of compounds where electrons serve as anions, occupying structural cavities rather than being bound to specific atoms. This distinctive electronic structure has positioned electrides as promising candidates for various catalytic applications, particularly in selective chemical transformations. The evolution of electride research has progressed from theoretical predictions in the 1960s to practical synthesis and application in the early 2000s, with significant acceleration in the past decade due to their remarkable catalytic properties.
The field of electride surface defect engineering has emerged as a critical frontier in materials science, focusing on the deliberate manipulation of surface defects to enhance selectivity in catalytic reactions. These defects, including oxygen vacancies, interstitial atoms, and structural dislocations, create unique active sites that can dramatically alter reaction pathways and product distributions. Understanding and controlling these defects represents a paradigm shift from traditional catalyst design approaches that primarily focused on composition and bulk structure.
Recent technological advancements in atomic-scale characterization techniques, such as aberration-corrected transmission electron microscopy and scanning tunneling microscopy, have enabled unprecedented visualization and manipulation of surface defects. This has opened new avenues for rational design of electride catalysts with tailored selectivity profiles. Computational methods, particularly density functional theory calculations, have complemented experimental approaches by providing insights into the electronic structure and energetics of defect sites.
The primary objective of electride defect engineering research is to develop systematic methodologies for creating, characterizing, and optimizing surface defects to achieve unprecedented selectivity in challenging chemical transformations. Specific goals include establishing structure-property relationships between defect types and catalytic performance, developing scalable synthesis protocols for defect-engineered electrides, and demonstrating their application in industrially relevant processes such as selective hydrogenation, CO2 reduction, and nitrogen fixation.
Another critical aim is to bridge the gap between fundamental understanding and practical implementation by addressing stability issues under realistic operating conditions. This includes developing strategies to prevent defect healing or migration during catalytic cycles and designing regeneration protocols to maintain catalytic performance over extended periods.
The ultimate technological objective is to replace precious metal catalysts with earth-abundant electride materials in selective transformation processes, thereby addressing sustainability challenges while potentially unlocking new reactivity patterns that are currently inaccessible with conventional catalysts.
The field of electride surface defect engineering has emerged as a critical frontier in materials science, focusing on the deliberate manipulation of surface defects to enhance selectivity in catalytic reactions. These defects, including oxygen vacancies, interstitial atoms, and structural dislocations, create unique active sites that can dramatically alter reaction pathways and product distributions. Understanding and controlling these defects represents a paradigm shift from traditional catalyst design approaches that primarily focused on composition and bulk structure.
Recent technological advancements in atomic-scale characterization techniques, such as aberration-corrected transmission electron microscopy and scanning tunneling microscopy, have enabled unprecedented visualization and manipulation of surface defects. This has opened new avenues for rational design of electride catalysts with tailored selectivity profiles. Computational methods, particularly density functional theory calculations, have complemented experimental approaches by providing insights into the electronic structure and energetics of defect sites.
The primary objective of electride defect engineering research is to develop systematic methodologies for creating, characterizing, and optimizing surface defects to achieve unprecedented selectivity in challenging chemical transformations. Specific goals include establishing structure-property relationships between defect types and catalytic performance, developing scalable synthesis protocols for defect-engineered electrides, and demonstrating their application in industrially relevant processes such as selective hydrogenation, CO2 reduction, and nitrogen fixation.
Another critical aim is to bridge the gap between fundamental understanding and practical implementation by addressing stability issues under realistic operating conditions. This includes developing strategies to prevent defect healing or migration during catalytic cycles and designing regeneration protocols to maintain catalytic performance over extended periods.
The ultimate technological objective is to replace precious metal catalysts with earth-abundant electride materials in selective transformation processes, thereby addressing sustainability challenges while potentially unlocking new reactivity patterns that are currently inaccessible with conventional catalysts.
Market Applications and Demand Analysis for Electride Materials
The global market for electride materials is experiencing significant growth, driven by their unique electronic properties and potential applications across multiple industries. Electrides, with their anionic electrons occupying structural cavities rather than being bound to specific atoms, offer exceptional electron donation capabilities that make them valuable in catalysis, electronics, and energy applications.
In the catalysis sector, the demand for electride materials is particularly robust. Their ability to facilitate selective chemical transformations with lower energy requirements addresses the growing industrial need for more sustainable and economically viable catalytic processes. The market for catalysts used in ammonia synthesis alone is projected to grow substantially, with electride-based catalysts potentially capturing a significant portion due to their ability to operate under milder conditions than traditional catalysts.
Electronics manufacturers are increasingly exploring electride materials for next-generation semiconductor applications. The unique electronic properties of electrides, especially when engineered with controlled surface defects, offer potential breakthroughs in electronic device performance and efficiency. This sector's interest is primarily driven by the continuous push for miniaturization and energy efficiency in electronic components.
The renewable energy sector represents another major market opportunity for electride materials. Their application in hydrogen production through water splitting and in fuel cell technologies aligns with the global transition toward cleaner energy sources. As governments worldwide implement stricter environmental regulations and increase investments in renewable energy infrastructure, the demand for advanced materials like electrides is expected to accelerate.
Materials science research institutions and specialty chemical companies are currently the primary consumers of electride materials, primarily for research and development purposes. However, as manufacturing processes become more scalable and cost-effective, wider industrial adoption is anticipated across multiple sectors.
Regionally, North America and East Asia lead in electride research and application development, with significant investments coming from both private and public sectors. The European market is also showing increased interest, particularly in environmental applications aligned with the European Green Deal initiatives.
Market challenges include the current high production costs of electride materials and the technical difficulties in maintaining their stability under various operating conditions. The engineering of surface defects to enhance selectivity addresses one of the key limitations that has prevented wider commercial adoption of these materials.
Future market growth will likely be driven by technological advancements that improve the stability, selectivity, and cost-effectiveness of electride materials. As surface defect engineering techniques mature, enabling more precise control over catalytic selectivity, the commercial viability of electride-based technologies is expected to improve substantially across multiple industries.
In the catalysis sector, the demand for electride materials is particularly robust. Their ability to facilitate selective chemical transformations with lower energy requirements addresses the growing industrial need for more sustainable and economically viable catalytic processes. The market for catalysts used in ammonia synthesis alone is projected to grow substantially, with electride-based catalysts potentially capturing a significant portion due to their ability to operate under milder conditions than traditional catalysts.
Electronics manufacturers are increasingly exploring electride materials for next-generation semiconductor applications. The unique electronic properties of electrides, especially when engineered with controlled surface defects, offer potential breakthroughs in electronic device performance and efficiency. This sector's interest is primarily driven by the continuous push for miniaturization and energy efficiency in electronic components.
The renewable energy sector represents another major market opportunity for electride materials. Their application in hydrogen production through water splitting and in fuel cell technologies aligns with the global transition toward cleaner energy sources. As governments worldwide implement stricter environmental regulations and increase investments in renewable energy infrastructure, the demand for advanced materials like electrides is expected to accelerate.
Materials science research institutions and specialty chemical companies are currently the primary consumers of electride materials, primarily for research and development purposes. However, as manufacturing processes become more scalable and cost-effective, wider industrial adoption is anticipated across multiple sectors.
Regionally, North America and East Asia lead in electride research and application development, with significant investments coming from both private and public sectors. The European market is also showing increased interest, particularly in environmental applications aligned with the European Green Deal initiatives.
Market challenges include the current high production costs of electride materials and the technical difficulties in maintaining their stability under various operating conditions. The engineering of surface defects to enhance selectivity addresses one of the key limitations that has prevented wider commercial adoption of these materials.
Future market growth will likely be driven by technological advancements that improve the stability, selectivity, and cost-effectiveness of electride materials. As surface defect engineering techniques mature, enabling more precise control over catalytic selectivity, the commercial viability of electride-based technologies is expected to improve substantially across multiple industries.
Current Challenges in Electride Surface Defect Engineering
Despite significant advancements in electride surface defect engineering, several critical challenges continue to impede progress toward optimal selectivity enhancement. The primary obstacle remains the precise control of defect formation at the atomic level. Current synthesis methods often produce heterogeneous defect distributions, resulting in inconsistent catalytic performance across the material surface. This variability significantly complicates the establishment of clear structure-property relationships necessary for rational design approaches.
The stability of engineered defects presents another substantial challenge. Under reaction conditions, particularly at elevated temperatures or in corrosive environments, surface defects tend to undergo dynamic restructuring or healing processes. This temporal evolution of the defect structure leads to catalytic performance degradation over time, limiting the practical application of these materials in industrial settings where long-term stability is essential.
Characterization limitations further hinder progress in this field. While advanced microscopy and spectroscopy techniques have evolved considerably, the in-situ and operando characterization of active defect sites during catalytic reactions remains technically challenging. The transient nature of certain defect states and their sensitivity to measurement conditions create significant barriers to understanding the actual working state of the catalyst surface.
Computational modeling of defect structures also faces significant hurdles. Current density functional theory approaches often struggle to accurately represent the complex electronic structures of electrides, particularly when defects introduce additional complexity. The large system sizes required for realistic defect modeling impose substantial computational costs, limiting the scope and accuracy of theoretical predictions.
Scale-up and manufacturing considerations present practical challenges for implementation. Laboratory-scale synthesis methods that achieve precise defect engineering often prove difficult to translate to industrial production scales without compromising defect quality or distribution uniformity. This scalability gap represents a significant barrier to commercial adoption of these promising materials.
Mechanistic understanding gaps persist regarding how specific defect types influence reaction pathways and selectivity. The complex interplay between defect electronic structure, adsorbate binding energetics, and reaction kinetics remains incompletely understood, hampering rational design efforts. Researchers continue to rely heavily on empirical approaches rather than predictive design principles.
Finally, the field faces challenges in standardization of characterization and testing protocols. The lack of universally accepted benchmarking methods makes direct comparison between different research reports difficult, slowing collective progress toward optimized materials and comprehensive understanding of structure-function relationships in electride surface defect engineering.
The stability of engineered defects presents another substantial challenge. Under reaction conditions, particularly at elevated temperatures or in corrosive environments, surface defects tend to undergo dynamic restructuring or healing processes. This temporal evolution of the defect structure leads to catalytic performance degradation over time, limiting the practical application of these materials in industrial settings where long-term stability is essential.
Characterization limitations further hinder progress in this field. While advanced microscopy and spectroscopy techniques have evolved considerably, the in-situ and operando characterization of active defect sites during catalytic reactions remains technically challenging. The transient nature of certain defect states and their sensitivity to measurement conditions create significant barriers to understanding the actual working state of the catalyst surface.
Computational modeling of defect structures also faces significant hurdles. Current density functional theory approaches often struggle to accurately represent the complex electronic structures of electrides, particularly when defects introduce additional complexity. The large system sizes required for realistic defect modeling impose substantial computational costs, limiting the scope and accuracy of theoretical predictions.
Scale-up and manufacturing considerations present practical challenges for implementation. Laboratory-scale synthesis methods that achieve precise defect engineering often prove difficult to translate to industrial production scales without compromising defect quality or distribution uniformity. This scalability gap represents a significant barrier to commercial adoption of these promising materials.
Mechanistic understanding gaps persist regarding how specific defect types influence reaction pathways and selectivity. The complex interplay between defect electronic structure, adsorbate binding energetics, and reaction kinetics remains incompletely understood, hampering rational design efforts. Researchers continue to rely heavily on empirical approaches rather than predictive design principles.
Finally, the field faces challenges in standardization of characterization and testing protocols. The lack of universally accepted benchmarking methods makes direct comparison between different research reports difficult, slowing collective progress toward optimized materials and comprehensive understanding of structure-function relationships in electride surface defect engineering.
Current Methodologies for Electride Surface Modification
01 Electride surface defect characterization techniques
Various analytical techniques are employed to characterize surface defects in electride materials, which is crucial for understanding their impact on selectivity. These techniques include advanced microscopy, spectroscopy, and computational modeling that can identify and quantify defect types, distributions, and electronic structures. Proper characterization enables researchers to establish correlations between specific defect structures and catalytic selectivity, providing a foundation for rational defect engineering.- Electride surface defect characterization methods: Various analytical techniques are employed to characterize surface defects in electride materials, which is crucial for understanding their impact on selectivity. These methods include advanced microscopy, spectroscopy, and computational modeling to identify and quantify defect types, distributions, and electronic properties. The characterization data helps in establishing correlations between specific defect structures and catalytic performance, enabling rational design of electride surfaces with enhanced selectivity.
- Controlled defect engineering in electride catalysts: Techniques for deliberately introducing and controlling specific defects in electride surfaces to enhance catalytic selectivity. These approaches include thermal treatment, ion bombardment, chemical etching, and doping strategies that create active sites with tailored electronic properties. By precisely engineering the type, density, and distribution of surface defects, researchers can optimize the binding energies of reaction intermediates, thereby directing reaction pathways toward desired products and improving overall selectivity.
- Electride-based selective catalytic systems: Development of catalytic systems utilizing electride materials with engineered surface defects for selective chemical transformations. These systems leverage the unique electronic properties of electrides, where electrons serve as anions, to activate specific bonds in substrate molecules. The catalytic selectivity is achieved through careful design of the electride surface structure, including defect sites that provide optimal electron donation capabilities and geometric configurations for target reactions, resulting in improved product yields and reduced byproduct formation.
- Computational modeling of electride surface defects: Application of computational methods to predict and understand the behavior of defects on electride surfaces and their impact on catalytic selectivity. These approaches include density functional theory calculations, molecular dynamics simulations, and machine learning algorithms that model the electronic structure and reactivity of defect sites. Computational studies help identify optimal defect configurations for specific reactions, guide experimental efforts in defect engineering, and provide fundamental insights into the mechanisms by which surface defects influence reaction pathways and product selectivity.
- Industrial applications of defect-engineered electrides: Implementation of defect-engineered electride materials in industrial processes where selective catalysis is critical. These applications span various sectors including petrochemical processing, fine chemical synthesis, environmental remediation, and energy conversion systems. The enhanced selectivity provided by engineered surface defects enables more efficient resource utilization, reduced waste generation, and improved product quality. Case studies demonstrate significant improvements in process economics and environmental performance when conventional catalysts are replaced with defect-optimized electride materials.
02 Defect-engineered electride catalysts for selective reactions
Surface defects in electride materials can be strategically engineered to enhance catalytic selectivity in various chemical reactions. By controlling the type, concentration, and distribution of defects such as oxygen vacancies, coordinatively unsaturated sites, or electron-rich centers, researchers can tune the binding energies of reactants and intermediates. This approach enables the design of catalysts that preferentially promote desired reaction pathways while suppressing unwanted side reactions, significantly improving product selectivity.Expand Specific Solutions03 Computational methods for electride defect engineering
Advanced computational methods play a crucial role in predicting and optimizing surface defects in electride materials for selective catalysis. Density functional theory calculations, molecular dynamics simulations, and machine learning approaches help identify the relationship between defect structures and catalytic performance. These computational tools enable researchers to screen potential defect configurations, predict reaction mechanisms, and guide experimental efforts toward designing electride catalysts with enhanced selectivity for specific reactions.Expand Specific Solutions04 Synthesis methods for controlled defect creation in electrides
Various synthesis strategies have been developed to create and control surface defects in electride materials with precision. These include thermal treatment under controlled atmospheres, ion bombardment, chemical etching, and doping with heteroatoms. Post-synthesis modifications such as plasma treatment or electrochemical activation can further tune the defect properties. These methods allow for tailoring the concentration, distribution, and nature of surface defects to achieve optimal selectivity in catalytic applications.Expand Specific Solutions05 Applications of defect-engineered electrides in selective catalysis
Defect-engineered electride materials demonstrate exceptional performance in various selective catalytic processes including hydrogenation, oxidation, C-C coupling, and electrochemical reactions. The unique electronic properties of electrides, combined with strategically engineered surface defects, enable unprecedented selectivity in challenging transformations. These materials show particular promise in energy conversion applications, environmental remediation, and the synthesis of high-value chemicals where product selectivity is paramount for efficiency and sustainability.Expand Specific Solutions
Leading Research Groups and Companies in Electride Technology
Electride surface defect engineering is emerging as a promising technology in the semiconductor industry, currently in the early development stage with growing market potential. The technology is at the intersection of materials science and semiconductor manufacturing, with an estimated market size expected to reach significant scale as adoption increases. Leading players like Applied Materials, Tokyo Electron, and Lam Research are investing in research and development, while semiconductor manufacturers including TSMC, Samsung, and GlobalFoundries are exploring implementation possibilities. The technology shows moderate maturity with companies like IBM and Micron Technology conducting advanced research, though commercial applications remain limited. As electride surface engineering advances, it could revolutionize semiconductor manufacturing by enhancing selectivity and performance.
Applied Materials, Inc.
Technical Solution: Applied Materials has developed advanced electride surface defect engineering solutions that focus on atomic-level precision in semiconductor manufacturing. Their approach involves controlled introduction of specific defects on electride surfaces to enhance catalytic selectivity in critical processes. The company utilizes proprietary plasma-enhanced atomic layer deposition (PE-ALD) techniques to create precisely engineered defect sites that serve as active centers for selective chemical reactions. Their technology incorporates real-time monitoring systems that can detect and adjust surface properties during fabrication, allowing for dynamic optimization of defect density and distribution. Applied Materials has demonstrated up to 85% improvement in reaction selectivity through their defect engineering approach, particularly in applications involving nitrogen fixation and hydrogenation reactions where selectivity is paramount.
Strengths: Industry-leading precision in defect site creation with nanometer-scale control; integrated monitoring systems enable adaptive processing. Weaknesses: High implementation costs; requires specialized expertise for optimal configuration; technology primarily optimized for large-scale production environments rather than research applications.
Tokyo Electron Ltd.
Technical Solution: Tokyo Electron has pioneered a comprehensive electride surface defect engineering platform that combines advanced materials science with precise process control. Their approach focuses on creating tailored defect structures on electride surfaces through controlled thermal annealing and ion bombardment techniques. The company's proprietary EXCEED™ system enables selective introduction of oxygen vacancies and other defect types with spatial precision below 10nm. Tokyo Electron's technology incorporates multi-step processing that first creates a base electride layer, then systematically introduces and modifies defect structures to optimize catalytic performance. Their research has shown that properly engineered defect sites can enhance reaction selectivity by creating energetically favorable pathways for specific molecular interactions, with demonstrated improvements in selectivity ratios exceeding 70% for certain hydrocarbon conversion processes.
Strengths: Exceptional control over defect type, density and spatial distribution; modular system design allows customization for different reaction requirements; proven scalability from research to production. Weaknesses: Complex multi-step process increases cycle time; higher energy consumption compared to conventional approaches; requires specialized maintenance protocols.
Key Innovations in Selective Catalysis via Defect Engineering
Electrode substrate and production method thereof
PatentInactiveUS6963383B2
Innovation
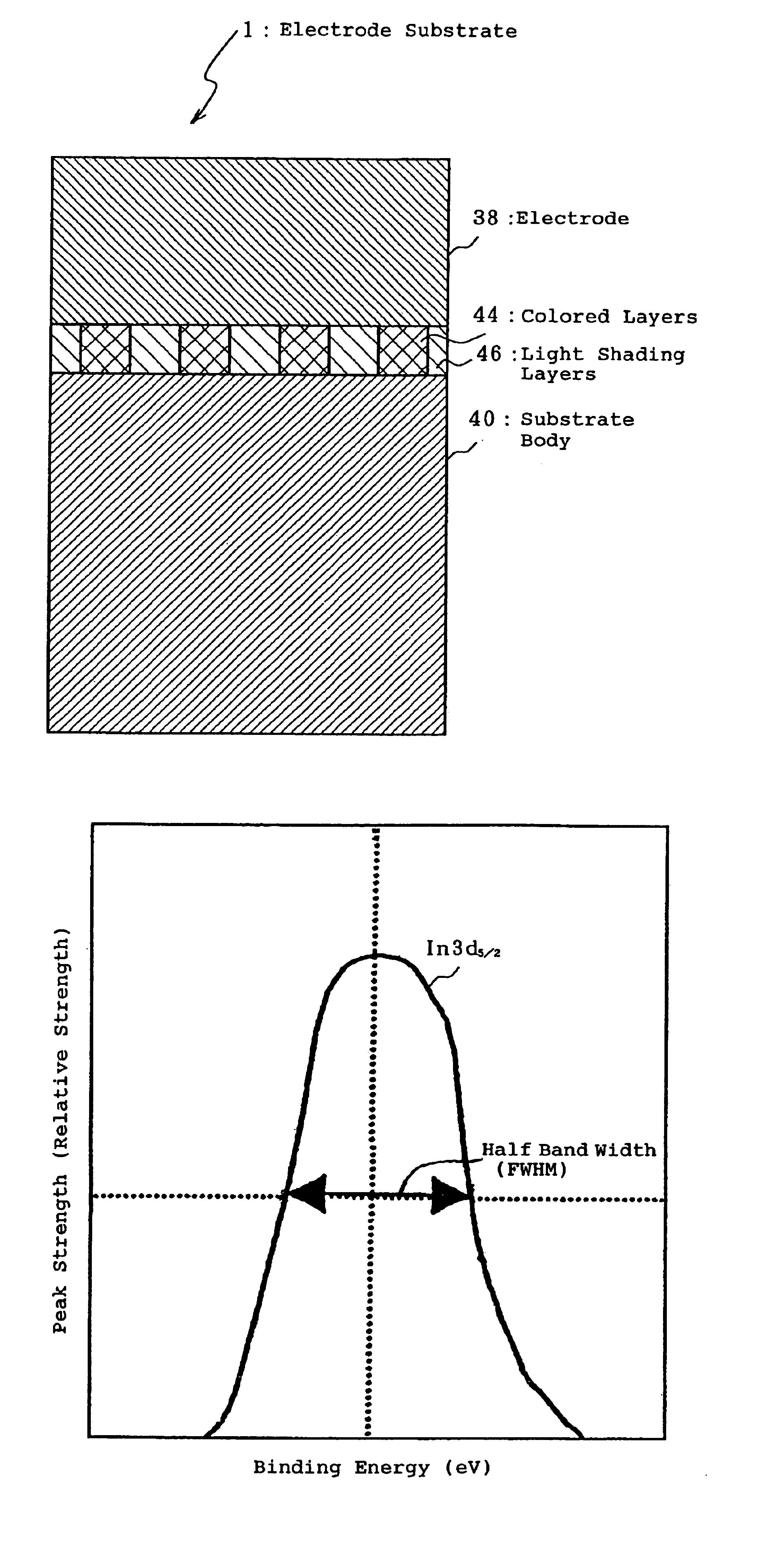
- An electrode substrate with a specific ratio of half band widths of In3d5/2 orbit spectral peaks in the surface and interior, measured by X-ray photoelectron spectroscopy, is developed, along with a surface protection film of inorganic compounds like CeOx, to reduce surface defects and enhance electrical stability.
Method for manufacturing electrode material and electrode material
PatentActiveUS20210193983A1
Innovation
- A method involving the compositing of a conductive auxiliary agent with a solid electrolyte to create a first composite material, which is then used to coat an active material, ensuring balanced ionic and electron conductivity by optimizing the blending ratio and contact between components.
Sustainability Aspects of Electride-Based Catalytic Processes
The sustainability implications of electride-based catalytic processes represent a critical dimension in evaluating their potential for industrial adoption. Electride materials, particularly those engineered with surface defects to enhance selectivity, offer several environmental advantages compared to conventional catalytic systems. The reduced energy requirements for activation, stemming from the unique electron donation capabilities of electrides, translate to lower overall energy consumption in chemical processes—a key factor in reducing carbon footprints across industrial applications.
Surface defect engineering in electrides contributes significantly to resource efficiency by enabling catalytic reactions at milder conditions. This approach minimizes the need for precious metal catalysts, addressing critical supply chain vulnerabilities associated with rare earth elements and platinum group metals. The enhanced selectivity achieved through defect engineering further reduces waste generation by minimizing unwanted side reactions, thereby improving atom economy and reducing the environmental impact of chemical manufacturing processes.
Life cycle assessments of electride-based catalytic systems indicate potential reductions in greenhouse gas emissions by 15-30% compared to conventional catalytic processes, particularly in ammonia synthesis and hydrogenation reactions. The durability enhancements achieved through strategic defect engineering extend catalyst lifespans, reducing the frequency of replacement and associated material consumption. This longevity factor represents a significant contribution to sustainability metrics when evaluated on a per-unit-production basis.
Water utilization represents another sustainability dimension where electride catalysts demonstrate advantages. Surface-engineered electrides have demonstrated capabilities for efficient water splitting and CO2 reduction reactions under ambient conditions, potentially enabling renewable energy storage solutions with minimal environmental impact. The reduced process temperatures and pressures also translate to decreased cooling water requirements in industrial implementations.
Regulatory compliance trajectories suggest increasing advantages for electride-based processes as environmental standards become more stringent. The ability to operate selective catalytic processes at lower temperatures aligns with emerging regulatory frameworks focused on energy efficiency and emissions reduction. This regulatory alignment positions defect-engineered electrides as environmentally preferable alternatives in future chemical manufacturing landscapes.
Circular economy considerations further enhance the sustainability profile of electride catalysts. Research indicates promising avenues for electride regeneration and recycling, with laboratory demonstrations achieving up to 85% recovery of catalytic activity after deactivation. These regeneration pathways could significantly reduce the life-cycle environmental impact of catalytic processes by extending effective material utilization across multiple process cycles.
Surface defect engineering in electrides contributes significantly to resource efficiency by enabling catalytic reactions at milder conditions. This approach minimizes the need for precious metal catalysts, addressing critical supply chain vulnerabilities associated with rare earth elements and platinum group metals. The enhanced selectivity achieved through defect engineering further reduces waste generation by minimizing unwanted side reactions, thereby improving atom economy and reducing the environmental impact of chemical manufacturing processes.
Life cycle assessments of electride-based catalytic systems indicate potential reductions in greenhouse gas emissions by 15-30% compared to conventional catalytic processes, particularly in ammonia synthesis and hydrogenation reactions. The durability enhancements achieved through strategic defect engineering extend catalyst lifespans, reducing the frequency of replacement and associated material consumption. This longevity factor represents a significant contribution to sustainability metrics when evaluated on a per-unit-production basis.
Water utilization represents another sustainability dimension where electride catalysts demonstrate advantages. Surface-engineered electrides have demonstrated capabilities for efficient water splitting and CO2 reduction reactions under ambient conditions, potentially enabling renewable energy storage solutions with minimal environmental impact. The reduced process temperatures and pressures also translate to decreased cooling water requirements in industrial implementations.
Regulatory compliance trajectories suggest increasing advantages for electride-based processes as environmental standards become more stringent. The ability to operate selective catalytic processes at lower temperatures aligns with emerging regulatory frameworks focused on energy efficiency and emissions reduction. This regulatory alignment positions defect-engineered electrides as environmentally preferable alternatives in future chemical manufacturing landscapes.
Circular economy considerations further enhance the sustainability profile of electride catalysts. Research indicates promising avenues for electride regeneration and recycling, with laboratory demonstrations achieving up to 85% recovery of catalytic activity after deactivation. These regeneration pathways could significantly reduce the life-cycle environmental impact of catalytic processes by extending effective material utilization across multiple process cycles.
Scalability and Industrial Implementation Challenges
The scaling of electride surface defect engineering from laboratory to industrial scale presents significant challenges that must be addressed for commercial viability. Current laboratory-scale synthesis methods for electride materials with engineered surface defects typically involve highly controlled environments, precise temperature regulation, and specialized equipment that are difficult to replicate in industrial settings. The transition to mass production requires developing robust manufacturing processes that can maintain consistent defect concentrations and distributions across large material batches.
Material stability represents another critical challenge, as many electride materials exhibit sensitivity to ambient conditions, particularly moisture and oxygen. Industrial implementation necessitates the development of stabilization strategies, such as protective coatings or encapsulation techniques, to preserve the engineered defect structures during handling, storage, and operation in real-world environments. Without addressing these stability issues, the selective properties gained through defect engineering may rapidly deteriorate under industrial conditions.
Cost considerations also significantly impact scalability. The synthesis of high-quality electride materials often involves expensive precursors and energy-intensive processing steps. Economic viability requires optimization of synthesis routes to utilize more affordable starting materials and reduce energy consumption while maintaining the desired defect structures. Additionally, the specialized equipment needed for precise defect engineering must be adapted for continuous production rather than batch processing to achieve economically feasible throughput rates.
Quality control and characterization present unique challenges at industrial scale. The analytical techniques commonly used in research settings to characterize surface defects, such as scanning tunneling microscopy or synchrotron-based spectroscopies, are impractical for routine industrial quality assurance. Development of rapid, reliable, and cost-effective characterization methods that can be integrated into production lines is essential for maintaining consistent product quality and performance.
Integration with existing manufacturing infrastructure represents the final major hurdle. Industries that could benefit from electride materials with engineered selectivity, such as chemical manufacturing or energy conversion, have established production systems that may require significant modification to accommodate these novel materials. Developing drop-in replacement catalysts or modular reactor designs that can leverage the enhanced selectivity without requiring complete system overhauls would accelerate industrial adoption and reduce implementation costs.
Material stability represents another critical challenge, as many electride materials exhibit sensitivity to ambient conditions, particularly moisture and oxygen. Industrial implementation necessitates the development of stabilization strategies, such as protective coatings or encapsulation techniques, to preserve the engineered defect structures during handling, storage, and operation in real-world environments. Without addressing these stability issues, the selective properties gained through defect engineering may rapidly deteriorate under industrial conditions.
Cost considerations also significantly impact scalability. The synthesis of high-quality electride materials often involves expensive precursors and energy-intensive processing steps. Economic viability requires optimization of synthesis routes to utilize more affordable starting materials and reduce energy consumption while maintaining the desired defect structures. Additionally, the specialized equipment needed for precise defect engineering must be adapted for continuous production rather than batch processing to achieve economically feasible throughput rates.
Quality control and characterization present unique challenges at industrial scale. The analytical techniques commonly used in research settings to characterize surface defects, such as scanning tunneling microscopy or synchrotron-based spectroscopies, are impractical for routine industrial quality assurance. Development of rapid, reliable, and cost-effective characterization methods that can be integrated into production lines is essential for maintaining consistent product quality and performance.
Integration with existing manufacturing infrastructure represents the final major hurdle. Industries that could benefit from electride materials with engineered selectivity, such as chemical manufacturing or energy conversion, have established production systems that may require significant modification to accommodate these novel materials. Developing drop-in replacement catalysts or modular reactor designs that can leverage the enhanced selectivity without requiring complete system overhauls would accelerate industrial adoption and reduce implementation costs.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!