How do cell-free systems innovate personalized diagnostics?
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cell-free Diagnostics Background and Objectives
Cell-free diagnostic systems represent a revolutionary approach in the field of molecular diagnostics, evolving from traditional laboratory techniques to portable, accessible platforms. These systems utilize biological components extracted from cells to detect specific biomarkers without requiring intact cellular structures. The development trajectory began with basic cell-free protein synthesis systems in the 1960s, progressing through significant advancements in synthetic biology and molecular engineering to today's sophisticated diagnostic applications.
The technological evolution of cell-free diagnostics has been accelerated by breakthroughs in biomolecular engineering, microfluidics, and biosensor development. Recent innovations have focused on enhancing sensitivity, specificity, and stability of these systems, while simultaneously reducing complexity and cost. The integration of CRISPR-Cas technologies has particularly revolutionized the field, enabling highly specific nucleic acid detection with minimal equipment requirements.
Current objectives in cell-free diagnostics center on achieving true personalization of diagnostic processes. This involves developing systems capable of detecting multiple biomarkers simultaneously, adapting to individual genetic profiles, and providing actionable health insights in real-time. The ultimate goal is to create diagnostic platforms that can be customized to individual patient needs, considering factors such as genetic predispositions, environmental exposures, and lifestyle factors.
The personalization objective extends beyond mere technical capabilities to encompass accessibility and usability. Researchers aim to develop cell-free diagnostic systems that can function effectively in diverse settings, from sophisticated hospital laboratories to resource-limited environments. This democratization of diagnostic technology represents a critical step toward global health equity and personalized medicine for all populations.
Technical objectives also include improving the shelf-stability of cell-free components, enhancing signal amplification methods for greater sensitivity, and developing standardized protocols for quality control. Additionally, there is significant focus on creating integrated systems that combine sample preparation, analysis, and data interpretation in single, user-friendly platforms.
The convergence of cell-free biology with digital technologies presents another frontier, with objectives centered on developing smart diagnostic systems capable of connecting to mobile devices, cloud computing, and artificial intelligence for data analysis. These integrated systems aim to transform raw biological signals into clinically relevant information, enabling personalized health monitoring and treatment optimization based on individual biomarker profiles.
The technological evolution of cell-free diagnostics has been accelerated by breakthroughs in biomolecular engineering, microfluidics, and biosensor development. Recent innovations have focused on enhancing sensitivity, specificity, and stability of these systems, while simultaneously reducing complexity and cost. The integration of CRISPR-Cas technologies has particularly revolutionized the field, enabling highly specific nucleic acid detection with minimal equipment requirements.
Current objectives in cell-free diagnostics center on achieving true personalization of diagnostic processes. This involves developing systems capable of detecting multiple biomarkers simultaneously, adapting to individual genetic profiles, and providing actionable health insights in real-time. The ultimate goal is to create diagnostic platforms that can be customized to individual patient needs, considering factors such as genetic predispositions, environmental exposures, and lifestyle factors.
The personalization objective extends beyond mere technical capabilities to encompass accessibility and usability. Researchers aim to develop cell-free diagnostic systems that can function effectively in diverse settings, from sophisticated hospital laboratories to resource-limited environments. This democratization of diagnostic technology represents a critical step toward global health equity and personalized medicine for all populations.
Technical objectives also include improving the shelf-stability of cell-free components, enhancing signal amplification methods for greater sensitivity, and developing standardized protocols for quality control. Additionally, there is significant focus on creating integrated systems that combine sample preparation, analysis, and data interpretation in single, user-friendly platforms.
The convergence of cell-free biology with digital technologies presents another frontier, with objectives centered on developing smart diagnostic systems capable of connecting to mobile devices, cloud computing, and artificial intelligence for data analysis. These integrated systems aim to transform raw biological signals into clinically relevant information, enabling personalized health monitoring and treatment optimization based on individual biomarker profiles.
Market Analysis for Personalized Diagnostic Solutions
The global personalized diagnostics market is experiencing unprecedented growth, driven by advancements in cell-free diagnostic technologies. Current market valuations place this sector at approximately $8.7 billion in 2023, with projections indicating a compound annual growth rate of 11.2% through 2030, potentially reaching $18.4 billion by the end of the decade.
Consumer demand for personalized healthcare solutions has created significant market opportunities, particularly in oncology, infectious diseases, and genetic disorder diagnostics. Cell-free systems, especially cell-free DNA/RNA testing, represent the fastest-growing segment within this market, with adoption rates increasing by nearly 25% annually in developed healthcare markets.
Regional analysis reveals North America currently dominates with approximately 42% market share, followed by Europe (28%) and Asia-Pacific (21%). However, the Asia-Pacific region demonstrates the highest growth potential, with China and India investing heavily in healthcare infrastructure and adopting innovative diagnostic technologies at accelerating rates.
The market segmentation shows distinct categories emerging: point-of-care diagnostics (36% market share), laboratory-based testing (41%), and direct-to-consumer solutions (23%). Cell-free diagnostic systems are particularly disrupting the point-of-care segment, offering rapid results without complex laboratory infrastructure, thereby expanding accessibility in resource-limited settings.
Key market drivers include aging global populations, rising chronic disease prevalence, increasing healthcare expenditure, and growing consumer awareness about preventive healthcare. The COVID-19 pandemic served as a significant catalyst, accelerating adoption of remote and rapid diagnostic solutions while demonstrating the value of personalized approaches to disease detection and monitoring.
Reimbursement policies represent both an opportunity and challenge for market growth. While some regions have established favorable coverage for novel diagnostic approaches, fragmented policies across different healthcare systems create market entry barriers. Countries with single-payer systems tend to demonstrate faster adoption rates once regulatory approval is secured.
Consumer willingness to pay for personalized diagnostic solutions varies significantly by region and application. Cancer-related diagnostics command premium pricing with higher acceptance rates, while preventive screening applications face more price sensitivity. The average consumer expenditure on personalized diagnostics has increased by 18% over the past three years, indicating growing market acceptance.
Consumer demand for personalized healthcare solutions has created significant market opportunities, particularly in oncology, infectious diseases, and genetic disorder diagnostics. Cell-free systems, especially cell-free DNA/RNA testing, represent the fastest-growing segment within this market, with adoption rates increasing by nearly 25% annually in developed healthcare markets.
Regional analysis reveals North America currently dominates with approximately 42% market share, followed by Europe (28%) and Asia-Pacific (21%). However, the Asia-Pacific region demonstrates the highest growth potential, with China and India investing heavily in healthcare infrastructure and adopting innovative diagnostic technologies at accelerating rates.
The market segmentation shows distinct categories emerging: point-of-care diagnostics (36% market share), laboratory-based testing (41%), and direct-to-consumer solutions (23%). Cell-free diagnostic systems are particularly disrupting the point-of-care segment, offering rapid results without complex laboratory infrastructure, thereby expanding accessibility in resource-limited settings.
Key market drivers include aging global populations, rising chronic disease prevalence, increasing healthcare expenditure, and growing consumer awareness about preventive healthcare. The COVID-19 pandemic served as a significant catalyst, accelerating adoption of remote and rapid diagnostic solutions while demonstrating the value of personalized approaches to disease detection and monitoring.
Reimbursement policies represent both an opportunity and challenge for market growth. While some regions have established favorable coverage for novel diagnostic approaches, fragmented policies across different healthcare systems create market entry barriers. Countries with single-payer systems tend to demonstrate faster adoption rates once regulatory approval is secured.
Consumer willingness to pay for personalized diagnostic solutions varies significantly by region and application. Cancer-related diagnostics command premium pricing with higher acceptance rates, while preventive screening applications face more price sensitivity. The average consumer expenditure on personalized diagnostics has increased by 18% over the past three years, indicating growing market acceptance.
Current Landscape and Technical Barriers
Cell-free systems represent a significant advancement in diagnostic technologies, offering a platform that operates outside the constraints of living cells. Currently, the landscape of cell-free diagnostics is rapidly evolving, with several key technologies gaining prominence. Paper-based cell-free systems have emerged as cost-effective solutions for point-of-care testing, while microfluidic platforms enable precise control over reaction conditions, enhancing sensitivity and specificity.
The integration of CRISPR-Cas systems with cell-free platforms has revolutionized nucleic acid detection, allowing for rapid identification of specific genetic sequences with minimal equipment. Synthetic biology approaches have further expanded capabilities through engineered genetic circuits that can process complex biological information and produce easily detectable outputs. Additionally, the incorporation of nanotechnology has improved signal amplification and detection limits.
Despite these advancements, significant technical barriers persist. Stability remains a primary challenge, as cell-free components often degrade rapidly, limiting shelf life and reliability in field conditions. Temperature sensitivity further complicates deployment in resource-limited settings where controlled environments may be unavailable. The standardization of production methods also presents difficulties, with batch-to-batch variability affecting reproducibility and reliability of diagnostic results.
Sensitivity and specificity limitations continue to challenge developers, particularly when detecting low-abundance biomarkers in complex biological samples. Background noise and cross-reactivity with non-target molecules frequently compromise accuracy. The complexity of sample preparation represents another substantial hurdle, as many current systems require extensive preprocessing steps that limit their utility in point-of-care settings.
Regulatory pathways for cell-free diagnostic systems remain underdeveloped, creating uncertainty for commercial development. The novel nature of these technologies often places them in regulatory gray areas, complicating validation and approval processes. Cost considerations also present barriers to widespread adoption, with many current systems requiring specialized reagents and equipment that increase per-test expenses.
Geographical disparities in technology development and implementation are evident, with most innovation concentrated in North America, Europe, and parts of Asia. This uneven distribution creates challenges for global access and adaptation to diverse healthcare contexts. The technical expertise required for implementation further limits adoption in regions with fewer specialized personnel.
The integration of CRISPR-Cas systems with cell-free platforms has revolutionized nucleic acid detection, allowing for rapid identification of specific genetic sequences with minimal equipment. Synthetic biology approaches have further expanded capabilities through engineered genetic circuits that can process complex biological information and produce easily detectable outputs. Additionally, the incorporation of nanotechnology has improved signal amplification and detection limits.
Despite these advancements, significant technical barriers persist. Stability remains a primary challenge, as cell-free components often degrade rapidly, limiting shelf life and reliability in field conditions. Temperature sensitivity further complicates deployment in resource-limited settings where controlled environments may be unavailable. The standardization of production methods also presents difficulties, with batch-to-batch variability affecting reproducibility and reliability of diagnostic results.
Sensitivity and specificity limitations continue to challenge developers, particularly when detecting low-abundance biomarkers in complex biological samples. Background noise and cross-reactivity with non-target molecules frequently compromise accuracy. The complexity of sample preparation represents another substantial hurdle, as many current systems require extensive preprocessing steps that limit their utility in point-of-care settings.
Regulatory pathways for cell-free diagnostic systems remain underdeveloped, creating uncertainty for commercial development. The novel nature of these technologies often places them in regulatory gray areas, complicating validation and approval processes. Cost considerations also present barriers to widespread adoption, with many current systems requiring specialized reagents and equipment that increase per-test expenses.
Geographical disparities in technology development and implementation are evident, with most innovation concentrated in North America, Europe, and parts of Asia. This uneven distribution creates challenges for global access and adaptation to diverse healthcare contexts. The technical expertise required for implementation further limits adoption in regions with fewer specialized personnel.
Existing Cell-free Diagnostic Platforms
01 Cell-free diagnostic systems for personalized medicine
Cell-free diagnostic systems enable personalized medicine approaches by analyzing biological samples without the need for intact cells. These systems can detect specific biomarkers, genetic mutations, or other molecular indicators that help tailor medical treatments to individual patients. The technology allows for rapid, sensitive detection of disease markers from bodily fluids like blood or saliva, facilitating early diagnosis and personalized treatment strategies.- Cell-free diagnostic systems for personalized medicine: Cell-free diagnostic systems enable personalized medicine approaches by analyzing biological samples without the need for intact cells. These systems can detect biomarkers, genetic mutations, and other molecular indicators to provide individualized diagnostic information. The technology allows for rapid, non-invasive testing that can be customized to patient-specific needs, facilitating more targeted treatment decisions and monitoring of disease progression.
- Point-of-care cell-free diagnostic platforms: Point-of-care cell-free diagnostic platforms provide rapid results in clinical settings without requiring sophisticated laboratory infrastructure. These systems integrate sample preparation, analysis, and result interpretation in compact, user-friendly devices. The technology enables immediate diagnostic decisions in various healthcare settings, from emergency rooms to remote locations, improving patient outcomes through faster intervention and treatment initiation.
- Cell-free nucleic acid analysis for personalized diagnostics: Cell-free nucleic acid analysis involves detecting and analyzing DNA and RNA fragments circulating in bodily fluids without requiring cell isolation. This approach enables non-invasive detection of genetic abnormalities, mutations, and disease markers for personalized diagnostic applications. The technology is particularly valuable for prenatal testing, cancer detection, and monitoring treatment response, offering insights into disease states with minimal patient discomfort.
- Automated systems for cell-free diagnostic sample processing: Automated systems for cell-free diagnostic sample processing streamline the preparation and analysis of biological specimens. These systems incorporate robotics, microfluidics, and integrated sensors to handle samples with minimal human intervention, reducing contamination risks and processing errors. The automation enables standardized, high-throughput analysis of cell-free samples, making personalized diagnostics more accessible and reliable for clinical applications.
- Cell-free protein expression systems for diagnostic applications: Cell-free protein expression systems enable the synthesis of proteins outside living cells for diagnostic purposes. These systems can rapidly produce biomarkers, antibodies, and other proteins needed for personalized diagnostic tests. The technology allows for quick adaptation to emerging disease variants and patient-specific requirements, facilitating the development of customized diagnostic tools that can detect unique disease signatures or drug response indicators.
02 Cell-free protein synthesis for diagnostic applications
Cell-free protein synthesis systems allow for the production of proteins outside living cells, which can be utilized in diagnostic applications. These systems can rapidly produce specific proteins or enzymes needed for diagnostic tests, enabling personalized diagnostic approaches. The technology offers advantages in speed, scalability, and the ability to produce proteins that might be toxic to living cells, making it valuable for developing personalized diagnostic tools.Expand Specific Solutions03 Cell-free nucleic acid analysis for personalized diagnostics
Cell-free nucleic acid analysis involves detecting and analyzing DNA or RNA fragments circulating in bodily fluids without requiring cell isolation. This approach enables non-invasive diagnostic testing for various conditions including cancer, infectious diseases, and genetic disorders. The technology allows for personalized diagnostic approaches by identifying specific genetic mutations, gene expression patterns, or other nucleic acid biomarkers that can inform individualized treatment decisions.Expand Specific Solutions04 Integrated microfluidic systems for cell-free diagnostics
Integrated microfluidic systems combine multiple laboratory functions on a single chip for cell-free diagnostic applications. These systems enable sample processing, analysis, and detection in a compact format, allowing for point-of-care personalized diagnostics. The technology integrates various components such as sample preparation, reagent mixing, and detection mechanisms to provide rapid, automated diagnostic results tailored to individual patients' needs.Expand Specific Solutions05 AI and data analysis for cell-free diagnostic interpretation
Artificial intelligence and advanced data analysis techniques enhance the interpretation of results from cell-free diagnostic systems. These computational approaches can identify patterns in complex diagnostic data, enabling more accurate personalized diagnostic conclusions. The technology leverages machine learning algorithms to analyze biomarker profiles, genetic information, and other diagnostic indicators, translating raw data into clinically meaningful insights for personalized patient care.Expand Specific Solutions
Leading Organizations in Cell-free Diagnostic Technologies
Cell-free diagnostic systems are revolutionizing personalized medicine, currently transitioning from early development to commercial growth phase. The market is expanding rapidly, projected to reach significant scale as these technologies enable point-of-care testing without traditional laboratory constraints. Leading innovators include GRAIL and Insitro leveraging machine learning for early cancer detection, while established players like Corning and Siemens Healthcare provide essential infrastructure. Academic institutions (Northwestern University, Harvard, Case Western) collaborate with commercial entities (Recursion Pharmaceuticals, Elucid Bioimaging) to advance the technology. The field is characterized by increasing integration of AI/ML capabilities with biological systems, creating diagnostic platforms that can detect disease biomarkers with unprecedented sensitivity and specificity in diverse healthcare settings.
Insitro, Inc.
Technical Solution: Insitro has developed an innovative approach to cell-free diagnostics by combining machine learning with high-throughput experimental methods. Their platform integrates cell-free expression systems with advanced computational models to predict patient responses to treatments and identify novel biomarkers. The company utilizes cell-free protein synthesis to rapidly produce and screen potential disease markers, enabling personalized diagnostic development at unprecedented speed. Their system employs automated laboratory processes to generate massive biological datasets from cell-free samples, which are then analyzed using proprietary machine learning algorithms to identify clinically relevant patterns. Insitro's technology can detect subtle variations in protein expression profiles from minimal biological samples, allowing for early disease detection and monitoring. The platform incorporates feedback loops between computational predictions and experimental validation, continuously improving diagnostic accuracy through iterative refinement. This approach has been particularly valuable for identifying biomarkers in complex conditions where traditional diagnostic methods have limited effectiveness[8][9].
Strengths: Integration of cutting-edge machine learning with biological data generation creates a powerful discovery engine; scalable platform capable of processing thousands of samples; ability to identify novel biomarkers not detectable through conventional methods; rapid iteration capability. Weaknesses: Requires substantial computational infrastructure; complex interpretability of machine learning models may present regulatory challenges; high initial development costs; dependence on high-quality training data.
GRAIL, Inc.
Technical Solution: GRAIL has pioneered cell-free DNA (cfDNA) technology for early cancer detection through their Galleri® multi-cancer early detection test. Their approach analyzes cell-free DNA fragments in blood samples using next-generation sequencing and machine learning algorithms to detect cancer signals across more than 50 types of cancer. The technology identifies methylation patterns in cfDNA that indicate the presence of cancer and can predict the cancer signal origin with high precision. GRAIL's system represents a significant advancement in liquid biopsy diagnostics, enabling detection at earlier stages when treatment may be more effective. Their proprietary methylation-based fragmentation analysis examines over 100,000 methylation sites across the genome, creating a highly personalized diagnostic approach that can be implemented in standard clinical workflows[1][2].
Strengths: Exceptional sensitivity and specificity for early-stage cancer detection across multiple cancer types simultaneously; ability to predict cancer origin with high accuracy; requires only a simple blood draw. Weaknesses: High cost of implementation; requires sophisticated laboratory infrastructure; potential for false positives requiring follow-up diagnostic procedures; limited accessibility in resource-constrained settings.
Key Patents and Breakthroughs
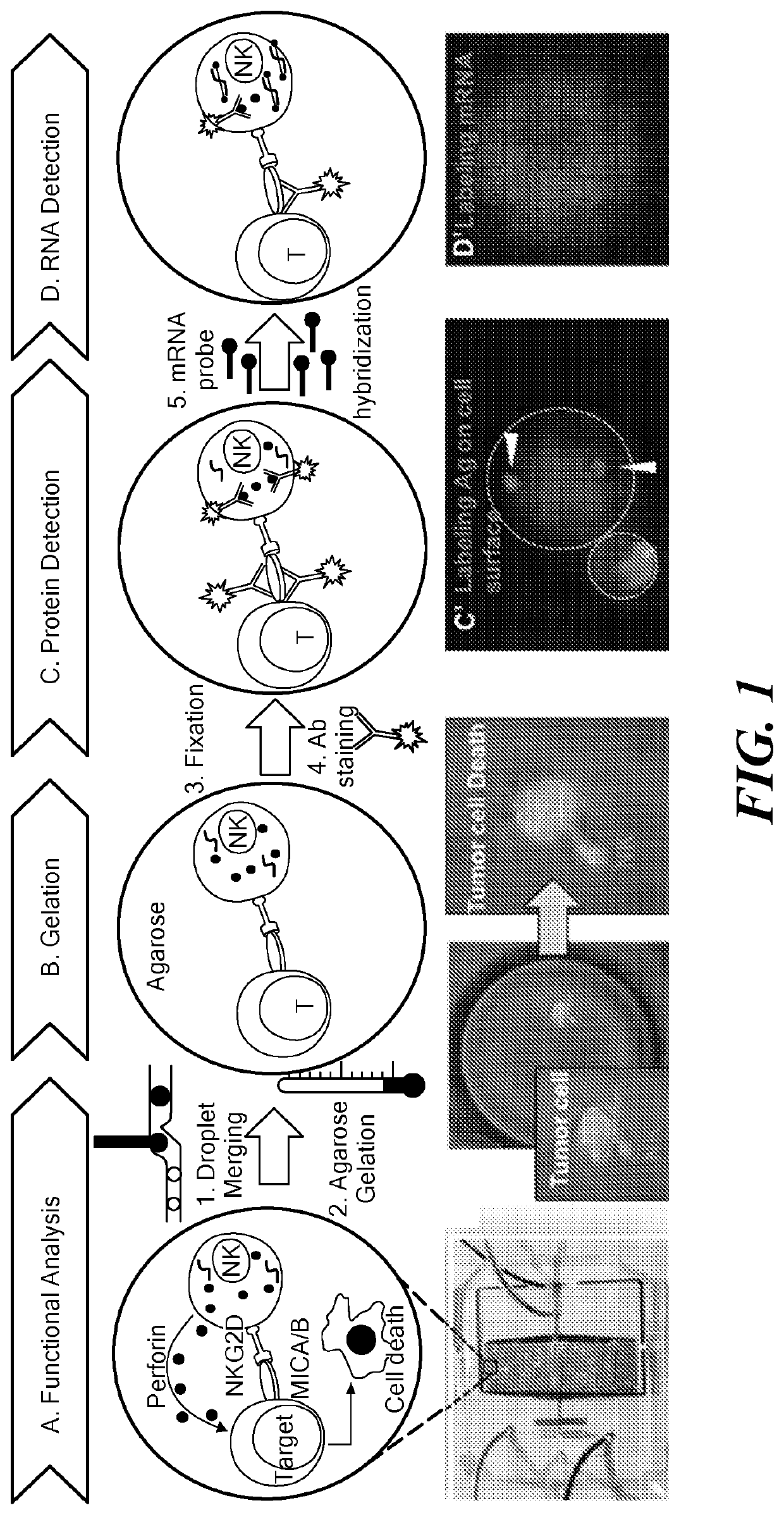
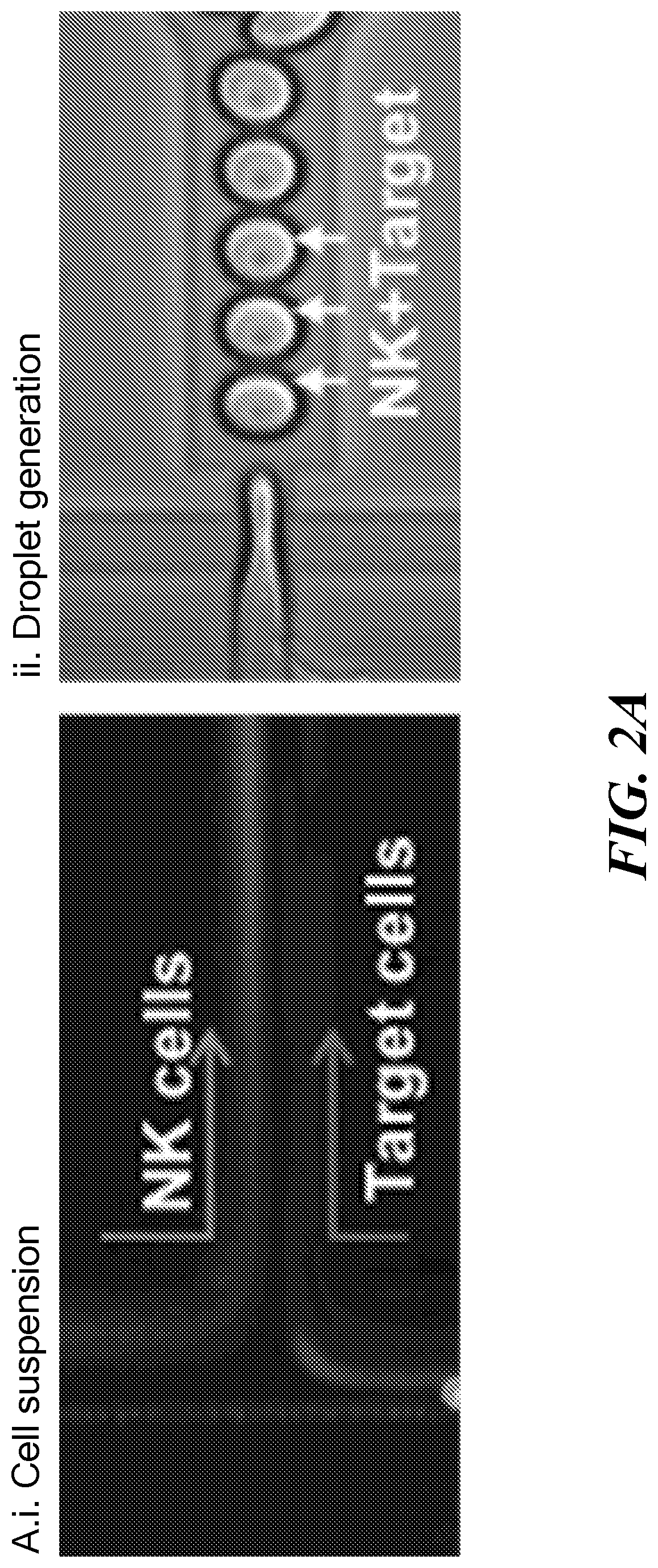
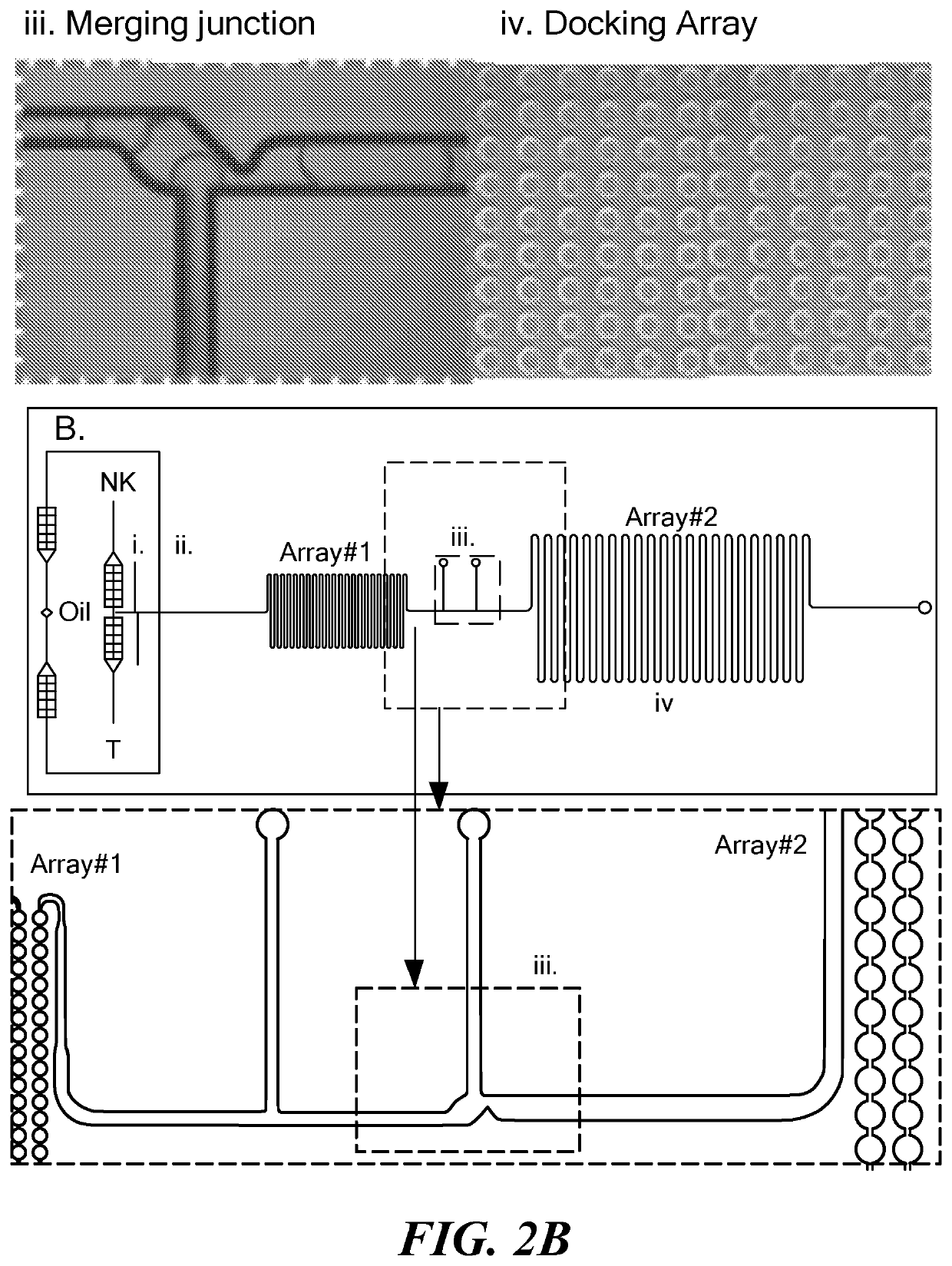
Single Cell Fluorescence in Situ Hybridization in Microfluidic Droplets
PatentActiveUS20210277455A1
Innovation
- A microfluidic device and method for performing on-chip single-molecule fluorescence in situ hybridization in aqueous microdroplets, allowing simultaneous detection of nucleic acid and protein species, and cell function analysis, using a dual-array system to form and analyze microdroplets containing cells within a biocompatible matrix, enabling rapid and automated detection of gene expression in single cells, including non-adherent cells.
Cell-free biosensors with DNA strand displacement circuits
PatentWO2022183102A1
Innovation
- Integration of toehold-mediated DNA strand displacement (TMSD) circuits with cell-free biosensing systems, utilizing allosteric transcription factors and engineered transcription templates to generate detectable signals through RNA polymerase-driven transcription and DNA strand displacement.
Regulatory Framework for Cell-free Diagnostics
The regulatory landscape for cell-free diagnostic systems presents a complex framework that continues to evolve as these innovative technologies advance. In the United States, the Food and Drug Administration (FDA) categorizes most cell-free diagnostic tests as in vitro diagnostic devices (IVDs), requiring premarket approval or clearance depending on their risk classification. Cell-free DNA (cfDNA) tests, particularly those used for non-invasive prenatal testing (NIPT), typically fall under Class II medical devices, necessitating 510(k) clearance with demonstrated substantial equivalence to predicate devices.
The European Union has implemented more stringent regulations through the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous directive in May 2022. This regulation introduces a new risk-based classification system where many cell-free diagnostic tests are classified as Class C (high individual risk) or Class D (high individual and public health risk), requiring notified body assessment and more comprehensive clinical evidence than previously required.
Regulatory challenges specific to cell-free diagnostics include validation of analytical performance across diverse patient populations, standardization of sample collection and processing protocols, and establishment of appropriate quality control measures. The decentralized nature of many cell-free diagnostic applications, particularly those aimed at point-of-care or home use, introduces additional regulatory considerations regarding usability, result interpretation, and data management.
Reimbursement pathways represent another critical regulatory hurdle, with many cell-free diagnostic tests struggling to secure coverage from public and private payers despite clinical utility. The Centers for Medicare & Medicaid Services (CMS) and private insurers typically require substantial evidence of clinical validity and utility before providing coverage, creating a challenging environment for novel cell-free diagnostic technologies.
Global regulatory harmonization efforts, including the International Medical Device Regulators Forum (IMDRF), are working to establish common principles for cell-free diagnostic evaluation, though significant regional differences persist. Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has implemented a Sakigake designation for breakthrough technologies, potentially accelerating approval for innovative cell-free diagnostic approaches.
Privacy regulations present additional complexity, particularly for cell-free diagnostics that analyze genetic information. The General Data Protection Regulation (GDPR) in Europe and various state-level genetic privacy laws in the US impose strict requirements on data handling, consent processes, and information disclosure that developers must navigate alongside traditional medical device regulations.
The European Union has implemented more stringent regulations through the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous directive in May 2022. This regulation introduces a new risk-based classification system where many cell-free diagnostic tests are classified as Class C (high individual risk) or Class D (high individual and public health risk), requiring notified body assessment and more comprehensive clinical evidence than previously required.
Regulatory challenges specific to cell-free diagnostics include validation of analytical performance across diverse patient populations, standardization of sample collection and processing protocols, and establishment of appropriate quality control measures. The decentralized nature of many cell-free diagnostic applications, particularly those aimed at point-of-care or home use, introduces additional regulatory considerations regarding usability, result interpretation, and data management.
Reimbursement pathways represent another critical regulatory hurdle, with many cell-free diagnostic tests struggling to secure coverage from public and private payers despite clinical utility. The Centers for Medicare & Medicaid Services (CMS) and private insurers typically require substantial evidence of clinical validity and utility before providing coverage, creating a challenging environment for novel cell-free diagnostic technologies.
Global regulatory harmonization efforts, including the International Medical Device Regulators Forum (IMDRF), are working to establish common principles for cell-free diagnostic evaluation, though significant regional differences persist. Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has implemented a Sakigake designation for breakthrough technologies, potentially accelerating approval for innovative cell-free diagnostic approaches.
Privacy regulations present additional complexity, particularly for cell-free diagnostics that analyze genetic information. The General Data Protection Regulation (GDPR) in Europe and various state-level genetic privacy laws in the US impose strict requirements on data handling, consent processes, and information disclosure that developers must navigate alongside traditional medical device regulations.
Clinical Implementation Challenges
The integration of cell-free diagnostic systems into clinical practice faces significant implementation barriers despite their promising potential for personalized medicine. Healthcare institutions must navigate complex regulatory frameworks that vary across regions, with FDA approval in the United States requiring extensive validation studies demonstrating both analytical and clinical validity. These regulatory hurdles often extend timelines for clinical adoption by 2-3 years compared to research applications.
Quality control represents another critical challenge, as cell-free diagnostics demand stringent standardization protocols to ensure consistent performance across different clinical settings. The inherent biological variability in patient samples necessitates robust calibration methods and reference standards that remain underdeveloped in many cell-free applications, particularly for complex biomarker panels.
Infrastructure limitations further complicate implementation, with many healthcare facilities lacking the specialized equipment and trained personnel required for cell-free diagnostic workflows. While some platforms aim for point-of-care simplicity, others require sophisticated laboratory setups with molecular biology capabilities that exceed current resources in many clinical environments, especially in resource-limited settings.
Data interpretation presents perhaps the most nuanced challenge, as clinicians must translate complex molecular signatures into actionable treatment decisions. The vast datasets generated by cell-free diagnostics often require advanced bioinformatics support and integration with electronic health records—capabilities that remain inconsistent across healthcare systems. This interpretation gap can lead to underutilization of diagnostic information or potential misapplication in clinical decision-making.
Cost considerations remain paramount, with current cell-free diagnostic technologies carrying significant per-test expenses ranging from $200 to several thousand dollars depending on complexity. Reimbursement pathways remain uncertain for many applications, creating financial disincentives for adoption despite potential long-term cost savings through improved treatment selection and reduced hospitalizations.
Ethical and privacy concerns also merit attention, particularly regarding incidental findings and data ownership. Cell-free diagnostics can reveal unexpected genetic information beyond the primary diagnostic target, raising questions about disclosure obligations and patient consent frameworks that clinical institutions must address through comprehensive policies and procedures.
Quality control represents another critical challenge, as cell-free diagnostics demand stringent standardization protocols to ensure consistent performance across different clinical settings. The inherent biological variability in patient samples necessitates robust calibration methods and reference standards that remain underdeveloped in many cell-free applications, particularly for complex biomarker panels.
Infrastructure limitations further complicate implementation, with many healthcare facilities lacking the specialized equipment and trained personnel required for cell-free diagnostic workflows. While some platforms aim for point-of-care simplicity, others require sophisticated laboratory setups with molecular biology capabilities that exceed current resources in many clinical environments, especially in resource-limited settings.
Data interpretation presents perhaps the most nuanced challenge, as clinicians must translate complex molecular signatures into actionable treatment decisions. The vast datasets generated by cell-free diagnostics often require advanced bioinformatics support and integration with electronic health records—capabilities that remain inconsistent across healthcare systems. This interpretation gap can lead to underutilization of diagnostic information or potential misapplication in clinical decision-making.
Cost considerations remain paramount, with current cell-free diagnostic technologies carrying significant per-test expenses ranging from $200 to several thousand dollars depending on complexity. Reimbursement pathways remain uncertain for many applications, creating financial disincentives for adoption despite potential long-term cost savings through improved treatment selection and reduced hospitalizations.
Ethical and privacy concerns also merit attention, particularly regarding incidental findings and data ownership. Cell-free diagnostics can reveal unexpected genetic information beyond the primary diagnostic target, raising questions about disclosure obligations and patient consent frameworks that clinical institutions must address through comprehensive policies and procedures.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!