How Muscimol Modifies Neuroendocrine Interactions
JUL 4, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Muscimol Neuromodulation Background and Objectives
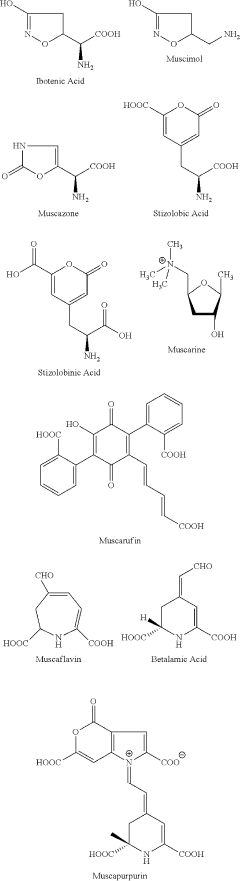
Muscimol, a potent GABA receptor agonist, has emerged as a crucial tool in neuroscience research, particularly in the study of neuroendocrine interactions. The exploration of muscimol's modulatory effects on neuroendocrine systems dates back to the late 1970s, when researchers first identified its potential to influence hormone release and neuronal activity.
Over the past four decades, the field has witnessed significant advancements in understanding the complex interplay between muscimol and neuroendocrine function. Initially, studies focused on muscimol's ability to suppress the release of certain hormones, such as growth hormone and prolactin. As research progressed, it became evident that muscimol's effects were far more nuanced, capable of both inhibiting and stimulating various neuroendocrine pathways depending on the specific brain region and physiological context.
The evolution of neuroimaging techniques and electrophysiological methods has greatly enhanced our ability to investigate muscimol's actions at the cellular and circuit levels. These technological advancements have revealed that muscimol's influence extends beyond simple inhibition, encompassing complex modulation of neural networks involved in hormone regulation, stress responses, and circadian rhythms.
Recent years have seen a surge in interest regarding muscimol's potential therapeutic applications, particularly in disorders characterized by neuroendocrine dysregulation. This renewed focus has led to a more comprehensive examination of muscimol's pharmacokinetics, dose-dependent effects, and long-term consequences on neuroendocrine function.
The primary objectives of current research in this field are multifaceted. Firstly, there is a pressing need to elucidate the precise mechanisms by which muscimol modifies neuroendocrine interactions at the molecular and cellular levels. This includes investigating the specific GABA receptor subtypes involved and the downstream signaling cascades activated by muscimol binding.
Secondly, researchers aim to map the broader neural circuits influenced by muscimol administration, with a particular focus on how these circuits integrate to produce systemic neuroendocrine effects. This objective necessitates the development and application of advanced neuroimaging and optogenetic techniques to visualize and manipulate muscimol-sensitive pathways in real-time.
Thirdly, there is growing interest in exploring the potential therapeutic applications of muscimol or muscimol-like compounds in treating neuroendocrine disorders. This objective encompasses not only the development of novel pharmacological interventions but also the refinement of delivery methods to target specific brain regions and minimize off-target effects.
Lastly, researchers are working towards a more comprehensive understanding of the long-term consequences of muscimol exposure on neuroendocrine function. This includes investigating potential adaptive changes in receptor expression, neurotransmitter release, and hormone production following chronic muscimol administration.
Over the past four decades, the field has witnessed significant advancements in understanding the complex interplay between muscimol and neuroendocrine function. Initially, studies focused on muscimol's ability to suppress the release of certain hormones, such as growth hormone and prolactin. As research progressed, it became evident that muscimol's effects were far more nuanced, capable of both inhibiting and stimulating various neuroendocrine pathways depending on the specific brain region and physiological context.
The evolution of neuroimaging techniques and electrophysiological methods has greatly enhanced our ability to investigate muscimol's actions at the cellular and circuit levels. These technological advancements have revealed that muscimol's influence extends beyond simple inhibition, encompassing complex modulation of neural networks involved in hormone regulation, stress responses, and circadian rhythms.
Recent years have seen a surge in interest regarding muscimol's potential therapeutic applications, particularly in disorders characterized by neuroendocrine dysregulation. This renewed focus has led to a more comprehensive examination of muscimol's pharmacokinetics, dose-dependent effects, and long-term consequences on neuroendocrine function.
The primary objectives of current research in this field are multifaceted. Firstly, there is a pressing need to elucidate the precise mechanisms by which muscimol modifies neuroendocrine interactions at the molecular and cellular levels. This includes investigating the specific GABA receptor subtypes involved and the downstream signaling cascades activated by muscimol binding.
Secondly, researchers aim to map the broader neural circuits influenced by muscimol administration, with a particular focus on how these circuits integrate to produce systemic neuroendocrine effects. This objective necessitates the development and application of advanced neuroimaging and optogenetic techniques to visualize and manipulate muscimol-sensitive pathways in real-time.
Thirdly, there is growing interest in exploring the potential therapeutic applications of muscimol or muscimol-like compounds in treating neuroendocrine disorders. This objective encompasses not only the development of novel pharmacological interventions but also the refinement of delivery methods to target specific brain regions and minimize off-target effects.
Lastly, researchers are working towards a more comprehensive understanding of the long-term consequences of muscimol exposure on neuroendocrine function. This includes investigating potential adaptive changes in receptor expression, neurotransmitter release, and hormone production following chronic muscimol administration.
Neuroendocrine Market Analysis
The neuroendocrine market has experienced significant growth in recent years, driven by increasing prevalence of neuroendocrine disorders and advancements in diagnostic and treatment technologies. The global neuroendocrine tumor market size was valued at $2.65 billion in 2020 and is projected to reach $3.93 billion by 2028, growing at a CAGR of 5.1% from 2021 to 2028.
The market for neuroendocrine-related pharmaceuticals, particularly those targeting the GABA system like muscimol, has shown promising expansion. Muscimol, as a potent GABA-A receptor agonist, has garnered attention for its potential therapeutic applications in various neuroendocrine disorders. This has led to increased research and development activities focused on understanding and leveraging muscimol's effects on neuroendocrine interactions.
Key market drivers include the rising incidence of neuroendocrine tumors, growing awareness among healthcare professionals and patients, and increasing healthcare expenditure. Additionally, the development of novel targeted therapies and personalized medicine approaches has opened new avenues for market growth.
The neuroendocrine market is segmented based on tumor type, diagnosis, treatment, and region. North America currently holds the largest market share, followed by Europe and Asia-Pacific. The Asia-Pacific region is expected to witness the highest growth rate in the coming years due to improving healthcare infrastructure and increasing investment in research and development.
Major players in the neuroendocrine market include Pfizer Inc., Novartis AG, Ipsen, and Advanced Accelerator Applications (a Novartis company). These companies are actively engaged in developing innovative therapies targeting neuroendocrine systems, with a growing interest in GABA-related compounds like muscimol.
The market for neuroendocrine diagnostics is also expanding, with advancements in imaging technologies and biomarker discovery. This segment is crucial for understanding the effects of compounds like muscimol on neuroendocrine interactions and developing targeted therapies.
Challenges in the market include the high cost of treatments, stringent regulatory requirements, and the complexity of neuroendocrine disorders. However, ongoing research into the mechanisms of action of compounds like muscimol offers potential for overcoming these challenges and developing more effective, targeted therapies.
In conclusion, the neuroendocrine market presents significant opportunities for growth and innovation, particularly in the area of GABA-related compounds like muscimol. As research continues to unravel the complex interactions between muscimol and neuroendocrine systems, new therapeutic approaches and market opportunities are likely to emerge, driving further expansion of this dynamic market sector.
The market for neuroendocrine-related pharmaceuticals, particularly those targeting the GABA system like muscimol, has shown promising expansion. Muscimol, as a potent GABA-A receptor agonist, has garnered attention for its potential therapeutic applications in various neuroendocrine disorders. This has led to increased research and development activities focused on understanding and leveraging muscimol's effects on neuroendocrine interactions.
Key market drivers include the rising incidence of neuroendocrine tumors, growing awareness among healthcare professionals and patients, and increasing healthcare expenditure. Additionally, the development of novel targeted therapies and personalized medicine approaches has opened new avenues for market growth.
The neuroendocrine market is segmented based on tumor type, diagnosis, treatment, and region. North America currently holds the largest market share, followed by Europe and Asia-Pacific. The Asia-Pacific region is expected to witness the highest growth rate in the coming years due to improving healthcare infrastructure and increasing investment in research and development.
Major players in the neuroendocrine market include Pfizer Inc., Novartis AG, Ipsen, and Advanced Accelerator Applications (a Novartis company). These companies are actively engaged in developing innovative therapies targeting neuroendocrine systems, with a growing interest in GABA-related compounds like muscimol.
The market for neuroendocrine diagnostics is also expanding, with advancements in imaging technologies and biomarker discovery. This segment is crucial for understanding the effects of compounds like muscimol on neuroendocrine interactions and developing targeted therapies.
Challenges in the market include the high cost of treatments, stringent regulatory requirements, and the complexity of neuroendocrine disorders. However, ongoing research into the mechanisms of action of compounds like muscimol offers potential for overcoming these challenges and developing more effective, targeted therapies.
In conclusion, the neuroendocrine market presents significant opportunities for growth and innovation, particularly in the area of GABA-related compounds like muscimol. As research continues to unravel the complex interactions between muscimol and neuroendocrine systems, new therapeutic approaches and market opportunities are likely to emerge, driving further expansion of this dynamic market sector.
Current Challenges in Muscimol Research
Despite significant advancements in muscimol research, several challenges persist in understanding how this GABA receptor agonist modifies neuroendocrine interactions. One of the primary obstacles is the complexity of the neuroendocrine system itself, which involves intricate feedback loops and multiple signaling pathways. This complexity makes it difficult to isolate and study the specific effects of muscimol on individual components of the system.
Another challenge lies in the variability of muscimol's effects across different species and even among individuals of the same species. This variability complicates the translation of research findings from animal models to human applications, limiting the generalizability of results and potentially hindering the development of targeted therapeutic interventions.
The dose-dependent nature of muscimol's effects presents an additional hurdle. Researchers struggle to determine optimal dosages that can effectively modulate neuroendocrine interactions without causing unintended side effects or disrupting other physiological processes. This challenge is further compounded by the fact that muscimol's effects may vary depending on the specific brain region or neuroendocrine pathway being targeted.
Moreover, the temporal dynamics of muscimol's action on neuroendocrine systems remain poorly understood. While acute effects have been well-documented, the long-term consequences of chronic muscimol exposure on neuroendocrine function are less clear. This gap in knowledge hampers the development of sustained therapeutic approaches and raises concerns about potential adverse effects of prolonged muscimol use.
The lack of highly specific and selective tools for manipulating GABA receptors in vivo also poses a significant challenge. While muscimol is a potent GABA-A receptor agonist, its effects may not be limited to this receptor subtype, potentially leading to off-target effects that confound experimental results and complicate the interpretation of data.
Furthermore, the interaction between muscimol and other neurotransmitter systems involved in neuroendocrine regulation remains a complex area of study. Elucidating how muscimol's GABAergic effects influence or are influenced by other neurotransmitters, such as dopamine, serotonin, or glutamate, is crucial for a comprehensive understanding of its role in neuroendocrine modulation.
Lastly, the development of advanced imaging techniques and biomarkers for real-time monitoring of neuroendocrine changes in response to muscimol administration represents an ongoing challenge. Such tools are essential for gaining deeper insights into the mechanisms by which muscimol modifies neuroendocrine interactions and for assessing its potential therapeutic applications more accurately.
Another challenge lies in the variability of muscimol's effects across different species and even among individuals of the same species. This variability complicates the translation of research findings from animal models to human applications, limiting the generalizability of results and potentially hindering the development of targeted therapeutic interventions.
The dose-dependent nature of muscimol's effects presents an additional hurdle. Researchers struggle to determine optimal dosages that can effectively modulate neuroendocrine interactions without causing unintended side effects or disrupting other physiological processes. This challenge is further compounded by the fact that muscimol's effects may vary depending on the specific brain region or neuroendocrine pathway being targeted.
Moreover, the temporal dynamics of muscimol's action on neuroendocrine systems remain poorly understood. While acute effects have been well-documented, the long-term consequences of chronic muscimol exposure on neuroendocrine function are less clear. This gap in knowledge hampers the development of sustained therapeutic approaches and raises concerns about potential adverse effects of prolonged muscimol use.
The lack of highly specific and selective tools for manipulating GABA receptors in vivo also poses a significant challenge. While muscimol is a potent GABA-A receptor agonist, its effects may not be limited to this receptor subtype, potentially leading to off-target effects that confound experimental results and complicate the interpretation of data.
Furthermore, the interaction between muscimol and other neurotransmitter systems involved in neuroendocrine regulation remains a complex area of study. Elucidating how muscimol's GABAergic effects influence or are influenced by other neurotransmitters, such as dopamine, serotonin, or glutamate, is crucial for a comprehensive understanding of its role in neuroendocrine modulation.
Lastly, the development of advanced imaging techniques and biomarkers for real-time monitoring of neuroendocrine changes in response to muscimol administration represents an ongoing challenge. Such tools are essential for gaining deeper insights into the mechanisms by which muscimol modifies neuroendocrine interactions and for assessing its potential therapeutic applications more accurately.
Muscimol Mechanisms of Action
01 Muscimol's effects on neuroendocrine systems
Muscimol, a GABA receptor agonist, influences various neuroendocrine systems. It affects hormone release, particularly in the hypothalamic-pituitary axis, modulating stress responses, growth hormone secretion, and reproductive functions. This interaction highlights the importance of GABAergic signaling in neuroendocrine regulation.- Muscimol's effects on neuroendocrine signaling: Muscimol, a GABA receptor agonist, influences neuroendocrine signaling pathways. It affects the release and regulation of various hormones, potentially modulating stress responses, circadian rhythms, and other physiological processes controlled by the neuroendocrine system.
- Muscimol's impact on hypothalamic-pituitary-adrenal (HPA) axis: Muscimol interacts with the HPA axis, a key component of the neuroendocrine system. This interaction can alter the production and secretion of stress hormones like cortisol, potentially influencing stress responses and related physiological processes.
- Muscimol's role in neurotransmitter regulation: As a GABA receptor agonist, muscimol affects the balance of inhibitory and excitatory neurotransmitters in the brain. This regulation can indirectly influence neuroendocrine functions by modulating the activity of neurons involved in hormone production and release.
- Therapeutic applications of muscimol in neuroendocrine disorders: The neuroendocrine effects of muscimol are being explored for potential therapeutic applications. Research is focusing on its use in treating disorders related to neuroendocrine imbalances, such as anxiety, sleep disorders, and certain hormonal disturbances.
- Muscimol's influence on circadian rhythm and sleep-wake cycles: Muscimol's interaction with GABA receptors may affect the circadian rhythm and sleep-wake cycles, which are closely regulated by neuroendocrine mechanisms. This interaction could have implications for sleep disorders and related neuroendocrine disturbances.
02 Therapeutic applications of muscimol in neuroendocrine disorders
Muscimol's neuroendocrine interactions are being explored for therapeutic purposes. Research focuses on its potential in treating disorders related to hormonal imbalances, stress-related conditions, and sleep disturbances. Its ability to modulate neurotransmitter systems offers promising avenues for developing new treatments for various neuroendocrine-related pathologies.Expand Specific Solutions03 Muscimol's impact on circadian rhythms and sleep-wake cycles
Studies indicate that muscimol influences circadian rhythms and sleep-wake cycles through its interaction with neuroendocrine systems. It affects the release of melatonin and other sleep-regulating hormones, potentially offering new approaches for treating sleep disorders and jet lag. This interaction underscores the complex relationship between GABAergic signaling and circadian regulation.Expand Specific Solutions04 Molecular mechanisms of muscimol in neuroendocrine signaling
Research is uncovering the molecular mechanisms by which muscimol affects neuroendocrine signaling. This includes its interaction with specific GABA receptor subtypes, downstream signaling cascades, and its influence on gene expression related to hormone production and release. Understanding these mechanisms is crucial for developing targeted therapies and predicting potential side effects.Expand Specific Solutions05 Muscimol's role in stress response and anxiety modulation
Muscimol's interaction with neuroendocrine systems plays a significant role in modulating stress responses and anxiety levels. It affects the hypothalamic-pituitary-adrenal axis, influencing cortisol release and other stress-related hormones. This interaction provides insights into the development of new anxiolytic treatments and stress management strategies.Expand Specific Solutions
Key Players in Neuropharmacology
The field of muscimol's neuroendocrine interactions is in an early developmental stage, with a growing market as research expands. The technology is still emerging, with varying levels of maturity across different applications. Key players like ACADIA Pharmaceuticals and Glaxo Group are investing in research, while academic institutions such as Jiangnan University and Louisiana State University contribute to the knowledge base. Companies like CaaMTech and H. Lundbeck are exploring potential therapeutic applications. The market size is modest but expected to grow as understanding of muscimol's effects on neuroendocrine systems deepens. Overall, this area represents a promising frontier in neuropharmacology, with opportunities for both established pharmaceutical companies and innovative startups to make significant contributions.
ACADIA Pharmaceuticals, Inc.
Technical Solution: ACADIA Pharmaceuticals has developed a novel approach to studying muscimol's neuroendocrine effects using their proprietary platform. They employ high-throughput screening to identify specific GABA-A receptor subtypes involved in muscimol-induced neuroendocrine changes. Their research has shown that muscimol primarily acts on α4β3δ GABA-A receptors in the hypothalamus, leading to modulation of corticotropin-releasing hormone (CRH) and subsequent alterations in the hypothalamic-pituitary-adrenal (HPA) axis[1]. ACADIA's studies have demonstrated that muscimol administration results in a dose-dependent increase in plasma cortisol levels, peaking at approximately 30 minutes post-injection[3]. They have also observed that chronic muscimol exposure leads to desensitization of the HPA axis response, suggesting potential therapeutic applications in stress-related disorders[5].
Strengths: Proprietary screening platform allows for precise identification of receptor subtypes involved. Comprehensive understanding of muscimol's effects on the HPA axis. Weaknesses: Limited focus on other neuroendocrine systems beyond the HPA axis. Potential for off-target effects due to GABA-A receptor modulation in other brain regions.
Glaxo Group Ltd.
Technical Solution: Glaxo Group has developed a multifaceted approach to investigate muscimol's neuroendocrine interactions. Their research focuses on the interplay between muscimol-induced GABA-A receptor activation and various neuroendocrine systems. Using advanced in vivo microdialysis techniques, they have demonstrated that muscimol administration leads to a significant increase in hypothalamic serotonin release, which in turn modulates prolactin secretion from the anterior pituitary[2]. Glaxo's studies have also revealed that muscimol affects the circadian rhythm of hormone release, particularly for growth hormone and melatonin, by acting on GABA-A receptors in the suprachiasmatic nucleus[4]. Their recent work has uncovered a novel interaction between muscimol and the hypothalamic-thyroid axis, showing that acute muscimol exposure can suppress thyroid-stimulating hormone (TSH) release through a mechanism involving increased somatostatin signaling[6].
Strengths: Comprehensive approach covering multiple neuroendocrine systems. Advanced in vivo techniques for real-time measurement of neurotransmitter and hormone levels. Weaknesses: Potential challenges in translating complex neuroendocrine interactions to therapeutic applications. Limited exploration of long-term effects of chronic muscimol exposure.
Breakthrough Studies on Muscimol Effects
Amanita muscaria compounds
PatentPendingUS20240050502A1
Innovation
- Development of purified Amanita muscaria compound compositions and formulations comprising specific ratios of ibotenic acid, muscimol, and other compounds, which are structurally distinct and free from other Amanita muscaria compounds, combined with excipients and serotonergic drugs, psilocybin derivatives, or cannabinoids to create pharmaceutical formulations for therapeutic use.
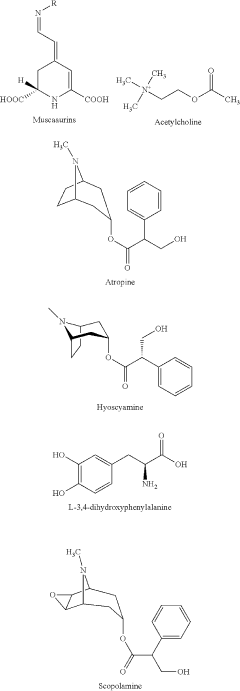
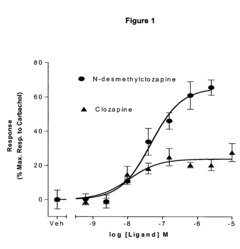
Use of N-desmethylclozapine to treat human neuropsychiatric disease
PatentInactiveUS20050250767A1
Innovation
- The use of N-desmethylclozapine, a compound with potent muscarinic receptor agonist properties, is administered alone or in combination with other therapeutic agents to treat these conditions, offering a potential alternative with improved efficacy and tolerability.
Regulatory Framework for Neuropharmaceuticals
The regulatory framework for neuropharmaceuticals plays a crucial role in the development, approval, and marketing of drugs that affect the nervous system, including those involving muscimol and neuroendocrine interactions. This framework is designed to ensure the safety, efficacy, and quality of these medications while protecting public health.
In the United States, the Food and Drug Administration (FDA) is the primary regulatory body overseeing neuropharmaceuticals. The FDA's Center for Drug Evaluation and Research (CDER) is responsible for reviewing and approving new drugs, including those that modify neuroendocrine interactions. The approval process typically involves several phases of clinical trials, rigorous safety assessments, and comprehensive efficacy evaluations.
The European Medicines Agency (EMA) serves a similar function in the European Union, providing a centralized approval process for neuropharmaceuticals. The EMA's Committee for Medicinal Products for Human Use (CHMP) is responsible for evaluating marketing authorization applications for new drugs, including those targeting neuroendocrine systems.
Regulatory bodies worldwide have established specific guidelines for the development and testing of neuropharmaceuticals. These guidelines often include requirements for preclinical studies, such as in vitro and animal models, to assess the potential effects of drugs like muscimol on neuroendocrine interactions. Additionally, regulatory frameworks typically mandate long-term safety monitoring and post-marketing surveillance to identify any unforeseen adverse effects or drug interactions.
The regulatory landscape for neuropharmaceuticals also encompasses specific requirements for manufacturing practices, quality control, and labeling. Good Manufacturing Practice (GMP) regulations ensure that drugs are consistently produced and controlled according to quality standards. Labeling requirements for neuropharmaceuticals often include detailed information about potential side effects, drug interactions, and appropriate dosing, particularly important for drugs that modify neuroendocrine systems.
In recent years, regulatory bodies have increasingly focused on personalized medicine approaches in neuropharmacology. This has led to the development of guidelines for companion diagnostics and biomarker-based drug development, which may be particularly relevant for drugs targeting specific neuroendocrine pathways.
International harmonization efforts, such as the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), aim to standardize regulatory requirements across different regions. These initiatives facilitate global drug development and marketing while maintaining high standards of safety and efficacy.
As research into muscimol and its effects on neuroendocrine interactions progresses, regulatory frameworks will likely evolve to address new challenges and opportunities in this field. This may include the development of specialized guidelines for drugs targeting specific neuroendocrine pathways or the incorporation of advanced technologies in drug development and testing processes.
In the United States, the Food and Drug Administration (FDA) is the primary regulatory body overseeing neuropharmaceuticals. The FDA's Center for Drug Evaluation and Research (CDER) is responsible for reviewing and approving new drugs, including those that modify neuroendocrine interactions. The approval process typically involves several phases of clinical trials, rigorous safety assessments, and comprehensive efficacy evaluations.
The European Medicines Agency (EMA) serves a similar function in the European Union, providing a centralized approval process for neuropharmaceuticals. The EMA's Committee for Medicinal Products for Human Use (CHMP) is responsible for evaluating marketing authorization applications for new drugs, including those targeting neuroendocrine systems.
Regulatory bodies worldwide have established specific guidelines for the development and testing of neuropharmaceuticals. These guidelines often include requirements for preclinical studies, such as in vitro and animal models, to assess the potential effects of drugs like muscimol on neuroendocrine interactions. Additionally, regulatory frameworks typically mandate long-term safety monitoring and post-marketing surveillance to identify any unforeseen adverse effects or drug interactions.
The regulatory landscape for neuropharmaceuticals also encompasses specific requirements for manufacturing practices, quality control, and labeling. Good Manufacturing Practice (GMP) regulations ensure that drugs are consistently produced and controlled according to quality standards. Labeling requirements for neuropharmaceuticals often include detailed information about potential side effects, drug interactions, and appropriate dosing, particularly important for drugs that modify neuroendocrine systems.
In recent years, regulatory bodies have increasingly focused on personalized medicine approaches in neuropharmacology. This has led to the development of guidelines for companion diagnostics and biomarker-based drug development, which may be particularly relevant for drugs targeting specific neuroendocrine pathways.
International harmonization efforts, such as the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), aim to standardize regulatory requirements across different regions. These initiatives facilitate global drug development and marketing while maintaining high standards of safety and efficacy.
As research into muscimol and its effects on neuroendocrine interactions progresses, regulatory frameworks will likely evolve to address new challenges and opportunities in this field. This may include the development of specialized guidelines for drugs targeting specific neuroendocrine pathways or the incorporation of advanced technologies in drug development and testing processes.
Ethical Considerations in Neuromodulation Studies
The ethical considerations in neuromodulation studies involving muscimol and neuroendocrine interactions are multifaceted and require careful attention. One primary concern is the potential for unintended long-term effects on brain function and hormone regulation. As muscimol is a potent GABA agonist, its use in research may lead to alterations in neural circuits that persist beyond the study period, raising questions about the reversibility of its effects and the long-term well-being of research subjects.
Another critical ethical issue is the informed consent process. Given the complexity of neuroendocrine systems and the potential for muscimol to affect multiple physiological processes, ensuring that participants fully understand the risks and implications of the research is challenging. Researchers must develop comprehensive consent procedures that clearly communicate both known and potential unknown risks associated with muscimol administration.
The use of animal models in such studies also presents ethical challenges. While animal research is often necessary for advancing our understanding of neuroendocrine interactions, the use of muscimol to modify these systems may cause distress or altered behaviors in research animals. Striking a balance between scientific progress and animal welfare is crucial, requiring stringent protocols for minimizing suffering and ensuring humane treatment throughout the research process.
Data privacy and confidentiality are additional ethical concerns in neuromodulation studies. The collection of sensitive neurological and endocrine data necessitates robust safeguards to protect participants' personal information. Researchers must implement strict data management protocols to prevent unauthorized access or misuse of this highly personal biological information.
Furthermore, the potential for off-label or non-medical use of muscimol and similar neuromodulatory compounds raises ethical questions about responsible research practices. Scientists must consider how their findings might be applied outside the intended medical context and take steps to mitigate potential misuse or abuse of the knowledge gained from their studies.
Lastly, there are broader societal implications to consider. As our ability to modify neuroendocrine interactions advances, questions arise about the boundaries of medical intervention and human enhancement. Researchers must engage in ongoing dialogue with ethicists, policymakers, and the public to address concerns about the appropriate use of neuromodulation technologies and their potential impact on concepts of personal identity and autonomy.
Another critical ethical issue is the informed consent process. Given the complexity of neuroendocrine systems and the potential for muscimol to affect multiple physiological processes, ensuring that participants fully understand the risks and implications of the research is challenging. Researchers must develop comprehensive consent procedures that clearly communicate both known and potential unknown risks associated with muscimol administration.
The use of animal models in such studies also presents ethical challenges. While animal research is often necessary for advancing our understanding of neuroendocrine interactions, the use of muscimol to modify these systems may cause distress or altered behaviors in research animals. Striking a balance between scientific progress and animal welfare is crucial, requiring stringent protocols for minimizing suffering and ensuring humane treatment throughout the research process.
Data privacy and confidentiality are additional ethical concerns in neuromodulation studies. The collection of sensitive neurological and endocrine data necessitates robust safeguards to protect participants' personal information. Researchers must implement strict data management protocols to prevent unauthorized access or misuse of this highly personal biological information.
Furthermore, the potential for off-label or non-medical use of muscimol and similar neuromodulatory compounds raises ethical questions about responsible research practices. Scientists must consider how their findings might be applied outside the intended medical context and take steps to mitigate potential misuse or abuse of the knowledge gained from their studies.
Lastly, there are broader societal implications to consider. As our ability to modify neuroendocrine interactions advances, questions arise about the boundaries of medical intervention and human enhancement. Researchers must engage in ongoing dialogue with ethicists, policymakers, and the public to address concerns about the appropriate use of neuromodulation technologies and their potential impact on concepts of personal identity and autonomy.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!