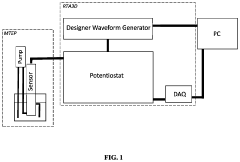
How to Analyze Tocopherol and Oryzanol Levels in Rice Bran Oil — HPLC Protocol
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Rice Bran Oil Analysis Background and Objectives
Rice bran oil (RBO) has emerged as a significant functional food oil over the past three decades, gaining prominence for its exceptional nutritional profile and health-promoting properties. The oil, extracted from the outer layer of rice grains, contains a unique composition of bioactive compounds, with tocopherols (vitamin E) and γ-oryzanol being particularly noteworthy for their antioxidant capabilities and potential health benefits.
The analysis of tocopherol and oryzanol levels in rice bran oil represents a critical aspect of quality control and nutritional assessment in the edible oil industry. Historically, various analytical methods have been employed, but High-Performance Liquid Chromatography (HPLC) has established itself as the gold standard due to its superior sensitivity, specificity, and reproducibility in quantifying these compounds.
The evolution of analytical techniques for RBO components has progressed significantly since the 1980s, when basic column chromatography methods were predominant. The 1990s saw the introduction of HPLC techniques specifically optimized for rice bran oil analysis, while the 2000s brought advancements in detection systems and column technology. Recent years have witnessed the integration of mass spectrometry with HPLC, enabling more precise identification and quantification of minor components.
The primary objective of developing standardized HPLC protocols for tocopherol and oryzanol analysis is to establish reliable, reproducible methodologies that can be implemented across research institutions and quality control laboratories in the food industry. Such standardization is essential for ensuring consistent product quality, facilitating regulatory compliance, and enabling meaningful comparison of research results across different studies.
Additionally, accurate quantification of these bioactive compounds is crucial for evaluating the nutritional value of rice bran oil products, validating health claims, and optimizing processing conditions to preserve these beneficial components. The stability of tocopherols and oryzanol during storage and processing represents another key area of investigation, as these compounds are susceptible to degradation under certain conditions.
From a market perspective, the growing consumer demand for functional foods with demonstrated health benefits has intensified interest in precise analytical methods for bioactive compounds. The ability to accurately measure and guarantee specific levels of tocopherols and oryzanol provides manufacturers with a competitive advantage in the premium edible oil segment.
The technical goals of this investigation include developing a robust HPLC protocol with optimized mobile phase composition, column selection, detection parameters, and sample preparation techniques specifically tailored for simultaneous analysis of tocopherols and oryzanol in rice bran oil matrices, while minimizing interference from other oil components.
The analysis of tocopherol and oryzanol levels in rice bran oil represents a critical aspect of quality control and nutritional assessment in the edible oil industry. Historically, various analytical methods have been employed, but High-Performance Liquid Chromatography (HPLC) has established itself as the gold standard due to its superior sensitivity, specificity, and reproducibility in quantifying these compounds.
The evolution of analytical techniques for RBO components has progressed significantly since the 1980s, when basic column chromatography methods were predominant. The 1990s saw the introduction of HPLC techniques specifically optimized for rice bran oil analysis, while the 2000s brought advancements in detection systems and column technology. Recent years have witnessed the integration of mass spectrometry with HPLC, enabling more precise identification and quantification of minor components.
The primary objective of developing standardized HPLC protocols for tocopherol and oryzanol analysis is to establish reliable, reproducible methodologies that can be implemented across research institutions and quality control laboratories in the food industry. Such standardization is essential for ensuring consistent product quality, facilitating regulatory compliance, and enabling meaningful comparison of research results across different studies.
Additionally, accurate quantification of these bioactive compounds is crucial for evaluating the nutritional value of rice bran oil products, validating health claims, and optimizing processing conditions to preserve these beneficial components. The stability of tocopherols and oryzanol during storage and processing represents another key area of investigation, as these compounds are susceptible to degradation under certain conditions.
From a market perspective, the growing consumer demand for functional foods with demonstrated health benefits has intensified interest in precise analytical methods for bioactive compounds. The ability to accurately measure and guarantee specific levels of tocopherols and oryzanol provides manufacturers with a competitive advantage in the premium edible oil segment.
The technical goals of this investigation include developing a robust HPLC protocol with optimized mobile phase composition, column selection, detection parameters, and sample preparation techniques specifically tailored for simultaneous analysis of tocopherols and oryzanol in rice bran oil matrices, while minimizing interference from other oil components.
Market Demand for Tocopherol and Oryzanol Analysis
The global market for rice bran oil analysis, particularly focusing on tocopherol and oryzanol content, has witnessed significant growth driven by increasing consumer awareness about health benefits of these bioactive compounds. Rice bran oil, rich in antioxidants like tocopherols (vitamin E) and gamma-oryzanol, has gained prominence in both food and pharmaceutical industries, creating substantial demand for reliable analytical methods.
The nutraceutical sector represents the largest market segment requiring tocopherol and oryzanol analysis, with an expanding consumer base seeking functional foods and supplements with proven health benefits. Research indicates that the global functional food market continues to grow steadily, with antioxidant-rich products commanding premium pricing and market positioning.
Food quality control laboratories constitute another significant market segment, as manufacturers increasingly emphasize nutritional content verification and product differentiation based on bioactive compound levels. The rising trend of clean-label products has further accelerated demand for precise quantification of natural antioxidants like tocopherols and oryzanol in rice bran oil.
Regulatory compliance represents a critical driver for analytical method demand. Various international food safety authorities have established guidelines for nutritional labeling that require accurate quantification of vitamin content, including tocopherols. This regulatory framework necessitates reliable analytical protocols that can be standardized across different laboratories and jurisdictions.
The pharmaceutical and cosmetic industries have also emerged as significant markets for tocopherol and oryzanol analysis, incorporating these compounds into various formulations for their antioxidant and skin-protective properties. The growing natural cosmetics sector particularly values rice bran derivatives with verified bioactive compound content.
Academic and research institutions form another substantial market segment, conducting studies on the health benefits of these compounds and developing improved extraction and analysis methodologies. The number of published research papers focusing on rice bran bioactives has increased substantially over the past decade, indicating growing scientific interest.
Geographically, Asia-Pacific represents the largest market for rice bran oil analysis, given the region's dominance in rice production and traditional usage of rice bran oil. However, North America and Europe show the fastest growth rates as awareness of rice bran oil benefits expands in these regions, creating new market opportunities for analytical service providers and equipment manufacturers.
The nutraceutical sector represents the largest market segment requiring tocopherol and oryzanol analysis, with an expanding consumer base seeking functional foods and supplements with proven health benefits. Research indicates that the global functional food market continues to grow steadily, with antioxidant-rich products commanding premium pricing and market positioning.
Food quality control laboratories constitute another significant market segment, as manufacturers increasingly emphasize nutritional content verification and product differentiation based on bioactive compound levels. The rising trend of clean-label products has further accelerated demand for precise quantification of natural antioxidants like tocopherols and oryzanol in rice bran oil.
Regulatory compliance represents a critical driver for analytical method demand. Various international food safety authorities have established guidelines for nutritional labeling that require accurate quantification of vitamin content, including tocopherols. This regulatory framework necessitates reliable analytical protocols that can be standardized across different laboratories and jurisdictions.
The pharmaceutical and cosmetic industries have also emerged as significant markets for tocopherol and oryzanol analysis, incorporating these compounds into various formulations for their antioxidant and skin-protective properties. The growing natural cosmetics sector particularly values rice bran derivatives with verified bioactive compound content.
Academic and research institutions form another substantial market segment, conducting studies on the health benefits of these compounds and developing improved extraction and analysis methodologies. The number of published research papers focusing on rice bran bioactives has increased substantially over the past decade, indicating growing scientific interest.
Geographically, Asia-Pacific represents the largest market for rice bran oil analysis, given the region's dominance in rice production and traditional usage of rice bran oil. However, North America and Europe show the fastest growth rates as awareness of rice bran oil benefits expands in these regions, creating new market opportunities for analytical service providers and equipment manufacturers.
Current HPLC Methodologies and Limitations
High-Performance Liquid Chromatography (HPLC) has emerged as the gold standard for quantitative analysis of tocopherols and oryzanol in rice bran oil due to its precision, sensitivity, and reproducibility. Current methodologies typically employ reverse-phase HPLC with C18 columns, which provide excellent separation of these compounds based on their polarity differences.
The most widely adopted protocol utilizes UV detection at wavelengths of 290-295 nm for tocopherols and 325 nm for oryzanol components. Fluorescence detection offers enhanced sensitivity for tocopherols, with excitation at 290-295 nm and emission at 330 nm. This dual detection approach allows for comprehensive profiling of both compound classes in a single analytical run.
Mobile phase composition represents a critical parameter in these analyses, with most methods employing gradient elution systems. Typical mobile phases consist of methanol, acetonitrile, or their mixtures with small additions of water. The addition of 0.1-0.5% acetic acid often improves peak resolution by suppressing ionization of analytes.
Sample preparation protocols generally involve simple dilution of rice bran oil in appropriate solvents like hexane or isopropanol, followed by filtration through 0.45 μm membrane filters. This straightforward approach minimizes analyte loss while ensuring chromatographic column protection.
Despite these established methodologies, several limitations persist in current HPLC protocols. Run times typically range from 20-40 minutes, limiting sample throughput in industrial quality control settings. The requirement for high-purity organic solvents also raises environmental and cost concerns, particularly for routine analyses.
Matrix effects present another significant challenge, as rice bran oil contains numerous compounds that can interfere with detection or alter retention times. This necessitates careful optimization of extraction procedures and chromatographic conditions for each specific oil source.
Quantification accuracy remains problematic due to the limited availability of certified reference standards, particularly for individual oryzanol components. Most laboratories rely on total oryzanol quantification using ferulic acid as a surrogate standard, which introduces potential quantification errors.
Instrument sensitivity limitations can also impact analysis of samples with low tocopherol or oryzanol concentrations, such as highly refined oils or processed products. This necessitates concentration steps that can introduce additional variability into the analytical workflow.
Method transferability between different HPLC systems and laboratories represents another persistent challenge, with variations in column chemistry, detector sensitivity, and data processing algorithms contributing to inconsistent results across different analytical platforms.
The most widely adopted protocol utilizes UV detection at wavelengths of 290-295 nm for tocopherols and 325 nm for oryzanol components. Fluorescence detection offers enhanced sensitivity for tocopherols, with excitation at 290-295 nm and emission at 330 nm. This dual detection approach allows for comprehensive profiling of both compound classes in a single analytical run.
Mobile phase composition represents a critical parameter in these analyses, with most methods employing gradient elution systems. Typical mobile phases consist of methanol, acetonitrile, or their mixtures with small additions of water. The addition of 0.1-0.5% acetic acid often improves peak resolution by suppressing ionization of analytes.
Sample preparation protocols generally involve simple dilution of rice bran oil in appropriate solvents like hexane or isopropanol, followed by filtration through 0.45 μm membrane filters. This straightforward approach minimizes analyte loss while ensuring chromatographic column protection.
Despite these established methodologies, several limitations persist in current HPLC protocols. Run times typically range from 20-40 minutes, limiting sample throughput in industrial quality control settings. The requirement for high-purity organic solvents also raises environmental and cost concerns, particularly for routine analyses.
Matrix effects present another significant challenge, as rice bran oil contains numerous compounds that can interfere with detection or alter retention times. This necessitates careful optimization of extraction procedures and chromatographic conditions for each specific oil source.
Quantification accuracy remains problematic due to the limited availability of certified reference standards, particularly for individual oryzanol components. Most laboratories rely on total oryzanol quantification using ferulic acid as a surrogate standard, which introduces potential quantification errors.
Instrument sensitivity limitations can also impact analysis of samples with low tocopherol or oryzanol concentrations, such as highly refined oils or processed products. This necessitates concentration steps that can introduce additional variability into the analytical workflow.
Method transferability between different HPLC systems and laboratories represents another persistent challenge, with variations in column chemistry, detector sensitivity, and data processing algorithms contributing to inconsistent results across different analytical platforms.
Established HPLC Protocols for Bioactive Compounds
01 HPLC methods for tocopherol analysis in rice bran oil
High-performance liquid chromatography (HPLC) techniques specifically optimized for the quantification of tocopherols in rice bran oil. These methods typically involve sample preparation through extraction and saponification, followed by separation on specialized columns. Detection is commonly performed using fluorescence or UV detectors at specific wavelengths optimal for tocopherol identification. These protocols allow for the accurate measurement of different tocopherol isomers (alpha, beta, gamma, and delta) that contribute to the antioxidant properties of rice bran oil.- HPLC methods for tocopherol analysis in rice bran oil: High-performance liquid chromatography (HPLC) methods specifically optimized for the quantification of tocopherols in rice bran oil. These protocols typically involve sample preparation through extraction or dilution, followed by separation on appropriate HPLC columns. Detection is commonly performed using fluorescence or UV detectors at specific wavelengths optimal for tocopherol compounds. The methods allow for the identification and quantification of different tocopherol isomers (alpha, beta, gamma, delta) present in rice bran oil samples.
- HPLC protocols for γ-oryzanol determination: Specialized HPLC methodologies for the accurate determination of γ-oryzanol content in rice bran oil. These protocols typically employ reverse-phase HPLC with C18 columns and UV detection at wavelengths around 315-330 nm. Sample preparation may involve dilution with appropriate solvents or extraction procedures to isolate the oryzanol components. The methods enable quantification of total oryzanol content as well as identification of individual oryzanol components, which are important bioactive compounds in rice bran oil.
- Simultaneous analysis of tocopherols and oryzanols: Integrated HPLC protocols that allow for the simultaneous determination of both tocopherols and oryzanols in a single analytical run. These methods typically employ gradient elution systems with carefully selected mobile phases to achieve separation of both compound classes. Detection systems may combine UV and fluorescence detection to optimize sensitivity for both compound types. This approach reduces analysis time and sample consumption while providing comprehensive profiling of these important bioactive components in rice bran oil.
- Sample preparation techniques for rice bran oil analysis: Specialized sample preparation methods for rice bran oil prior to HPLC analysis of tocopherols and oryzanols. These techniques may include solvent extraction, saponification, solid-phase extraction, or direct dilution approaches depending on the specific analytical requirements. Proper sample preparation is crucial for removing interfering compounds, concentrating analytes of interest, and ensuring reproducible results. The methods are designed to maximize recovery of target compounds while minimizing degradation during the preparation process.
- Validation and application of analytical methods: Validation protocols and practical applications of HPLC methods for tocopherol and oryzanol analysis in rice bran oil. These include determination of analytical parameters such as linearity, precision, accuracy, limits of detection and quantification, and robustness. The validated methods are applied to various scenarios including quality control of commercial rice bran oil products, stability studies during processing and storage, comparison of extraction methods, and evaluation of rice bran oil from different rice varieties or processing conditions.
02 HPLC protocols for γ-oryzanol quantification
Specialized HPLC methodologies for the detection and quantification of γ-oryzanol components in rice bran oil. These protocols typically employ reverse-phase columns with specific mobile phase compositions optimized for oryzanol separation. UV detection at wavelengths around 315-330 nm is commonly used due to the characteristic absorption of oryzanol compounds. The methods allow for identification of the multiple ferulic acid esters that comprise the oryzanol complex, providing detailed compositional analysis of this bioactive component in rice bran oil.Expand Specific Solutions03 Simultaneous determination of tocopherols and oryzanols
Integrated HPLC protocols that enable the concurrent analysis of both tocopherols and oryzanols in a single run. These methods typically involve optimized extraction procedures, specialized column selection, and gradient elution systems with carefully selected mobile phases. Detection systems often combine UV and fluorescence detection to capture the different spectral properties of tocopherols and oryzanols. These simultaneous determination methods improve efficiency and allow for comprehensive evaluation of the antioxidant profile of rice bran oil samples.Expand Specific Solutions04 Sample preparation techniques for rice bran oil analysis
Specialized preparation methods for rice bran oil samples prior to HPLC analysis of tocopherols and oryzanols. These techniques include various extraction protocols using organic solvents, saponification procedures to release bound compounds, and cleanup steps to remove interfering substances. Solid-phase extraction and other purification methods may be employed to concentrate the analytes of interest. Proper sample preparation is crucial for accurate quantification and helps overcome challenges associated with the complex matrix of rice bran oil.Expand Specific Solutions05 Validation and standardization of analytical methods
Protocols for validating and standardizing HPLC methods used in the analysis of tocopherols and oryzanols in rice bran oil. These include procedures for determining method parameters such as linearity, precision, accuracy, limits of detection and quantification, and robustness. Standard reference materials and internal standards are often employed to ensure reliability and comparability of results across different laboratories. Method validation ensures that the analytical procedures provide consistent and reliable data for quality control and research applications.Expand Specific Solutions
Leading Laboratories and Equipment Manufacturers
The rice bran oil analysis market is in a growth phase, driven by increasing consumer demand for healthier oils with functional benefits. The global market size for specialized edible oils, including rice bran oil, is expanding at a CAGR of approximately 5-7%. Technologically, HPLC protocols for tocopherol and oryzanol analysis have reached moderate maturity, with ongoing refinements. Key players include established agricultural research institutions like CSIR and CSIRO, major agribusiness corporations such as Archer-Daniels-Midland and Riceland Foods, specialized oil processors like Tsuno Food Industrial, and academic research centers including Jiangnan University and Zhejiang University. These organizations are advancing analytical methodologies while commercial entities focus on quality control applications and product development to capitalize on rice bran oil's nutritional properties.
Council of Scientific & Industrial Research
Technical Solution: The Council of Scientific & Industrial Research (CSIR) has developed a comprehensive HPLC protocol for analyzing tocopherol and oryzanol levels in rice bran oil that balances analytical performance with practical applicability. Their method employs a two-dimensional chromatographic approach that first separates major oil components on a silica guard column before transferring the analytes of interest to an analytical C18 column (250mm × 4.6mm, 5μm) for final separation. The mobile phase system consists of methanol/acetonitrile/water (50:40:10 v/v/v) with 0.1% acetic acid at a flow rate of 1.0 mL/min. For detection, CSIR utilizes a photodiode array detector operating at multiple wavelengths (292nm for tocopherols, 325nm for oryzanol) with full spectral scanning from 200-400nm for compound confirmation. Their sample preparation involves a modified liquid-liquid extraction procedure using methanol and hexane that effectively removes interfering triglycerides while maintaining high recovery rates for both tocopherols and oryzanol. The method includes an automated standard addition procedure to compensate for matrix effects, with calibration curves constructed using five concentration levels spanning the expected range in rice bran oil samples (typically 5-500 μg/g for tocopherols and 1000-3000 μg/g for oryzanol).
Strengths: The two-dimensional approach significantly reduces interference from matrix components, improving accuracy in complex samples. The method demonstrates excellent recovery rates (95-102%) and precision (RSD < 3%) across a wide concentration range. The spectral confirmation capability reduces the risk of misidentification in complex samples. Weaknesses: The two-column setup increases method complexity and requires specialized switching valves. The analysis time of approximately 25 minutes per sample is longer than some single-column approaches, and the method requires careful optimization of transfer parameters to maintain reproducibility.
Archer-Daniels-Midland Co.
Technical Solution: Archer-Daniels-Midland (ADM) has developed a robust HPLC protocol for simultaneous determination of tocopherols and oryzanol in rice bran oil that emphasizes accuracy and reproducibility. Their method utilizes a specialized phenyl-hexyl column (250mm × 4.6mm, 5μm) that provides excellent separation of both polar tocopherols and the less polar oryzanol components. The mobile phase consists of a gradient system of acetonitrile/methanol/water with 0.1% phosphoric acid, starting at 50:45:5 and transitioning to 45:55:0 over 25 minutes at a flow rate of 1.2 mL/min. For detection, ADM employs a dual-detection system with a fluorescence detector for tocopherols (excitation 290nm, emission 330nm) and UV detection at 325nm for oryzanol. Their sample preparation protocol involves direct dilution of rice bran oil in the mobile phase (1:50) followed by filtration through a 0.22μm membrane, eliminating the need for extraction steps that could lead to compound loss. The method includes calibration using certified reference materials for all four tocopherol isomers (α, β, γ, δ) and major oryzanol components, ensuring traceability to international standards.
Strengths: The method achieves exceptional separation of all tocopherol isomers and major oryzanol components with resolution values >1.5, allowing precise quantification of individual compounds. The direct dilution approach minimizes sample preparation time and reduces potential analyte losses. Validation studies show excellent linearity (r² > 0.999) and precision (RSD < 2%). Weaknesses: The specialized column and dual-detection system increase equipment costs. The method requires careful temperature control (±0.5°C) to maintain reproducibility, and the analysis time of 30 minutes per sample is longer than some alternative approaches.
Key Innovations in Chromatographic Separation
Method for simultaneously determining fat-soluble vitamins and carotenoids in serum
PatentActiveUS20210190733A1
Innovation
- A method using ionic liquids or binary mixed solvents with organic solvents for liquid-liquid extraction, followed by HPLC analysis with retinyl acetate and trans-β-apo-8′-carotenal as internal standards, simplifying the pretreatment and enabling simultaneous determination of fat-soluble vitamins and carotenoids in serum.
System for the Simultaneous Monitoring of Constituents of an Electroplating Bath
PatentPendingUS20240133074A1
Innovation
- The development of novel second-order, consolidated voltammetric waveforms combined with chemometric analysis and data compression techniques allows for the simultaneous measurement and analysis of all electroplating bath constituents without pretreatment, using a multi-frequency, variable amplitude waveform to generate a diagnostic voltammetric output that captures the synergistic interactions and maintains process control within the electroplating process.
Regulatory Standards for Edible Oil Analysis
The regulatory landscape for edible oil analysis, particularly for rice bran oil components like tocopherol and oryzanol, is governed by various international and national standards. These regulations ensure consumer safety, product quality, and fair trade practices in the global edible oil market.
The Codex Alimentarius Commission, established by FAO and WHO, provides international food standards including specific parameters for rice bran oil analysis. Their guidelines (CODEX STAN 210-1999) specify acceptable levels of tocopherols and other bioactive compounds in edible oils, requiring precise analytical methods like HPLC for verification.
In the United States, the FDA regulates edible oils under 21 CFR Part 101, with specific provisions for nutrient content claims related to tocopherols (vitamin E). The AOAC International provides official methods for tocopherol analysis in oils, with Method 992.03 being particularly relevant for HPLC determination of tocopherols in vegetable oils.
The European Union enforces Regulation (EU) No 1169/2011 for food information to consumers, including nutritional labeling of tocopherol content. The European Committee for Standardization (CEN) has established EN 12822 for vitamin E (tocopherol) determination using HPLC, which applies to rice bran oil analysis.
In Asia, where rice bran oil is widely produced and consumed, countries have developed specific standards. Japan's Ministry of Health, Labour and Welfare enforces the Japanese Agricultural Standard (JAS) for edible oils, with specific provisions for rice bran oil components. Similarly, India's Food Safety and Standards Authority (FSSAI) has established standards under FSSAI Regulations 2011 for rice bran oil, specifying acceptable ranges for oryzanol content (minimum 1.0%).
International Organization for Standardization (ISO) provides globally recognized methods for oil analysis, including ISO 9936 for determination of tocopherols and tocotrienols by HPLC. These standards specify sample preparation procedures, chromatographic conditions, and validation parameters essential for accurate quantification.
Regulatory compliance requires laboratories to maintain strict quality control measures, including regular calibration of HPLC systems, use of certified reference materials, and participation in proficiency testing programs. Method validation parameters such as linearity, precision, accuracy, and limits of detection must meet regulatory requirements to ensure reliable analysis of tocopherol and oryzanol in rice bran oil.
The Codex Alimentarius Commission, established by FAO and WHO, provides international food standards including specific parameters for rice bran oil analysis. Their guidelines (CODEX STAN 210-1999) specify acceptable levels of tocopherols and other bioactive compounds in edible oils, requiring precise analytical methods like HPLC for verification.
In the United States, the FDA regulates edible oils under 21 CFR Part 101, with specific provisions for nutrient content claims related to tocopherols (vitamin E). The AOAC International provides official methods for tocopherol analysis in oils, with Method 992.03 being particularly relevant for HPLC determination of tocopherols in vegetable oils.
The European Union enforces Regulation (EU) No 1169/2011 for food information to consumers, including nutritional labeling of tocopherol content. The European Committee for Standardization (CEN) has established EN 12822 for vitamin E (tocopherol) determination using HPLC, which applies to rice bran oil analysis.
In Asia, where rice bran oil is widely produced and consumed, countries have developed specific standards. Japan's Ministry of Health, Labour and Welfare enforces the Japanese Agricultural Standard (JAS) for edible oils, with specific provisions for rice bran oil components. Similarly, India's Food Safety and Standards Authority (FSSAI) has established standards under FSSAI Regulations 2011 for rice bran oil, specifying acceptable ranges for oryzanol content (minimum 1.0%).
International Organization for Standardization (ISO) provides globally recognized methods for oil analysis, including ISO 9936 for determination of tocopherols and tocotrienols by HPLC. These standards specify sample preparation procedures, chromatographic conditions, and validation parameters essential for accurate quantification.
Regulatory compliance requires laboratories to maintain strict quality control measures, including regular calibration of HPLC systems, use of certified reference materials, and participation in proficiency testing programs. Method validation parameters such as linearity, precision, accuracy, and limits of detection must meet regulatory requirements to ensure reliable analysis of tocopherol and oryzanol in rice bran oil.
Method Validation and Quality Assurance Protocols
Method validation is essential for ensuring the reliability and reproducibility of HPLC protocols for tocopherol and oryzanol analysis in rice bran oil. The validation process should follow international guidelines such as ICH Q2(R1) or USP <1225>, encompassing parameters including specificity, linearity, accuracy, precision, detection limit, quantitation limit, and robustness.
Specificity validation requires demonstrating that the analytical method can accurately distinguish tocopherols (α, β, γ, δ) and oryzanol components from other substances in rice bran oil. This is typically achieved through peak purity assessment using diode array detection or mass spectrometry.
Linearity must be established across a concentration range of at least 80-120% of the expected analyte levels, with correlation coefficients (r²) exceeding 0.995 considered acceptable. For tocopherols, this typically spans 5-500 μg/mL, while oryzanol requires 10-1000 μg/mL ranges.
Accuracy validation involves recovery studies at three concentration levels (low, medium, high), with acceptance criteria of 98-102% recovery for both tocopherols and oryzanol. Precision evaluation includes repeatability (intra-day) and intermediate precision (inter-day), with relative standard deviation (RSD) values below 2% for both analytes.
Detection and quantitation limits should be determined using signal-to-noise ratios, with typical LOD values of 0.1-0.5 μg/mL for tocopherols and 0.5-2.0 μg/mL for oryzanol components in rice bran oil matrices.
Robustness testing must evaluate the method's stability under varied conditions, including minor changes in mobile phase composition (±2%), flow rate (±0.1 mL/min), column temperature (±2°C), and detection wavelength (±2 nm). System suitability parameters should be established, including resolution between critical pairs (Rs > 1.5), tailing factors (Tf < 2.0), and theoretical plate counts (N > 2000).
Quality assurance protocols should incorporate regular system suitability testing before sample analysis, including blank injections, standard solution stability verification, and calibration curve validation. Control charts monitoring retention times, peak areas, and resolution factors help identify system drift over time.
Reference standards require proper certification, storage, and handling protocols. Working standards should be prepared fresh or validated for stability under storage conditions. Regular participation in proficiency testing programs and use of certified reference materials (CRMs) provide external validation of method performance.
Documentation must be comprehensive, including detailed standard operating procedures (SOPs), analyst training records, equipment qualification reports, and method validation documentation. Electronic data systems should comply with 21 CFR Part 11 or equivalent regulations regarding data integrity and audit trails.
Specificity validation requires demonstrating that the analytical method can accurately distinguish tocopherols (α, β, γ, δ) and oryzanol components from other substances in rice bran oil. This is typically achieved through peak purity assessment using diode array detection or mass spectrometry.
Linearity must be established across a concentration range of at least 80-120% of the expected analyte levels, with correlation coefficients (r²) exceeding 0.995 considered acceptable. For tocopherols, this typically spans 5-500 μg/mL, while oryzanol requires 10-1000 μg/mL ranges.
Accuracy validation involves recovery studies at three concentration levels (low, medium, high), with acceptance criteria of 98-102% recovery for both tocopherols and oryzanol. Precision evaluation includes repeatability (intra-day) and intermediate precision (inter-day), with relative standard deviation (RSD) values below 2% for both analytes.
Detection and quantitation limits should be determined using signal-to-noise ratios, with typical LOD values of 0.1-0.5 μg/mL for tocopherols and 0.5-2.0 μg/mL for oryzanol components in rice bran oil matrices.
Robustness testing must evaluate the method's stability under varied conditions, including minor changes in mobile phase composition (±2%), flow rate (±0.1 mL/min), column temperature (±2°C), and detection wavelength (±2 nm). System suitability parameters should be established, including resolution between critical pairs (Rs > 1.5), tailing factors (Tf < 2.0), and theoretical plate counts (N > 2000).
Quality assurance protocols should incorporate regular system suitability testing before sample analysis, including blank injections, standard solution stability verification, and calibration curve validation. Control charts monitoring retention times, peak areas, and resolution factors help identify system drift over time.
Reference standards require proper certification, storage, and handling protocols. Working standards should be prepared fresh or validated for stability under storage conditions. Regular participation in proficiency testing programs and use of certified reference materials (CRMs) provide external validation of method performance.
Documentation must be comprehensive, including detailed standard operating procedures (SOPs), analyst training records, equipment qualification reports, and method validation documentation. Electronic data systems should comply with 21 CFR Part 11 or equivalent regulations regarding data integrity and audit trails.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!