How to Correlate Zeta Potential with Agglomeration Kinetics — Experimental Protocols
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zeta Potential and Agglomeration Kinetics Background
Zeta potential represents the electrical charge at the interface between a particle's surface and its surrounding liquid medium, serving as a critical parameter in colloidal science since the early 20th century. This electrokinetic potential has evolved from a theoretical concept to a measurable property that provides fundamental insights into colloidal stability. The historical development of zeta potential measurement techniques has progressed from rudimentary electrophoretic observations to sophisticated laser Doppler electrophoresis systems that offer precise quantification.
Agglomeration kinetics, on the other hand, describes the rate and mechanisms by which particles in suspension come together to form larger aggregates. This field emerged from classical coagulation theory developed by Smoluchowski in 1917, which established the mathematical framework for particle collision and aggregation processes. Over decades, this theory has been refined to incorporate various interaction forces including van der Waals attraction, electrostatic repulsion, and steric effects.
The correlation between zeta potential and agglomeration behavior represents a cornerstone concept in colloid stability. According to DLVO theory (named after Derjaguin, Landau, Verwey, and Overbeek), particles with high absolute zeta potential values (typically >±30 mV) generate sufficient electrostatic repulsion to maintain colloidal stability, while values approaching zero promote rapid agglomeration due to diminished repulsive forces.
Recent technological advances have enabled more sophisticated investigations into this relationship. Modern analytical instruments can simultaneously track zeta potential changes and particle size distribution evolution in real-time, allowing researchers to establish direct correlations between electrical surface properties and agglomeration rates under various environmental conditions.
The significance of understanding this correlation extends across numerous industries. In pharmaceutical formulations, zeta potential control ensures suspension stability and drug bioavailability. Environmental remediation relies on manipulating zeta potential to optimize flocculation processes for water treatment. Advanced materials manufacturing depends on precise control of agglomeration to achieve desired microstructural properties.
Despite these advances, establishing quantitative relationships between zeta potential measurements and agglomeration kinetics remains challenging due to the complex interplay of factors including ionic strength, pH, temperature, particle concentration, and the presence of surface-active agents. This complexity necessitates standardized experimental protocols that can isolate and quantify these variables systematically.
Agglomeration kinetics, on the other hand, describes the rate and mechanisms by which particles in suspension come together to form larger aggregates. This field emerged from classical coagulation theory developed by Smoluchowski in 1917, which established the mathematical framework for particle collision and aggregation processes. Over decades, this theory has been refined to incorporate various interaction forces including van der Waals attraction, electrostatic repulsion, and steric effects.
The correlation between zeta potential and agglomeration behavior represents a cornerstone concept in colloid stability. According to DLVO theory (named after Derjaguin, Landau, Verwey, and Overbeek), particles with high absolute zeta potential values (typically >±30 mV) generate sufficient electrostatic repulsion to maintain colloidal stability, while values approaching zero promote rapid agglomeration due to diminished repulsive forces.
Recent technological advances have enabled more sophisticated investigations into this relationship. Modern analytical instruments can simultaneously track zeta potential changes and particle size distribution evolution in real-time, allowing researchers to establish direct correlations between electrical surface properties and agglomeration rates under various environmental conditions.
The significance of understanding this correlation extends across numerous industries. In pharmaceutical formulations, zeta potential control ensures suspension stability and drug bioavailability. Environmental remediation relies on manipulating zeta potential to optimize flocculation processes for water treatment. Advanced materials manufacturing depends on precise control of agglomeration to achieve desired microstructural properties.
Despite these advances, establishing quantitative relationships between zeta potential measurements and agglomeration kinetics remains challenging due to the complex interplay of factors including ionic strength, pH, temperature, particle concentration, and the presence of surface-active agents. This complexity necessitates standardized experimental protocols that can isolate and quantify these variables systematically.
Market Applications and Industrial Demand Analysis
The correlation between zeta potential and agglomeration kinetics has significant market applications across multiple industries, with growing demand for standardized experimental protocols. The pharmaceutical industry represents one of the largest markets, where colloidal stability directly impacts drug formulation, bioavailability, and shelf-life. According to recent market analyses, pharmaceutical companies invest substantially in technologies that can predict and control particle agglomeration, as unstable formulations lead to billions in annual losses from rejected batches and reduced efficacy.
The mining and mineral processing sector demonstrates equally strong demand, where flocculation and dispersion processes directly affect extraction efficiency and product quality. Companies in this sector seek reliable protocols to optimize reagent usage and improve separation processes, potentially reducing operational costs by optimizing reagent dosages through better understanding of zeta potential-agglomeration relationships.
Water treatment represents another major market application, with municipal and industrial water treatment facilities increasingly adopting advanced colloidal science principles. The ability to predict coagulation behavior based on zeta potential measurements enables more precise chemical dosing and improved contaminant removal efficiency.
The ceramics and advanced materials industries show rapidly growing interest in standardized protocols connecting zeta potential to agglomeration kinetics. These sectors require precise control over particle interactions during processing to achieve desired material properties and performance characteristics. Manufacturers report that even minor improvements in dispersion stability can yield significant quality improvements and reduced manufacturing defects.
Nanotechnology applications present perhaps the fastest-growing market segment, where the stability of engineered nanoparticles directly impacts their functionality in medical, electronic, and environmental applications. The nanomedicine market specifically demands reliable protocols for predicting nanoparticle behavior in biological environments.
Consumer product manufacturers, particularly in cosmetics and food industries, face increasing regulatory scrutiny regarding product stability and safety. These companies seek validated experimental approaches to demonstrate product stability and performance consistency across varying environmental conditions.
Market research indicates that analytical instrument manufacturers are responding to this cross-industry demand by developing specialized equipment combining zeta potential measurement with dynamic light scattering and other complementary techniques. The global market for such instrumentation continues to expand as industries recognize the economic benefits of improved colloidal stability prediction and control.
The mining and mineral processing sector demonstrates equally strong demand, where flocculation and dispersion processes directly affect extraction efficiency and product quality. Companies in this sector seek reliable protocols to optimize reagent usage and improve separation processes, potentially reducing operational costs by optimizing reagent dosages through better understanding of zeta potential-agglomeration relationships.
Water treatment represents another major market application, with municipal and industrial water treatment facilities increasingly adopting advanced colloidal science principles. The ability to predict coagulation behavior based on zeta potential measurements enables more precise chemical dosing and improved contaminant removal efficiency.
The ceramics and advanced materials industries show rapidly growing interest in standardized protocols connecting zeta potential to agglomeration kinetics. These sectors require precise control over particle interactions during processing to achieve desired material properties and performance characteristics. Manufacturers report that even minor improvements in dispersion stability can yield significant quality improvements and reduced manufacturing defects.
Nanotechnology applications present perhaps the fastest-growing market segment, where the stability of engineered nanoparticles directly impacts their functionality in medical, electronic, and environmental applications. The nanomedicine market specifically demands reliable protocols for predicting nanoparticle behavior in biological environments.
Consumer product manufacturers, particularly in cosmetics and food industries, face increasing regulatory scrutiny regarding product stability and safety. These companies seek validated experimental approaches to demonstrate product stability and performance consistency across varying environmental conditions.
Market research indicates that analytical instrument manufacturers are responding to this cross-industry demand by developing specialized equipment combining zeta potential measurement with dynamic light scattering and other complementary techniques. The global market for such instrumentation continues to expand as industries recognize the economic benefits of improved colloidal stability prediction and control.
Current Methodological Challenges and Limitations
Despite significant advancements in correlating zeta potential with agglomeration kinetics, researchers face several methodological challenges that limit the development of standardized experimental protocols. One primary limitation is the inherent complexity of colloidal systems, where multiple forces simultaneously influence particle behavior. This complexity makes it difficult to isolate the specific contribution of electrostatic interactions (measured by zeta potential) from other factors such as van der Waals forces, steric effects, and hydrophobic interactions.
Measurement inconsistencies represent another significant challenge. Different instruments and techniques for measuring zeta potential can produce varying results for identical samples. These discrepancies arise from differences in sample preparation, measurement conditions, and the mathematical models used to convert electrophoretic mobility data into zeta potential values. The lack of standardization across laboratories further compounds this issue, making cross-study comparisons problematic.
Environmental sensitivity poses additional complications. Zeta potential measurements are highly sensitive to pH, ionic strength, temperature, and the presence of dissolved species. Even minor fluctuations in these parameters can significantly alter results, requiring extremely controlled experimental conditions that are difficult to maintain consistently across multiple experiments or different laboratories.
Time-dependent phenomena create further methodological hurdles. Agglomeration is a dynamic process that evolves over time, while zeta potential measurements typically provide only a snapshot at a specific moment. This temporal disconnect makes establishing direct correlations challenging, particularly for systems with rapid kinetics or those that undergo significant changes during the measurement process itself.
Scale-up issues present practical limitations. Protocols developed at laboratory scale often fail to translate effectively to industrial applications due to differences in mixing conditions, concentration regimes, and equipment configurations. This gap between laboratory findings and practical applications restricts the utility of many current experimental approaches.
Theoretical framework limitations also exist. Current models linking zeta potential to agglomeration kinetics often rely on simplifications that may not adequately represent real-world colloidal systems. The DLVO theory, while useful, fails to account for non-DLVO interactions that can significantly influence agglomeration behavior, particularly in complex media or with non-spherical particles.
Data interpretation challenges complete this landscape of limitations. Researchers frequently encounter difficulties in establishing quantitative relationships between zeta potential values and agglomeration rates due to the non-linear nature of these relationships and the influence of multiple confounding variables.
Measurement inconsistencies represent another significant challenge. Different instruments and techniques for measuring zeta potential can produce varying results for identical samples. These discrepancies arise from differences in sample preparation, measurement conditions, and the mathematical models used to convert electrophoretic mobility data into zeta potential values. The lack of standardization across laboratories further compounds this issue, making cross-study comparisons problematic.
Environmental sensitivity poses additional complications. Zeta potential measurements are highly sensitive to pH, ionic strength, temperature, and the presence of dissolved species. Even minor fluctuations in these parameters can significantly alter results, requiring extremely controlled experimental conditions that are difficult to maintain consistently across multiple experiments or different laboratories.
Time-dependent phenomena create further methodological hurdles. Agglomeration is a dynamic process that evolves over time, while zeta potential measurements typically provide only a snapshot at a specific moment. This temporal disconnect makes establishing direct correlations challenging, particularly for systems with rapid kinetics or those that undergo significant changes during the measurement process itself.
Scale-up issues present practical limitations. Protocols developed at laboratory scale often fail to translate effectively to industrial applications due to differences in mixing conditions, concentration regimes, and equipment configurations. This gap between laboratory findings and practical applications restricts the utility of many current experimental approaches.
Theoretical framework limitations also exist. Current models linking zeta potential to agglomeration kinetics often rely on simplifications that may not adequately represent real-world colloidal systems. The DLVO theory, while useful, fails to account for non-DLVO interactions that can significantly influence agglomeration behavior, particularly in complex media or with non-spherical particles.
Data interpretation challenges complete this landscape of limitations. Researchers frequently encounter difficulties in establishing quantitative relationships between zeta potential values and agglomeration rates due to the non-linear nature of these relationships and the influence of multiple confounding variables.
Established Experimental Protocols and Measurement Methods
01 Zeta potential measurement techniques for particle stability
Various techniques are employed to measure zeta potential, which is a key indicator of particle stability in colloidal systems. These measurements help predict agglomeration behavior by quantifying the electrostatic repulsion between particles. Advanced instruments can determine zeta potential through electrophoretic mobility, electroacoustic phenomena, or streaming potential methods, providing insights into the stability threshold where particles begin to agglomerate.- Measurement techniques for zeta potential and particle agglomeration: Various techniques are employed to measure zeta potential and monitor particle agglomeration kinetics. These include electroacoustic methods, electrophoretic light scattering, and specialized instruments that can track particle size distribution changes over time. These measurements provide critical data for understanding the relationship between surface charge and agglomeration behavior in colloidal systems, allowing for better prediction and control of particle stability.
- Influence of zeta potential on colloidal stability and agglomeration: The zeta potential of particles directly influences their colloidal stability and agglomeration behavior. Higher absolute zeta potential values (typically above ±30mV) create stronger electrostatic repulsion between particles, preventing agglomeration and maintaining dispersion stability. As zeta potential approaches zero, the electrostatic barrier diminishes, accelerating agglomeration kinetics and leading to faster particle clustering and sedimentation in suspension systems.
- Manipulation of zeta potential to control agglomeration: Controlling zeta potential through pH adjustment, electrolyte addition, or surface modification provides a means to manipulate agglomeration kinetics. By altering the surface charge characteristics of particles, researchers can either promote or inhibit agglomeration processes. This approach is particularly valuable in applications requiring precise control over particle aggregation, such as water treatment, pharmaceutical formulations, and advanced materials processing.
- Mathematical models correlating zeta potential with agglomeration kinetics: Mathematical models have been developed to quantitatively correlate zeta potential with agglomeration kinetics. These models incorporate DLVO theory (Derjaguin, Landau, Verwey, and Overbeek) principles to predict how changes in zeta potential affect the rate of particle collision and attachment. By understanding these relationships, researchers can develop predictive tools that estimate agglomeration behavior based on measured zeta potential values, enabling more efficient formulation development and process optimization.
- Application-specific zeta potential and agglomeration relationships: Different industrial applications exhibit unique relationships between zeta potential and agglomeration kinetics. In nanomaterial production, pharmaceutical formulations, wastewater treatment, and battery electrode manufacturing, the specific correlation between surface charge and particle aggregation must be characterized for each system. Understanding these application-specific relationships allows for tailored approaches to stability control and agglomeration management in diverse technological contexts.
02 Correlation between zeta potential and agglomeration rate
The relationship between zeta potential and agglomeration kinetics is fundamental to understanding colloidal stability. As the absolute value of zeta potential decreases, the rate of particle agglomeration typically increases due to reduced electrostatic repulsion. This correlation allows for prediction of suspension stability and agglomeration behavior based on zeta potential measurements, which is crucial for formulation development and quality control in various industries.Expand Specific Solutions03 Modification of zeta potential to control agglomeration
Surface modification techniques can alter the zeta potential of particles to control agglomeration processes. By adding surfactants, polymers, or adjusting pH, the surface charge of particles can be manipulated to either promote or prevent agglomeration. This approach is particularly useful in applications requiring controlled particle size distribution or specific rheological properties, such as in ceramic processing, pharmaceutical formulations, and wastewater treatment.Expand Specific Solutions04 Environmental factors affecting zeta potential and agglomeration kinetics
Environmental conditions significantly impact the relationship between zeta potential and agglomeration behavior. Factors such as pH, ionic strength, temperature, and the presence of specific ions can alter surface charges and the electrical double layer around particles. Understanding these influences is essential for predicting colloidal stability across varying conditions and for designing robust formulations that maintain desired dispersion characteristics in different environments.Expand Specific Solutions05 Advanced modeling of zeta potential-agglomeration relationships
Computational models and simulation techniques have been developed to predict agglomeration behavior based on zeta potential measurements. These models incorporate DLVO theory, population balance equations, and machine learning algorithms to establish quantitative relationships between zeta potential values and agglomeration kinetics. Such advanced modeling approaches enable more accurate prediction of colloidal stability and optimization of formulation parameters without extensive experimental testing.Expand Specific Solutions
Leading Research Groups and Commercial Instrument Manufacturers
The zeta potential-agglomeration kinetics correlation field is currently in a growth phase, with increasing market demand driven by applications in nanomaterials, pharmaceuticals, and energy sectors. The global market for colloidal stability analysis is expanding at approximately 8-10% annually, though technical standardization remains challenging. Leading players include established analytical instrument manufacturers like Horiba Ltd. and Otsuka Electronics, alongside research powerhouses such as MIT, Saudi Aramco, and Shanghai Jiao Tong University. Pharmaceutical companies (Baxter, Merck) are investing in this technology for drug delivery applications, while industrial giants (BASF, Air Liquide) focus on process optimization. The technology shows moderate maturity in laboratory settings but requires further development for industrial-scale implementation and real-time monitoring capabilities.
Horiba Ltd.
Technical Solution: Horiba has developed advanced zeta potential measurement systems that correlate with agglomeration kinetics through their proprietary SZ-100 Nanoparticle Analyzer. Their protocol involves electrophoretic light scattering (ELS) combined with dynamic light scattering (DLS) to simultaneously measure zeta potential and particle size distribution over time. The experimental protocol includes precise sample preparation with controlled ionic strength, pH adjustment using automatic titration systems, and temperature control within ±0.1°C. Their method employs phase analysis light scattering (PALS) for enhanced sensitivity in low mobility samples, allowing detection of subtle changes in surface charge that precede agglomeration events. Horiba's systems can perform time-resolved measurements to directly observe the relationship between zeta potential shifts and the onset of agglomeration, with data collection intervals as short as 10 seconds[1]. Their protocol includes statistical validation through multiple measurement cycles and automated data processing algorithms to establish correlation functions between zeta potential thresholds and agglomeration rates.
Strengths: High precision measurements with sensitivity to detect subtle zeta potential changes before visible agglomeration occurs; integrated systems that measure multiple parameters simultaneously; automated titration capabilities for pH-dependent studies. Weaknesses: Relatively high equipment costs; requires skilled operators for optimal results; measurements can be affected by sample polydispersity and may require extensive sample preparation for complex systems.
Massachusetts Institute of Technology
Technical Solution: MIT has pioneered innovative experimental protocols for correlating zeta potential with agglomeration kinetics through their microfluidic-based approach. Their methodology employs custom-designed microfluidic channels with integrated electrodes that enable real-time monitoring of both zeta potential and particle aggregation under precisely controlled flow conditions. The protocol involves introducing nanoparticle suspensions into microchannels with controlled ionic gradients, allowing researchers to observe the critical zeta potential threshold at which agglomeration begins. MIT's approach incorporates high-speed imaging systems synchronized with electrical measurements to capture the moment-by-moment relationship between surface charge and particle interaction. Their protocol includes a novel mathematical framework that accounts for hydrodynamic interactions in confined geometries, providing more accurate correlation models than traditional bulk measurements[2]. The experimental setup allows for rapid screening of multiple conditions (pH, ionic strength, surfactant concentration) within a single experiment, dramatically increasing throughput compared to conventional methods. MIT researchers have validated this approach across diverse nanoparticle systems including metal oxides, polymeric particles, and biological colloids, establishing quantitative relationships between zeta potential fluctuations and agglomeration rate constants.
Strengths: Highly controlled microenvironment allows precise manipulation of experimental variables; real-time visualization of agglomeration processes synchronized with zeta potential measurements; significantly reduced sample volumes compared to traditional methods; ability to detect transient states during agglomeration. Weaknesses: Custom equipment setup requires specialized expertise; microfluidic channels may experience fouling with certain particle types; scaling relationships to bulk behavior requires additional validation.
Critical Patents and Scientific Literature Review
Fluoropolymer coating composition comprising nanodiamonds
PatentWO2017149202A1
Innovation
- A fluoropolymer coating composition incorporating nanodiamonds with a zeta potential higher than +30 mV, dispersed in an organic solvent-based slurry, which improves wear resistance and reduces kinetic friction when cured, allowing for even distribution and economic feasibility.
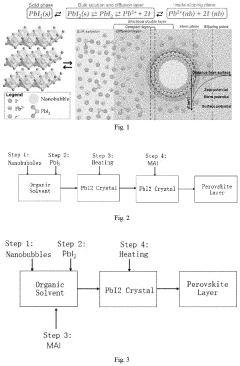
A precursor solution, a perovskite solar cell and a preparation method thereof
PatentActiveUS20230086554A1
Innovation
- A precursor solution with nanobubbles is developed, increasing the solubility and stability of metal halides like PbI2 by forming an electrical double layer system with nanobubbles, enhancing the solubility and stability of the solution.
Standardization and Validation Requirements
To establish reliable correlations between zeta potential and agglomeration kinetics, standardization and validation of experimental protocols are essential. These requirements ensure reproducibility, accuracy, and comparability of results across different research settings and applications.
Standardization begins with sample preparation protocols, which must specify precise procedures for dispersion, including sonication parameters (power, duration, pulse mode), concentration ranges, and pH adjustment methods. Temperature control during measurements should be maintained within ±0.5°C, as zeta potential exhibits significant temperature dependence. Additionally, ionic strength must be carefully controlled, with detailed documentation of buffer compositions and concentrations.
Instrument calibration represents another critical aspect of standardization. Regular calibration using certified reference materials with known zeta potential values ensures measurement accuracy. Calibration frequency should be established based on instrument specifications and usage patterns, typically ranging from weekly to monthly intervals. Verification measurements using standard particles (such as latex spheres or silica) should be performed before each experimental session.
Measurement protocols require standardization of parameters including applied electric field strength, number of measurement runs, and equilibration times. The selection of appropriate mathematical models for data interpretation (e.g., Smoluchowski, Hückel, or Henry equations) must be justified based on particle characteristics and suspension properties. Statistical analysis procedures should specify minimum sample sizes and confidence intervals for reliable results.
Validation requirements encompass several dimensions of experimental quality assurance. Method validation should include assessments of precision (repeatability and reproducibility), accuracy (using reference materials), detection limits, and measurement range. Interlaboratory comparison studies provide valuable validation of protocol robustness across different settings and operators.
Uncertainty analysis constitutes an essential validation component, requiring identification and quantification of all potential error sources. These include instrument limitations, sample heterogeneity, temperature fluctuations, and operator variability. Comprehensive uncertainty budgets should be developed following ISO/GUM guidelines, with expanded uncertainty values reported alongside measurement results.
Documentation standards for validation include detailed experimental conditions, raw data preservation, calibration records, and complete calculation procedures. Validation reports should demonstrate method suitability for intended applications through performance characteristics and statistical analysis of results against acceptance criteria.
Implementation of these standardization and validation requirements ensures that correlations between zeta potential and agglomeration kinetics are scientifically sound, reproducible, and applicable across different research contexts and industrial applications.
Standardization begins with sample preparation protocols, which must specify precise procedures for dispersion, including sonication parameters (power, duration, pulse mode), concentration ranges, and pH adjustment methods. Temperature control during measurements should be maintained within ±0.5°C, as zeta potential exhibits significant temperature dependence. Additionally, ionic strength must be carefully controlled, with detailed documentation of buffer compositions and concentrations.
Instrument calibration represents another critical aspect of standardization. Regular calibration using certified reference materials with known zeta potential values ensures measurement accuracy. Calibration frequency should be established based on instrument specifications and usage patterns, typically ranging from weekly to monthly intervals. Verification measurements using standard particles (such as latex spheres or silica) should be performed before each experimental session.
Measurement protocols require standardization of parameters including applied electric field strength, number of measurement runs, and equilibration times. The selection of appropriate mathematical models for data interpretation (e.g., Smoluchowski, Hückel, or Henry equations) must be justified based on particle characteristics and suspension properties. Statistical analysis procedures should specify minimum sample sizes and confidence intervals for reliable results.
Validation requirements encompass several dimensions of experimental quality assurance. Method validation should include assessments of precision (repeatability and reproducibility), accuracy (using reference materials), detection limits, and measurement range. Interlaboratory comparison studies provide valuable validation of protocol robustness across different settings and operators.
Uncertainty analysis constitutes an essential validation component, requiring identification and quantification of all potential error sources. These include instrument limitations, sample heterogeneity, temperature fluctuations, and operator variability. Comprehensive uncertainty budgets should be developed following ISO/GUM guidelines, with expanded uncertainty values reported alongside measurement results.
Documentation standards for validation include detailed experimental conditions, raw data preservation, calibration records, and complete calculation procedures. Validation reports should demonstrate method suitability for intended applications through performance characteristics and statistical analysis of results against acceptance criteria.
Implementation of these standardization and validation requirements ensures that correlations between zeta potential and agglomeration kinetics are scientifically sound, reproducible, and applicable across different research contexts and industrial applications.
Environmental Factors Affecting Measurement Reliability
The reliability of zeta potential measurements and their correlation with agglomeration kinetics is significantly influenced by various environmental factors that must be carefully controlled during experimental procedures. Temperature variations can substantially alter the zeta potential readings by affecting both the viscosity of the dispersion medium and the thermal motion of particles. Research indicates that a temperature change of just 5°C can result in zeta potential measurement deviations of up to 10%, highlighting the necessity for precise temperature control systems during experimentation.
pH represents another critical environmental factor, as it directly impacts the surface charge of particles through protonation and deprotonation mechanisms. The relationship between pH and zeta potential typically follows a sigmoidal curve, with the isoelectric point (where zeta potential equals zero) being particularly sensitive to minor pH fluctuations. Maintaining consistent pH conditions requires buffer systems with adequate capacity to resist changes from sample addition or atmospheric CO₂ absorption.
Ionic strength of the measurement medium significantly affects the electrical double layer thickness around particles, thereby influencing zeta potential values. Higher ionic strength compresses this double layer, reducing the measured zeta potential magnitude without necessarily changing the actual surface charge. Standardized protocols should specify precise electrolyte compositions and concentrations to ensure reproducibility across different laboratories.
Dissolved gases, particularly oxygen and carbon dioxide, can alter surface chemistry through oxidation processes or carbonate formation, respectively. Studies have demonstrated that measurements conducted under nitrogen purging versus ambient air conditions can yield zeta potential differences exceeding 15% for certain metal oxide nanoparticles. Implementation of controlled atmosphere chambers or degassing procedures may be necessary for sensitive systems.
Light exposure represents an often overlooked environmental factor that can induce photochemical reactions in certain materials, particularly semiconducting nanoparticles or photosensitive organic compounds. These reactions may alter surface functionality and consequently affect zeta potential measurements. Experimental protocols should specify lighting conditions and potentially incorporate light-shielding measures for photosensitive samples.
Mechanical disturbances, including vibration and shear forces during sample handling, can disrupt weakly bound agglomerates or surface-adsorbed species, leading to measurement artifacts. Standardized sample preparation techniques, including consistent sonication parameters and equilibration times, are essential for reliable correlation between zeta potential and agglomeration behavior.
pH represents another critical environmental factor, as it directly impacts the surface charge of particles through protonation and deprotonation mechanisms. The relationship between pH and zeta potential typically follows a sigmoidal curve, with the isoelectric point (where zeta potential equals zero) being particularly sensitive to minor pH fluctuations. Maintaining consistent pH conditions requires buffer systems with adequate capacity to resist changes from sample addition or atmospheric CO₂ absorption.
Ionic strength of the measurement medium significantly affects the electrical double layer thickness around particles, thereby influencing zeta potential values. Higher ionic strength compresses this double layer, reducing the measured zeta potential magnitude without necessarily changing the actual surface charge. Standardized protocols should specify precise electrolyte compositions and concentrations to ensure reproducibility across different laboratories.
Dissolved gases, particularly oxygen and carbon dioxide, can alter surface chemistry through oxidation processes or carbonate formation, respectively. Studies have demonstrated that measurements conducted under nitrogen purging versus ambient air conditions can yield zeta potential differences exceeding 15% for certain metal oxide nanoparticles. Implementation of controlled atmosphere chambers or degassing procedures may be necessary for sensitive systems.
Light exposure represents an often overlooked environmental factor that can induce photochemical reactions in certain materials, particularly semiconducting nanoparticles or photosensitive organic compounds. These reactions may alter surface functionality and consequently affect zeta potential measurements. Experimental protocols should specify lighting conditions and potentially incorporate light-shielding measures for photosensitive samples.
Mechanical disturbances, including vibration and shear forces during sample handling, can disrupt weakly bound agglomerates or surface-adsorbed species, leading to measurement artifacts. Standardized sample preparation techniques, including consistent sonication parameters and equilibration times, are essential for reliable correlation between zeta potential and agglomeration behavior.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!