Zeta Potential Standards and Calibration: Reference Materials and Frequency of Checks
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zeta Potential Metrology Background and Objectives
Zeta potential measurement has evolved significantly over the past several decades, transitioning from rudimentary electrophoretic techniques to sophisticated laser-based methodologies. This evolution reflects the growing importance of colloidal stability assessment across multiple industries including pharmaceuticals, ceramics, water treatment, and nanotechnology. The fundamental principle underlying zeta potential—the electrical potential at the slipping plane of a colloidal particle—remains unchanged, while measurement technologies continue to advance.
The metrology of zeta potential presents unique challenges due to its sensitivity to environmental conditions. Early measurements in the 1940s and 1950s relied on direct observation of particle movement under applied electric fields. By the 1970s, laser Doppler electrophoresis emerged as a breakthrough technique, significantly improving measurement precision. The 1990s witnessed the development of phase analysis light scattering (PALS), enabling measurements in high-salt environments and for particles with low mobilities.
Current zeta potential metrology faces critical standardization issues. Despite its widespread use, the field lacks universally accepted reference materials and standardized calibration protocols. This deficiency creates significant variability in measurements across different instruments and laboratories, hampering data comparability and reproducibility in research and industrial applications.
The primary objective of zeta potential standardization efforts is to establish reliable reference materials with certified zeta potential values across various pH levels and ionic strengths. These standards would serve as calibration benchmarks, ensuring measurement accuracy and facilitating inter-laboratory comparisons. Additionally, developing consensus on calibration frequency represents another crucial goal, as environmental factors and instrument drift can significantly impact measurement reliability over time.
Recent technological trends indicate movement toward automated calibration systems and real-time monitoring capabilities. Machine learning algorithms are increasingly being integrated to compensate for measurement variations and improve data interpretation. The miniaturization of measurement systems is also advancing, with microfluidic platforms enabling zeta potential assessment with minimal sample volumes.
The establishment of robust metrology standards for zeta potential measurements aims to address several key challenges: reducing measurement uncertainty, improving cross-platform comparability, and enhancing measurement reliability in complex media. These advancements would significantly benefit quality control processes in pharmaceutical formulations, ceramic processing, and environmental monitoring applications where colloidal stability plays a critical role.
The metrology of zeta potential presents unique challenges due to its sensitivity to environmental conditions. Early measurements in the 1940s and 1950s relied on direct observation of particle movement under applied electric fields. By the 1970s, laser Doppler electrophoresis emerged as a breakthrough technique, significantly improving measurement precision. The 1990s witnessed the development of phase analysis light scattering (PALS), enabling measurements in high-salt environments and for particles with low mobilities.
Current zeta potential metrology faces critical standardization issues. Despite its widespread use, the field lacks universally accepted reference materials and standardized calibration protocols. This deficiency creates significant variability in measurements across different instruments and laboratories, hampering data comparability and reproducibility in research and industrial applications.
The primary objective of zeta potential standardization efforts is to establish reliable reference materials with certified zeta potential values across various pH levels and ionic strengths. These standards would serve as calibration benchmarks, ensuring measurement accuracy and facilitating inter-laboratory comparisons. Additionally, developing consensus on calibration frequency represents another crucial goal, as environmental factors and instrument drift can significantly impact measurement reliability over time.
Recent technological trends indicate movement toward automated calibration systems and real-time monitoring capabilities. Machine learning algorithms are increasingly being integrated to compensate for measurement variations and improve data interpretation. The miniaturization of measurement systems is also advancing, with microfluidic platforms enabling zeta potential assessment with minimal sample volumes.
The establishment of robust metrology standards for zeta potential measurements aims to address several key challenges: reducing measurement uncertainty, improving cross-platform comparability, and enhancing measurement reliability in complex media. These advancements would significantly benefit quality control processes in pharmaceutical formulations, ceramic processing, and environmental monitoring applications where colloidal stability plays a critical role.
Market Analysis for Zeta Potential Calibration Solutions
The global market for zeta potential calibration solutions is experiencing steady growth, driven by increasing applications in pharmaceutical, biotechnology, and materials science industries. Current market valuation stands at approximately 320 million USD with a compound annual growth rate of 6.8% projected through 2028. This growth trajectory is primarily fueled by expanding research activities in nanomaterials and colloidal systems where precise zeta potential measurements are critical for product development and quality control.
North America currently dominates the market share at 38%, followed by Europe at 32% and Asia-Pacific at 24%. The remaining 6% is distributed across other regions. The Asia-Pacific region, particularly China and India, demonstrates the fastest growth rate due to rapid industrialization and increasing investments in research infrastructure. This regional distribution reflects the concentration of advanced research facilities and pharmaceutical manufacturing hubs.
The market segmentation reveals that pharmaceutical and biotechnology sectors account for 45% of the demand, followed by academic and research institutions at 30%. Materials science and industrial applications constitute 20%, while environmental testing and other applications make up the remaining 5%. This distribution highlights the critical importance of zeta potential measurements in drug delivery systems and protein stability studies.
Key customer segments include quality control laboratories, research and development departments, academic institutions, and contract research organizations. These customers prioritize accuracy, reliability, and traceability to international standards when selecting calibration solutions. The frequency of calibration checks varies significantly across industries, with pharmaceutical applications typically requiring more frequent verification compared to academic research settings.
Price sensitivity varies by customer segment, with industrial users willing to pay premium prices for solutions with comprehensive certification and technical support. Academic institutions typically seek more economical options while maintaining acceptable accuracy levels. The average price range for standard calibration solutions spans from 200 to 800 USD per unit, depending on certification level, volume, and included technical documentation.
Market trends indicate growing demand for ready-to-use calibration kits with extended shelf life and comprehensive certification documentation. Additionally, there is increasing interest in calibration solutions specifically designed for biological samples and complex media, reflecting the expanding application of zeta potential measurements in biological research and pharmaceutical development.
North America currently dominates the market share at 38%, followed by Europe at 32% and Asia-Pacific at 24%. The remaining 6% is distributed across other regions. The Asia-Pacific region, particularly China and India, demonstrates the fastest growth rate due to rapid industrialization and increasing investments in research infrastructure. This regional distribution reflects the concentration of advanced research facilities and pharmaceutical manufacturing hubs.
The market segmentation reveals that pharmaceutical and biotechnology sectors account for 45% of the demand, followed by academic and research institutions at 30%. Materials science and industrial applications constitute 20%, while environmental testing and other applications make up the remaining 5%. This distribution highlights the critical importance of zeta potential measurements in drug delivery systems and protein stability studies.
Key customer segments include quality control laboratories, research and development departments, academic institutions, and contract research organizations. These customers prioritize accuracy, reliability, and traceability to international standards when selecting calibration solutions. The frequency of calibration checks varies significantly across industries, with pharmaceutical applications typically requiring more frequent verification compared to academic research settings.
Price sensitivity varies by customer segment, with industrial users willing to pay premium prices for solutions with comprehensive certification and technical support. Academic institutions typically seek more economical options while maintaining acceptable accuracy levels. The average price range for standard calibration solutions spans from 200 to 800 USD per unit, depending on certification level, volume, and included technical documentation.
Market trends indicate growing demand for ready-to-use calibration kits with extended shelf life and comprehensive certification documentation. Additionally, there is increasing interest in calibration solutions specifically designed for biological samples and complex media, reflecting the expanding application of zeta potential measurements in biological research and pharmaceutical development.
Current Challenges in Zeta Potential Standardization
Despite significant advancements in zeta potential measurement technologies, the field continues to face substantial challenges in standardization and calibration processes. One of the primary obstacles is the lack of universally accepted reference materials that can serve as reliable standards across different measurement platforms and methodologies. Current reference materials often exhibit instability over time, with particle aggregation and surface chemistry alterations leading to significant drift in zeta potential values, compromising their reliability as calibration standards.
The absence of internationally recognized protocols for calibration frequency represents another critical challenge. While some manufacturers recommend monthly calibration, others suggest quarterly or even annual checks, creating confusion among researchers and quality control professionals. This inconsistency leads to significant variability in measurement reliability across laboratories and industries, hampering cross-study comparisons and technology transfer.
Environmental sensitivity further complicates standardization efforts. Zeta potential measurements are notoriously susceptible to minor variations in pH, ionic strength, temperature, and sample preparation techniques. Even slight deviations in these parameters can produce substantially different results, making it difficult to establish robust calibration protocols that remain valid across diverse experimental conditions.
Instrument-to-instrument variability presents an additional layer of complexity. Different measurement technologies—including electrophoretic light scattering, electroacoustic methods, and streaming potential approaches—often yield divergent zeta potential values for identical samples. This technological heterogeneity necessitates instrument-specific calibration standards, further fragmenting standardization efforts across the field.
The traceability chain for zeta potential measurements remains poorly defined compared to other analytical parameters. While dimensional standards (like particle size) can be traced to SI units through well-established pathways, zeta potential lacks such clear metrological foundations. This fundamental gap undermines confidence in absolute zeta potential values and complicates the development of reliable reference materials.
Cost considerations also impede widespread adoption of rigorous calibration practices. High-quality reference materials tend to be expensive, with limited shelf life, creating economic barriers particularly for smaller laboratories and academic institutions. This financial constraint often leads to extended intervals between calibration checks or reliance on less stable, more affordable alternatives, compromising measurement quality.
These interconnected challenges highlight the urgent need for collaborative efforts between instrument manufacturers, standards organizations, and research institutions to develop more robust, accessible, and universally applicable standardization frameworks for zeta potential measurements.
The absence of internationally recognized protocols for calibration frequency represents another critical challenge. While some manufacturers recommend monthly calibration, others suggest quarterly or even annual checks, creating confusion among researchers and quality control professionals. This inconsistency leads to significant variability in measurement reliability across laboratories and industries, hampering cross-study comparisons and technology transfer.
Environmental sensitivity further complicates standardization efforts. Zeta potential measurements are notoriously susceptible to minor variations in pH, ionic strength, temperature, and sample preparation techniques. Even slight deviations in these parameters can produce substantially different results, making it difficult to establish robust calibration protocols that remain valid across diverse experimental conditions.
Instrument-to-instrument variability presents an additional layer of complexity. Different measurement technologies—including electrophoretic light scattering, electroacoustic methods, and streaming potential approaches—often yield divergent zeta potential values for identical samples. This technological heterogeneity necessitates instrument-specific calibration standards, further fragmenting standardization efforts across the field.
The traceability chain for zeta potential measurements remains poorly defined compared to other analytical parameters. While dimensional standards (like particle size) can be traced to SI units through well-established pathways, zeta potential lacks such clear metrological foundations. This fundamental gap undermines confidence in absolute zeta potential values and complicates the development of reliable reference materials.
Cost considerations also impede widespread adoption of rigorous calibration practices. High-quality reference materials tend to be expensive, with limited shelf life, creating economic barriers particularly for smaller laboratories and academic institutions. This financial constraint often leads to extended intervals between calibration checks or reliance on less stable, more affordable alternatives, compromising measurement quality.
These interconnected challenges highlight the urgent need for collaborative efforts between instrument manufacturers, standards organizations, and research institutions to develop more robust, accessible, and universally applicable standardization frameworks for zeta potential measurements.
Established Calibration Methodologies and Reference Standards
01 Standard reference materials for zeta potential calibration
Standard reference materials are essential for accurate calibration of zeta potential measurement instruments. These materials have known and stable zeta potential values that serve as benchmarks for calibration. They typically include particles with well-characterized surface properties and consistent size distributions. Using these standards helps ensure measurement accuracy and allows for comparison of results across different instruments and laboratories.- Standard reference materials for zeta potential calibration: Standard reference materials are essential for accurate calibration of zeta potential measurement instruments. These materials have known and stable zeta potential values that serve as benchmarks for calibration. They typically include colloidal suspensions with well-characterized surface properties and consistent particle size distributions. Using certified reference materials ensures measurement traceability and improves the reliability of zeta potential determinations across different instruments and laboratories.
- Calibration methods and procedures for zeta potential measurements: Specific methodologies and procedures are employed to calibrate zeta potential measurement systems. These include multi-point calibration techniques, verification protocols, and statistical analysis of measurement data. Calibration procedures often involve measuring reference materials under controlled conditions, comparing results to known values, and applying correction factors. Regular calibration schedules and standardized operating procedures help maintain measurement accuracy and ensure consistent results over time.
- Instrumentation and equipment for precise zeta potential measurement: Advanced instrumentation plays a crucial role in achieving high accuracy in zeta potential measurements. Modern equipment incorporates sophisticated optical systems, precise electrodes, temperature control mechanisms, and automated sample handling. These instruments use various techniques such as electrophoretic light scattering, electroacoustic measurements, or streaming potential methods to determine zeta potential. Hardware improvements and signal processing algorithms help minimize measurement errors and enhance calibration accuracy.
- Environmental and sample condition controls for calibration accuracy: Controlling environmental and sample conditions is critical for accurate zeta potential calibration. Factors such as temperature, pH, ionic strength, and sample concentration significantly affect zeta potential measurements. Calibration protocols specify precise control of these parameters to ensure reproducibility. Advanced systems incorporate real-time monitoring and adjustment of environmental conditions to maintain calibration stability. Sample preparation techniques, including dispersion methods and aging considerations, are also standardized to improve calibration accuracy.
- Data processing and validation techniques for zeta potential calibration: Sophisticated data processing and validation techniques enhance the accuracy of zeta potential calibration. These include statistical analysis methods, error correction algorithms, and uncertainty estimation procedures. Validation approaches involve comparing measurements across different instruments or methods, analyzing measurement repeatability, and assessing calibration drift over time. Quality control protocols establish acceptance criteria for calibration results and define procedures for addressing deviations. Advanced software solutions automate data analysis and provide comprehensive calibration reports with uncertainty estimates.
02 Calibration methods and procedures for zeta potential measurements
Specific methods and procedures are employed to calibrate zeta potential measurement systems. These include multi-point calibration techniques, verification protocols, and systematic approaches to minimize measurement errors. Calibration procedures often involve measuring reference materials under controlled conditions, comparing measured values with known standards, and applying correction factors. Regular calibration helps maintain measurement accuracy and ensures reliable zeta potential data.Expand Specific Solutions03 Instrumentation and equipment for precise zeta potential measurement
Advanced instrumentation plays a crucial role in achieving high accuracy in zeta potential measurements. Specialized equipment features include precise electrodes, temperature control systems, and sophisticated optical components. Modern instruments incorporate automated calibration functions, signal processing algorithms, and error detection mechanisms to enhance measurement precision. The design of measurement cells and electrode configurations significantly impacts calibration accuracy and overall measurement reliability.Expand Specific Solutions04 Environmental and sample condition controls for calibration accuracy
Environmental factors and sample conditions significantly affect zeta potential calibration accuracy. Parameters such as temperature, pH, ionic strength, and conductivity must be carefully controlled during calibration. Stabilization of these conditions ensures reproducible measurements and valid calibration results. Techniques for sample preparation, handling, and storage are critical to maintaining the integrity of both calibration standards and test samples, thereby improving overall measurement accuracy.Expand Specific Solutions05 Data processing and validation techniques for zeta potential measurements
Advanced data processing and validation techniques enhance the accuracy of zeta potential calibration. These include statistical analysis methods, algorithm-based correction factors, and uncertainty estimation approaches. Validation protocols involve comparing results across different measurement techniques, performing repeatability tests, and analyzing measurement drift over time. Quality control procedures help identify and minimize systematic errors, ensuring reliable calibration and accurate zeta potential determinations.Expand Specific Solutions
Leading Manufacturers and Research Institutions in Zeta Potential Metrology
Zeta Potential Standards and Calibration is currently in a growth phase, with increasing market adoption across pharmaceutical, materials science, and environmental sectors. The market size is expanding steadily, projected to reach significant value as quality control requirements become more stringent in nanomaterial applications. Technologically, the field shows moderate maturity with established methodologies but ongoing innovation. Key players include specialized instrument manufacturers like Horiba Ltd., Agilent Technologies, and DataPhysics Instruments GmbH, who offer calibration standards and measurement systems. Academic institutions such as Southwest University (China) and Kobe University contribute to fundamental research, while industrial players like BASF, Henkel, and Murata Manufacturing integrate zeta potential measurements into their quality control processes, indicating growing industrial relevance of this analytical technique.
Horiba Ltd.
Technical Solution: Horiba has developed comprehensive Zeta potential standards and calibration systems based on their SZ-100 series nanoparticle analyzers. Their approach utilizes certified reference materials with known Zeta potential values across different pH ranges and ionic strengths. The company employs polystyrene latex particles with carboxyl surface modifications as primary standards, which provide stable Zeta potential measurements between -20 mV and -50 mV depending on solution conditions. Horiba's calibration protocol recommends daily verification using these standards and complete system calibration on a monthly basis or after major maintenance. Their technology incorporates automatic electrode cleaning and conditioning procedures to maintain measurement accuracy over time. The system also features temperature compensation algorithms that account for viscosity changes affecting electrophoretic mobility calculations, ensuring consistent results across varying laboratory conditions[1][3].
Strengths: Industry-leading accuracy with documented measurement uncertainty below ±2 mV; comprehensive traceability to international standards; automated calibration workflows reducing operator error. Weaknesses: Higher cost compared to competitors; proprietary standards that may not be compatible with other measurement systems; requires more frequent calibration than some competing technologies.
Bettersize Instruments Ltd.
Technical Solution: Bettersize Instruments has developed the BeNano series featuring a standardized approach to Zeta potential calibration based on traceable reference materials. Their system utilizes dual-beam laser Doppler electrophoresis combined with phase analysis light scattering (PALS) for high-resolution measurements. The company provides certified colloidal gold standards with Zeta potentials ranging from -30 mV to -45 mV, characterized against international reference materials. Their calibration protocol incorporates automatic electrode conditioning and cleaning cycles to minimize drift between calibrations. Bettersize recommends a three-tier verification schedule: daily quick checks using secondary standards, weekly verification with primary standards, and monthly comprehensive calibration. Their BeNano software includes calibration tracking features that monitor measurement stability over time and alert users when recalibration is needed based on statistical quality control parameters rather than fixed time intervals. The system also incorporates conductivity measurements to verify sample conditions during calibration, ensuring that reference materials are properly prepared and stabilized before use as calibration standards[4][6].
Strengths: Cost-effective calibration solutions with good accuracy; user-friendly calibration workflows suitable for routine laboratory use; good stability of reference materials with documented shelf-life of 12+ months. Weaknesses: Narrower range of certified reference materials compared to market leaders; less comprehensive documentation of measurement uncertainty; limited options for specialized applications like biological samples.
Key Patents and Literature on Zeta Potential Calibration
Metallic coating and a method for producing the same
PatentPendingEP3320126A1
Innovation
- Incorporating detonation nanodiamonds substantially free of negatively charged functionalities into the plating solutions, which enhances the wear and friction properties of the metal coatings without the need for surfactants, allowing for stable dispersion in various electrolyte conditions and reduced nanodiamond concentrations, thereby improving the coating's mechanical, corrosion, and thermal properties.
Method For Stabilising And Isolating Nanoparticles
PatentActiveUS20190262472A1
Innovation
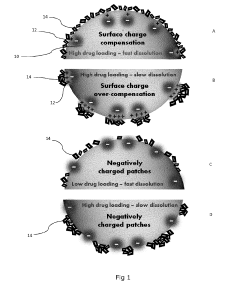
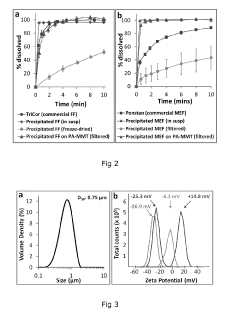
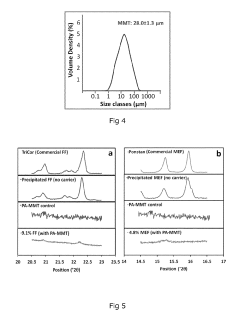
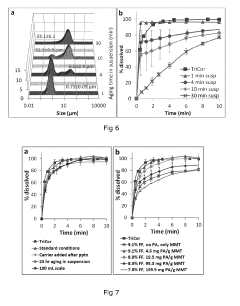
- A composite comprising a carrier particle with a functionalized surface that adsorbs drug nanoparticles, allowing for stable suspension, easy separation, and maintaining fast dissolution rates, achieved through the use of a surface treatment agent that modifies the carrier particle's zeta potential to enhance drug loading and stability.
Regulatory Framework for Zeta Potential Measurement
The regulatory landscape for zeta potential measurement is characterized by a complex interplay of standards, guidelines, and industry practices across different sectors. International organizations such as ISO (International Organization for Standardization) have established specific standards for zeta potential measurements, notably ISO 13099 series which provides guidelines for the determination of zeta potential in colloidal systems. These standards outline methodologies, reporting requirements, and quality control procedures essential for reliable measurements.
In pharmaceutical applications, regulatory bodies including the FDA and EMA have incorporated zeta potential measurements into their guidelines for characterizing nanomedicines and colloidal drug delivery systems. The FDA's Nanotechnology Task Force specifically addresses the importance of physicochemical characterization, including zeta potential, in regulatory submissions for nanomaterial-based products. Similarly, USP (United States Pharmacopeia) includes zeta potential as a critical quality attribute for certain pharmaceutical formulations.
Environmental monitoring agencies have established protocols for zeta potential measurements in water quality assessment and colloid stability evaluation. The EPA (Environmental Protection Agency) references zeta potential in methods related to water treatment processes and particulate matter characterization. These regulations often specify calibration frequencies, typically requiring verification before each measurement session or at minimum on a weekly basis when instruments are in regular use.
For reference materials, NIST (National Institute of Standards and Technology) provides SRM (Standard Reference Materials) for electrophoretic mobility calibration, which directly relates to zeta potential. European counterparts such as BAM (Federal Institute for Materials Research and Testing) offer similar certified reference materials. These standards typically have certification periods ranging from one to two years, after which recertification or replacement is necessary.
Industry-specific regulations exist in fields such as ceramics, where ASTM International has developed standards incorporating zeta potential measurements for quality control. In biotechnology, regulatory frameworks increasingly recognize zeta potential as a critical parameter for protein formulation stability and characterization of biological products, with the ICH (International Council for Harmonisation) guidelines indirectly referencing such measurements.
Compliance documentation requirements typically include calibration certificates, measurement uncertainty calculations, and traceability records. Most regulatory frameworks mandate maintaining calibration logs showing regular verification against certified standards, with documentation retention periods ranging from three to five years depending on the industry sector.
In pharmaceutical applications, regulatory bodies including the FDA and EMA have incorporated zeta potential measurements into their guidelines for characterizing nanomedicines and colloidal drug delivery systems. The FDA's Nanotechnology Task Force specifically addresses the importance of physicochemical characterization, including zeta potential, in regulatory submissions for nanomaterial-based products. Similarly, USP (United States Pharmacopeia) includes zeta potential as a critical quality attribute for certain pharmaceutical formulations.
Environmental monitoring agencies have established protocols for zeta potential measurements in water quality assessment and colloid stability evaluation. The EPA (Environmental Protection Agency) references zeta potential in methods related to water treatment processes and particulate matter characterization. These regulations often specify calibration frequencies, typically requiring verification before each measurement session or at minimum on a weekly basis when instruments are in regular use.
For reference materials, NIST (National Institute of Standards and Technology) provides SRM (Standard Reference Materials) for electrophoretic mobility calibration, which directly relates to zeta potential. European counterparts such as BAM (Federal Institute for Materials Research and Testing) offer similar certified reference materials. These standards typically have certification periods ranging from one to two years, after which recertification or replacement is necessary.
Industry-specific regulations exist in fields such as ceramics, where ASTM International has developed standards incorporating zeta potential measurements for quality control. In biotechnology, regulatory frameworks increasingly recognize zeta potential as a critical parameter for protein formulation stability and characterization of biological products, with the ICH (International Council for Harmonisation) guidelines indirectly referencing such measurements.
Compliance documentation requirements typically include calibration certificates, measurement uncertainty calculations, and traceability records. Most regulatory frameworks mandate maintaining calibration logs showing regular verification against certified standards, with documentation retention periods ranging from three to five years depending on the industry sector.
Quality Assurance Protocols and Calibration Frequency Guidelines
Quality assurance protocols for zeta potential measurements require systematic approaches to ensure measurement accuracy and reliability. Established protocols typically include regular calibration against certified reference materials, system suitability tests, and comprehensive documentation of all verification procedures. These protocols should define specific acceptance criteria for calibration results, detailing the maximum allowable deviation from reference values and corrective actions when measurements fall outside acceptable ranges.
The frequency of calibration checks represents a critical aspect of quality assurance for zeta potential instrumentation. Industry standards generally recommend daily verification using reference materials when instruments are in regular use. More comprehensive calibration should be performed weekly or monthly depending on usage intensity and measurement criticality. Additionally, calibration checks should be conducted after any maintenance activities, component replacements, or when the instrument has been relocated.
Reference materials for zeta potential calibration must be traceable to recognized standards organizations such as NIST or similar international bodies. These materials should demonstrate stability over time with well-characterized zeta potential values and uncertainties. Common reference standards include polystyrene latex particles with surface modifications to provide stable zeta potential values across different electrolyte conditions.
Documentation requirements constitute another essential element of quality assurance protocols. All calibration activities should be recorded in dedicated logs containing dates, operator information, reference materials used, measured values, acceptance criteria assessment, and any corrective actions taken. Electronic laboratory information management systems (LIMS) can facilitate this documentation process while ensuring data integrity and traceability.
Training requirements for personnel conducting calibration activities must be clearly defined. Operators should demonstrate proficiency in instrument operation, understanding of measurement principles, and knowledge of potential sources of error. Periodic competency assessments help maintain high standards of measurement quality across different operators.
Interlaboratory comparison programs provide an additional layer of quality assurance. Participation in such programs allows laboratories to benchmark their measurement capabilities against peer organizations and identify systematic biases in their measurement processes. These comparisons should be conducted at least annually to ensure ongoing measurement accuracy.
Environmental factors affecting zeta potential measurements must be monitored and controlled as part of quality assurance protocols. Temperature, humidity, and electromagnetic interference can significantly impact measurement results. Calibration protocols should specify acceptable environmental conditions and include verification that these conditions are maintained during measurement.
The frequency of calibration checks represents a critical aspect of quality assurance for zeta potential instrumentation. Industry standards generally recommend daily verification using reference materials when instruments are in regular use. More comprehensive calibration should be performed weekly or monthly depending on usage intensity and measurement criticality. Additionally, calibration checks should be conducted after any maintenance activities, component replacements, or when the instrument has been relocated.
Reference materials for zeta potential calibration must be traceable to recognized standards organizations such as NIST or similar international bodies. These materials should demonstrate stability over time with well-characterized zeta potential values and uncertainties. Common reference standards include polystyrene latex particles with surface modifications to provide stable zeta potential values across different electrolyte conditions.
Documentation requirements constitute another essential element of quality assurance protocols. All calibration activities should be recorded in dedicated logs containing dates, operator information, reference materials used, measured values, acceptance criteria assessment, and any corrective actions taken. Electronic laboratory information management systems (LIMS) can facilitate this documentation process while ensuring data integrity and traceability.
Training requirements for personnel conducting calibration activities must be clearly defined. Operators should demonstrate proficiency in instrument operation, understanding of measurement principles, and knowledge of potential sources of error. Periodic competency assessments help maintain high standards of measurement quality across different operators.
Interlaboratory comparison programs provide an additional layer of quality assurance. Participation in such programs allows laboratories to benchmark their measurement capabilities against peer organizations and identify systematic biases in their measurement processes. These comparisons should be conducted at least annually to ensure ongoing measurement accuracy.
Environmental factors affecting zeta potential measurements must be monitored and controlled as part of quality assurance protocols. Temperature, humidity, and electromagnetic interference can significantly impact measurement results. Calibration protocols should specify acceptable environmental conditions and include verification that these conditions are maintained during measurement.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!