How to Use Zeta Potential to Optimize Nanoparticle Formulations for Drug Delivery
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zeta Potential in Nanoparticle Drug Delivery: Background and Objectives
Zeta potential has emerged as a critical parameter in the development and optimization of nanoparticle-based drug delivery systems over the past three decades. Initially conceptualized in colloidal science, zeta potential represents the electrical potential at the slipping plane of a particle in suspension, effectively measuring the magnitude of electrostatic repulsion or attraction between particles. This fundamental property significantly influences nanoparticle stability, cellular interactions, biodistribution, and ultimately therapeutic efficacy.
The evolution of zeta potential applications in nanomedicine has paralleled the broader development of nanotechnology in pharmaceutical sciences. Early research in the 1990s established basic correlations between surface charge and nanoparticle behavior in biological systems. By the early 2000s, researchers began systematically manipulating zeta potential to enhance drug delivery outcomes, leading to the current sophisticated understanding of its multifaceted role in nanomedicine.
Recent technological advances have enabled more precise measurement and control of zeta potential, facilitating its integration into rational nanoparticle design strategies. Contemporary research demonstrates that optimal zeta potential values vary significantly depending on the target tissue, administration route, and therapeutic payload, necessitating tailored approaches rather than one-size-fits-all solutions.
The primary objective of zeta potential optimization in nanoparticle formulations is to achieve an ideal balance between colloidal stability and biological performance. Specifically, this involves developing nanocarriers with sufficient charge to prevent aggregation during storage and administration while enabling favorable interactions with biological barriers and target cells. Secondary objectives include enhancing drug loading efficiency, controlling release kinetics, and minimizing non-specific interactions with serum proteins and immune system components.
Current trends indicate growing interest in dynamic or switchable zeta potential systems that respond to biological microenvironments, allowing nanoparticles to adapt their surface properties throughout their journey in the body. This represents a paradigm shift from static to responsive design principles, potentially overcoming longstanding challenges in targeted drug delivery.
The technical trajectory suggests continued refinement of zeta potential manipulation strategies, with increasing emphasis on correlating in vitro measurements with in vivo performance. Emerging computational models are beginning to predict optimal zeta potential values for specific applications, potentially accelerating formulation development and reducing reliance on empirical testing.
The evolution of zeta potential applications in nanomedicine has paralleled the broader development of nanotechnology in pharmaceutical sciences. Early research in the 1990s established basic correlations between surface charge and nanoparticle behavior in biological systems. By the early 2000s, researchers began systematically manipulating zeta potential to enhance drug delivery outcomes, leading to the current sophisticated understanding of its multifaceted role in nanomedicine.
Recent technological advances have enabled more precise measurement and control of zeta potential, facilitating its integration into rational nanoparticle design strategies. Contemporary research demonstrates that optimal zeta potential values vary significantly depending on the target tissue, administration route, and therapeutic payload, necessitating tailored approaches rather than one-size-fits-all solutions.
The primary objective of zeta potential optimization in nanoparticle formulations is to achieve an ideal balance between colloidal stability and biological performance. Specifically, this involves developing nanocarriers with sufficient charge to prevent aggregation during storage and administration while enabling favorable interactions with biological barriers and target cells. Secondary objectives include enhancing drug loading efficiency, controlling release kinetics, and minimizing non-specific interactions with serum proteins and immune system components.
Current trends indicate growing interest in dynamic or switchable zeta potential systems that respond to biological microenvironments, allowing nanoparticles to adapt their surface properties throughout their journey in the body. This represents a paradigm shift from static to responsive design principles, potentially overcoming longstanding challenges in targeted drug delivery.
The technical trajectory suggests continued refinement of zeta potential manipulation strategies, with increasing emphasis on correlating in vitro measurements with in vivo performance. Emerging computational models are beginning to predict optimal zeta potential values for specific applications, potentially accelerating formulation development and reducing reliance on empirical testing.
Market Analysis of Nanoparticle-Based Drug Delivery Systems
The global market for nanoparticle-based drug delivery systems has experienced remarkable growth, reaching approximately $41.3 billion in 2021 and projected to reach $118.6 billion by 2027, with a compound annual growth rate (CAGR) of 19.1%. This substantial growth is primarily driven by the increasing prevalence of chronic diseases, advancements in nanotechnology, and the rising demand for targeted drug delivery systems.
North America currently dominates the market, accounting for nearly 40% of the global share, followed by Europe and Asia-Pacific. However, the Asia-Pacific region is expected to witness the highest growth rate during the forecast period due to increasing healthcare expenditure, growing research activities, and rising investments in pharmaceutical R&D in countries like China, India, and Japan.
The market segmentation reveals that liposomes hold the largest market share (approximately 31%) among nanoparticle types used in drug delivery, followed by polymeric nanoparticles (24%), solid lipid nanoparticles (18%), and inorganic nanoparticles (15%). This distribution highlights the importance of surface charge properties, including zeta potential, in formulation development and market success.
Oncology applications represent the largest therapeutic segment (36% of market share), followed by infectious diseases (22%), cardiovascular diseases (15%), and neurology (12%). The preference for nanoparticle formulations in cancer treatment underscores the critical role of zeta potential optimization in enhancing tumor targeting and reducing off-target effects.
Key market drivers include the growing demand for minimally invasive drug delivery methods, increasing research funding, and the rising adoption of personalized medicine approaches. The COVID-19 pandemic has further accelerated market growth, particularly for mRNA vaccine delivery systems utilizing lipid nanoparticles, where zeta potential plays a crucial role in stability and cellular uptake.
Major challenges facing the market include stringent regulatory requirements, high development costs, and manufacturing scalability issues. The complex relationship between zeta potential and in vivo performance represents a significant technical hurdle that companies must overcome to achieve commercial success.
Emerging trends include the development of stimuli-responsive nanoparticles, combination therapies, and AI-driven formulation optimization platforms that can predict optimal zeta potential values for specific applications. The increasing focus on sustainable and biodegradable materials for nanoparticle formulation is also gaining traction, particularly in European markets where environmental regulations are more stringent.
North America currently dominates the market, accounting for nearly 40% of the global share, followed by Europe and Asia-Pacific. However, the Asia-Pacific region is expected to witness the highest growth rate during the forecast period due to increasing healthcare expenditure, growing research activities, and rising investments in pharmaceutical R&D in countries like China, India, and Japan.
The market segmentation reveals that liposomes hold the largest market share (approximately 31%) among nanoparticle types used in drug delivery, followed by polymeric nanoparticles (24%), solid lipid nanoparticles (18%), and inorganic nanoparticles (15%). This distribution highlights the importance of surface charge properties, including zeta potential, in formulation development and market success.
Oncology applications represent the largest therapeutic segment (36% of market share), followed by infectious diseases (22%), cardiovascular diseases (15%), and neurology (12%). The preference for nanoparticle formulations in cancer treatment underscores the critical role of zeta potential optimization in enhancing tumor targeting and reducing off-target effects.
Key market drivers include the growing demand for minimally invasive drug delivery methods, increasing research funding, and the rising adoption of personalized medicine approaches. The COVID-19 pandemic has further accelerated market growth, particularly for mRNA vaccine delivery systems utilizing lipid nanoparticles, where zeta potential plays a crucial role in stability and cellular uptake.
Major challenges facing the market include stringent regulatory requirements, high development costs, and manufacturing scalability issues. The complex relationship between zeta potential and in vivo performance represents a significant technical hurdle that companies must overcome to achieve commercial success.
Emerging trends include the development of stimuli-responsive nanoparticles, combination therapies, and AI-driven formulation optimization platforms that can predict optimal zeta potential values for specific applications. The increasing focus on sustainable and biodegradable materials for nanoparticle formulation is also gaining traction, particularly in European markets where environmental regulations are more stringent.
Current Challenges in Zeta Potential Measurement and Control
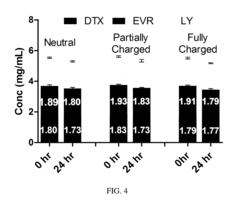
Despite significant advancements in zeta potential measurement technologies, researchers and pharmaceutical developers face several persistent challenges when applying this parameter to nanoparticle drug delivery systems. The fundamental issue lies in measurement reproducibility across different instruments and laboratories. Environmental factors such as temperature, pH, ionic strength, and sample concentration significantly influence zeta potential readings, making standardization difficult. Even minor variations in these conditions can lead to substantially different results, complicating cross-study comparisons.
Sample preparation presents another major hurdle. Nanoparticle suspensions often require specific dilution protocols to achieve optimal measurement conditions without altering the surface properties being measured. Aggregation during preparation can dramatically affect readings, while the presence of multiple particle populations with varying surface characteristics creates heterogeneity that standard measurement techniques struggle to characterize accurately.
The biological relevance of in vitro zeta potential measurements remains questionable. Laboratory conditions poorly simulate the complex biological environments encountered in vivo, where protein corona formation rapidly alters nanoparticle surface properties upon contact with biological fluids. This phenomenon, known as biofouling, can completely transform the effective zeta potential of nanoparticles, rendering pre-administration measurements potentially misleading for predicting in vivo behavior.
Technical limitations of current measurement technologies further complicate the landscape. Most commercial instruments employ light scattering techniques that perform optimally with specific particle size ranges and concentrations. Highly concentrated or polydisperse samples often yield unreliable results. Additionally, non-spherical particles present particular challenges as their orientation during measurement affects the detected electrophoretic mobility.
Real-time monitoring of zeta potential during formulation development or storage remains largely unachievable with current technologies. The inability to track dynamic changes in surface charge characteristics limits understanding of stability mechanisms and aging processes in nanoparticle formulations.
Control challenges are equally significant. Maintaining consistent zeta potential during scale-up from laboratory to industrial production presents formidable difficulties. Manufacturing processes introduce variables that can alter surface properties, while sterilization procedures may significantly impact the surface chemistry governing zeta potential. Furthermore, achieving targeted zeta potential values while simultaneously optimizing other critical quality attributes creates a complex multi-parameter optimization problem that current methodological approaches struggle to solve efficiently.
Sample preparation presents another major hurdle. Nanoparticle suspensions often require specific dilution protocols to achieve optimal measurement conditions without altering the surface properties being measured. Aggregation during preparation can dramatically affect readings, while the presence of multiple particle populations with varying surface characteristics creates heterogeneity that standard measurement techniques struggle to characterize accurately.
The biological relevance of in vitro zeta potential measurements remains questionable. Laboratory conditions poorly simulate the complex biological environments encountered in vivo, where protein corona formation rapidly alters nanoparticle surface properties upon contact with biological fluids. This phenomenon, known as biofouling, can completely transform the effective zeta potential of nanoparticles, rendering pre-administration measurements potentially misleading for predicting in vivo behavior.
Technical limitations of current measurement technologies further complicate the landscape. Most commercial instruments employ light scattering techniques that perform optimally with specific particle size ranges and concentrations. Highly concentrated or polydisperse samples often yield unreliable results. Additionally, non-spherical particles present particular challenges as their orientation during measurement affects the detected electrophoretic mobility.
Real-time monitoring of zeta potential during formulation development or storage remains largely unachievable with current technologies. The inability to track dynamic changes in surface charge characteristics limits understanding of stability mechanisms and aging processes in nanoparticle formulations.
Control challenges are equally significant. Maintaining consistent zeta potential during scale-up from laboratory to industrial production presents formidable difficulties. Manufacturing processes introduce variables that can alter surface properties, while sterilization procedures may significantly impact the surface chemistry governing zeta potential. Furthermore, achieving targeted zeta potential values while simultaneously optimizing other critical quality attributes creates a complex multi-parameter optimization problem that current methodological approaches struggle to solve efficiently.
Established Methodologies for Zeta Potential Optimization
01 Zeta potential measurement techniques
Various techniques and devices are used to measure zeta potential in colloidal systems. These include electrokinetic analyzers, electrophoretic light scattering, and specialized instruments that can determine the surface charge of particles in suspension. These measurement techniques are crucial for understanding and optimizing zeta potential in different applications, providing data that can be used to adjust formulations and processing conditions.- Zeta potential measurement techniques: Various techniques and devices have been developed for measuring zeta potential in colloidal systems. These include electroacoustic methods, electrophoretic light scattering, and specialized sensors that can provide accurate measurements of surface charge characteristics. These measurement techniques are crucial for optimizing zeta potential in different applications, as they provide the baseline data needed for subsequent modifications.
- Zeta potential modification in water treatment: Optimization of zeta potential plays a critical role in water treatment processes, particularly in filtration and purification systems. By adjusting the zeta potential of suspended particles, coagulation and flocculation processes can be enhanced, leading to more efficient removal of contaminants. This involves the addition of specific chemicals or the application of electric fields to modify the surface charge of particles in the water.
- Zeta potential control in battery and fuel cell applications: In energy storage and conversion devices such as batteries and fuel cells, zeta potential optimization is essential for improving performance and durability. By controlling the zeta potential of electrode materials and electrolytes, ion transport can be enhanced, and unwanted reactions at interfaces can be minimized. This leads to improved energy efficiency, longer cycle life, and better overall performance of these electrochemical systems.
- Pharmaceutical and biomedical applications of zeta potential optimization: In pharmaceutical formulations and biomedical applications, zeta potential optimization is crucial for controlling drug delivery, stability of suspensions, and biocompatibility of materials. By adjusting the zeta potential of drug carriers or implant surfaces, their interaction with biological systems can be controlled, leading to improved therapeutic outcomes. This includes enhancing cellular uptake, reducing immunogenicity, and increasing circulation time in the bloodstream.
- Industrial separation processes using zeta potential control: Zeta potential optimization is widely used in industrial separation processes such as mineral processing, oil recovery, and waste treatment. By manipulating the zeta potential of particles in suspension, selective separation can be achieved through techniques like flotation, sedimentation, and electrophoresis. This allows for more efficient resource recovery and waste management in various industries, reducing costs and environmental impact.
02 Zeta potential control in water treatment
Optimizing zeta potential is critical in water and wastewater treatment processes. By adjusting the zeta potential of suspended particles, coagulation and flocculation processes can be enhanced, leading to improved separation of contaminants. This involves the addition of specific chemicals that alter the surface charge of particles, causing them to aggregate and settle more efficiently, resulting in cleaner water output.Expand Specific Solutions03 Zeta potential modification in battery materials
In battery technology, particularly for lithium-ion batteries, zeta potential optimization plays a crucial role in electrode material stability and performance. By controlling the surface charge of electrode particles, researchers can improve dispersion, prevent agglomeration, and enhance electrochemical properties. This leads to batteries with higher capacity, better cycling stability, and improved overall performance.Expand Specific Solutions04 Surfactant and pH adjustment for zeta potential control
The addition of surfactants and adjustment of pH are common methods for optimizing zeta potential in colloidal systems. Surfactants can adsorb onto particle surfaces, altering their surface charge and stability. Similarly, pH adjustments can change the ionization state of surface groups, affecting the zeta potential. These approaches are widely used in industries ranging from pharmaceuticals to ceramics to achieve desired dispersion properties.Expand Specific Solutions05 Zeta potential applications in nanomaterials and drug delivery
Zeta potential optimization is essential in the development of nanomaterials and drug delivery systems. By controlling the surface charge of nanoparticles, researchers can enhance stability, prevent aggregation, and improve interactions with biological systems. This is particularly important for targeted drug delivery, where the zeta potential affects how nanocarriers interact with cell membranes and proteins in the bloodstream.Expand Specific Solutions
Leading Companies and Research Institutions in Nanopharmaceuticals
The zeta potential optimization for nanoparticle drug delivery systems is currently in a growth phase, with the global nanomedicine market expanding at approximately 12% CAGR. This technology has reached moderate maturity, with leading academic institutions (MIT, Johns Hopkins University, Shanghai Jiao Tong University) establishing fundamental research frameworks, while pharmaceutical companies (Lemonex, Samyang Holdings, New Phase Ltd.) are advancing commercial applications. Industry leaders like Moderna and Sirnaomics are leveraging zeta potential optimization to enhance RNA delivery systems, while research hospitals (Brigham & Women's, Rhode Island Hospital) are conducting clinical validation studies. The competitive landscape shows a balanced distribution between academic innovation and industrial implementation, with increasing cross-sector collaborations accelerating technology transfer and clinical translation.
Massachusetts Institute of Technology
Technical Solution: MIT has developed advanced nanoparticle formulation techniques that utilize zeta potential measurements to optimize drug delivery systems. Their approach involves precise control of surface charge through polymer coating modifications, particularly using poly(ethylene glycol) (PEG) and other biodegradable polymers. MIT researchers have established correlations between zeta potential values and nanoparticle stability, circulation time, and cellular uptake efficiency. They employ layer-by-layer assembly techniques to create multi-functional nanoparticles with tailored surface charges for targeting specific tissues. Their research has demonstrated that maintaining zeta potential values between -10 mV and -30 mV can significantly improve nanoparticle stability in physiological conditions while reducing non-specific protein adsorption[1]. MIT has also pioneered methods to dynamically alter nanoparticle zeta potential in response to environmental stimuli such as pH changes in tumor microenvironments, enhancing targeted drug delivery capabilities[3].
Strengths: Exceptional interdisciplinary approach combining materials science, bioengineering, and pharmaceutical expertise; access to state-of-the-art characterization equipment; strong industry partnerships accelerating translation. Weaknesses: Complex formulations may face manufacturing scalability challenges; higher production costs compared to conventional drug delivery systems.
ModernaTX, Inc.
Technical Solution: ModernaTX has developed proprietary lipid nanoparticle (LNP) delivery systems with optimized zeta potential for mRNA therapeutics. Their approach involves precise engineering of ionizable lipids that undergo charge transitions in response to pH changes, allowing for neutral or slightly negative zeta potential in circulation (approximately -5 to -10 mV) and positive charge in acidic endosomal environments. This pH-responsive zeta potential shift facilitates both stable circulation and efficient cellular uptake[2]. ModernaTX employs high-throughput screening methods to identify optimal lipid compositions that achieve desired zeta potential profiles across physiological pH ranges. Their formulation process includes controlled mixing techniques using microfluidic devices to ensure consistent particle size distribution and zeta potential values. The company has developed specialized analytical methods for real-time monitoring of zeta potential during manufacturing to maintain tight quality control specifications[4]. Their LNP systems incorporate PEG-lipids at precisely controlled molar ratios to shield positive charges and achieve the target zeta potential range.
Strengths: Proven clinical success with mRNA delivery platforms; extensive manufacturing infrastructure; proprietary lipid chemistry expertise; established regulatory pathway. Weaknesses: Technology primarily optimized for nucleic acid delivery rather than small molecules; potential cold chain requirements for some formulations; higher production costs compared to conventional formulations.
Key Patents and Literature on Zeta Potential-Controlled Drug Delivery
Nanocarrier drug delivery platform
PatentActiveUS20170087095A1
Innovation
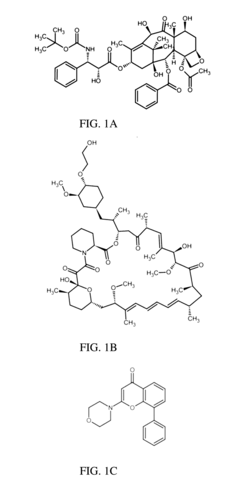
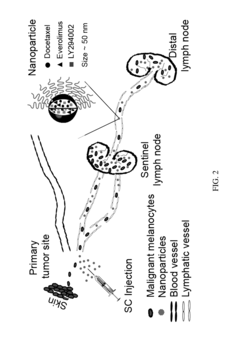
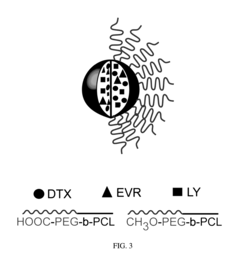
- A composition comprising a nanoparticle system with specific size and zeta potential, made of polyethyleneglycol-block-poly(ε-caprolactone), encapsulating drugs like docetaxel, everolimus, and LY294002, designed for subcutaneous or intramuscular administration to facilitate uptake and dissemination within the lymphatic system, targeting both proximal and distal lymph nodes.
Nanocarriers and their processing for diagnostics and therapeutics
PatentWO2015139051A2
Innovation
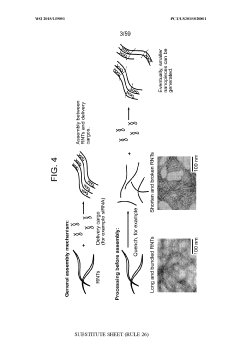
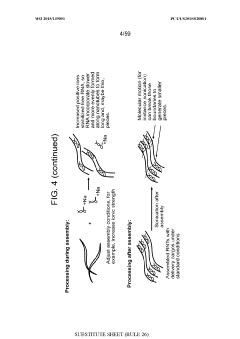
- Development of nanoparticles with specific surface charges and sizes that preferentially localize to target tissues, such as rosette nanotubes and lipid nanoparticles, which are designed to penetrate and deliver cargo compounds to specific bodily tissues, including those with extracellular matrix, by forming complexes with therapeutic or diagnostic agents.
Regulatory Considerations for Nanoparticle-Based Pharmaceuticals
The regulatory landscape for nanoparticle-based pharmaceuticals is complex and evolving, with significant implications for zeta potential optimization in drug delivery systems. Regulatory bodies worldwide, including the FDA, EMA, and NMPA, have established specific frameworks for evaluating the safety and efficacy of nanomedicines, with particular attention to physicochemical properties such as zeta potential.
These regulatory agencies require comprehensive characterization of nanoparticle formulations, including zeta potential measurements as part of quality control and batch-to-batch consistency verification. The FDA's Nanotechnology Task Force specifically addresses the importance of surface charge characteristics in determining the biological behavior of nanoparticles, requiring documentation of zeta potential values and their stability over time.
Regulatory submissions must demonstrate how zeta potential optimization contributes to the intended therapeutic outcome while minimizing potential risks. This includes providing evidence that the selected zeta potential range ensures appropriate biodistribution, cellular uptake, and reduced toxicity profiles. Stability studies must show that zeta potential remains within specified ranges throughout the product's shelf life.
International harmonization efforts, such as those by the International Council for Harmonisation (ICH) and ISO/TC 229 (Nanotechnologies), are working to standardize testing methodologies for nanoparticle characterization, including zeta potential measurements. Adherence to these emerging standards is increasingly important for global regulatory approval.
Environmental risk assessments are becoming mandatory in many jurisdictions, requiring manufacturers to evaluate how the zeta potential of their nanoformulations might influence environmental fate and potential ecological impacts after patient excretion or disposal.
Good Manufacturing Practice (GMP) guidelines for nanomedicines emphasize the need for validated analytical methods for zeta potential determination, with defined acceptance criteria and appropriate calibration standards. Regulatory bodies expect manufacturers to implement robust in-process controls to monitor zeta potential during production.
Post-market surveillance requirements are particularly stringent for nanomedicines, with regulatory authorities often requesting ongoing monitoring of physicochemical properties, including zeta potential, to detect any drift that might impact safety or efficacy profiles over time.
Researchers and manufacturers must navigate these regulatory considerations early in development, incorporating zeta potential optimization strategies that not only enhance therapeutic outcomes but also satisfy increasingly sophisticated regulatory requirements for nanomedicine characterization, manufacturing consistency, and long-term stability.
These regulatory agencies require comprehensive characterization of nanoparticle formulations, including zeta potential measurements as part of quality control and batch-to-batch consistency verification. The FDA's Nanotechnology Task Force specifically addresses the importance of surface charge characteristics in determining the biological behavior of nanoparticles, requiring documentation of zeta potential values and their stability over time.
Regulatory submissions must demonstrate how zeta potential optimization contributes to the intended therapeutic outcome while minimizing potential risks. This includes providing evidence that the selected zeta potential range ensures appropriate biodistribution, cellular uptake, and reduced toxicity profiles. Stability studies must show that zeta potential remains within specified ranges throughout the product's shelf life.
International harmonization efforts, such as those by the International Council for Harmonisation (ICH) and ISO/TC 229 (Nanotechnologies), are working to standardize testing methodologies for nanoparticle characterization, including zeta potential measurements. Adherence to these emerging standards is increasingly important for global regulatory approval.
Environmental risk assessments are becoming mandatory in many jurisdictions, requiring manufacturers to evaluate how the zeta potential of their nanoformulations might influence environmental fate and potential ecological impacts after patient excretion or disposal.
Good Manufacturing Practice (GMP) guidelines for nanomedicines emphasize the need for validated analytical methods for zeta potential determination, with defined acceptance criteria and appropriate calibration standards. Regulatory bodies expect manufacturers to implement robust in-process controls to monitor zeta potential during production.
Post-market surveillance requirements are particularly stringent for nanomedicines, with regulatory authorities often requesting ongoing monitoring of physicochemical properties, including zeta potential, to detect any drift that might impact safety or efficacy profiles over time.
Researchers and manufacturers must navigate these regulatory considerations early in development, incorporating zeta potential optimization strategies that not only enhance therapeutic outcomes but also satisfy increasingly sophisticated regulatory requirements for nanomedicine characterization, manufacturing consistency, and long-term stability.
Biocompatibility and Safety Assessment Frameworks
The assessment of biocompatibility and safety represents a critical framework in the development of nanoparticle formulations for drug delivery systems. When utilizing zeta potential measurements to optimize these formulations, researchers must integrate comprehensive safety evaluation protocols that address both immediate and long-term biological interactions. Established regulatory frameworks from organizations such as the FDA, EMA, and ISO provide structured approaches for evaluating nanoparticle safety, with specific consideration for surface charge characteristics.
Zeta potential directly influences the interaction between nanoparticles and biological systems, making it a key parameter in safety assessment. Particles with extreme positive charges often demonstrate higher cytotoxicity due to strong interactions with negatively charged cell membranes. Conversely, neutral or slightly negative zeta potentials typically exhibit improved biocompatibility profiles. These relationships must be systematically evaluated through standardized in vitro and in vivo testing protocols.
Current biocompatibility assessment frameworks incorporate multiple tiers of evaluation, beginning with in vitro cytotoxicity assays using relevant cell lines. These include MTT/MTS assays, LDH release tests, and ROS generation measurements—all of which can reveal correlations between zeta potential values and cellular responses. Hemolysis assays are particularly important for intravenously administered formulations, as surface charge significantly affects interactions with blood components.
Advanced frameworks now emphasize the importance of protein corona formation assessment, as zeta potential directly influences protein adsorption patterns that ultimately determine biological identity and clearance mechanisms. Techniques such as dynamic light scattering combined with zeta potential measurements before and after serum exposure provide valuable insights into this phenomenon.
Immunological response evaluation has emerged as another critical component, with protocols examining complement activation, cytokine production, and macrophage uptake as functions of surface charge characteristics. Long-term safety assessments must address potential accumulation in organs, particularly for formulations with zeta potential values that promote extended circulation times or specific tissue affinities.
Regulatory bodies increasingly require comprehensive characterization of zeta potential stability under physiological conditions as part of safety documentation. This includes measurements in various biological media that simulate in vivo environments. The ICH Q8(R2) guideline on pharmaceutical development specifically recommends identifying critical quality attributes, with zeta potential now recognized as one such parameter for nanomedicine formulations.
Zeta potential directly influences the interaction between nanoparticles and biological systems, making it a key parameter in safety assessment. Particles with extreme positive charges often demonstrate higher cytotoxicity due to strong interactions with negatively charged cell membranes. Conversely, neutral or slightly negative zeta potentials typically exhibit improved biocompatibility profiles. These relationships must be systematically evaluated through standardized in vitro and in vivo testing protocols.
Current biocompatibility assessment frameworks incorporate multiple tiers of evaluation, beginning with in vitro cytotoxicity assays using relevant cell lines. These include MTT/MTS assays, LDH release tests, and ROS generation measurements—all of which can reveal correlations between zeta potential values and cellular responses. Hemolysis assays are particularly important for intravenously administered formulations, as surface charge significantly affects interactions with blood components.
Advanced frameworks now emphasize the importance of protein corona formation assessment, as zeta potential directly influences protein adsorption patterns that ultimately determine biological identity and clearance mechanisms. Techniques such as dynamic light scattering combined with zeta potential measurements before and after serum exposure provide valuable insights into this phenomenon.
Immunological response evaluation has emerged as another critical component, with protocols examining complement activation, cytokine production, and macrophage uptake as functions of surface charge characteristics. Long-term safety assessments must address potential accumulation in organs, particularly for formulations with zeta potential values that promote extended circulation times or specific tissue affinities.
Regulatory bodies increasingly require comprehensive characterization of zeta potential stability under physiological conditions as part of safety documentation. This includes measurements in various biological media that simulate in vivo environments. The ICH Q8(R2) guideline on pharmaceutical development specifically recommends identifying critical quality attributes, with zeta potential now recognized as one such parameter for nanomedicine formulations.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!