How to Use Zeta Potential Data in Regulatory Dossiers for Nanomaterials — Recommended Sections
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Nanomaterial Zeta Potential Background and Objectives
Zeta potential measurement has emerged as a critical parameter in the characterization of nanomaterials, evolving from its origins in colloid science to becoming an essential tool in regulatory science. The concept was first developed in the early 20th century to describe the electrokinetic potential in colloidal systems, but has gained significant prominence in nanomaterial assessment over the past two decades as nanotechnology applications have proliferated across industries.
The evolution of zeta potential measurement techniques has paralleled advances in nanotechnology, moving from basic electrophoretic light scattering methods to sophisticated approaches capable of measuring complex nanomaterial systems under various environmental conditions. This progression has been driven by the recognition that surface charge characteristics significantly influence nanomaterial behavior in biological systems, environmental fate, and product performance.
Current regulatory frameworks for nanomaterials, including those established by the FDA, EMA, and ECHA, increasingly recognize zeta potential as a key physicochemical parameter. However, there remains considerable variation in how this data should be presented, interpreted, and utilized within regulatory submissions. This inconsistency creates challenges for both industry stakeholders and regulatory authorities.
The primary objective of this technical assessment is to establish standardized approaches for incorporating zeta potential data in regulatory dossiers for nanomaterials. Specifically, we aim to identify the most appropriate sections within regulatory submissions where zeta potential data provides maximum value, determine optimal measurement protocols that ensure data reliability and reproducibility, and develop guidelines for data interpretation that account for the complex interplay between nanomaterial properties and biological systems.
Additionally, this assessment seeks to explore how zeta potential measurements can be leveraged to predict nanomaterial behavior in biological environments, potentially reducing the need for extensive in vivo testing. This aligns with global regulatory trends toward alternative testing strategies and the 3Rs principle (Replacement, Reduction, Refinement) in toxicological assessments.
The technological trajectory suggests that zeta potential measurements will become increasingly sophisticated, potentially incorporating real-time monitoring capabilities and integration with other characterization techniques. These advancements will likely enhance the predictive power of zeta potential data in regulatory risk assessments, particularly for complex nanomaterial formulations used in medical applications, advanced materials, and consumer products.
The evolution of zeta potential measurement techniques has paralleled advances in nanotechnology, moving from basic electrophoretic light scattering methods to sophisticated approaches capable of measuring complex nanomaterial systems under various environmental conditions. This progression has been driven by the recognition that surface charge characteristics significantly influence nanomaterial behavior in biological systems, environmental fate, and product performance.
Current regulatory frameworks for nanomaterials, including those established by the FDA, EMA, and ECHA, increasingly recognize zeta potential as a key physicochemical parameter. However, there remains considerable variation in how this data should be presented, interpreted, and utilized within regulatory submissions. This inconsistency creates challenges for both industry stakeholders and regulatory authorities.
The primary objective of this technical assessment is to establish standardized approaches for incorporating zeta potential data in regulatory dossiers for nanomaterials. Specifically, we aim to identify the most appropriate sections within regulatory submissions where zeta potential data provides maximum value, determine optimal measurement protocols that ensure data reliability and reproducibility, and develop guidelines for data interpretation that account for the complex interplay between nanomaterial properties and biological systems.
Additionally, this assessment seeks to explore how zeta potential measurements can be leveraged to predict nanomaterial behavior in biological environments, potentially reducing the need for extensive in vivo testing. This aligns with global regulatory trends toward alternative testing strategies and the 3Rs principle (Replacement, Reduction, Refinement) in toxicological assessments.
The technological trajectory suggests that zeta potential measurements will become increasingly sophisticated, potentially incorporating real-time monitoring capabilities and integration with other characterization techniques. These advancements will likely enhance the predictive power of zeta potential data in regulatory risk assessments, particularly for complex nanomaterial formulations used in medical applications, advanced materials, and consumer products.
Regulatory Requirements Analysis for Nanomaterial Characterization
The regulatory landscape for nanomaterials has evolved significantly over the past decade, with various jurisdictions implementing specific frameworks to address the unique properties and potential risks associated with these materials. Zeta potential measurement has emerged as a critical characterization parameter for nanomaterials in regulatory submissions, providing valuable information about surface charge, stability, and behavior in biological systems.
In the European Union, the REACH regulation (Registration, Evaluation, Authorization and Restriction of Chemicals) has incorporated specific provisions for nanomaterials, requiring comprehensive physicochemical characterization including zeta potential data. The European Chemicals Agency (ECHA) has published guidance documents that explicitly mention zeta potential as a recommended parameter for nanomaterial characterization.
The US Food and Drug Administration (FDA) has established guidelines for nanomaterial-containing products, particularly for pharmaceutical and medical applications. Their approach emphasizes a case-by-case evaluation, with zeta potential data being increasingly requested as part of the submission package to assess stability and biological interactions.
International standards organizations, including ISO and ASTM, have developed specific protocols for nanomaterial characterization. ISO/TR 13014:2012 specifically addresses physicochemical characterization of nanomaterials and includes zeta potential as a key parameter. These standards are often referenced in regulatory frameworks globally.
Asian regulatory bodies, particularly in Japan and South Korea, have implemented nanomaterial-specific regulations that align with international standards. China has recently strengthened its regulatory framework for nanomaterials, incorporating zeta potential measurements as part of the required characterization data.
Regulatory requirements typically vary based on the intended application of nanomaterials. For pharmaceutical applications, zeta potential data is often required in sections related to drug substance characterization, formulation development, and stability studies. For medical devices, the data may be included in biocompatibility assessments and material characterization sections.
Environmental regulations increasingly demand zeta potential data to assess the potential mobility and fate of nanomaterials in environmental matrices. The OECD has developed testing guidelines that incorporate zeta potential measurements for environmental risk assessment of nanomaterials.
The trend in regulatory frameworks is moving toward harmonization of requirements across different jurisdictions, with initiatives such as the OECD Working Party on Manufactured Nanomaterials working to establish consistent approaches to nanomaterial characterization and risk assessment.
Recent regulatory developments indicate an increasing emphasis on the quality and reliability of zeta potential measurements, with specific requirements for method validation, reference materials, and reporting of measurement conditions. This reflects the growing recognition of zeta potential as a critical parameter in understanding nanomaterial behavior and safety.
In the European Union, the REACH regulation (Registration, Evaluation, Authorization and Restriction of Chemicals) has incorporated specific provisions for nanomaterials, requiring comprehensive physicochemical characterization including zeta potential data. The European Chemicals Agency (ECHA) has published guidance documents that explicitly mention zeta potential as a recommended parameter for nanomaterial characterization.
The US Food and Drug Administration (FDA) has established guidelines for nanomaterial-containing products, particularly for pharmaceutical and medical applications. Their approach emphasizes a case-by-case evaluation, with zeta potential data being increasingly requested as part of the submission package to assess stability and biological interactions.
International standards organizations, including ISO and ASTM, have developed specific protocols for nanomaterial characterization. ISO/TR 13014:2012 specifically addresses physicochemical characterization of nanomaterials and includes zeta potential as a key parameter. These standards are often referenced in regulatory frameworks globally.
Asian regulatory bodies, particularly in Japan and South Korea, have implemented nanomaterial-specific regulations that align with international standards. China has recently strengthened its regulatory framework for nanomaterials, incorporating zeta potential measurements as part of the required characterization data.
Regulatory requirements typically vary based on the intended application of nanomaterials. For pharmaceutical applications, zeta potential data is often required in sections related to drug substance characterization, formulation development, and stability studies. For medical devices, the data may be included in biocompatibility assessments and material characterization sections.
Environmental regulations increasingly demand zeta potential data to assess the potential mobility and fate of nanomaterials in environmental matrices. The OECD has developed testing guidelines that incorporate zeta potential measurements for environmental risk assessment of nanomaterials.
The trend in regulatory frameworks is moving toward harmonization of requirements across different jurisdictions, with initiatives such as the OECD Working Party on Manufactured Nanomaterials working to establish consistent approaches to nanomaterial characterization and risk assessment.
Recent regulatory developments indicate an increasing emphasis on the quality and reliability of zeta potential measurements, with specific requirements for method validation, reference materials, and reporting of measurement conditions. This reflects the growing recognition of zeta potential as a critical parameter in understanding nanomaterial behavior and safety.
Current Challenges in Zeta Potential Measurement and Reporting
Despite significant advancements in nanotechnology, the measurement and reporting of zeta potential for regulatory purposes remains fraught with challenges. The lack of standardized protocols across different industries and regulatory bodies creates significant inconsistencies in data collection and interpretation. Laboratories often employ varying sample preparation methods, buffer compositions, and measurement parameters, leading to results that cannot be reliably compared across studies or institutions.
Instrument variability presents another substantial hurdle. Different measurement technologies (electrophoretic light scattering, electroacoustic methods, etc.) can produce divergent results for identical samples. Even within the same technology platform, variations in hardware specifications and software algorithms between manufacturers contribute to measurement discrepancies that complicate regulatory submissions.
Environmental factors significantly impact zeta potential measurements but are frequently inadequately controlled or reported. Parameters such as pH, ionic strength, temperature, and the presence of dissolved gases can dramatically alter results. Many regulatory submissions fail to comprehensively document these conditions, undermining the reproducibility and validity of the data presented.
The complex nature of nanomaterial dispersions introduces additional complications. Polydisperse samples, particle aggregation, and surface adsorption phenomena can distort measurements and lead to misinterpretation of stability characteristics. Current measurement techniques struggle to account for these dynamic behaviors, particularly in biologically relevant media that more closely approximate in vivo conditions.
Reporting practices represent perhaps the most significant challenge. Many dossiers include zeta potential values without sufficient methodological details, statistical analysis, or measurement uncertainties. The absence of this contextual information severely limits the utility of the data for regulatory decision-making. Furthermore, there is no consensus on how to present zeta potential data in relation to other physicochemical properties or biological endpoints.
Regulatory agencies themselves often lack clear guidance on how zeta potential data should be evaluated or what threshold values indicate acceptable stability for different nanomaterial applications. This regulatory ambiguity creates uncertainty for manufacturers and can lead to inconsistent assessment outcomes across different jurisdictions or even within the same agency.
The scientific community has yet to establish robust correlations between zeta potential measurements in simplified laboratory media and actual nanomaterial behavior in complex biological environments. This knowledge gap undermines confidence in using zeta potential as a predictive parameter for safety and efficacy assessments in regulatory contexts.
Instrument variability presents another substantial hurdle. Different measurement technologies (electrophoretic light scattering, electroacoustic methods, etc.) can produce divergent results for identical samples. Even within the same technology platform, variations in hardware specifications and software algorithms between manufacturers contribute to measurement discrepancies that complicate regulatory submissions.
Environmental factors significantly impact zeta potential measurements but are frequently inadequately controlled or reported. Parameters such as pH, ionic strength, temperature, and the presence of dissolved gases can dramatically alter results. Many regulatory submissions fail to comprehensively document these conditions, undermining the reproducibility and validity of the data presented.
The complex nature of nanomaterial dispersions introduces additional complications. Polydisperse samples, particle aggregation, and surface adsorption phenomena can distort measurements and lead to misinterpretation of stability characteristics. Current measurement techniques struggle to account for these dynamic behaviors, particularly in biologically relevant media that more closely approximate in vivo conditions.
Reporting practices represent perhaps the most significant challenge. Many dossiers include zeta potential values without sufficient methodological details, statistical analysis, or measurement uncertainties. The absence of this contextual information severely limits the utility of the data for regulatory decision-making. Furthermore, there is no consensus on how to present zeta potential data in relation to other physicochemical properties or biological endpoints.
Regulatory agencies themselves often lack clear guidance on how zeta potential data should be evaluated or what threshold values indicate acceptable stability for different nanomaterial applications. This regulatory ambiguity creates uncertainty for manufacturers and can lead to inconsistent assessment outcomes across different jurisdictions or even within the same agency.
The scientific community has yet to establish robust correlations between zeta potential measurements in simplified laboratory media and actual nanomaterial behavior in complex biological environments. This knowledge gap undermines confidence in using zeta potential as a predictive parameter for safety and efficacy assessments in regulatory contexts.
Standard Protocols for Zeta Potential Data Inclusion
01 Zeta potential measurement systems for regulatory compliance
Systems for measuring zeta potential are essential for ensuring regulatory compliance in various industries. These systems provide data on particle surface charge and stability, which is critical for quality control and meeting regulatory standards. Advanced measurement technologies enable accurate and reproducible zeta potential data that can be used to demonstrate compliance with regulatory requirements for particle characterization and stability assessment.- Regulatory compliance frameworks for zeta potential data: Regulatory frameworks govern the collection, storage, and reporting of zeta potential data in various industries. These frameworks ensure that the data meets quality standards and is handled in compliance with relevant regulations. Compliance systems may include validation protocols, data integrity measures, and reporting requirements specific to zeta potential measurements used in pharmaceutical, environmental, or materials science applications.
- Data management systems for zeta potential compliance: Specialized data management systems are designed to handle zeta potential measurements in a compliant manner. These systems incorporate features such as audit trails, electronic signatures, and data security measures to ensure regulatory compliance. They facilitate the proper documentation, storage, and retrieval of zeta potential data while maintaining data integrity throughout the product lifecycle.
- Validation methodologies for zeta potential measurements: Validation methodologies ensure that zeta potential measurement techniques and instruments meet regulatory requirements. These methodologies include system suitability tests, calibration procedures, and performance verification protocols. Validated methods help establish the reliability and reproducibility of zeta potential data, which is critical for regulatory submissions and quality control in regulated industries.
- Risk assessment and compliance monitoring for zeta potential data: Risk assessment frameworks help identify potential compliance issues related to zeta potential data. These frameworks evaluate factors such as measurement variability, instrument calibration, and data handling procedures. Continuous monitoring systems track compliance metrics and alert stakeholders to potential deviations, enabling proactive management of regulatory risks associated with zeta potential measurements.
- Electronic documentation and reporting systems for regulatory submissions: Electronic documentation and reporting systems facilitate the submission of zeta potential data to regulatory authorities. These systems ensure that data is formatted according to regulatory requirements and includes all necessary metadata. They provide secure transmission channels and maintain records of submissions for future reference, streamlining the regulatory approval process for products where zeta potential is a critical quality attribute.
02 Data management systems for regulatory compliance reporting
Specialized data management systems are designed to collect, store, and process zeta potential data for regulatory compliance purposes. These systems include features for data integrity, audit trails, and automated reporting to meet regulatory requirements. They facilitate the organization of complex zeta potential measurements and other analytical data, ensuring that information is readily available for submission to regulatory authorities and for internal compliance verification.Expand Specific Solutions03 Risk assessment frameworks incorporating zeta potential data
Risk assessment frameworks that incorporate zeta potential data help organizations evaluate compliance risks and implement appropriate controls. These frameworks analyze zeta potential measurements alongside other parameters to identify potential stability issues or formulation problems that could lead to regulatory non-compliance. By integrating zeta potential data into risk management processes, companies can proactively address stability concerns before they become regulatory issues.Expand Specific Solutions04 Blockchain and secure systems for zeta potential data integrity
Blockchain technology and other secure systems are being implemented to ensure the integrity and traceability of zeta potential data for regulatory compliance. These technologies create immutable records of measurement data, preventing tampering and providing a verifiable chain of custody. Such systems are particularly valuable in regulated industries where data integrity is crucial for demonstrating compliance with good manufacturing practices and other regulatory requirements.Expand Specific Solutions05 Automated compliance monitoring systems for zeta potential specifications
Automated systems monitor zeta potential measurements against predefined regulatory specifications and alert users to potential compliance issues. These systems continuously track measurement data, compare it against acceptable ranges, and generate compliance reports. By automating the monitoring process, organizations can ensure consistent adherence to regulatory requirements for particle stability and surface characteristics, reducing the risk of non-compliance and facilitating timely corrective actions.Expand Specific Solutions
Key Regulatory Bodies and Industry Stakeholders
The zeta potential data regulatory landscape for nanomaterials is currently in a growth phase, with increasing standardization efforts emerging across global markets. The market size for nanomaterial characterization technologies is expanding rapidly as regulatory frameworks evolve, particularly in pharmaceutical, chemical, and consumer product sectors. Technical maturity varies significantly among key players, with established chemical giants like BASF SE, Saudi Arabian Oil Co., and General Electric Company demonstrating advanced capabilities in nanomaterial characterization. Research institutions including Massachusetts Institute of Technology, California Institute of Technology, and King Abdullah University of Science & Technology are driving innovation in measurement methodologies. Companies like JSR Corp. and Seiko Epson are developing specialized instrumentation for zeta potential analysis, while collaborative efforts between industry and academia are accelerating standardization of regulatory submission protocols.
The Georgia Tech Research Corp.
Technical Solution: Georgia Tech has developed a systematic approach for integrating zeta potential measurements into regulatory submissions for nanomaterials. Their methodology focuses on establishing clear relationships between surface charge characteristics and functional properties relevant to safety assessment. Georgia Tech researchers employ a tiered testing strategy that begins with baseline zeta potential measurements in standard media, followed by increasingly complex biological and environmental matrices to simulate real-world conditions[4]. Their technical approach includes advanced statistical analysis of zeta potential distributions rather than simple mean values, providing insights into surface heterogeneity and potential for differential biological interactions. For regulatory dossiers, Georgia Tech recommends including zeta potential data in the physicochemical characterization section with cross-references to stability assessments and toxicological evaluations. Their framework includes specific guidance on measurement conditions (pH, ionic strength, temperature) that should be reported to ensure reproducibility and regulatory acceptance. Additionally, they've developed predictive models that correlate zeta potential with cellular uptake mechanisms, allowing for more informed safety assessments without extensive biological testing[7].
Strengths: Their tiered testing approach efficiently allocates resources while providing comprehensive characterization. The statistical analysis of zeta potential distributions offers deeper insights than conventional mean values. Weaknesses: The complex statistical approaches may present interpretation challenges for regulatory reviewers without specialized training. Their predictive models, while valuable, may require additional validation for novel nanomaterial classes.
California Institute of Technology
Technical Solution: Caltech has developed a precision-focused approach to zeta potential characterization for nanomaterial regulatory submissions. Their methodology emphasizes high-resolution measurements across physiologically relevant conditions, particularly focusing on protein corona formation and its impact on effective surface charge. Caltech researchers utilize advanced electrokinetic analyzers combined with microfluidic systems to capture zeta potential under flow conditions that better simulate in vivo environments[1]. Their technical framework includes correlation analyses between zeta potential and other surface properties (hydrophobicity, surface chemistry) to create comprehensive surface interaction profiles. For regulatory dossiers, Caltech recommends including zeta potential data in three main sections: (1) Physicochemical Characterization, with detailed measurement protocols and quality control parameters; (2) Environmental Fate and Behavior, demonstrating how surface charge influences environmental transport and transformation; and (3) Toxicological Assessment, linking zeta potential to cellular uptake mechanisms and biodistribution patterns. Their approach includes mathematical modeling to extrapolate zeta potential behavior across different biological compartments, enhancing predictive capabilities for regulatory risk assessment[5].
Strengths: Highly precise measurement techniques provide exceptional data quality and reproducibility. Their integration of zeta potential with protein corona formation offers valuable insights into biological behavior. Weaknesses: The sophisticated measurement systems may not be widely accessible, potentially limiting broader adoption. The approach may be unnecessarily complex for simpler nanomaterial applications with well-established safety profiles.
Critical Technical Specifications for Zeta Potential Documentation
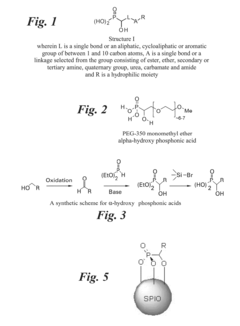
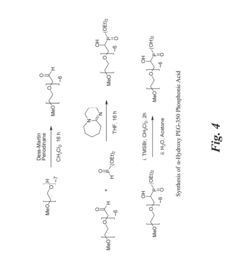
Treating water insoluble nanoparticles with hydrophilic alpha-hydroxyphosphonic acid conjugates, the so modified nanoparticles and their use as contrast agents
PatentActiveUS20160038617A1
Innovation
- Conjugates of α-hydroxyphosphonic acid linked via the carbon atom carrying the α-hydroxy group with hydrophilic moieties, such as polyethylene glycol, are used to enhance the hydrophilicity of nanoparticles, maintaining their stability in aqueous media and avoiding interactions with human tissue by maintaining a neutral zeta potential.
Case Studies of Successful Regulatory Submissions
Several regulatory submissions have successfully incorporated zeta potential data to gain approval for nanomaterial-based products. The European Medicines Agency (EMA) approved a liposomal doxorubicin formulation where manufacturers provided comprehensive zeta potential characterization across different physiological conditions. This data demonstrated stability in blood circulation and helped establish consistent quality control parameters, which proved critical for regulatory acceptance.
In the United States, the FDA approved a nanoscale iron oxide contrast agent after reviewing extensive zeta potential measurements that correlated with in vivo biodistribution patterns. The submission included stability data under various pH conditions and protein environments, establishing clear relationships between surface charge characteristics and biological behavior. This case highlighted how zeta potential data can bridge physicochemical properties with biological performance.
A notable agricultural nanopesticide submission to the Environmental Protection Agency incorporated zeta potential measurements to address environmental fate concerns. The data demonstrated how surface charge influenced soil adsorption and aquatic mobility, allowing regulators to make informed decisions about environmental risk. The submission established threshold values for zeta potential that predicted environmental behavior, creating a precedent for future agricultural nanomaterial evaluations.
In Australia, the Therapeutic Goods Administration approved a nanosilver-based wound dressing after manufacturers provided zeta potential data showing consistent surface properties across production batches. The submission correlated zeta potential with antimicrobial efficacy and demonstrated minimal variation in critical quality attributes, establishing this parameter as a key quality control metric.
The European Chemicals Agency accepted a registration for engineered silica nanoparticles where zeta potential measurements across different environmental conditions helped establish the material's stability profile. The submission included isoelectric point determination and aggregation behavior predictions based on zeta potential, which informed exposure assessment models.
These case studies demonstrate that successful regulatory submissions typically include zeta potential data that: (1) spans relevant physiological or environmental conditions, (2) correlates with functional performance or safety parameters, (3) establishes batch-to-batch consistency, and (4) provides mechanistic understanding of biological or environmental interactions. Regulatory authorities increasingly view comprehensive zeta potential characterization as evidence of thorough product understanding and quality control.
In the United States, the FDA approved a nanoscale iron oxide contrast agent after reviewing extensive zeta potential measurements that correlated with in vivo biodistribution patterns. The submission included stability data under various pH conditions and protein environments, establishing clear relationships between surface charge characteristics and biological behavior. This case highlighted how zeta potential data can bridge physicochemical properties with biological performance.
A notable agricultural nanopesticide submission to the Environmental Protection Agency incorporated zeta potential measurements to address environmental fate concerns. The data demonstrated how surface charge influenced soil adsorption and aquatic mobility, allowing regulators to make informed decisions about environmental risk. The submission established threshold values for zeta potential that predicted environmental behavior, creating a precedent for future agricultural nanomaterial evaluations.
In Australia, the Therapeutic Goods Administration approved a nanosilver-based wound dressing after manufacturers provided zeta potential data showing consistent surface properties across production batches. The submission correlated zeta potential with antimicrobial efficacy and demonstrated minimal variation in critical quality attributes, establishing this parameter as a key quality control metric.
The European Chemicals Agency accepted a registration for engineered silica nanoparticles where zeta potential measurements across different environmental conditions helped establish the material's stability profile. The submission included isoelectric point determination and aggregation behavior predictions based on zeta potential, which informed exposure assessment models.
These case studies demonstrate that successful regulatory submissions typically include zeta potential data that: (1) spans relevant physiological or environmental conditions, (2) correlates with functional performance or safety parameters, (3) establishes batch-to-batch consistency, and (4) provides mechanistic understanding of biological or environmental interactions. Regulatory authorities increasingly view comprehensive zeta potential characterization as evidence of thorough product understanding and quality control.
International Harmonization of Nanomaterial Reporting Standards
The global landscape of nanomaterial regulation presents significant challenges due to the lack of standardized reporting requirements across different jurisdictions. Currently, regulatory frameworks for nanomaterials vary substantially between regions such as the European Union, United States, Japan, and China, creating compliance burdens for manufacturers and potentially impeding international trade and scientific collaboration.
Efforts toward international harmonization of nanomaterial reporting standards have gained momentum in recent years, with several key initiatives leading the way. The Organization for Economic Cooperation and Development (OECD) Working Party on Manufactured Nanomaterials has established testing guidelines and standardized approaches for characterizing nanomaterials, including protocols for zeta potential measurement.
The International Organization for Standardization (ISO) Technical Committee 229 has developed numerous standards specifically addressing nanomaterial characterization, terminology, and measurement techniques. These standards provide a foundation for consistent reporting across borders, though their adoption in regulatory frameworks remains inconsistent.
Regulatory cooperation mechanisms such as the International Cooperation on Cosmetics Regulation (ICCR) and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) have begun incorporating nanomaterial-specific considerations into their guidelines, recognizing the need for alignment in this rapidly evolving field.
Zeta potential data represents a critical area where harmonization efforts could yield significant benefits. Currently, different regulatory bodies request varying levels of detail regarding zeta potential measurements, from simple numerical values to comprehensive methodological descriptions including buffer composition, pH conditions, and measurement parameters.
A promising development is the emergence of mutual recognition agreements between regulatory authorities regarding nanomaterial characterization data. These agreements allow for the acceptance of test results across jurisdictions when performed according to mutually recognized protocols, reducing redundant testing requirements and associated costs.
Challenges to harmonization include differing regional priorities, varying definitions of what constitutes a nanomaterial, and the rapid pace of technological advancement that continually introduces new materials requiring regulatory consideration. Additionally, developing nations may lack the technical infrastructure to implement sophisticated characterization requirements, creating potential trade barriers.
Moving forward, international consensus building on minimum reporting requirements for zeta potential data would significantly benefit global nanomaterial regulation. This would ideally include standardized measurement conditions, reporting formats, and quality assurance protocols that balance scientific rigor with practical implementation considerations.
Efforts toward international harmonization of nanomaterial reporting standards have gained momentum in recent years, with several key initiatives leading the way. The Organization for Economic Cooperation and Development (OECD) Working Party on Manufactured Nanomaterials has established testing guidelines and standardized approaches for characterizing nanomaterials, including protocols for zeta potential measurement.
The International Organization for Standardization (ISO) Technical Committee 229 has developed numerous standards specifically addressing nanomaterial characterization, terminology, and measurement techniques. These standards provide a foundation for consistent reporting across borders, though their adoption in regulatory frameworks remains inconsistent.
Regulatory cooperation mechanisms such as the International Cooperation on Cosmetics Regulation (ICCR) and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) have begun incorporating nanomaterial-specific considerations into their guidelines, recognizing the need for alignment in this rapidly evolving field.
Zeta potential data represents a critical area where harmonization efforts could yield significant benefits. Currently, different regulatory bodies request varying levels of detail regarding zeta potential measurements, from simple numerical values to comprehensive methodological descriptions including buffer composition, pH conditions, and measurement parameters.
A promising development is the emergence of mutual recognition agreements between regulatory authorities regarding nanomaterial characterization data. These agreements allow for the acceptance of test results across jurisdictions when performed according to mutually recognized protocols, reducing redundant testing requirements and associated costs.
Challenges to harmonization include differing regional priorities, varying definitions of what constitutes a nanomaterial, and the rapid pace of technological advancement that continually introduces new materials requiring regulatory consideration. Additionally, developing nations may lack the technical infrastructure to implement sophisticated characterization requirements, creating potential trade barriers.
Moving forward, international consensus building on minimum reporting requirements for zeta potential data would significantly benefit global nanomaterial regulation. This would ideally include standardized measurement conditions, reporting formats, and quality assurance protocols that balance scientific rigor with practical implementation considerations.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!