Zeta Potential Measurement Troubleshooting: Electrode, Conductivity and Instrument Checks
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zeta Potential Measurement Background and Objectives
Zeta potential measurement has evolved significantly since its conceptual development in the early 20th century. Initially proposed as part of the DLVO theory (Derjaguin, Landau, Verwey, and Overbeek), this measurement technique has become fundamental in understanding colloidal stability and particle surface characteristics. The progression from rudimentary electrophoretic mobility measurements to today's sophisticated laser Doppler electrophoresis systems represents a remarkable technological advancement in the field of colloid science.
The primary objective of zeta potential measurement is to quantify the electrical potential at the slipping plane of a colloidal particle, which serves as a critical indicator of suspension stability. This parameter provides essential insights into the electrostatic repulsion between particles, helping predict aggregation behavior in various dispersion systems. Modern applications span numerous industries including pharmaceuticals, ceramics, water treatment, and nanotechnology, where precise control of surface properties is paramount.
Current zeta potential measurement technologies predominantly rely on electrokinetic phenomena, particularly electrophoresis, where particle movement in an applied electric field is analyzed. The technological evolution has focused on improving measurement accuracy, reproducibility, and applicability across diverse sample types. Recent advancements have enabled measurements in concentrated suspensions and non-aqueous media, expanding the technique's utility in complex formulations.
The troubleshooting aspects of zeta potential measurement, particularly regarding electrodes, conductivity, and instrument checks, represent critical challenges in maintaining measurement integrity. Electrode degradation can significantly impact measurement accuracy, while sample conductivity variations may interfere with proper field application. Instrument calibration and maintenance remain ongoing concerns for laboratories seeking consistent results.
Our technical research aims to comprehensively analyze the current state of zeta potential measurement troubleshooting methodologies, with particular emphasis on electrode maintenance protocols, conductivity compensation techniques, and instrument validation procedures. By examining these aspects, we seek to establish standardized approaches that enhance measurement reliability across different instrument platforms and sample conditions.
The expected outcomes include identifying optimal electrode materials and configurations that minimize degradation effects, developing robust conductivity correction algorithms, and establishing comprehensive instrument check protocols that ensure measurement consistency. These advancements would address persistent challenges in the field, particularly for complex biological samples, high-salt environments, and non-aqueous systems where traditional measurement approaches often falter.
As emerging technologies like microfluidic zeta potential analyzers and artificial intelligence-assisted data interpretation gain prominence, understanding fundamental troubleshooting principles becomes increasingly important to ensure proper implementation and result interpretation in next-generation measurement systems.
The primary objective of zeta potential measurement is to quantify the electrical potential at the slipping plane of a colloidal particle, which serves as a critical indicator of suspension stability. This parameter provides essential insights into the electrostatic repulsion between particles, helping predict aggregation behavior in various dispersion systems. Modern applications span numerous industries including pharmaceuticals, ceramics, water treatment, and nanotechnology, where precise control of surface properties is paramount.
Current zeta potential measurement technologies predominantly rely on electrokinetic phenomena, particularly electrophoresis, where particle movement in an applied electric field is analyzed. The technological evolution has focused on improving measurement accuracy, reproducibility, and applicability across diverse sample types. Recent advancements have enabled measurements in concentrated suspensions and non-aqueous media, expanding the technique's utility in complex formulations.
The troubleshooting aspects of zeta potential measurement, particularly regarding electrodes, conductivity, and instrument checks, represent critical challenges in maintaining measurement integrity. Electrode degradation can significantly impact measurement accuracy, while sample conductivity variations may interfere with proper field application. Instrument calibration and maintenance remain ongoing concerns for laboratories seeking consistent results.
Our technical research aims to comprehensively analyze the current state of zeta potential measurement troubleshooting methodologies, with particular emphasis on electrode maintenance protocols, conductivity compensation techniques, and instrument validation procedures. By examining these aspects, we seek to establish standardized approaches that enhance measurement reliability across different instrument platforms and sample conditions.
The expected outcomes include identifying optimal electrode materials and configurations that minimize degradation effects, developing robust conductivity correction algorithms, and establishing comprehensive instrument check protocols that ensure measurement consistency. These advancements would address persistent challenges in the field, particularly for complex biological samples, high-salt environments, and non-aqueous systems where traditional measurement approaches often falter.
As emerging technologies like microfluidic zeta potential analyzers and artificial intelligence-assisted data interpretation gain prominence, understanding fundamental troubleshooting principles becomes increasingly important to ensure proper implementation and result interpretation in next-generation measurement systems.
Market Applications and Demand Analysis
Zeta potential measurement technology has witnessed significant market growth across various industries due to its critical role in understanding colloidal stability and particle interactions. The global market for zeta potential analyzers is currently valued at approximately 350 million USD, with projections indicating a compound annual growth rate of 5.7% through 2028, driven primarily by expanding applications in pharmaceuticals and advanced materials.
The pharmaceutical and biotechnology sectors represent the largest market segment, accounting for nearly 40% of the total demand. These industries rely heavily on zeta potential measurements for drug formulation stability, protein aggregation studies, and quality control processes. The increasing development of complex biologics and nanomedicines has further intensified the need for precise zeta potential analysis to ensure product efficacy and safety.
Water treatment and environmental monitoring constitute another substantial market segment. Municipal facilities and environmental agencies utilize zeta potential measurements to optimize coagulation processes, monitor colloid stability in natural waters, and assess the effectiveness of water purification systems. This application area has seen increased regulatory pressure, driving demand for more reliable troubleshooting protocols and instrument validation methods.
The academic and research sector demonstrates steady growth in demand, particularly in nanomaterial development and colloidal science. Universities and research institutions require dependable zeta potential measurement systems with comprehensive troubleshooting capabilities to ensure experimental reproducibility and validity of scientific findings.
Electronics and semiconductor manufacturing represents an emerging high-value market segment. As device miniaturization continues, manufacturers increasingly rely on zeta potential measurements to control electroplating processes, chemical mechanical polishing, and particle contamination. The stringent quality requirements in this sector necessitate advanced troubleshooting protocols for electrode maintenance and conductivity calibration.
Regional analysis reveals North America and Europe as dominant markets, collectively accounting for approximately 65% of global demand. However, the Asia-Pacific region, particularly China and India, shows the fastest growth rate at 7.8% annually, driven by expanding pharmaceutical manufacturing and increasing environmental regulations.
Market research indicates that customers increasingly prioritize instrument reliability and ease of troubleshooting over initial acquisition costs. Survey data shows that 78% of users consider electrode maintenance protocols and conductivity calibration features as critical factors in purchasing decisions, highlighting the importance of comprehensive troubleshooting capabilities in modern zeta potential measurement systems.
The pharmaceutical and biotechnology sectors represent the largest market segment, accounting for nearly 40% of the total demand. These industries rely heavily on zeta potential measurements for drug formulation stability, protein aggregation studies, and quality control processes. The increasing development of complex biologics and nanomedicines has further intensified the need for precise zeta potential analysis to ensure product efficacy and safety.
Water treatment and environmental monitoring constitute another substantial market segment. Municipal facilities and environmental agencies utilize zeta potential measurements to optimize coagulation processes, monitor colloid stability in natural waters, and assess the effectiveness of water purification systems. This application area has seen increased regulatory pressure, driving demand for more reliable troubleshooting protocols and instrument validation methods.
The academic and research sector demonstrates steady growth in demand, particularly in nanomaterial development and colloidal science. Universities and research institutions require dependable zeta potential measurement systems with comprehensive troubleshooting capabilities to ensure experimental reproducibility and validity of scientific findings.
Electronics and semiconductor manufacturing represents an emerging high-value market segment. As device miniaturization continues, manufacturers increasingly rely on zeta potential measurements to control electroplating processes, chemical mechanical polishing, and particle contamination. The stringent quality requirements in this sector necessitate advanced troubleshooting protocols for electrode maintenance and conductivity calibration.
Regional analysis reveals North America and Europe as dominant markets, collectively accounting for approximately 65% of global demand. However, the Asia-Pacific region, particularly China and India, shows the fastest growth rate at 7.8% annually, driven by expanding pharmaceutical manufacturing and increasing environmental regulations.
Market research indicates that customers increasingly prioritize instrument reliability and ease of troubleshooting over initial acquisition costs. Survey data shows that 78% of users consider electrode maintenance protocols and conductivity calibration features as critical factors in purchasing decisions, highlighting the importance of comprehensive troubleshooting capabilities in modern zeta potential measurement systems.
Current Challenges in Zeta Potential Measurement
Zeta potential measurement faces significant challenges that impact the accuracy and reliability of results across various applications. The primary issue stems from electrode degradation, which occurs gradually with repeated use. Electrodes can become contaminated with sample residues, leading to inconsistent readings and reduced sensitivity. This degradation is particularly problematic when measuring samples with high ionic strength or extreme pH values, which accelerate electrode wear.
Conductivity-related challenges represent another major obstacle in zeta potential measurements. Samples with very high conductivity often produce excessive joule heating during measurement, distorting results through thermal gradients and convection effects. Conversely, extremely low conductivity samples may not generate sufficient signal for accurate detection. The relationship between conductivity and measurement quality follows a non-linear pattern, creating a narrow optimal measurement window that varies by instrument and application.
Instrument calibration and maintenance present ongoing challenges for laboratories. Many facilities lack standardized protocols for regular calibration verification, leading to undetected drift in measurements over time. The absence of universally accepted reference materials for zeta potential calibration compounds this problem, making cross-laboratory comparisons difficult and reducing result reproducibility across different research groups.
Sample preparation inconsistencies further complicate measurements. Parameters such as concentration, dispersion method, and equilibration time significantly impact zeta potential values but are often inadequately controlled or reported. This variability introduces systematic errors that can be difficult to identify and correct, particularly when comparing results from different studies or laboratories.
Environmental factors, including temperature fluctuations and electromagnetic interference, can substantially affect measurement stability. Modern instruments incorporate various compensation mechanisms, but these corrections introduce their own uncertainties and may not fully account for all environmental variables, especially in challenging measurement environments.
Data interpretation presents perhaps the most subtle challenge. The conversion of electrophoretic mobility to zeta potential relies on theoretical models with inherent assumptions that may not hold for complex biological samples, nanoparticle suspensions, or non-spherical particles. The Henry, Smoluchowski, and Hückel equations each have specific validity domains that are frequently overlooked in practical applications, leading to misinterpretation of results and incorrect conclusions about colloidal stability.
Conductivity-related challenges represent another major obstacle in zeta potential measurements. Samples with very high conductivity often produce excessive joule heating during measurement, distorting results through thermal gradients and convection effects. Conversely, extremely low conductivity samples may not generate sufficient signal for accurate detection. The relationship between conductivity and measurement quality follows a non-linear pattern, creating a narrow optimal measurement window that varies by instrument and application.
Instrument calibration and maintenance present ongoing challenges for laboratories. Many facilities lack standardized protocols for regular calibration verification, leading to undetected drift in measurements over time. The absence of universally accepted reference materials for zeta potential calibration compounds this problem, making cross-laboratory comparisons difficult and reducing result reproducibility across different research groups.
Sample preparation inconsistencies further complicate measurements. Parameters such as concentration, dispersion method, and equilibration time significantly impact zeta potential values but are often inadequately controlled or reported. This variability introduces systematic errors that can be difficult to identify and correct, particularly when comparing results from different studies or laboratories.
Environmental factors, including temperature fluctuations and electromagnetic interference, can substantially affect measurement stability. Modern instruments incorporate various compensation mechanisms, but these corrections introduce their own uncertainties and may not fully account for all environmental variables, especially in challenging measurement environments.
Data interpretation presents perhaps the most subtle challenge. The conversion of electrophoretic mobility to zeta potential relies on theoretical models with inherent assumptions that may not hold for complex biological samples, nanoparticle suspensions, or non-spherical particles. The Henry, Smoluchowski, and Hückel equations each have specific validity domains that are frequently overlooked in practical applications, leading to misinterpretation of results and incorrect conclusions about colloidal stability.
Electrode and Conductivity Troubleshooting Solutions
01 Sample preparation and handling issues
Proper sample preparation is critical for accurate zeta potential measurements. Issues such as contamination, improper dilution, or inadequate dispersion can lead to erroneous results. Solutions include using clean equipment, appropriate sample concentration, and ensuring stable dispersions. Proper handling procedures and standardized preparation protocols can significantly improve measurement reliability and reproducibility.- Sample preparation and handling for zeta potential measurements: Proper sample preparation is critical for accurate zeta potential measurements. This includes techniques for handling colloidal suspensions, nanoparticles, and other materials to ensure reliable results. Factors such as sample concentration, dispersion methods, and contamination prevention significantly impact measurement accuracy. Specialized preparation protocols may be required for different material types to maintain sample stability during measurement.
- Electrode and cell maintenance in zeta potential analyzers: Maintenance of electrodes and measurement cells is essential for troubleshooting zeta potential measurement issues. Problems such as electrode fouling, degradation, or contamination can lead to erroneous results. Regular cleaning protocols, electrode replacement schedules, and proper storage conditions help maintain measurement accuracy. Cell design features that minimize bubble formation and ensure proper sample flow are also important considerations for reliable measurements.
- Signal processing and data analysis techniques: Advanced signal processing and data analysis methods are crucial for interpreting zeta potential measurements correctly. This includes algorithms for noise reduction, peak identification, and statistical analysis of measurement data. Software solutions that can identify measurement artifacts, detect outliers, and provide quality control indicators help troubleshoot measurement issues. Calibration procedures and reference standards are also important for validating measurement accuracy.
- Environmental factors affecting measurement accuracy: Environmental conditions significantly impact zeta potential measurements and require careful control. Temperature fluctuations, pH variations, ionic strength changes, and electromagnetic interference can all lead to measurement errors. Stabilization systems for controlling these parameters during measurement help ensure reproducible results. Shielding techniques and laboratory environment controls are essential troubleshooting considerations for obtaining consistent measurements.
- Instrument calibration and validation methods: Regular calibration and validation procedures are essential for maintaining zeta potential measurement accuracy. This includes the use of standard reference materials with known zeta potential values, verification protocols, and system performance checks. Automated calibration systems help identify instrument drift and systematic errors. Comparative analysis between different measurement techniques can validate results and identify instrument-specific issues that may affect measurement reliability.
02 Electrode and cell maintenance
Electrode fouling, cell contamination, and wear can significantly affect zeta potential measurements. Regular cleaning, maintenance, and calibration of measurement cells and electrodes are essential for accurate results. Protocols for detecting electrode degradation and methods for restoring electrode performance help ensure measurement consistency over time.Expand Specific Solutions03 Environmental and experimental conditions
Zeta potential measurements are highly sensitive to environmental factors such as temperature, pH, ionic strength, and conductivity. Controlling these parameters during measurement is crucial for obtaining reliable results. Techniques for stabilizing measurement conditions and accounting for environmental variations can improve data quality and reproducibility.Expand Specific Solutions04 Instrument calibration and validation
Regular calibration and validation of zeta potential measurement instruments are essential for accurate results. This includes using standard reference materials, performing system suitability tests, and implementing quality control procedures. Proper calibration protocols help identify instrument drift and ensure measurement accuracy across different samples and conditions.Expand Specific Solutions05 Data analysis and interpretation challenges
Interpreting zeta potential measurement data requires understanding various factors that can affect results. Challenges include distinguishing between multiple populations in polydisperse samples, identifying artifacts, and applying appropriate mathematical models. Advanced data processing techniques and software solutions can help overcome these challenges and extract meaningful information from complex measurements.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The zeta potential measurement technology market is currently in a growth phase, characterized by increasing adoption across pharmaceutical, environmental, and materials science sectors. The global market size is estimated to be around $300-400 million, with projected annual growth of 6-8%. Technologically, the field has reached moderate maturity with established measurement principles, though innovations continue in electrode design and conductivity management. Key players include Horiba Ltd. and Anton Paar GmbH as market leaders with comprehensive instrument portfolios, while Bettersize Instruments and DataPhysics Instruments offer specialized solutions. Academic institutions like Osaka University and Carnegie Mellon contribute significant research advancements, particularly in troubleshooting methodologies for electrode degradation and conductivity interference issues.
Horiba Ltd.
Technical Solution: Horiba's Zeta potential measurement troubleshooting system incorporates advanced electrode monitoring technology that continuously evaluates electrode condition during measurements. Their SZ-100 series utilizes a patented carbon electrode design that minimizes electrode polarization effects and extends electrode life significantly compared to traditional gold electrodes. The system features automatic conductivity adjustment algorithms that compensate for sample conductivity variations in real-time, ensuring accurate measurements across diverse sample types. Horiba's troubleshooting protocol includes comprehensive self-diagnostic routines that identify common issues such as electrode fouling, bubble formation, and sample contamination. Their latest instruments incorporate machine learning algorithms that can predict electrode degradation before measurement quality is compromised, allowing preventative maintenance rather than reactive troubleshooting[1][3].
Strengths: Superior electrode longevity with carbon electrode technology; comprehensive self-diagnostics; predictive maintenance capabilities. Weaknesses: Higher initial cost compared to competitors; requires more frequent calibration for high-precision measurements; proprietary software may limit integration with third-party systems.
Anton Paar GmbH
Technical Solution: Anton Paar's approach to Zeta potential measurement troubleshooting centers on their proprietary Omega electrode design, which features a unique geometry that minimizes polarization effects and extends electrode lifespan. Their Litesizer series incorporates automated electrode check protocols that evaluate electrode performance before each measurement cycle, detecting issues before they affect results. The system employs a multi-frequency measurement technique that allows for conductivity compensation across a wide range (0.1-200 mS/cm), significantly broader than industry standards. Their instruments feature a patented optical alignment verification system that ensures proper cell positioning, eliminating a common source of measurement errors. Anton Paar has developed specialized algorithms for identifying and filtering outlier data points caused by large particles or aggregates that could otherwise skew zeta potential results[2][4]. The company's troubleshooting software includes visual guidance for common issues, with step-by-step resolution procedures tailored to specific error codes.
Strengths: Exceptional measurement range for conductivity; sophisticated optical alignment verification; comprehensive automated checks before measurements; intuitive troubleshooting interface. Weaknesses: Premium pricing positions instruments at the higher end of the market; complex systems require more extensive training; replacement electrodes are proprietary and relatively expensive.
Critical Technologies in Zeta Potential Instrumentation
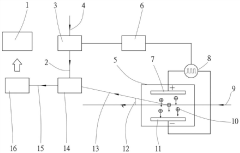
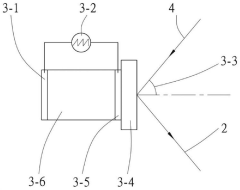
Zeta potential measurement method and measurement device
PatentActiveUS12313590B2
Innovation
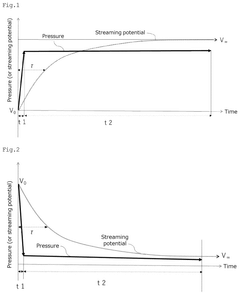
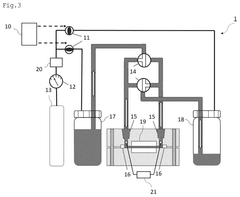
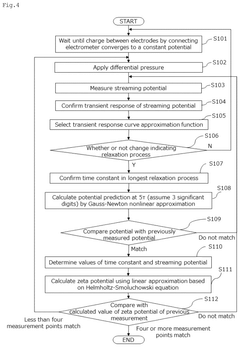
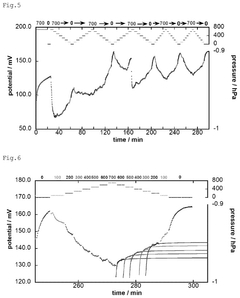
- A method and device for measuring zeta potential using a step-wise pressure change profile, where the pressure is changed in a rising or falling manner for a short time followed by a steady state phase lasting longer than the relaxation time, allowing for the calculation of zeta potential from the asymptotic value of the streaming potential.
Method and device for measuring Zeta potential through cosine fitting technology
PatentActiveCN115508421A
Innovation
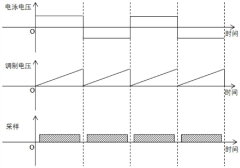
- Cosine fitting technology is used to measure the Zeta potential. The reference light is modulated with a fixed frequency shift and an alternating square wave voltage is applied to the electrophoresis electrode to make the charged particles move at a constant speed under positive and negative electric fields. The automatic response of each string of signals is calculated. The correlation function removes the low-frequency terms and obtains the angular frequencies of the positive and negative signals through cosine function fitting to calculate the Zeta potential.
Calibration and Quality Control Protocols
Effective calibration and quality control protocols are essential for ensuring reliable zeta potential measurements. These protocols should be implemented systematically to maintain instrument accuracy and data integrity. Regular calibration using certified reference materials with known zeta potential values provides a baseline for instrument performance assessment. Industry standards recommend calibration at least monthly, with more frequent checks when measuring critical samples or after maintenance procedures.
A comprehensive calibration protocol should include verification of electrode condition, which can be assessed through impedance measurements. Electrodes typically show optimal performance when impedance values fall within manufacturer-specified ranges, usually between 100-500 kΩ. Deviations from these values often indicate electrode degradation or contamination requiring immediate attention.
Conductivity calibration represents another critical aspect of quality control. Standard solutions with precisely known conductivity values (typically KCl solutions ranging from 0.01 to 0.1 M) should be used to verify the accuracy of conductivity measurements. This calibration is particularly important as sample conductivity directly affects the electric field applied during zeta potential measurements, potentially introducing systematic errors if improperly calibrated.
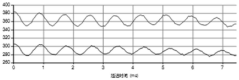
System suitability tests using reference particles (such as polystyrene latex standards) should be performed regularly to verify overall instrument performance. These tests should demonstrate measurement reproducibility within ±5% of expected values. Documentation of these tests in instrument logbooks provides traceability and helps identify long-term drift or sudden performance changes.
Statistical quality control methods enhance measurement reliability. Implementation of control charts tracking key parameters like reference material measurements allows for early detection of instrument drift. Establishing warning and action limits (typically at ±2σ and ±3σ from the mean) provides objective criteria for instrument maintenance decisions.
Cross-validation between different measurement techniques or instruments offers additional quality assurance. Periodic comparison of results with alternative methods such as electrophoretic light scattering or streaming potential measurements helps identify method-specific biases and ensures measurement accuracy across platforms.
Environmental monitoring during measurements should be incorporated into quality control protocols. Temperature fluctuations exceeding ±0.5°C can significantly impact zeta potential values, necessitating strict temperature control and monitoring. Similarly, pH stability should be verified before and after measurements to detect potential drift that could affect interpretation of results.
A comprehensive calibration protocol should include verification of electrode condition, which can be assessed through impedance measurements. Electrodes typically show optimal performance when impedance values fall within manufacturer-specified ranges, usually between 100-500 kΩ. Deviations from these values often indicate electrode degradation or contamination requiring immediate attention.
Conductivity calibration represents another critical aspect of quality control. Standard solutions with precisely known conductivity values (typically KCl solutions ranging from 0.01 to 0.1 M) should be used to verify the accuracy of conductivity measurements. This calibration is particularly important as sample conductivity directly affects the electric field applied during zeta potential measurements, potentially introducing systematic errors if improperly calibrated.
System suitability tests using reference particles (such as polystyrene latex standards) should be performed regularly to verify overall instrument performance. These tests should demonstrate measurement reproducibility within ±5% of expected values. Documentation of these tests in instrument logbooks provides traceability and helps identify long-term drift or sudden performance changes.
Statistical quality control methods enhance measurement reliability. Implementation of control charts tracking key parameters like reference material measurements allows for early detection of instrument drift. Establishing warning and action limits (typically at ±2σ and ±3σ from the mean) provides objective criteria for instrument maintenance decisions.
Cross-validation between different measurement techniques or instruments offers additional quality assurance. Periodic comparison of results with alternative methods such as electrophoretic light scattering or streaming potential measurements helps identify method-specific biases and ensures measurement accuracy across platforms.
Environmental monitoring during measurements should be incorporated into quality control protocols. Temperature fluctuations exceeding ±0.5°C can significantly impact zeta potential values, necessitating strict temperature control and monitoring. Similarly, pH stability should be verified before and after measurements to detect potential drift that could affect interpretation of results.
Sample Preparation Best Practices
Sample preparation is a critical factor in obtaining accurate and reproducible zeta potential measurements. The quality of sample preparation directly impacts the reliability of results when troubleshooting electrode, conductivity, and instrument issues. Proper sample preparation begins with understanding the physical and chemical properties of the sample material, including particle size, concentration, and stability in the dispersing medium.
For colloidal suspensions, maintaining an appropriate concentration is essential. Overly concentrated samples can lead to multiple scattering effects and particle interactions that distort measurements, while excessively dilute samples may produce insufficient signal strength. Generally, a concentration range of 0.01% to 0.1% by weight is recommended for most applications, though this may vary depending on the optical properties of the particles.
The dispersing medium must be carefully selected based on compatibility with the sample. Ideally, the medium should maintain sample stability while providing suitable conductivity for zeta potential measurements. For aqueous systems, deionized water with controlled ionic strength is commonly used. The addition of background electrolytes may be necessary to achieve appropriate conductivity levels, typically between 0.01 mS/cm and 5 mS/cm.
Filtration is another crucial step in sample preparation. All solutions should be filtered through appropriate membrane filters (typically 0.2-0.45 μm) to remove dust and large contaminants that could interfere with measurements. For the sample itself, gentle filtration or centrifugation may be required to achieve the desired particle size distribution, particularly when dealing with polydisperse systems.
Temperature stabilization prior to measurement is often overlooked but remains essential. Samples should be equilibrated to the measurement temperature for at least 15 minutes before analysis to prevent thermal gradients that could induce convection currents and affect electrophoretic mobility measurements.
pH adjustment and control represent another critical aspect of sample preparation. Since surface charge is highly dependent on pH, maintaining consistent pH conditions is vital for reproducible measurements. Buffer solutions may be employed, but their ionic contribution must be considered when interpreting results.
Sonication may be necessary to disperse agglomerates, but excessive sonication can alter surface properties or damage particles. A standardized sonication protocol should be established based on sample characteristics, typically involving short bursts (30-60 seconds) with cooling intervals to prevent overheating.
Finally, proper handling and storage of prepared samples is essential. Freshly prepared samples generally yield the most reliable results, as aging can lead to aggregation, surface modification, or contamination that affects zeta potential values.
For colloidal suspensions, maintaining an appropriate concentration is essential. Overly concentrated samples can lead to multiple scattering effects and particle interactions that distort measurements, while excessively dilute samples may produce insufficient signal strength. Generally, a concentration range of 0.01% to 0.1% by weight is recommended for most applications, though this may vary depending on the optical properties of the particles.
The dispersing medium must be carefully selected based on compatibility with the sample. Ideally, the medium should maintain sample stability while providing suitable conductivity for zeta potential measurements. For aqueous systems, deionized water with controlled ionic strength is commonly used. The addition of background electrolytes may be necessary to achieve appropriate conductivity levels, typically between 0.01 mS/cm and 5 mS/cm.
Filtration is another crucial step in sample preparation. All solutions should be filtered through appropriate membrane filters (typically 0.2-0.45 μm) to remove dust and large contaminants that could interfere with measurements. For the sample itself, gentle filtration or centrifugation may be required to achieve the desired particle size distribution, particularly when dealing with polydisperse systems.
Temperature stabilization prior to measurement is often overlooked but remains essential. Samples should be equilibrated to the measurement temperature for at least 15 minutes before analysis to prevent thermal gradients that could induce convection currents and affect electrophoretic mobility measurements.
pH adjustment and control represent another critical aspect of sample preparation. Since surface charge is highly dependent on pH, maintaining consistent pH conditions is vital for reproducible measurements. Buffer solutions may be employed, but their ionic contribution must be considered when interpreting results.
Sonication may be necessary to disperse agglomerates, but excessive sonication can alter surface properties or damage particles. A standardized sonication protocol should be established based on sample characteristics, typically involving short bursts (30-60 seconds) with cooling intervals to prevent overheating.
Finally, proper handling and storage of prepared samples is essential. Freshly prepared samples generally yield the most reliable results, as aging can lead to aggregation, surface modification, or contamination that affects zeta potential values.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!