Zeta Potential — Reporting Table Template: Fields, Units and Acceptance Criteria for QC
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zeta Potential QC Background and Objectives
Zeta potential measurement has emerged as a critical quality control parameter in various industries, particularly in pharmaceutical, colloidal systems, and nanomaterial production. The concept originated in the early 20th century with the development of electrokinetic theories, but has gained significant practical importance over the past three decades with the advancement of measurement technologies and increased understanding of colloidal stability mechanisms.
The evolution of zeta potential measurement techniques has progressed from rudimentary electrophoretic mobility observations to sophisticated laser Doppler electrophoresis systems capable of providing rapid, accurate measurements. This technological progression has enabled zeta potential to transition from a purely academic parameter to an essential quality control metric in industrial settings.
The primary objective of implementing zeta potential measurements in quality control processes is to ensure consistent product performance through monitoring colloidal stability. For pharmaceutical formulations, maintaining a specific zeta potential range is crucial for preventing aggregation, ensuring proper drug delivery, and extending shelf life. Similarly, in nanomaterial production, zeta potential serves as an indicator of surface modification success and batch-to-batch consistency.
Current quality control practices involving zeta potential measurements often lack standardized reporting formats, leading to inconsistencies in data interpretation and difficulty in establishing meaningful specification limits. The absence of industry-wide reporting templates has created challenges in regulatory compliance, particularly for pharmaceutical and medical device manufacturers who must demonstrate consistent product quality to regulatory authorities.
The development of a comprehensive zeta potential reporting table template for quality control aims to address these challenges by standardizing measurement conditions, data presentation, and interpretation guidelines. Such standardization would facilitate more meaningful comparisons between batches, enable more effective troubleshooting of production issues, and support regulatory submissions with well-organized, consistent data.
Additionally, as industries continue to develop more complex colloidal systems and nanomaterials, the importance of zeta potential as a quality attribute is expected to increase. Establishing robust reporting mechanisms now will provide a foundation for future advancements in quality control methodologies and support the integration of zeta potential measurements with other analytical techniques for comprehensive product characterization.
This research initiative seeks to evaluate existing reporting practices across industries, identify key parameters that should be included in a standardized template, and develop a flexible yet comprehensive reporting format that can be adapted to various industrial applications while maintaining scientific rigor and regulatory compliance.
The evolution of zeta potential measurement techniques has progressed from rudimentary electrophoretic mobility observations to sophisticated laser Doppler electrophoresis systems capable of providing rapid, accurate measurements. This technological progression has enabled zeta potential to transition from a purely academic parameter to an essential quality control metric in industrial settings.
The primary objective of implementing zeta potential measurements in quality control processes is to ensure consistent product performance through monitoring colloidal stability. For pharmaceutical formulations, maintaining a specific zeta potential range is crucial for preventing aggregation, ensuring proper drug delivery, and extending shelf life. Similarly, in nanomaterial production, zeta potential serves as an indicator of surface modification success and batch-to-batch consistency.
Current quality control practices involving zeta potential measurements often lack standardized reporting formats, leading to inconsistencies in data interpretation and difficulty in establishing meaningful specification limits. The absence of industry-wide reporting templates has created challenges in regulatory compliance, particularly for pharmaceutical and medical device manufacturers who must demonstrate consistent product quality to regulatory authorities.
The development of a comprehensive zeta potential reporting table template for quality control aims to address these challenges by standardizing measurement conditions, data presentation, and interpretation guidelines. Such standardization would facilitate more meaningful comparisons between batches, enable more effective troubleshooting of production issues, and support regulatory submissions with well-organized, consistent data.
Additionally, as industries continue to develop more complex colloidal systems and nanomaterials, the importance of zeta potential as a quality attribute is expected to increase. Establishing robust reporting mechanisms now will provide a foundation for future advancements in quality control methodologies and support the integration of zeta potential measurements with other analytical techniques for comprehensive product characterization.
This research initiative seeks to evaluate existing reporting practices across industries, identify key parameters that should be included in a standardized template, and develop a flexible yet comprehensive reporting format that can be adapted to various industrial applications while maintaining scientific rigor and regulatory compliance.
Market Demand Analysis for Zeta Potential QC Solutions
The global market for Zeta Potential measurement solutions in quality control applications has experienced significant growth over the past five years, driven primarily by increasing demands for precise particle characterization in pharmaceutical, biotechnology, and advanced materials industries. Current market estimates value this segment at approximately 320 million USD with a compound annual growth rate of 7.8% projected through 2028.
Pharmaceutical and biopharmaceutical sectors represent the largest market share, accounting for nearly 42% of the total demand. This dominance stems from stringent regulatory requirements for colloidal stability assessment in drug formulations, particularly for injectable medications, liposomal drug delivery systems, and protein-based therapeutics. The FDA and EMA's emphasis on comprehensive physicochemical characterization has made Zeta Potential measurement a critical quality control parameter.
Academic and research institutions constitute the second-largest market segment at 27%, where Zeta Potential measurements are essential for fundamental research in colloid science, nanomaterials, and interface phenomena. The remaining market share is distributed among environmental monitoring (14%), food and beverage industries (11%), and other industrial applications (6%).
Geographically, North America leads the market with approximately 38% share, followed by Europe (31%), Asia-Pacific (24%), and rest of the world (7%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth trajectory due to expanding pharmaceutical manufacturing capabilities and increasing adoption of advanced analytical technologies.
Key market drivers include the rising production of nanomaterials and nanoparticle-based products, which require precise stability characterization. Additionally, the growing complexity of formulations in pharmaceuticals and cosmetics necessitates more sophisticated quality control protocols incorporating Zeta Potential measurements.
Customer pain points identified through market research include the need for standardized reporting formats that facilitate regulatory compliance, challenges in data interpretation across different sample types, and difficulties in establishing meaningful specification limits for diverse product categories. Survey data indicates that 78% of quality control professionals desire improved reporting templates that can streamline documentation and decision-making processes.
The market shows increasing demand for integrated solutions that combine Zeta Potential measurements with complementary techniques such as particle size analysis and rheological characterization. Furthermore, there is growing interest in automated systems that can reduce operator variability and enhance measurement reproducibility across manufacturing sites.
Pharmaceutical and biopharmaceutical sectors represent the largest market share, accounting for nearly 42% of the total demand. This dominance stems from stringent regulatory requirements for colloidal stability assessment in drug formulations, particularly for injectable medications, liposomal drug delivery systems, and protein-based therapeutics. The FDA and EMA's emphasis on comprehensive physicochemical characterization has made Zeta Potential measurement a critical quality control parameter.
Academic and research institutions constitute the second-largest market segment at 27%, where Zeta Potential measurements are essential for fundamental research in colloid science, nanomaterials, and interface phenomena. The remaining market share is distributed among environmental monitoring (14%), food and beverage industries (11%), and other industrial applications (6%).
Geographically, North America leads the market with approximately 38% share, followed by Europe (31%), Asia-Pacific (24%), and rest of the world (7%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth trajectory due to expanding pharmaceutical manufacturing capabilities and increasing adoption of advanced analytical technologies.
Key market drivers include the rising production of nanomaterials and nanoparticle-based products, which require precise stability characterization. Additionally, the growing complexity of formulations in pharmaceuticals and cosmetics necessitates more sophisticated quality control protocols incorporating Zeta Potential measurements.
Customer pain points identified through market research include the need for standardized reporting formats that facilitate regulatory compliance, challenges in data interpretation across different sample types, and difficulties in establishing meaningful specification limits for diverse product categories. Survey data indicates that 78% of quality control professionals desire improved reporting templates that can streamline documentation and decision-making processes.
The market shows increasing demand for integrated solutions that combine Zeta Potential measurements with complementary techniques such as particle size analysis and rheological characterization. Furthermore, there is growing interest in automated systems that can reduce operator variability and enhance measurement reproducibility across manufacturing sites.
Current Challenges in Zeta Potential Measurement
Despite significant advancements in zeta potential measurement technologies, several persistent challenges continue to impede accurate and reliable measurements in quality control environments. The variability in sample preparation protocols represents a major obstacle, as inconsistent sample handling, concentration variations, and contamination can significantly alter zeta potential readings. This lack of standardization across industries makes it difficult to establish universal quality control benchmarks.
Instrument calibration and validation present another significant challenge. Many laboratories struggle with maintaining properly calibrated equipment, and validation procedures often vary between instrument manufacturers. The absence of universally accepted reference materials specifically designed for zeta potential calibration further complicates this issue, leading to potential measurement discrepancies between different laboratories or even within the same facility using different instruments.
Environmental factors exert considerable influence on zeta potential measurements. Parameters such as temperature, pH, ionic strength, and the presence of dissolved gases can dramatically affect results. Controlling these variables in industrial settings proves particularly challenging, especially in facilities where environmental conditions fluctuate throughout production cycles. This environmental sensitivity undermines measurement reproducibility and complicates trend analysis in quality control applications.
Data interpretation remains problematic due to the complex relationship between zeta potential and colloidal stability. Quality control personnel often lack specialized training in colloid science, leading to misinterpretation of measurement data. The absence of standardized reporting formats and acceptance criteria across industries further exacerbates this challenge, making it difficult to establish meaningful quality control thresholds.
Sample-specific challenges also persist, particularly with complex formulations containing multiple components or highly concentrated systems. Many commercial instruments struggle with accurate measurements of samples with high polydispersity or those containing large particles. Additionally, certain sample types, such as those with extreme conductivity values or non-aqueous systems, present unique measurement difficulties that current technologies have not fully addressed.
Reporting inconsistencies represent a significant obstacle to effective quality control implementation. The lack of standardized reporting templates results in variable data presentation formats across laboratories, complicating cross-facility comparisons and trend analysis. Critical measurement parameters such as applied voltage, count rate, and conductivity are frequently omitted from reports, limiting the ability to troubleshoot anomalous results or verify measurement validity.
Instrument calibration and validation present another significant challenge. Many laboratories struggle with maintaining properly calibrated equipment, and validation procedures often vary between instrument manufacturers. The absence of universally accepted reference materials specifically designed for zeta potential calibration further complicates this issue, leading to potential measurement discrepancies between different laboratories or even within the same facility using different instruments.
Environmental factors exert considerable influence on zeta potential measurements. Parameters such as temperature, pH, ionic strength, and the presence of dissolved gases can dramatically affect results. Controlling these variables in industrial settings proves particularly challenging, especially in facilities where environmental conditions fluctuate throughout production cycles. This environmental sensitivity undermines measurement reproducibility and complicates trend analysis in quality control applications.
Data interpretation remains problematic due to the complex relationship between zeta potential and colloidal stability. Quality control personnel often lack specialized training in colloid science, leading to misinterpretation of measurement data. The absence of standardized reporting formats and acceptance criteria across industries further exacerbates this challenge, making it difficult to establish meaningful quality control thresholds.
Sample-specific challenges also persist, particularly with complex formulations containing multiple components or highly concentrated systems. Many commercial instruments struggle with accurate measurements of samples with high polydispersity or those containing large particles. Additionally, certain sample types, such as those with extreme conductivity values or non-aqueous systems, present unique measurement difficulties that current technologies have not fully addressed.
Reporting inconsistencies represent a significant obstacle to effective quality control implementation. The lack of standardized reporting templates results in variable data presentation formats across laboratories, complicating cross-facility comparisons and trend analysis. Critical measurement parameters such as applied voltage, count rate, and conductivity are frequently omitted from reports, limiting the ability to troubleshoot anomalous results or verify measurement validity.
Current Reporting Methodologies for Zeta Potential QC
01 Zeta potential measurement methods and apparatus
Various methods and apparatus for measuring zeta potential in colloidal systems are described. These include electrophoretic light scattering techniques, electroacoustic methods, and specialized equipment designed to measure the electrical potential at the slipping plane of particles in suspension. The measurements are typically reported in standardized table formats showing particle mobility, zeta potential values, and related parameters under different conditions.- Zeta potential measurement methods and devices: Various methods and devices are used for measuring zeta potential of particles in suspension. These include electrophoretic light scattering techniques, electroacoustic methods, and specialized instruments that track particle movement under an electric field. These measurements are critical for understanding colloidal stability and surface charge characteristics of particles in different media.
- Data reporting formats for zeta potential analysis: Standardized formats for reporting zeta potential data include tabular templates that capture key parameters such as sample identification, measurement conditions (pH, ionic strength, temperature), measured zeta potential values, standard deviations, and statistical significance. These reporting formats ensure consistency and reproducibility in zeta potential analysis across different laboratories and research settings.
- Zeta potential applications in water treatment and filtration: Zeta potential measurements are crucial in water treatment and filtration processes. The data helps optimize coagulation, flocculation, and membrane filtration by monitoring the surface charge of particles and contaminants. Reporting templates for these applications typically include parameters specific to water quality assessment, such as turbidity correlation, filter performance metrics, and treatment efficiency indicators.
- Zeta potential monitoring in pharmaceutical and biomedical applications: In pharmaceutical and biomedical fields, zeta potential reporting templates are designed to track the stability of drug formulations, protein solutions, and nanoparticle-based therapeutics. These templates include sections for recording changes in zeta potential over time, under different storage conditions, and in various biological media, which are critical for predicting drug stability and biocompatibility.
- Quality control and standardization of zeta potential measurements: Quality control procedures for zeta potential measurements involve standardized reporting templates that include calibration data, reference material measurements, and system suitability tests. These templates help ensure the reliability and comparability of zeta potential data across different instruments and laboratories, often incorporating statistical analysis tools to evaluate measurement uncertainty and reproducibility.
02 Data presentation formats for zeta potential analysis
Standardized templates for reporting zeta potential data include tabular formats with columns for sample identification, measurement conditions (pH, ionic strength, temperature), measured zeta potential values, standard deviations, and statistical significance indicators. These templates often include graphical representations showing zeta potential as a function of various parameters to facilitate interpretation of colloidal stability.Expand Specific Solutions03 Zeta potential analysis in water treatment applications
Zeta potential measurements are crucial in water and wastewater treatment processes, where reporting templates include parameters specific to filtration efficiency, coagulation optimization, and membrane fouling prevention. These templates typically include columns for raw water quality, coagulant dosage, resulting zeta potential, and treatment efficiency metrics to establish correlations between surface charge and treatment effectiveness.Expand Specific Solutions04 Pharmaceutical and biomedical zeta potential reporting
In pharmaceutical and biomedical applications, zeta potential reporting templates are designed to track the stability of drug formulations, protein solutions, and nanoparticle-based delivery systems. These templates include fields for drug loading efficiency, particle size distribution alongside zeta potential values, and stability indicators over time under various storage conditions to predict shelf-life and biocompatibility.Expand Specific Solutions05 Quality control and standardization of zeta potential measurements
Standardized reporting templates for quality control purposes include reference materials measurements, calibration data, and instrument performance metrics. These templates are designed to ensure reproducibility and comparability of zeta potential measurements across different laboratories and instruments, with fields for system suitability tests, verification of measurement accuracy, and traceability to international standards.Expand Specific Solutions
Key Industry Players in Colloidal Stability Analysis
The zeta potential measurement market for quality control is in a growth phase, driven by increasing demand for precise particle characterization in pharmaceutical, biotechnology, and environmental applications. The market size is expanding steadily, with projections indicating significant growth due to rising quality control requirements across industries. Technologically, the field shows moderate maturity with established players like Bio-Rad Laboratories and Life Technologies offering sophisticated solutions, while newer entrants such as Shanghai Kunlai Biological Technology are introducing innovative approaches. Companies like Shimadzu and Applied Biosystems demonstrate the market's evolution through integration of zeta potential measurements with comprehensive analytical platforms, reflecting the trend toward multifunctional quality control systems that enhance reproducibility and reporting capabilities.
Bio-Rad Laboratories, Inc.
Technical Solution: Bio-Rad's zeta potential quality control reporting system integrates with their DelsaMax PRO and ZetaSizer instruments to provide comprehensive colloidal stability analysis. Their template-based reporting system automatically captures critical zeta potential parameters including mean, standard deviation, and mobility distributions across multiple measurements. The system incorporates statistical process control (SPC) charts that track zeta potential measurements over time, allowing for immediate identification of trending issues before they affect product quality. Their reporting templates include customizable acceptance criteria with color-coded indicators for pass/fail status, and automated calculation of control limits based on historical data. The system also features multi-user accessibility with role-based permissions to maintain data integrity throughout the quality control process.
Strengths: Seamless integration with their analytical instruments provides end-to-end workflow solutions; robust statistical analysis capabilities enable trend identification; customizable reporting templates adapt to specific industry requirements. Weaknesses: Proprietary system may limit compatibility with third-party instruments; higher implementation costs compared to generic reporting solutions; requires specialized training for optimal utilization.
Life Technologies Corp.
Technical Solution: Life Technologies has developed a specialized zeta potential quality control reporting system tailored for biological and pharmaceutical applications. Their approach integrates zeta potential measurements with their broader bioprocess monitoring platform to provide context-rich quality assessment. The reporting templates incorporate multi-parameter analysis that correlates zeta potential with critical quality attributes such as protein stability, formulation consistency, and batch-to-batch variability. Their system features automated data collection from multiple measurement points throughout production processes, with real-time statistical analysis to identify deviations from established control parameters. The reporting templates include visualization tools that present zeta potential trends alongside process parameters, enabling root cause analysis of quality deviations. Additionally, their system incorporates machine learning algorithms that analyze historical zeta potential data to predict potential quality issues before they manifest.
Strengths: Specialized focus on biological applications provides industry-specific insights; integration with broader bioprocess monitoring creates comprehensive quality picture; predictive analytics capabilities enable proactive quality management. Weaknesses: Narrower application scope limits utility outside biological/pharmaceutical industries; complex implementation may require significant technical support; higher ongoing maintenance costs for advanced analytical features.
Critical Parameters in Zeta Potential Data Interpretation
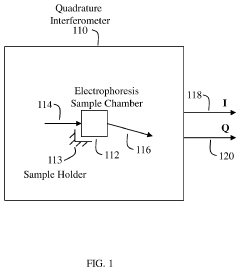
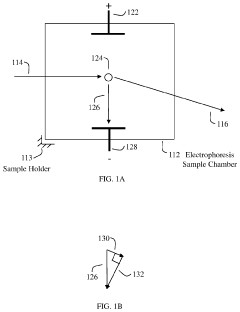
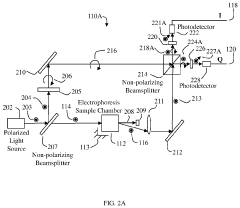
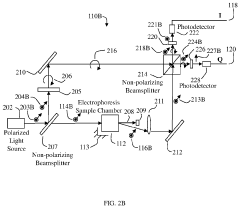
Stationary devices for determination of magnitude and polarity of electrophoretic mobility and zeta potential
PatentActiveUS11181503B1
Innovation
- The use of an optical quadrature interferometer that generates quadrature signals from scattered and reference light, allowing for the determination of particle speeds and directions without moving optical components, thereby eliminating the need for phase modulators or frequency shifters and simplifying the instrument design.
Standardization and Validation Protocols
Standardization of zeta potential measurement protocols is essential for ensuring consistent and reliable quality control across different laboratories and manufacturing facilities. The development of robust validation protocols begins with the establishment of standard operating procedures (SOPs) that detail sample preparation, instrument calibration, measurement parameters, and data analysis methodologies. These SOPs must specify environmental conditions such as temperature, pH, and ionic strength, which significantly influence zeta potential measurements.
Validation protocols should incorporate a multi-tiered approach, starting with system suitability tests using certified reference materials with known zeta potential values. These reference materials, typically polystyrene latex beads or silica particles, provide a benchmark for instrument performance verification. The protocol should define acceptance criteria for these reference measurements, typically within ±10% of the certified value, to confirm system readiness before sample analysis.
Method validation parameters must include precision (repeatability and intermediate precision), accuracy, specificity, linearity, range, and robustness. Repeatability should be assessed through multiple measurements of the same sample preparation, while intermediate precision evaluates variability across different days, operators, and instruments. For quality control applications, establishing control charts with defined action and warning limits is crucial for monitoring measurement stability over time.
Statistical analysis frameworks should be integrated into the validation protocol, specifying appropriate statistical tests for evaluating measurement uncertainty and determining significant differences between samples. This includes calculation of standard deviation, coefficient of variation, and confidence intervals. The protocol should also define minimum sample sizes required for achieving statistically significant results based on expected variability.
Interlaboratory comparison studies represent a critical component of validation, allowing for assessment of method transferability and identification of systematic biases between different measurement systems. These studies should follow established guidelines such as ISO 5725 or ASTM E691, with results analyzed using statistical methods like z-scores or Youden plots to identify outliers and systematic errors.
Documentation requirements constitute the final essential element of standardization protocols. These should include detailed reporting templates capturing all relevant measurement parameters, calibration data, sample characteristics, and environmental conditions. The reporting format should facilitate traceability through unique sample identifiers, time stamps, operator information, and instrument identification, ensuring complete data provenance for regulatory compliance and quality assurance purposes.
Validation protocols should incorporate a multi-tiered approach, starting with system suitability tests using certified reference materials with known zeta potential values. These reference materials, typically polystyrene latex beads or silica particles, provide a benchmark for instrument performance verification. The protocol should define acceptance criteria for these reference measurements, typically within ±10% of the certified value, to confirm system readiness before sample analysis.
Method validation parameters must include precision (repeatability and intermediate precision), accuracy, specificity, linearity, range, and robustness. Repeatability should be assessed through multiple measurements of the same sample preparation, while intermediate precision evaluates variability across different days, operators, and instruments. For quality control applications, establishing control charts with defined action and warning limits is crucial for monitoring measurement stability over time.
Statistical analysis frameworks should be integrated into the validation protocol, specifying appropriate statistical tests for evaluating measurement uncertainty and determining significant differences between samples. This includes calculation of standard deviation, coefficient of variation, and confidence intervals. The protocol should also define minimum sample sizes required for achieving statistically significant results based on expected variability.
Interlaboratory comparison studies represent a critical component of validation, allowing for assessment of method transferability and identification of systematic biases between different measurement systems. These studies should follow established guidelines such as ISO 5725 or ASTM E691, with results analyzed using statistical methods like z-scores or Youden plots to identify outliers and systematic errors.
Documentation requirements constitute the final essential element of standardization protocols. These should include detailed reporting templates capturing all relevant measurement parameters, calibration data, sample characteristics, and environmental conditions. The reporting format should facilitate traceability through unique sample identifiers, time stamps, operator information, and instrument identification, ensuring complete data provenance for regulatory compliance and quality assurance purposes.
Regulatory Compliance for Zeta Potential QC Documentation
Regulatory compliance for Zeta Potential quality control documentation requires adherence to multiple international standards and guidelines. The FDA's 21 CFR Part 11 mandates electronic record integrity and validation for pharmaceutical and biotech industries, necessitating secure data management systems for Zeta Potential measurements with audit trails and electronic signatures. Similarly, ISO 13099 specifically addresses methods for Zeta Potential determination, providing standardized protocols that must be reflected in QC documentation templates.
The European Medicines Agency (EMA) guidelines on quality control for nanomedicines and colloidal systems explicitly require Zeta Potential characterization with comprehensive documentation. These guidelines emphasize the need for detailed method validation, instrument qualification, and consistent reporting formats to ensure data reliability and comparability across batches.
USP <1430> and <429> chapters provide specific guidance on particle characterization methods including Zeta Potential measurements, outlining acceptance criteria and documentation requirements that must be incorporated into reporting templates. For medical devices, ISO 10993-18 requires physicochemical characterization including surface charge properties, with documentation that demonstrates biocompatibility assessment.
ICH Q8, Q9, and Q10 guidelines collectively emphasize a quality-by-design approach, requiring risk assessment documentation that includes critical quality attributes like Zeta Potential for colloidal systems. This necessitates templates that capture not only measurement results but also their interpretation within the product's quality control strategy.
GMP regulations across jurisdictions require traceability in all quality control documentation. For Zeta Potential measurements, this translates to templates that document calibration status, reference standards used, operator qualifications, and environmental conditions during testing. The templates must also include deviation reporting mechanisms and CAPA documentation sections.
ASTM E2490 provides standard guidance for measurement of particle size distribution and Zeta Potential characterization, requiring specific documentation formats that demonstrate method suitability. Compliance with these standards necessitates reporting templates that include statistical analysis of measurement uncertainty and variability.
For global regulatory submissions, harmonized documentation approaches are increasingly important. Zeta Potential QC templates should therefore be designed with flexibility to accommodate regional regulatory requirements while maintaining core scientific integrity and data comparability across markets.
The European Medicines Agency (EMA) guidelines on quality control for nanomedicines and colloidal systems explicitly require Zeta Potential characterization with comprehensive documentation. These guidelines emphasize the need for detailed method validation, instrument qualification, and consistent reporting formats to ensure data reliability and comparability across batches.
USP <1430> and <429> chapters provide specific guidance on particle characterization methods including Zeta Potential measurements, outlining acceptance criteria and documentation requirements that must be incorporated into reporting templates. For medical devices, ISO 10993-18 requires physicochemical characterization including surface charge properties, with documentation that demonstrates biocompatibility assessment.
ICH Q8, Q9, and Q10 guidelines collectively emphasize a quality-by-design approach, requiring risk assessment documentation that includes critical quality attributes like Zeta Potential for colloidal systems. This necessitates templates that capture not only measurement results but also their interpretation within the product's quality control strategy.
GMP regulations across jurisdictions require traceability in all quality control documentation. For Zeta Potential measurements, this translates to templates that document calibration status, reference standards used, operator qualifications, and environmental conditions during testing. The templates must also include deviation reporting mechanisms and CAPA documentation sections.
ASTM E2490 provides standard guidance for measurement of particle size distribution and Zeta Potential characterization, requiring specific documentation formats that demonstrate method suitability. Compliance with these standards necessitates reporting templates that include statistical analysis of measurement uncertainty and variability.
For global regulatory submissions, harmonized documentation approaches are increasingly important. Zeta Potential QC templates should therefore be designed with flexibility to accommodate regional regulatory requirements while maintaining core scientific integrity and data comparability across markets.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!