ICP-MS vs CE-MS: Essential Comparison in Multi-Element Detection
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
ICP-MS and CE-MS Technology Evolution and Objectives
The evolution of analytical techniques for multi-element detection has been marked by significant technological advancements over the past several decades. Inductively Coupled Plasma Mass Spectrometry (ICP-MS) emerged in the early 1980s as a revolutionary technique for elemental analysis, offering unprecedented sensitivity and multi-element capabilities. The technology evolved from earlier atomic emission spectroscopy methods, with commercial instruments becoming widely available by the mid-1980s.
ICP-MS development has progressed through several generations, each addressing limitations of previous iterations. Early systems faced challenges with matrix effects and polyatomic interferences, leading to innovations such as collision/reaction cells in the late 1990s and triple quadrupole systems in the 2010s that significantly improved detection limits and analytical precision.
Concurrently, Capillary Electrophoresis Mass Spectrometry (CE-MS) developed along a different trajectory. While electrophoresis techniques date back to the 1930s, the coupling of capillary electrophoresis with mass spectrometry gained momentum in the 1990s. This integration addressed the need for both separation efficiency and structural identification capabilities, particularly for complex biological samples.
CE-MS has undergone substantial refinement in interface design, with the introduction of electrospray ionization (ESI) interfaces in the mid-1990s representing a critical breakthrough. Subsequent developments focused on improving sensitivity, reproducibility, and compatibility with various sample types.
The technological objectives for both techniques have evolved in response to emerging analytical challenges. For ICP-MS, current development goals include enhancing sensitivity for ultra-trace analysis, reducing sample volume requirements, improving isotope ratio precision, and developing more effective interference removal strategies. Single-particle and single-cell capabilities represent frontier applications pushing the technology's limits.
For CE-MS, objectives center on improving concentration sensitivity, enhancing separation efficiency for complex mixtures, developing more robust interfaces, and expanding applicability across diverse sample matrices. Integration with microfluidic platforms and miniaturization represent significant development directions.
Both technologies are converging toward greater automation, improved data processing capabilities, and integration with complementary techniques to provide comprehensive analytical solutions. The ultimate goal is to develop analytical platforms that combine the elemental specificity of ICP-MS with the speciation capabilities of CE-MS, offering comprehensive characterization of complex samples across environmental, clinical, and industrial applications.
ICP-MS development has progressed through several generations, each addressing limitations of previous iterations. Early systems faced challenges with matrix effects and polyatomic interferences, leading to innovations such as collision/reaction cells in the late 1990s and triple quadrupole systems in the 2010s that significantly improved detection limits and analytical precision.
Concurrently, Capillary Electrophoresis Mass Spectrometry (CE-MS) developed along a different trajectory. While electrophoresis techniques date back to the 1930s, the coupling of capillary electrophoresis with mass spectrometry gained momentum in the 1990s. This integration addressed the need for both separation efficiency and structural identification capabilities, particularly for complex biological samples.
CE-MS has undergone substantial refinement in interface design, with the introduction of electrospray ionization (ESI) interfaces in the mid-1990s representing a critical breakthrough. Subsequent developments focused on improving sensitivity, reproducibility, and compatibility with various sample types.
The technological objectives for both techniques have evolved in response to emerging analytical challenges. For ICP-MS, current development goals include enhancing sensitivity for ultra-trace analysis, reducing sample volume requirements, improving isotope ratio precision, and developing more effective interference removal strategies. Single-particle and single-cell capabilities represent frontier applications pushing the technology's limits.
For CE-MS, objectives center on improving concentration sensitivity, enhancing separation efficiency for complex mixtures, developing more robust interfaces, and expanding applicability across diverse sample matrices. Integration with microfluidic platforms and miniaturization represent significant development directions.
Both technologies are converging toward greater automation, improved data processing capabilities, and integration with complementary techniques to provide comprehensive analytical solutions. The ultimate goal is to develop analytical platforms that combine the elemental specificity of ICP-MS with the speciation capabilities of CE-MS, offering comprehensive characterization of complex samples across environmental, clinical, and industrial applications.
Market Demand Analysis for Multi-Element Detection
The multi-element detection market has experienced substantial growth over the past decade, driven primarily by increasing demands in environmental monitoring, pharmaceutical research, food safety testing, and clinical diagnostics. The global analytical instrumentation market, which encompasses multi-element detection technologies, was valued at approximately $58 billion in 2022 and is projected to reach $81 billion by 2027, with a compound annual growth rate of 6.8%.
Within this broader market, the demand for advanced elemental analysis techniques like ICP-MS (Inductively Coupled Plasma Mass Spectrometry) and CE-MS (Capillary Electrophoresis Mass Spectrometry) continues to expand as regulatory requirements become more stringent across industries. Environmental agencies worldwide have implemented stricter monitoring protocols for heavy metals and trace elements in water, soil, and air samples, creating a steady demand for high-sensitivity detection methods.
The pharmaceutical and biotechnology sectors represent the fastest-growing segment for multi-element detection technologies, with a market growth rate exceeding 8% annually. This acceleration is attributed to the increasing complexity of biopharmaceutical products and the need for comprehensive characterization of metal impurities in drug substances. The implementation of ICH Q3D guidelines for elemental impurities has significantly boosted the adoption of advanced analytical techniques in pharmaceutical quality control.
Food safety testing represents another substantial market driver, particularly in developed economies where consumer awareness regarding contaminants has heightened. The global food safety testing market reached $19.5 billion in 2022, with elemental analysis comprising approximately 15% of this market. Regulatory bodies including the FDA, EFSA, and various national food safety authorities have established maximum permissible limits for toxic elements in food products, necessitating sensitive and reliable detection methods.
Clinical diagnostics applications for multi-element detection are expanding beyond traditional heavy metal toxicity testing to include nutritional status assessment and biomarker discovery. The clinical elemental analysis market segment is growing at 7.2% annually, with particular emphasis on technologies that can handle complex biological matrices with minimal sample preparation.
Geographically, North America and Europe currently dominate the market for advanced multi-element detection technologies, accounting for approximately 65% of global revenue. However, the Asia-Pacific region, particularly China, India, and South Korea, is experiencing the highest growth rate at 9.5% annually, driven by expanding industrial bases, increasing environmental concerns, and strengthening regulatory frameworks.
The market trend clearly indicates a shift toward technologies offering higher sensitivity, improved selectivity, lower detection limits, and the ability to handle complex sample matrices with minimal preparation. This trend positions both ICP-MS and CE-MS as critical technologies in the analytical instrumentation landscape, with their respective strengths addressing different segments of the growing multi-element detection market.
Within this broader market, the demand for advanced elemental analysis techniques like ICP-MS (Inductively Coupled Plasma Mass Spectrometry) and CE-MS (Capillary Electrophoresis Mass Spectrometry) continues to expand as regulatory requirements become more stringent across industries. Environmental agencies worldwide have implemented stricter monitoring protocols for heavy metals and trace elements in water, soil, and air samples, creating a steady demand for high-sensitivity detection methods.
The pharmaceutical and biotechnology sectors represent the fastest-growing segment for multi-element detection technologies, with a market growth rate exceeding 8% annually. This acceleration is attributed to the increasing complexity of biopharmaceutical products and the need for comprehensive characterization of metal impurities in drug substances. The implementation of ICH Q3D guidelines for elemental impurities has significantly boosted the adoption of advanced analytical techniques in pharmaceutical quality control.
Food safety testing represents another substantial market driver, particularly in developed economies where consumer awareness regarding contaminants has heightened. The global food safety testing market reached $19.5 billion in 2022, with elemental analysis comprising approximately 15% of this market. Regulatory bodies including the FDA, EFSA, and various national food safety authorities have established maximum permissible limits for toxic elements in food products, necessitating sensitive and reliable detection methods.
Clinical diagnostics applications for multi-element detection are expanding beyond traditional heavy metal toxicity testing to include nutritional status assessment and biomarker discovery. The clinical elemental analysis market segment is growing at 7.2% annually, with particular emphasis on technologies that can handle complex biological matrices with minimal sample preparation.
Geographically, North America and Europe currently dominate the market for advanced multi-element detection technologies, accounting for approximately 65% of global revenue. However, the Asia-Pacific region, particularly China, India, and South Korea, is experiencing the highest growth rate at 9.5% annually, driven by expanding industrial bases, increasing environmental concerns, and strengthening regulatory frameworks.
The market trend clearly indicates a shift toward technologies offering higher sensitivity, improved selectivity, lower detection limits, and the ability to handle complex sample matrices with minimal preparation. This trend positions both ICP-MS and CE-MS as critical technologies in the analytical instrumentation landscape, with their respective strengths addressing different segments of the growing multi-element detection market.
Technical Status and Challenges in Mass Spectrometry
Mass spectrometry technology has evolved significantly over the past decades, with various methodologies developed to address specific analytical challenges. Currently, two prominent techniques in multi-element detection are Inductively Coupled Plasma Mass Spectrometry (ICP-MS) and Capillary Electrophoresis coupled with Mass Spectrometry (CE-MS), each with distinct capabilities and limitations.
ICP-MS represents the gold standard for trace element analysis, offering detection limits in the parts-per-trillion range for most elements. The technology has matured considerably since its commercial introduction in the 1980s, with modern instruments featuring collision/reaction cells to minimize polyatomic interferences. However, challenges persist in handling complex matrices and distinguishing between certain isobaric interferences.
CE-MS, while less established for multi-element analysis, has gained traction for its exceptional separation capabilities prior to mass detection. This technique excels in analyzing charged species and provides superior resolution for complex mixtures. The primary technical hurdle for CE-MS remains its lower sensitivity compared to ICP-MS, particularly for certain metal ions, and challenges in maintaining stable electrospray interfaces.
Geographically, development of advanced mass spectrometry technologies remains concentrated in North America, Europe, and Japan, with emerging contributions from China. The United States leads in innovation, particularly through academic-industrial partnerships, while European research centers focus on specialized applications and methodological refinements.
A significant technical challenge facing both technologies is the need for improved sample introduction systems. For ICP-MS, enhancing ionization efficiency for elements with high ionization potentials remains problematic. CE-MS struggles with sample loading capacity limitations, which directly impacts detection sensitivity for trace elements.
Data processing represents another frontier, with the exponential growth in data volume from high-resolution instruments outpacing analytical software capabilities. Machine learning approaches are being explored to address this bottleneck, though standardization of these methods remains incomplete.
Miniaturization efforts face different obstacles for each technology. ICP-MS miniaturization is constrained by the fundamental requirements of plasma generation and maintenance, while CE-MS shows more promise for portable applications but sacrifices sensitivity in smaller formats.
The integration of these technologies with complementary techniques represents both a challenge and opportunity. Hyphenated systems combining chromatographic separation with mass detection show promise but require sophisticated interface engineering and synchronization protocols to maintain analytical performance.
ICP-MS represents the gold standard for trace element analysis, offering detection limits in the parts-per-trillion range for most elements. The technology has matured considerably since its commercial introduction in the 1980s, with modern instruments featuring collision/reaction cells to minimize polyatomic interferences. However, challenges persist in handling complex matrices and distinguishing between certain isobaric interferences.
CE-MS, while less established for multi-element analysis, has gained traction for its exceptional separation capabilities prior to mass detection. This technique excels in analyzing charged species and provides superior resolution for complex mixtures. The primary technical hurdle for CE-MS remains its lower sensitivity compared to ICP-MS, particularly for certain metal ions, and challenges in maintaining stable electrospray interfaces.
Geographically, development of advanced mass spectrometry technologies remains concentrated in North America, Europe, and Japan, with emerging contributions from China. The United States leads in innovation, particularly through academic-industrial partnerships, while European research centers focus on specialized applications and methodological refinements.
A significant technical challenge facing both technologies is the need for improved sample introduction systems. For ICP-MS, enhancing ionization efficiency for elements with high ionization potentials remains problematic. CE-MS struggles with sample loading capacity limitations, which directly impacts detection sensitivity for trace elements.
Data processing represents another frontier, with the exponential growth in data volume from high-resolution instruments outpacing analytical software capabilities. Machine learning approaches are being explored to address this bottleneck, though standardization of these methods remains incomplete.
Miniaturization efforts face different obstacles for each technology. ICP-MS miniaturization is constrained by the fundamental requirements of plasma generation and maintenance, while CE-MS shows more promise for portable applications but sacrifices sensitivity in smaller formats.
The integration of these technologies with complementary techniques represents both a challenge and opportunity. Hyphenated systems combining chromatographic separation with mass detection show promise but require sophisticated interface engineering and synchronization protocols to maintain analytical performance.
Current Technical Solutions for Multi-Element Detection
01 ICP-MS technology for multi-element detection
Inductively Coupled Plasma Mass Spectrometry (ICP-MS) is a powerful analytical technique used for multi-element detection with high sensitivity and accuracy. It allows for the simultaneous detection of multiple elements in various sample types. The technology involves ionizing the sample with inductively coupled plasma and then using mass spectrometry to separate and quantify the ions according to their mass-to-charge ratio. This technique is particularly valuable for trace element analysis in complex matrices.- ICP-MS technology for multi-element detection: Inductively Coupled Plasma Mass Spectrometry (ICP-MS) is a powerful analytical technique used for multi-element detection with high sensitivity and accuracy. This technology enables simultaneous detection of multiple elements in various sample matrices, offering low detection limits and wide dynamic range. ICP-MS systems typically include sample introduction systems, plasma generation components, mass analyzers, and detection systems optimized for trace element analysis in environmental, biological, and industrial samples.
- CE-MS integration for enhanced separation and detection: Capillary Electrophoresis coupled with Mass Spectrometry (CE-MS) provides complementary capabilities for multi-element analysis by combining the high separation efficiency of CE with the sensitive detection capabilities of MS. This integration allows for the separation of complex mixtures based on charge-to-mass ratio before mass spectrometric detection, enabling improved selectivity and specificity. CE-MS systems are particularly valuable for analyzing charged species, biomolecules, and metal complexes in complex matrices.
- Sample preparation techniques for multi-element analysis: Effective sample preparation methods are crucial for accurate multi-element detection using ICP-MS and CE-MS. These techniques include digestion procedures, extraction methods, preconcentration steps, and matrix removal strategies to minimize interferences and enhance detection sensitivity. Specialized sample introduction systems and microfluidic devices have been developed to improve sample throughput, reduce sample consumption, and maintain analytical performance for challenging sample types.
- Interference reduction and calibration strategies: Advanced methods for reducing spectral and non-spectral interferences in multi-element detection systems improve measurement accuracy and precision. These include collision/reaction cell technologies, mathematical correction algorithms, and optimized instrument parameters. Calibration strategies such as internal standardization, isotope dilution, and standard addition methods are employed to compensate for matrix effects and instrument drift, ensuring reliable quantitative results across diverse sample types and concentration ranges.
- Applications in environmental and biological analysis: ICP-MS and CE-MS multi-element detection technologies are widely applied in environmental monitoring, clinical diagnostics, food safety testing, and pharmaceutical analysis. These techniques enable the determination of trace elements, speciation analysis, and isotope ratio measurements in complex matrices such as water, soil, biological fluids, and tissues. Recent advancements include portable and miniaturized systems for field analysis, automated high-throughput platforms, and integration with separation techniques for comprehensive elemental profiling.
02 CE-MS technology for multi-element analysis
Capillary Electrophoresis Mass Spectrometry (CE-MS) combines the separation capabilities of capillary electrophoresis with the detection power of mass spectrometry for multi-element analysis. This technique separates ions based on their electrophoretic mobility and then identifies them using mass spectrometry. CE-MS offers advantages such as high resolution, minimal sample consumption, and the ability to analyze complex mixtures. It is particularly useful for analyzing charged species and has applications in various fields including environmental monitoring and bioanalysis.Expand Specific Solutions03 Sample preparation methods for ICP-MS and CE-MS
Effective sample preparation is crucial for accurate multi-element detection using ICP-MS and CE-MS. Various methods have been developed to prepare samples for analysis, including digestion techniques, extraction procedures, and preconcentration methods. These preparation steps help to eliminate matrix effects, reduce interferences, and improve detection limits. The choice of sample preparation method depends on the sample type, target elements, and required sensitivity. Proper sample preparation ensures reliable and reproducible results in multi-element analysis.Expand Specific Solutions04 Instrumentation improvements for multi-element detection
Advancements in instrumentation have significantly enhanced the capabilities of ICP-MS and CE-MS for multi-element detection. These improvements include the development of high-resolution mass analyzers, more efficient ion sources, and better interface designs. Modern instruments feature enhanced sensitivity, reduced interferences, and improved stability. Additionally, automation and integration with sample introduction systems have streamlined the analytical process. These technological advancements have expanded the application range and improved the performance of multi-element detection systems.Expand Specific Solutions05 Applications of ICP-MS and CE-MS in various fields
ICP-MS and CE-MS multi-element detection techniques have wide-ranging applications across numerous fields. In environmental analysis, they are used to monitor pollutants and trace elements in water, soil, and air samples. In the pharmaceutical industry, these techniques help ensure product quality and safety. They are also valuable in clinical diagnostics, food safety testing, geochemical analysis, and materials science. The ability to detect multiple elements simultaneously with high sensitivity makes these techniques indispensable tools for research and quality control in various industries.Expand Specific Solutions
Key Industry Players and Competitive Landscape
ICP-MS vs CE-MS technology comparison reveals a competitive landscape in the multi-element detection market, currently in a growth phase with increasing applications across pharmaceutical, environmental, and clinical sectors. The global market is expanding rapidly, driven by demand for higher sensitivity and throughput in elemental analysis. Technologically, ICP-MS is more mature, with industry leaders like Thermo Fisher Scientific and Agilent Technologies offering established platforms with high sensitivity for trace element detection. CE-MS represents an emerging complementary technology, with companies like Mosaiques Diagnostics pioneering applications in clinical proteomics. Academic institutions including ETH Zurich and McMaster University are advancing both technologies through research collaborations with industry partners, while Chinese companies like Bioyong Technology are rapidly entering the market with cost-competitive solutions.
Thermo Fisher Scientific (Bremen) GmbH
Technical Solution: Thermo Fisher Scientific has developed advanced ICP-MS systems like the iCAP RQ and iCAP TQ that offer triple quadrupole technology for interference removal and enhanced sensitivity. Their ICP-MS solutions provide detection limits in the sub-ppt range with a linear dynamic range spanning up to 10 orders of magnitude. For CE-MS integration, they've created specialized interfaces that maintain electrical connectivity while providing efficient ion transfer to the mass analyzer. Their Orbitrap technology, when coupled with CE, delivers high-resolution accurate mass capabilities particularly valuable for metabolomics and proteomics applications where both charge and mass information are critical. Thermo's systems incorporate intelligent software that automates method development and simplifies the comparison between ICP-MS and CE-MS data for comprehensive elemental profiling.
Strengths: Industry-leading sensitivity in ICP-MS with triple quadrupole technology; seamless integration between separation and detection systems; comprehensive software ecosystem. Weaknesses: Higher initial investment costs compared to competitors; CE-MS interfaces may require more specialized expertise for optimal operation.
Agilent Technologies, Inc.
Technical Solution: Agilent Technologies has pioneered integrated solutions for both ICP-MS and CE-MS analysis. Their 8900 Triple Quadrupole ICP-MS system offers MS/MS capability with helium collision mode that effectively removes polyatomic interferences while maintaining sensitivity for multi-element detection. For CE-MS applications, Agilent has developed specialized CE-ESI-MS interfaces that provide stable electrospray conditions while maintaining separation efficiency. Their CE-MS systems feature proprietary sheath-flow technology that enhances ionization efficiency while preserving CE resolution. Agilent's MassHunter software platform enables seamless method transfer between ICP-MS and CE-MS workflows, allowing researchers to leverage complementary data from both techniques. The company has also developed application-specific kits for metallomics studies that combine the quantitative strengths of ICP-MS with the speciation capabilities of CE-MS.
Strengths: Comprehensive portfolio covering both technologies; excellent interference management in ICP-MS; innovative CE-MS interfaces with optimized ionization efficiency; unified software platform. Weaknesses: CE-MS solutions have more limited dynamic range compared to their ICP-MS offerings; higher operational complexity when switching between techniques.
Critical Patents and Innovations in Mass Spectrometry
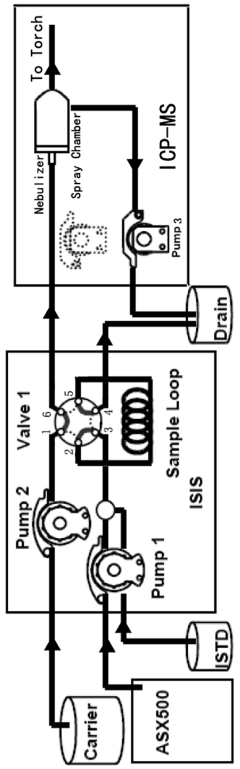
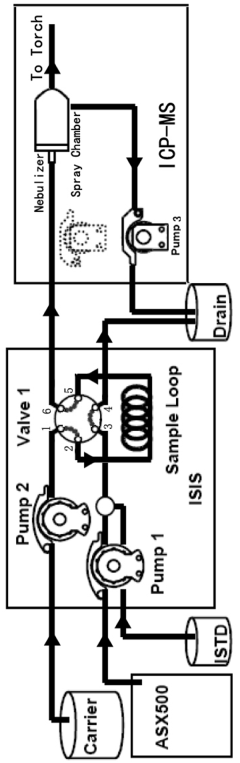
ICP-ms method for multi-element determination by direct sample injection of high salt water
PatentActiveNL2033445A
Innovation
- A Discrete Flow Injection Sampling (DFIS) method using a six-way valve and peristaltic pumps to directly inject seawater samples into an ICP-MS system, optimizing parameters like sample loop diameter, carrier acidity, and pumping speed to minimize salt deposition and ensure stable sample delivery, allowing simultaneous analysis of multiple elements.
Analytical Performance Comparison: Sensitivity and Precision
When comparing ICP-MS and CE-MS for multi-element detection, analytical performance metrics—particularly sensitivity and precision—are critical differentiating factors that determine their suitability for various applications.
ICP-MS demonstrates exceptional sensitivity with detection limits typically in the parts-per-trillion (ppt) to parts-per-quadrillion (ppq) range for most elements. This superior sensitivity stems from the complete atomization and ionization processes occurring in the high-temperature plasma (6,000-10,000K). The technique achieves impressive precision levels, with relative standard deviations (RSDs) commonly below 2% for most elements under optimized conditions.
CE-MS, while generally less sensitive than ICP-MS, offers detection limits in the parts-per-billion (ppb) range for many elements. Its sensitivity is highly dependent on the specific interface design connecting the capillary electrophoresis system to the mass spectrometer. Recent technological advancements in interface design have significantly improved CE-MS sensitivity, narrowing the gap with ICP-MS for certain applications.
Precision represents another key performance metric where these techniques differ substantially. ICP-MS typically delivers superior precision in quantitative analysis, with day-to-day reproducibility often better than 5% RSD. CE-MS historically struggles with precision, showing RSDs between 5-15% in many applications, primarily due to injection volume variations and interface stability issues.
The linear dynamic range further distinguishes these techniques. ICP-MS routinely achieves a linear dynamic range spanning 8-9 orders of magnitude, allowing simultaneous measurement of major, minor, and trace elements without sample dilution. CE-MS typically offers a more limited dynamic range of 3-4 orders of magnitude, requiring careful consideration of sample concentration during method development.
Matrix effects impact both techniques differently. ICP-MS suffers from well-documented polyatomic interferences and matrix suppression effects, though these can be mitigated through collision/reaction cell technology. CE-MS experiences fewer spectral interferences but remains susceptible to matrix-induced migration time shifts and ionization suppression, particularly with complex biological samples.
Sample throughput considerations reveal ICP-MS as the higher-throughput option, capable of analyzing 60-100 samples per hour in standard configurations. CE-MS typically processes 6-12 samples per hour due to longer separation times, though this limitation is offset by its superior separation capabilities for complex mixtures.
Recent technological innovations continue to enhance the analytical performance of both techniques, with ICP-MS benefiting from triple-quadrupole configurations and CE-MS from improved interfaces and microchip implementations that promise enhanced sensitivity and precision.
ICP-MS demonstrates exceptional sensitivity with detection limits typically in the parts-per-trillion (ppt) to parts-per-quadrillion (ppq) range for most elements. This superior sensitivity stems from the complete atomization and ionization processes occurring in the high-temperature plasma (6,000-10,000K). The technique achieves impressive precision levels, with relative standard deviations (RSDs) commonly below 2% for most elements under optimized conditions.
CE-MS, while generally less sensitive than ICP-MS, offers detection limits in the parts-per-billion (ppb) range for many elements. Its sensitivity is highly dependent on the specific interface design connecting the capillary electrophoresis system to the mass spectrometer. Recent technological advancements in interface design have significantly improved CE-MS sensitivity, narrowing the gap with ICP-MS for certain applications.
Precision represents another key performance metric where these techniques differ substantially. ICP-MS typically delivers superior precision in quantitative analysis, with day-to-day reproducibility often better than 5% RSD. CE-MS historically struggles with precision, showing RSDs between 5-15% in many applications, primarily due to injection volume variations and interface stability issues.
The linear dynamic range further distinguishes these techniques. ICP-MS routinely achieves a linear dynamic range spanning 8-9 orders of magnitude, allowing simultaneous measurement of major, minor, and trace elements without sample dilution. CE-MS typically offers a more limited dynamic range of 3-4 orders of magnitude, requiring careful consideration of sample concentration during method development.
Matrix effects impact both techniques differently. ICP-MS suffers from well-documented polyatomic interferences and matrix suppression effects, though these can be mitigated through collision/reaction cell technology. CE-MS experiences fewer spectral interferences but remains susceptible to matrix-induced migration time shifts and ionization suppression, particularly with complex biological samples.
Sample throughput considerations reveal ICP-MS as the higher-throughput option, capable of analyzing 60-100 samples per hour in standard configurations. CE-MS typically processes 6-12 samples per hour due to longer separation times, though this limitation is offset by its superior separation capabilities for complex mixtures.
Recent technological innovations continue to enhance the analytical performance of both techniques, with ICP-MS benefiting from triple-quadrupole configurations and CE-MS from improved interfaces and microchip implementations that promise enhanced sensitivity and precision.
Regulatory Standards and Method Validation Requirements
Regulatory frameworks governing analytical methods for multi-element detection vary significantly across different regions and industries, necessitating thorough understanding of applicable standards. For ICP-MS, the US Environmental Protection Agency (EPA) has established Method 6020 for elemental analysis in environmental samples, while the Food and Drug Administration (FDA) provides guidelines for elemental impurities in pharmaceutical products through ICH Q3D. These regulatory standards specify detection limits, quality control procedures, and validation parameters that laboratories must adhere to when implementing ICP-MS methodologies.
CE-MS, being a relatively newer technique for multi-element analysis, has fewer dedicated regulatory standards. However, it falls under general mass spectrometry validation guidelines provided by organizations such as the International Conference on Harmonisation (ICH) and the Association of Official Analytical Chemists (AOAC). The European Medicines Agency (EMA) also provides guidance applicable to CE-MS implementations in pharmaceutical analysis through its guideline on bioanalytical method validation.
Method validation requirements for both techniques share common parameters including accuracy, precision, specificity, detection limit, quantitation limit, linearity, and robustness. However, ICP-MS validation protocols typically emphasize interference management and calibration stability across wide concentration ranges, while CE-MS validation focuses more on migration time reproducibility and system suitability testing specific to electrophoretic separations.
For environmental applications, ICP-MS methods must comply with ISO 17025 accreditation requirements and demonstrate performance characteristics meeting regional environmental monitoring standards. The EPA requires specific quality control measures including calibration verification, internal standardization, and interference checks that are tailored to ICP-MS technology.
Pharmaceutical industry applications face particularly stringent requirements under USP <232>, <233>, and ICH Q3D guidelines for elemental impurities analysis. These standards specify permitted daily exposure limits for various elements and require validated methods capable of reliably detecting these elements at or below specified thresholds. While ICP-MS has established protocols under these frameworks, laboratories employing CE-MS for similar applications must develop and validate custom protocols that demonstrate equivalent performance.
Clinical diagnostic applications of both techniques must comply with Clinical Laboratory Improvement Amendments (CLIA) regulations in the US or equivalent standards internationally. These regulations mandate regular proficiency testing, quality control procedures, and thorough documentation of method validation parameters to ensure reliable patient results.
CE-MS, being a relatively newer technique for multi-element analysis, has fewer dedicated regulatory standards. However, it falls under general mass spectrometry validation guidelines provided by organizations such as the International Conference on Harmonisation (ICH) and the Association of Official Analytical Chemists (AOAC). The European Medicines Agency (EMA) also provides guidance applicable to CE-MS implementations in pharmaceutical analysis through its guideline on bioanalytical method validation.
Method validation requirements for both techniques share common parameters including accuracy, precision, specificity, detection limit, quantitation limit, linearity, and robustness. However, ICP-MS validation protocols typically emphasize interference management and calibration stability across wide concentration ranges, while CE-MS validation focuses more on migration time reproducibility and system suitability testing specific to electrophoretic separations.
For environmental applications, ICP-MS methods must comply with ISO 17025 accreditation requirements and demonstrate performance characteristics meeting regional environmental monitoring standards. The EPA requires specific quality control measures including calibration verification, internal standardization, and interference checks that are tailored to ICP-MS technology.
Pharmaceutical industry applications face particularly stringent requirements under USP <232>, <233>, and ICH Q3D guidelines for elemental impurities analysis. These standards specify permitted daily exposure limits for various elements and require validated methods capable of reliably detecting these elements at or below specified thresholds. While ICP-MS has established protocols under these frameworks, laboratories employing CE-MS for similar applications must develop and validate custom protocols that demonstrate equivalent performance.
Clinical diagnostic applications of both techniques must comply with Clinical Laboratory Improvement Amendments (CLIA) regulations in the US or equivalent standards internationally. These regulations mandate regular proficiency testing, quality control procedures, and thorough documentation of method validation parameters to ensure reliable patient results.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!