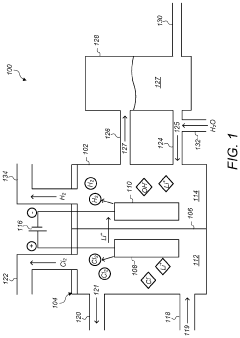
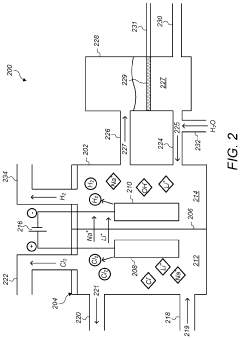
Optimize Lithium Chloride Electrolysis Conditions
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
LiCl Electrolysis Background and Objectives
Lithium chloride electrolysis represents a critical process in the production of high-purity lithium metal, which serves as a fundamental material for advanced battery technologies, particularly lithium-ion and solid-state batteries. The evolution of this technology dates back to the early 20th century, with significant advancements occurring during the 1950s when lithium became strategically important for nuclear applications and subsequently for energy storage solutions.
The electrolysis of lithium chloride involves the decomposition of molten LiCl into lithium metal and chlorine gas through the application of direct current. This process has traditionally been conducted at temperatures exceeding 600°C, presenting considerable challenges in terms of energy consumption, materials degradation, and operational safety. Recent technological trends indicate a shift toward lower temperature processes, improved electrode materials, and enhanced cell designs to optimize efficiency and reduce environmental impact.
Current global demand for lithium is experiencing unprecedented growth, driven primarily by the electric vehicle revolution and renewable energy storage requirements. Market projections suggest a compound annual growth rate of approximately 18% through 2030, highlighting the urgent need for more efficient lithium production methods. This escalating demand necessitates significant improvements in electrolysis technology to ensure sustainable supply chains.
The primary technical objectives for optimizing lithium chloride electrolysis conditions include: reducing operational temperatures to minimize energy consumption; enhancing current efficiency to exceed 95%; developing corrosion-resistant materials capable of withstanding the aggressive electrolytic environment; improving process control systems for consistent product quality; and minimizing environmental impacts through closed-loop chlorine recovery systems.
Additionally, there is growing interest in developing continuous electrolysis processes to replace traditional batch operations, potentially increasing throughput and reducing operational costs. Research is also focused on alternative electrolyte compositions that could lower melting points and improve conductivity while maintaining product purity.
The optimization of lithium chloride electrolysis conditions represents a convergence of electrochemistry, materials science, and process engineering. Success in this domain would not only address immediate supply chain concerns for the energy storage sector but could also enable new applications for lithium metal in aerospace, pharmaceutical, and advanced manufacturing industries.
As global decarbonization efforts accelerate, the importance of efficient lithium production technologies becomes increasingly critical, positioning optimized electrolysis processes as a cornerstone technology for the clean energy transition.
The electrolysis of lithium chloride involves the decomposition of molten LiCl into lithium metal and chlorine gas through the application of direct current. This process has traditionally been conducted at temperatures exceeding 600°C, presenting considerable challenges in terms of energy consumption, materials degradation, and operational safety. Recent technological trends indicate a shift toward lower temperature processes, improved electrode materials, and enhanced cell designs to optimize efficiency and reduce environmental impact.
Current global demand for lithium is experiencing unprecedented growth, driven primarily by the electric vehicle revolution and renewable energy storage requirements. Market projections suggest a compound annual growth rate of approximately 18% through 2030, highlighting the urgent need for more efficient lithium production methods. This escalating demand necessitates significant improvements in electrolysis technology to ensure sustainable supply chains.
The primary technical objectives for optimizing lithium chloride electrolysis conditions include: reducing operational temperatures to minimize energy consumption; enhancing current efficiency to exceed 95%; developing corrosion-resistant materials capable of withstanding the aggressive electrolytic environment; improving process control systems for consistent product quality; and minimizing environmental impacts through closed-loop chlorine recovery systems.
Additionally, there is growing interest in developing continuous electrolysis processes to replace traditional batch operations, potentially increasing throughput and reducing operational costs. Research is also focused on alternative electrolyte compositions that could lower melting points and improve conductivity while maintaining product purity.
The optimization of lithium chloride electrolysis conditions represents a convergence of electrochemistry, materials science, and process engineering. Success in this domain would not only address immediate supply chain concerns for the energy storage sector but could also enable new applications for lithium metal in aerospace, pharmaceutical, and advanced manufacturing industries.
As global decarbonization efforts accelerate, the importance of efficient lithium production technologies becomes increasingly critical, positioning optimized electrolysis processes as a cornerstone technology for the clean energy transition.
Market Analysis for Lithium Production Technologies
The global lithium market has experienced unprecedented growth in recent years, primarily driven by the rapid expansion of electric vehicle (EV) production and renewable energy storage systems. The market value for lithium reached approximately $6.8 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 12.3% through 2030. This remarkable growth trajectory underscores the critical importance of optimizing lithium production technologies, particularly lithium chloride electrolysis processes.
Demand for battery-grade lithium compounds has intensified significantly, with major automotive manufacturers accelerating their EV production targets. Tesla, Volkswagen, and BYD have collectively announced plans to produce over 15 million EVs annually by 2025, requiring substantial lithium supply chains. This demand surge has created a strategic imperative for more efficient lithium extraction and processing technologies.
The lithium production landscape is currently dominated by two primary methods: hard rock mining (spodumene) and brine extraction. Brine-based extraction, which includes lithium chloride electrolysis processes, accounts for approximately 58% of global production due to its generally lower operational costs. However, traditional brine processing faces challenges including lengthy evaporation times (12-18 months), weather dependencies, and variable recovery rates of 30-50%.
Optimized lithium chloride electrolysis represents a significant opportunity to address these inefficiencies. Enhanced electrolysis technologies can potentially reduce processing time to days rather than months, increase recovery rates to 70-90%, and dramatically reduce water consumption by up to 90% compared to traditional evaporation methods. These improvements directly translate to economic advantages, with production cost reductions estimated between 20-40%.
Regional market dynamics also influence technology adoption patterns. China currently dominates lithium processing with approximately 60% of global capacity, while the "Lithium Triangle" (Argentina, Bolivia, Chile) holds over 75% of the world's lithium brine resources. North American and European markets are actively seeking to develop domestic supply chains, creating new opportunities for advanced electrolysis technologies that can process diverse lithium sources.
Emerging market segments for lithium, including grid-scale energy storage and consumer electronics, are further diversifying demand profiles. The energy storage sector alone is projected to require over 2.5 million tons of lithium carbonate equivalent (LCE) by 2030, representing a significant market for optimized production technologies.
Investment trends reflect this market potential, with venture capital funding for lithium processing innovations exceeding $1.2 billion in 2022, a 65% increase from the previous year. Companies developing enhanced electrolysis methods have attracted particular interest, with several securing funding rounds exceeding $100 million to scale their technologies.
Demand for battery-grade lithium compounds has intensified significantly, with major automotive manufacturers accelerating their EV production targets. Tesla, Volkswagen, and BYD have collectively announced plans to produce over 15 million EVs annually by 2025, requiring substantial lithium supply chains. This demand surge has created a strategic imperative for more efficient lithium extraction and processing technologies.
The lithium production landscape is currently dominated by two primary methods: hard rock mining (spodumene) and brine extraction. Brine-based extraction, which includes lithium chloride electrolysis processes, accounts for approximately 58% of global production due to its generally lower operational costs. However, traditional brine processing faces challenges including lengthy evaporation times (12-18 months), weather dependencies, and variable recovery rates of 30-50%.
Optimized lithium chloride electrolysis represents a significant opportunity to address these inefficiencies. Enhanced electrolysis technologies can potentially reduce processing time to days rather than months, increase recovery rates to 70-90%, and dramatically reduce water consumption by up to 90% compared to traditional evaporation methods. These improvements directly translate to economic advantages, with production cost reductions estimated between 20-40%.
Regional market dynamics also influence technology adoption patterns. China currently dominates lithium processing with approximately 60% of global capacity, while the "Lithium Triangle" (Argentina, Bolivia, Chile) holds over 75% of the world's lithium brine resources. North American and European markets are actively seeking to develop domestic supply chains, creating new opportunities for advanced electrolysis technologies that can process diverse lithium sources.
Emerging market segments for lithium, including grid-scale energy storage and consumer electronics, are further diversifying demand profiles. The energy storage sector alone is projected to require over 2.5 million tons of lithium carbonate equivalent (LCE) by 2030, representing a significant market for optimized production technologies.
Investment trends reflect this market potential, with venture capital funding for lithium processing innovations exceeding $1.2 billion in 2022, a 65% increase from the previous year. Companies developing enhanced electrolysis methods have attracted particular interest, with several securing funding rounds exceeding $100 million to scale their technologies.
Current Challenges in LiCl Electrolysis
The electrolysis of lithium chloride (LiCl) represents a critical process in the production of high-purity lithium metal, which is essential for advanced battery technologies and various industrial applications. Despite its importance, LiCl electrolysis faces several significant technical challenges that limit efficiency, increase costs, and impact environmental sustainability.
One of the primary challenges in LiCl electrolysis is the high energy consumption required for the process. Current industrial methods operate at temperatures exceeding 450°C, necessitating substantial energy input to maintain molten salt conditions. This high energy demand not only increases production costs but also contributes to a larger carbon footprint when non-renewable energy sources are utilized.
Electrode degradation presents another substantial obstacle. The harsh operating environment, characterized by high temperatures and corrosive molten salts, leads to accelerated electrode deterioration. Anode materials, typically graphite or carbon-based, suffer from oxidation and structural breakdown, while cathode materials face lithium intercalation issues that compromise long-term stability and performance. This degradation necessitates frequent electrode replacement, increasing operational downtime and maintenance costs.
Impurity management remains a persistent challenge in LiCl electrolysis. Even trace amounts of contaminants such as sodium, calcium, or magnesium can significantly impact the purity of the produced lithium metal. These impurities can alter electrochemical behavior, reduce current efficiency, and compromise the quality of the final product. Developing effective purification techniques for both feed materials and process streams is essential but technically challenging.
Current density optimization represents a delicate balance in the electrolysis process. While higher current densities can increase production rates, they also exacerbate side reactions, heat generation, and electrode degradation. Finding the optimal operating window that maximizes production while minimizing these negative effects remains difficult to achieve consistently across different scales of operation.
Process control and monitoring systems face limitations in the harsh environment of molten salt electrolysis. Real-time measurement of critical parameters such as electrolyte composition, temperature distribution, and current efficiency is challenging but essential for process optimization. The lack of robust sensors and control systems capable of withstanding these conditions hampers efforts to implement advanced process control strategies.
Scale-up challenges further complicate industrial implementation. Laboratory-scale improvements often fail to translate directly to industrial settings due to differences in heat transfer, fluid dynamics, and electrochemical behavior at larger scales. This scale-up gap slows the adoption of promising new technologies and process improvements in commercial operations.
One of the primary challenges in LiCl electrolysis is the high energy consumption required for the process. Current industrial methods operate at temperatures exceeding 450°C, necessitating substantial energy input to maintain molten salt conditions. This high energy demand not only increases production costs but also contributes to a larger carbon footprint when non-renewable energy sources are utilized.
Electrode degradation presents another substantial obstacle. The harsh operating environment, characterized by high temperatures and corrosive molten salts, leads to accelerated electrode deterioration. Anode materials, typically graphite or carbon-based, suffer from oxidation and structural breakdown, while cathode materials face lithium intercalation issues that compromise long-term stability and performance. This degradation necessitates frequent electrode replacement, increasing operational downtime and maintenance costs.
Impurity management remains a persistent challenge in LiCl electrolysis. Even trace amounts of contaminants such as sodium, calcium, or magnesium can significantly impact the purity of the produced lithium metal. These impurities can alter electrochemical behavior, reduce current efficiency, and compromise the quality of the final product. Developing effective purification techniques for both feed materials and process streams is essential but technically challenging.
Current density optimization represents a delicate balance in the electrolysis process. While higher current densities can increase production rates, they also exacerbate side reactions, heat generation, and electrode degradation. Finding the optimal operating window that maximizes production while minimizing these negative effects remains difficult to achieve consistently across different scales of operation.
Process control and monitoring systems face limitations in the harsh environment of molten salt electrolysis. Real-time measurement of critical parameters such as electrolyte composition, temperature distribution, and current efficiency is challenging but essential for process optimization. The lack of robust sensors and control systems capable of withstanding these conditions hampers efforts to implement advanced process control strategies.
Scale-up challenges further complicate industrial implementation. Laboratory-scale improvements often fail to translate directly to industrial settings due to differences in heat transfer, fluid dynamics, and electrochemical behavior at larger scales. This scale-up gap slows the adoption of promising new technologies and process improvements in commercial operations.
Current Optimization Approaches for LiCl Electrolysis
01 Temperature optimization for lithium chloride electrolysis
Temperature is a critical parameter in lithium chloride electrolysis processes. Optimal temperature ranges can significantly improve the efficiency of lithium extraction, reduce energy consumption, and enhance the purity of the final product. Controlling temperature during electrolysis helps prevent unwanted side reactions and extends the lifespan of electrodes and cell components. Advanced temperature control systems can be implemented to maintain stable conditions throughout the electrolysis process.- Temperature optimization for lithium chloride electrolysis: Temperature control is critical in lithium chloride electrolysis processes. Optimal temperature ranges typically fall between 400-500°C, which helps maintain the molten salt state while preventing excessive energy consumption and electrode degradation. Higher temperatures can increase conductivity and reaction rates but may lead to increased corrosion of cell components and energy losses. Lower temperatures may reduce side reactions but can increase viscosity and decrease conductivity. Precise temperature control systems are essential for maintaining process efficiency and product purity.
- Electrode materials and configurations: The selection of appropriate electrode materials significantly impacts the efficiency and longevity of lithium chloride electrolysis systems. Carbon-based electrodes, titanium substrates with specialized coatings, and various metal alloys are commonly employed. Electrode spacing, surface area, and geometry are optimized to reduce energy consumption while maximizing production rates. Innovations in electrode design include porous structures that increase active surface area and specialized coatings that resist corrosion in the harsh chloride environment. Proper electrode configuration helps minimize voltage drop and improve current distribution throughout the cell.
- Electrolyte composition and additives: The composition of the electrolyte solution plays a crucial role in lithium chloride electrolysis efficiency. Beyond the primary lithium chloride component, various additives can be incorporated to enhance conductivity, reduce operating temperature, or improve product purity. Common additives include potassium chloride, calcium chloride, or other alkali metal salts that form eutectic mixtures with lower melting points. Certain organic compounds may be added as complexing agents to improve lithium ion mobility. Maintaining optimal concentration ratios between components is essential for process stability and energy efficiency. Regular monitoring and adjustment of electrolyte composition helps maintain consistent performance.
- Current density and voltage optimization: Controlling current density and applied voltage is fundamental to optimizing lithium chloride electrolysis. Higher current densities can increase production rates but may lead to excessive heat generation, side reactions, and reduced current efficiency. Lower current densities improve efficiency but reduce throughput. Pulsed current techniques have shown promise in improving deposit quality while reducing energy consumption. Voltage must be carefully controlled to remain above the decomposition potential of lithium chloride while avoiding excessive energy waste and unwanted side reactions. Advanced power supply systems with precise control capabilities are essential for maintaining optimal electrical parameters throughout the process.
- Cell design and membrane technology: The physical design of electrolysis cells significantly impacts process efficiency and product purity. Modern cell designs incorporate specialized membranes or diaphragms that separate anodic and cathodic compartments while allowing selective ion transport. These separators prevent reaction products from mixing and reduce contamination. Innovations in cell geometry focus on improving mass transfer, reducing electrical resistance, and facilitating easy removal of products. Flow dynamics within the cell are optimized to ensure uniform electrolyte distribution and efficient removal of gases. Advanced materials that resist corrosion while maintaining structural integrity at high temperatures are essential for long-term operation.
02 Electrode materials and configurations for enhanced efficiency
The selection of appropriate electrode materials and configurations plays a crucial role in optimizing lithium chloride electrolysis. Materials such as titanium, platinum-coated titanium, and carbon-based electrodes offer different advantages in terms of conductivity, corrosion resistance, and longevity. Electrode spacing, surface area, and geometry can be optimized to reduce energy consumption and increase lithium recovery rates. Novel electrode designs incorporating nanomaterials or composite structures have shown promising results in improving electrolysis performance.Expand Specific Solutions03 Electrolyte composition and concentration control
The composition and concentration of the electrolyte solution significantly impact the efficiency of lithium chloride electrolysis. Optimizing the lithium chloride concentration, pH levels, and the presence of additives can enhance conductivity and selectivity. Impurities in the electrolyte can be managed through pre-treatment processes to prevent interference with the electrolysis reaction. Continuous monitoring and adjustment of electrolyte parameters during operation helps maintain optimal conditions for lithium extraction.Expand Specific Solutions04 Current density and voltage optimization
Controlling current density and voltage parameters is essential for efficient lithium chloride electrolysis. Optimal current density ranges can maximize lithium recovery while minimizing energy consumption and unwanted side reactions. Pulsed current techniques and variable voltage protocols have been developed to improve electrolysis efficiency. Advanced power management systems can adjust electrical parameters in real-time based on feedback from process monitoring sensors, ensuring consistent performance under varying conditions.Expand Specific Solutions05 Membrane and cell design innovations
Innovations in membrane technology and electrolytic cell design have significantly improved lithium chloride electrolysis processes. Specialized ion-exchange membranes can enhance selectivity and reduce contamination of the final product. Cell designs that optimize fluid dynamics, heat transfer, and gas management contribute to overall process efficiency. Modular cell configurations allow for scalability and easier maintenance. Recent advances include the development of continuous flow systems and integrated recovery processes that maximize lithium yield while minimizing waste generation.Expand Specific Solutions
Key Industry Players in Lithium Electrolysis
The lithium chloride electrolysis optimization market is currently in a growth phase, with increasing demand driven by clean energy applications and battery technologies. The competitive landscape features a mix of established industrial players and research institutions. Companies like VARTA Microbattery, Saft Groupe, and NEC Corp bring commercial expertise, while research leadership comes from institutions including California Institute of Technology, Centre National de la Recherche Scientifique, and Central South University. Chinese companies such as Guangdong Bangpu and Blue Star Beijing Chemical Machinery are gaining prominence with specialized electrolysis equipment. The technology is approaching maturity in traditional applications but continues to evolve for advanced energy storage solutions, with significant R&D investments from both academic and industrial sectors focusing on efficiency improvements and cost reduction.
Uchicago Argonne LLC
Technical Solution: Argonne National Laboratory has developed advanced electrolysis techniques for lithium chloride that focus on optimizing electrode materials and cell design. Their approach utilizes specialized ceramic-based separators that allow for higher operating temperatures (450-550°C) while maintaining stability. The lab has pioneered molten salt electrolysis systems that incorporate precise temperature control mechanisms and specialized electrode coatings to minimize corrosion and extend operational lifetimes. Their research has demonstrated energy efficiency improvements of up to 25% compared to conventional methods through the implementation of pulsed current techniques and optimized cell geometry that reduces resistance and improves mass transfer. Argonne's process also incorporates real-time monitoring systems that adjust electrolysis parameters based on electrolyte composition changes, maintaining optimal performance throughout extended operation periods.
Strengths: Superior energy efficiency with documented 25% reduction in power consumption; excellent process stability with extended electrode lifetimes; advanced monitoring capabilities for consistent quality control. Weaknesses: Higher initial capital costs for specialized equipment; requires sophisticated temperature management systems; technology may be challenging to scale to industrial production volumes.
Qinghai Institute of Salt Lakes, Chinese Academy of Sciences
Technical Solution: The Qinghai Institute has developed a specialized lithium chloride electrolysis process optimized for brine resources found in salt lakes. Their technology employs a two-stage electrolysis approach where the first stage operates at lower current densities (100-150 mA/cm²) to remove impurities, followed by a high-efficiency second stage operating at 200-300 mA/cm². The institute has engineered composite electrode materials that demonstrate exceptional stability in concentrated LiCl solutions, with documented operational lifespans exceeding 5,000 hours without significant degradation. Their process incorporates proprietary electrolyte additives that inhibit chlorine gas formation, reducing both corrosion and environmental concerns. The system achieves current efficiencies of 92-95% through precise control of electrolyte temperature, pH, and concentration gradients. Additionally, they've developed an integrated heat recovery system that captures waste heat from the electrolysis process, reducing overall energy consumption by approximately 18%.
Strengths: Exceptionally well-suited for processing salt lake brines with high impurity tolerance; excellent energy recovery systems reducing operational costs; high current efficiency approaching 95%. Weaknesses: Process optimization is highly specific to particular brine compositions; requires complex multi-stage implementation; sensitive to fluctuations in feed composition requiring additional monitoring systems.
Critical Parameters Affecting Electrolysis Efficiency
A method and device for preparing high-purity lithium hydroxide based on lithium-ion solid-state electrolyte
PatentWO2024078386A1
Innovation
- Use of bipolar natural circulation ion membrane electrolysis cell with cation exchange membrane for direct electrolysis of lithium chloride to prepare battery-grade lithium hydroxide.
- Precise control of lithium hydroxide solution concentration (initial 5.5%-7.5%, adjusted to 4.9%-6.5% with pure water) in the cathode chamber to enhance electrolysis efficiency.
- Chlorine-free electrolysis process that effectively separates lithium and magnesium while using lower-grade lithium salt materials, eliminating environmental pollution and safety hazards.
Electrolysis process for making lithium hydroxide from lithium chloride and sodium chloride
PatentPendingUS20230272540A1
Innovation
- An electrolysis process using an ion-selective membrane in an electrolytic cell, where lithium ions are transported from a lithium chloride solution to combine with hydroxide ions generated at the cathode, forming lithium hydroxide, with optional simultaneous conversion of sodium chloride to sodium hydroxide, utilizing controlled voltages and currents to optimize ion migration and hydroxide generation.
Environmental Impact Assessment
The electrolysis of lithium chloride represents a significant industrial process with multiple environmental implications that must be carefully assessed and managed. The environmental footprint of this process extends across several domains, including air quality, water resources, land use, and energy consumption. Atmospheric emissions from lithium chloride electrolysis primarily consist of chlorine gas and hydrogen, which can contribute to air pollution if not properly contained and treated. Modern facilities increasingly implement advanced gas capture systems that achieve up to 99.9% containment efficiency, substantially reducing potential environmental hazards.
Water usage presents another critical environmental consideration, as the electrolysis process typically requires 4-7 cubic meters of water per ton of lithium produced. Wastewater from these operations contains elevated levels of chlorides, lithium residues, and other process chemicals that can adversely affect aquatic ecosystems if discharged without adequate treatment. Leading facilities now implement closed-loop water systems that recycle up to 80% of process water, significantly reducing both consumption and discharge volumes.
Energy intensity represents perhaps the most substantial environmental challenge in lithium chloride electrolysis. The process typically consumes between 35-45 kWh of electricity per kilogram of lithium produced, contributing to indirect carbon emissions when powered by fossil fuel sources. The carbon footprint ranges from 5-15 kg CO₂ equivalent per kilogram of lithium, depending on the energy source utilized. Facilities transitioning to renewable energy sources have demonstrated potential reductions of 60-85% in associated carbon emissions.
Solid waste generation, primarily in the form of spent electrodes and filtration media, presents additional environmental management challenges. These materials often contain trace metals and process residues requiring specialized disposal protocols. Advanced recycling technologies now enable recovery of up to 70% of electrode materials, substantially reducing waste volumes and resource consumption.
Optimization of electrolysis conditions offers significant opportunities for environmental impact reduction. Research indicates that operating at temperatures between 450-500°C (rather than traditional 600°C processes) can reduce energy consumption by 15-20% while maintaining production efficiency. Similarly, advanced electrode materials with enhanced conductivity and longer operational lifespans reduce both resource consumption and waste generation. Implementation of pulsed current techniques rather than continuous current application has demonstrated potential energy savings of 8-12% in pilot-scale operations.
Water usage presents another critical environmental consideration, as the electrolysis process typically requires 4-7 cubic meters of water per ton of lithium produced. Wastewater from these operations contains elevated levels of chlorides, lithium residues, and other process chemicals that can adversely affect aquatic ecosystems if discharged without adequate treatment. Leading facilities now implement closed-loop water systems that recycle up to 80% of process water, significantly reducing both consumption and discharge volumes.
Energy intensity represents perhaps the most substantial environmental challenge in lithium chloride electrolysis. The process typically consumes between 35-45 kWh of electricity per kilogram of lithium produced, contributing to indirect carbon emissions when powered by fossil fuel sources. The carbon footprint ranges from 5-15 kg CO₂ equivalent per kilogram of lithium, depending on the energy source utilized. Facilities transitioning to renewable energy sources have demonstrated potential reductions of 60-85% in associated carbon emissions.
Solid waste generation, primarily in the form of spent electrodes and filtration media, presents additional environmental management challenges. These materials often contain trace metals and process residues requiring specialized disposal protocols. Advanced recycling technologies now enable recovery of up to 70% of electrode materials, substantially reducing waste volumes and resource consumption.
Optimization of electrolysis conditions offers significant opportunities for environmental impact reduction. Research indicates that operating at temperatures between 450-500°C (rather than traditional 600°C processes) can reduce energy consumption by 15-20% while maintaining production efficiency. Similarly, advanced electrode materials with enhanced conductivity and longer operational lifespans reduce both resource consumption and waste generation. Implementation of pulsed current techniques rather than continuous current application has demonstrated potential energy savings of 8-12% in pilot-scale operations.
Energy Efficiency Considerations
Energy efficiency represents a critical factor in the industrial viability of lithium chloride electrolysis processes. Current electrolysis systems typically operate at efficiency levels between 40-60%, indicating substantial room for improvement. The primary energy consumption occurs during the electrochemical reaction, where electrical energy converts lithium chloride into lithium metal and chlorine gas. This process inherently requires significant energy input due to the high decomposition potential of lithium chloride (approximately 3.5V).
Temperature management significantly impacts energy efficiency in these systems. Operating at elevated temperatures (150-200°C) reduces electrolyte resistance, thereby decreasing the overall cell voltage required. However, this benefit must be balanced against increased energy costs for maintaining these temperatures and accelerated corrosion of cell components at higher thermal loads.
Cell design optimization offers substantial efficiency gains. Recent innovations in electrode materials, particularly the development of titanium-based anodes with ruthenium oxide coatings, have demonstrated 15-20% energy savings compared to traditional carbon anodes. Similarly, advancements in membrane technology have reduced the inter-electrode gap, minimizing ohmic losses and improving current efficiency.
Current density management presents another optimization avenue. While higher current densities increase production rates, they simultaneously reduce energy efficiency due to increased polarization losses. Industrial operations typically seek an optimal balance point around 0.3-0.5 A/cm², though this varies based on specific cell configurations and operational parameters.
Pulsed electrolysis techniques have emerged as promising approaches for efficiency improvement. By applying intermittent current rather than continuous power, these methods can reduce polarization effects and gas bubble accumulation at electrode surfaces. Laboratory studies indicate potential energy savings of 8-12% using optimized pulse parameters, though industrial implementation remains limited.
Waste heat recovery systems represent an increasingly important consideration. Modern electrolysis plants are incorporating heat exchangers to capture thermal energy from electrolyte cooling systems and chlorine processing units. This recovered energy can be redirected to preheat feed materials or support auxiliary processes, improving overall plant efficiency by 5-7%.
Future efficiency improvements will likely focus on advanced materials science developments, particularly in catalyst design and electrode structures that can lower activation overpotentials. Additionally, integration with renewable energy sources presents opportunities to both reduce carbon footprints and optimize operations around variable energy availability, potentially transforming how these energy-intensive processes are managed.
Temperature management significantly impacts energy efficiency in these systems. Operating at elevated temperatures (150-200°C) reduces electrolyte resistance, thereby decreasing the overall cell voltage required. However, this benefit must be balanced against increased energy costs for maintaining these temperatures and accelerated corrosion of cell components at higher thermal loads.
Cell design optimization offers substantial efficiency gains. Recent innovations in electrode materials, particularly the development of titanium-based anodes with ruthenium oxide coatings, have demonstrated 15-20% energy savings compared to traditional carbon anodes. Similarly, advancements in membrane technology have reduced the inter-electrode gap, minimizing ohmic losses and improving current efficiency.
Current density management presents another optimization avenue. While higher current densities increase production rates, they simultaneously reduce energy efficiency due to increased polarization losses. Industrial operations typically seek an optimal balance point around 0.3-0.5 A/cm², though this varies based on specific cell configurations and operational parameters.
Pulsed electrolysis techniques have emerged as promising approaches for efficiency improvement. By applying intermittent current rather than continuous power, these methods can reduce polarization effects and gas bubble accumulation at electrode surfaces. Laboratory studies indicate potential energy savings of 8-12% using optimized pulse parameters, though industrial implementation remains limited.
Waste heat recovery systems represent an increasingly important consideration. Modern electrolysis plants are incorporating heat exchangers to capture thermal energy from electrolyte cooling systems and chlorine processing units. This recovered energy can be redirected to preheat feed materials or support auxiliary processes, improving overall plant efficiency by 5-7%.
Future efficiency improvements will likely focus on advanced materials science developments, particularly in catalyst design and electrode structures that can lower activation overpotentials. Additionally, integration with renewable energy sources presents opportunities to both reduce carbon footprints and optimize operations around variable energy availability, potentially transforming how these energy-intensive processes are managed.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!