What Are the Regulatory Considerations for Soft Pneumatic Actuators
OCT 11, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Soft Pneumatic Actuators Background and Objectives
Soft Pneumatic Actuators (SPAs) represent a revolutionary advancement in the field of robotics and automation, emerging from the broader domain of soft robotics. Unlike traditional rigid actuators, SPAs utilize pneumatic pressure to create controlled deformation in flexible materials, enabling complex movements and adaptable interactions with the environment. The evolution of this technology can be traced back to the early 2000s, with significant acceleration in research and development occurring over the past decade.
The technological trajectory of SPAs has been characterized by progressive improvements in material science, manufacturing techniques, and control systems. Initially limited by material constraints and fabrication challenges, modern SPAs now benefit from advanced elastomers, composite structures, and sophisticated 3D printing methodologies that enable precise geometric configurations and enhanced performance characteristics.
Current research trends indicate a growing focus on biomimetic designs, multi-material fabrication, and integrated sensing capabilities. These developments are pushing SPAs toward more sophisticated applications requiring precise control, durability, and reliability in diverse operating environments.
The primary technical objectives for SPA development include enhancing operational longevity, improving force-to-weight ratios, developing standardized performance metrics, and establishing comprehensive regulatory frameworks. Particularly critical is the need to address the regulatory considerations that will govern the safe and effective deployment of these systems across various industries.
Regulatory objectives specifically aim to establish clear safety standards, performance benchmarks, and compliance protocols that can accommodate the unique characteristics of soft pneumatic systems. This includes developing testing methodologies that accurately assess failure modes, operational limits, and potential hazards associated with pressurized soft systems in human-interactive environments.
Furthermore, there is a pressing need to harmonize emerging SPA technologies with existing regulatory frameworks in medical devices, industrial automation, and consumer products. This harmonization process must address the inherent variability in SPA performance characteristics, which differs significantly from the more predictable behavior of conventional rigid actuators.
The long-term technological goal for SPAs involves creating a comprehensive ecosystem that encompasses materials, manufacturing processes, control systems, and regulatory standards. This ecosystem would support the seamless integration of SPAs into critical applications such as medical devices, assistive technologies, and collaborative robotics, where their inherent compliance and adaptability offer significant advantages over traditional actuation methods.
The technological trajectory of SPAs has been characterized by progressive improvements in material science, manufacturing techniques, and control systems. Initially limited by material constraints and fabrication challenges, modern SPAs now benefit from advanced elastomers, composite structures, and sophisticated 3D printing methodologies that enable precise geometric configurations and enhanced performance characteristics.
Current research trends indicate a growing focus on biomimetic designs, multi-material fabrication, and integrated sensing capabilities. These developments are pushing SPAs toward more sophisticated applications requiring precise control, durability, and reliability in diverse operating environments.
The primary technical objectives for SPA development include enhancing operational longevity, improving force-to-weight ratios, developing standardized performance metrics, and establishing comprehensive regulatory frameworks. Particularly critical is the need to address the regulatory considerations that will govern the safe and effective deployment of these systems across various industries.
Regulatory objectives specifically aim to establish clear safety standards, performance benchmarks, and compliance protocols that can accommodate the unique characteristics of soft pneumatic systems. This includes developing testing methodologies that accurately assess failure modes, operational limits, and potential hazards associated with pressurized soft systems in human-interactive environments.
Furthermore, there is a pressing need to harmonize emerging SPA technologies with existing regulatory frameworks in medical devices, industrial automation, and consumer products. This harmonization process must address the inherent variability in SPA performance characteristics, which differs significantly from the more predictable behavior of conventional rigid actuators.
The long-term technological goal for SPAs involves creating a comprehensive ecosystem that encompasses materials, manufacturing processes, control systems, and regulatory standards. This ecosystem would support the seamless integration of SPAs into critical applications such as medical devices, assistive technologies, and collaborative robotics, where their inherent compliance and adaptability offer significant advantages over traditional actuation methods.
Market Demand Analysis for Soft Robotics Applications
The global market for soft robotics applications is experiencing significant growth, driven by increasing demand across multiple sectors. The soft pneumatic actuators market, valued at approximately $1.2 billion in 2022, is projected to reach $3.5 billion by 2028, representing a compound annual growth rate of 19.6%. This growth is primarily fueled by the inherent advantages of soft pneumatic actuators, including their flexibility, safety in human interaction, and adaptability to irregular surfaces.
Healthcare represents the largest market segment, accounting for nearly 35% of the total soft robotics market. Within healthcare, rehabilitation devices and surgical assistants utilizing soft pneumatic actuators are gaining substantial traction. The aging population in developed economies and increasing prevalence of mobility disorders have created a robust demand for assistive devices that can safely interact with human tissues without causing damage.
Manufacturing and logistics sectors follow closely, with approximately 28% market share. Companies are increasingly adopting soft robotic grippers and manipulators for handling delicate or irregularly shaped objects. The food processing industry, in particular, has shown keen interest in soft pneumatic systems that can handle fragile products without damage, addressing a long-standing challenge in automation.
Consumer electronics and wearable technology markets are emerging as promising growth areas, currently representing about 15% of the market but growing at over 25% annually. The demand for haptic feedback devices, adaptive wearables, and human-machine interfaces is driving innovation in miniaturized soft pneumatic actuators with precise control capabilities.
Regional analysis indicates North America leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (24%). However, the Asia-Pacific region is demonstrating the fastest growth rate at 23% annually, primarily due to rapid industrialization in China, Japan, and South Korea, coupled with increasing healthcare expenditure.
Market surveys indicate that end-users prioritize regulatory compliance (87%), operational safety (92%), and durability (78%) when selecting soft pneumatic actuator technologies. This highlights the critical importance of addressing regulatory considerations in product development strategies. Companies that can navigate the complex regulatory landscape while delivering innovative solutions stand to capture significant market share.
The market is also witnessing a shift toward integrated systems that combine soft pneumatic actuators with advanced sensing and control technologies. This trend is expected to accelerate as artificial intelligence and machine learning capabilities become more accessible, enabling more sophisticated and responsive soft robotic applications across industries.
Healthcare represents the largest market segment, accounting for nearly 35% of the total soft robotics market. Within healthcare, rehabilitation devices and surgical assistants utilizing soft pneumatic actuators are gaining substantial traction. The aging population in developed economies and increasing prevalence of mobility disorders have created a robust demand for assistive devices that can safely interact with human tissues without causing damage.
Manufacturing and logistics sectors follow closely, with approximately 28% market share. Companies are increasingly adopting soft robotic grippers and manipulators for handling delicate or irregularly shaped objects. The food processing industry, in particular, has shown keen interest in soft pneumatic systems that can handle fragile products without damage, addressing a long-standing challenge in automation.
Consumer electronics and wearable technology markets are emerging as promising growth areas, currently representing about 15% of the market but growing at over 25% annually. The demand for haptic feedback devices, adaptive wearables, and human-machine interfaces is driving innovation in miniaturized soft pneumatic actuators with precise control capabilities.
Regional analysis indicates North America leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (24%). However, the Asia-Pacific region is demonstrating the fastest growth rate at 23% annually, primarily due to rapid industrialization in China, Japan, and South Korea, coupled with increasing healthcare expenditure.
Market surveys indicate that end-users prioritize regulatory compliance (87%), operational safety (92%), and durability (78%) when selecting soft pneumatic actuator technologies. This highlights the critical importance of addressing regulatory considerations in product development strategies. Companies that can navigate the complex regulatory landscape while delivering innovative solutions stand to capture significant market share.
The market is also witnessing a shift toward integrated systems that combine soft pneumatic actuators with advanced sensing and control technologies. This trend is expected to accelerate as artificial intelligence and machine learning capabilities become more accessible, enabling more sophisticated and responsive soft robotic applications across industries.
Global Regulatory Landscape and Technical Challenges
The global regulatory landscape for soft pneumatic actuators (SPAs) presents a complex and evolving framework that varies significantly across regions. In the United States, the FDA has established specific guidelines for medical applications of SPAs, requiring extensive safety testing and biocompatibility assessments before approval. These regulations focus primarily on material safety, mechanical reliability, and failure mode analysis, creating substantial barriers for new market entrants.
The European Union employs the CE marking system through the Medical Device Regulation (MDR) and Machinery Directive, which demand comprehensive technical documentation and risk management procedures. Unlike the US approach, the EU regulatory framework places greater emphasis on post-market surveillance and real-world performance data, requiring manufacturers to implement robust tracking systems for deployed actuators.
In Asia, regulatory approaches diverge significantly. Japan's PMDA has developed specialized protocols for soft robotics technologies, while China's NMPA focuses on domestic production capabilities alongside safety considerations. This regional variation creates significant challenges for global manufacturers seeking multi-market deployment of SPA technologies.
A critical technical challenge in meeting these diverse regulatory requirements is the standardization of testing methodologies. Current testing protocols were largely developed for rigid actuators and fail to adequately address the unique characteristics of soft pneumatic systems, such as non-linear deformation, material fatigue under repeated inflation cycles, and long-term performance degradation.
Material compliance represents another significant hurdle. Many high-performance elastomers used in SPAs contain phthalates or other potentially harmful plasticizers that face increasing restrictions globally. The development of alternative materials that maintain desired mechanical properties while meeting biocompatibility standards remains an ongoing challenge.
Pressure containment safety standards also vary widely across jurisdictions, with some regions requiring redundant safety mechanisms while others focus on operational pressure limitations. This inconsistency complicates design processes and increases development costs for manufacturers targeting multiple markets.
The lack of harmonized international standards specifically addressing soft robotics technologies further exacerbates these challenges. While ISO/TC 299 has begun developing robotics standards that include flexible systems, these efforts remain in early stages and have not yet produced comprehensive guidelines for SPAs. This regulatory gap creates uncertainty for manufacturers and potentially slows innovation in the field.
The European Union employs the CE marking system through the Medical Device Regulation (MDR) and Machinery Directive, which demand comprehensive technical documentation and risk management procedures. Unlike the US approach, the EU regulatory framework places greater emphasis on post-market surveillance and real-world performance data, requiring manufacturers to implement robust tracking systems for deployed actuators.
In Asia, regulatory approaches diverge significantly. Japan's PMDA has developed specialized protocols for soft robotics technologies, while China's NMPA focuses on domestic production capabilities alongside safety considerations. This regional variation creates significant challenges for global manufacturers seeking multi-market deployment of SPA technologies.
A critical technical challenge in meeting these diverse regulatory requirements is the standardization of testing methodologies. Current testing protocols were largely developed for rigid actuators and fail to adequately address the unique characteristics of soft pneumatic systems, such as non-linear deformation, material fatigue under repeated inflation cycles, and long-term performance degradation.
Material compliance represents another significant hurdle. Many high-performance elastomers used in SPAs contain phthalates or other potentially harmful plasticizers that face increasing restrictions globally. The development of alternative materials that maintain desired mechanical properties while meeting biocompatibility standards remains an ongoing challenge.
Pressure containment safety standards also vary widely across jurisdictions, with some regions requiring redundant safety mechanisms while others focus on operational pressure limitations. This inconsistency complicates design processes and increases development costs for manufacturers targeting multiple markets.
The lack of harmonized international standards specifically addressing soft robotics technologies further exacerbates these challenges. While ISO/TC 299 has begun developing robotics standards that include flexible systems, these efforts remain in early stages and have not yet produced comprehensive guidelines for SPAs. This regulatory gap creates uncertainty for manufacturers and potentially slows innovation in the field.
Current Regulatory Compliance Solutions
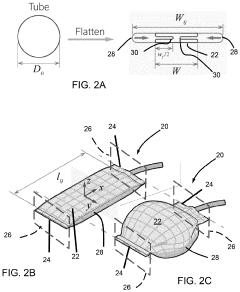
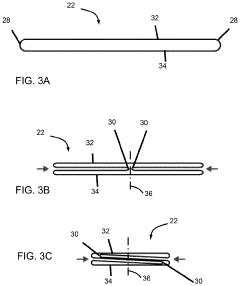
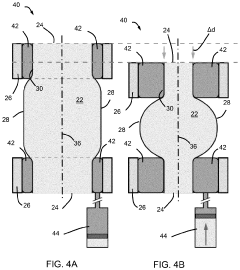
01 Design and fabrication of soft pneumatic actuators
Soft pneumatic actuators are designed and fabricated using flexible materials that can deform when pressurized with air. These actuators typically consist of chambers or channels within elastomeric materials that expand or contract when air pressure is applied. The fabrication methods include molding, 3D printing, and layered manufacturing techniques to create the internal air channels and external structure. These design approaches enable the development of actuators with specific motion profiles and force capabilities.- Design and structure of soft pneumatic actuators: Soft pneumatic actuators are designed with flexible materials that can deform and change shape when pressurized with air. These actuators typically consist of chambers or channels within elastomeric materials that expand when inflated, creating movement or force. The structural design elements include the arrangement of air chambers, material selection for optimal flexibility and durability, and integration of reinforcement components to control deformation patterns.
- Applications in robotics and automation: Soft pneumatic actuators are increasingly used in robotics and automation systems where traditional rigid actuators are unsuitable. These applications include soft robotic grippers for handling delicate objects, wearable assistive devices, medical robots for minimally invasive procedures, and biomimetic robots that replicate natural movements. The inherent compliance of these actuators makes them safer for human interaction and more adaptable to irregular environments and objects.
- Control systems and sensing integration: Advanced control systems are essential for precise operation of soft pneumatic actuators. These systems often incorporate pressure sensors, position feedback mechanisms, and sophisticated algorithms to manage air flow and pressure distribution. Integration of embedded sensors within the soft structure allows for closed-loop control and adaptive behavior. Some designs feature distributed sensing elements that can detect contact forces, deformation states, or environmental interactions.
- Manufacturing techniques and materials: Innovative manufacturing methods for soft pneumatic actuators include 3D printing, molding processes, and hybrid fabrication techniques. Materials commonly used include silicone elastomers, thermoplastic polyurethanes, and composite structures that combine flexible and rigid components. Recent advancements focus on developing materials with programmable stiffness, self-healing properties, or environmentally responsive characteristics to enhance actuator performance and longevity.
- Performance optimization and energy efficiency: Research in soft pneumatic actuators focuses on optimizing performance metrics such as force output, speed, range of motion, and energy efficiency. Strategies include geometric optimization of internal channels, multi-material designs that strategically combine rigid and flexible components, and development of valving systems that minimize air consumption. Some designs incorporate passive elements like springs or bistable mechanisms to reduce the energy required for actuation or to maintain positions with minimal pressure.
02 Applications in robotics and automation
Soft pneumatic actuators are widely used in robotics and automation systems where compliant, safe interaction with the environment or humans is required. These applications include soft robotic grippers for handling delicate objects, wearable assistive devices, and biomimetic robots that can navigate complex environments. The inherent compliance of soft actuators makes them particularly suitable for collaborative robots and systems operating in unstructured environments where traditional rigid actuators might cause damage or injury.Expand Specific Solutions03 Control systems and sensing integration
Advanced control systems are developed for soft pneumatic actuators to achieve precise movement and force control. These systems often incorporate pressure sensors, position sensors, and embedded electronics to provide feedback about the actuator's state. Machine learning algorithms and model-based control strategies are employed to compensate for the nonlinear behavior of soft materials. Integration of sensing elements within the soft structure enables closed-loop control and adaptive behavior in response to environmental interactions.Expand Specific Solutions04 Novel materials and composite structures
Research in soft pneumatic actuators focuses on developing novel materials and composite structures to enhance performance characteristics such as force output, speed, and durability. These include fiber-reinforced elastomers, variable stiffness materials, and multi-material composites that can change properties during actuation. Specialized coatings and embedded elements are used to improve air retention, reduce friction, and enable specific functionalities like electro-adhesion or thermal regulation.Expand Specific Solutions05 Energy efficiency and portable power systems
Improving energy efficiency is a key focus in soft pneumatic actuator development, addressing challenges related to air leakage and the energy requirements of pressurization systems. Innovations include optimized valve designs, regenerative pneumatic circuits, and compact, portable air supply systems. Research also explores alternative actuation methods that can be combined with pneumatics, such as electroactive polymers or phase-change materials, to reduce overall energy consumption while maintaining the benefits of soft actuation.Expand Specific Solutions
Key Industry Players and Competitive Landscape
The regulatory landscape for Soft Pneumatic Actuators (SPAs) is evolving as this technology transitions from early research to commercial applications. Currently, the market is in a growth phase with increasing adoption across medical, industrial, and wearable robotics sectors. Leading academic institutions including Harvard, MIT, and Zhejiang University are driving fundamental research, while companies like Oxipital AI, Bioliberty Ltd., and Toyota are commercializing applications. The regulatory framework remains fragmented, with medical applications facing stricter FDA/CE requirements compared to industrial uses. Key players are addressing safety standards, biocompatibility (for medical devices), and performance reliability. The technology's inherent safety advantages through compliance and low operating pressures may facilitate regulatory approval compared to traditional rigid actuators, though standardization efforts are still developing across global markets.
President & Fellows of Harvard College
Technical Solution: Harvard's Wyss Institute has pioneered soft pneumatic actuator regulatory frameworks through their comprehensive safety-by-design approach. Their technical solution integrates biocompatibility testing protocols specifically adapted for soft robotic materials that contact human tissue, establishing standardized mechanical failure testing procedures for pneumatic systems under various pressure conditions. Harvard researchers have developed a three-tier regulatory compliance system that addresses FDA medical device classifications (Class I-III) for soft pneumatic actuators in healthcare applications, with particular attention to skin-contact, minimally invasive, and implantable soft robotic systems. Their regulatory approach includes documented risk assessment methodologies that quantify potential failure modes and establish acceptable safety margins for pneumatic pressure systems in human-interactive applications.
Strengths: Established leadership in soft robotics regulatory frameworks with strong FDA relationships and comprehensive biocompatibility testing protocols. Weaknesses: Regulatory frameworks primarily focused on medical applications rather than industrial or consumer contexts, potentially limiting broader applicability.
The Regents of the University of California
Technical Solution: UC Berkeley and UC San Diego have jointly developed a comprehensive regulatory framework for soft pneumatic actuators through their robotics and bioengineering departments. Their technical solution includes a multi-tiered safety classification system specifically designed for wearable soft pneumatic devices that interface with human users. The UC system has established standardized testing protocols for pneumatic pressure containment that address both acute failure risks and long-term material degradation concerns. Their approach incorporates human factors engineering principles into regulatory considerations, with particular attention to user misuse scenarios and fail-safe design requirements. UC researchers have developed specialized regulatory guidance for soft pneumatic actuators in rehabilitation applications, working closely with FDA medical device regulators to establish appropriate safety margins and clinical testing requirements for therapeutic soft robotic systems. Their framework includes detailed documentation requirements for material selection, manufacturing processes, and quality control measures specific to soft pneumatic systems.
Strengths: Strong integration of human factors engineering into regulatory frameworks and established relationships with medical device regulators. Comprehensive documentation templates for regulatory submissions. Weaknesses: Regulatory approach sometimes emphasizes theoretical safety concerns over practical implementation considerations.
Critical Patents and Technical Standards Analysis
Pneumatic soft actuators with tunable force-displacement relation and methods and machines therefor
PatentPendingUS20230373082A1
Innovation
- A pneumatic soft actuator with an inflatable pouch featuring symmetrical folds at its ends, allowing for active modification of the end geometry through a branched tendon and spool mechanism, enabling adjustment of the force-strain relationship and range of motion without altering the pouch's composition or structure.
Safety and Risk Assessment Frameworks
The safety and risk assessment frameworks for soft pneumatic actuators (SPAs) represent a critical component in their regulatory pathway. Currently, these frameworks are primarily adapted from traditional robotics and medical device regulations, creating a hybrid approach that addresses the unique characteristics of SPAs. ISO 13482:2014 for personal care robots and IEC 60601 for medical electrical equipment provide foundational safety principles, though neither fully encompasses the compliant nature of pneumatic systems.
Risk assessment methodologies for SPAs typically follow a three-tiered structure: hazard identification, risk estimation, and risk evaluation. The compliant nature of these actuators introduces unique safety considerations not present in rigid systems, particularly regarding unpredictable deformation under varying loads and potential failure modes such as ballooning or rupture under pressure. These characteristics necessitate specialized testing protocols beyond conventional mechanical safety standards.
Failure Mode and Effects Analysis (FMEA) has emerged as a preferred methodology for SPA risk assessment, with particular emphasis on pressure-related failures, material degradation pathways, and environmental sensitivity. The FDA's guidance on benefit-risk determinations for medical devices provides additional structure when SPAs are intended for healthcare applications, requiring quantitative assessment of both probability and severity of harm.
Testing frameworks for SPAs typically include cyclic fatigue testing, pressure boundary testing, and environmental stress testing. These protocols must account for the non-linear material properties and viscoelastic behaviors characteristic of soft materials. The lack of standardized testing methodologies specifically for soft robotics remains a significant regulatory gap, with many manufacturers developing proprietary testing protocols that may not be universally accepted.
Recent collaborative efforts between industry stakeholders and regulatory bodies have begun addressing these gaps through the development of consensus standards. The International Organization for Standardization (ISO) has established working groups focused on soft robotics safety, while the ASTM F42 committee has initiated discussions on testing standards specific to compliant actuators. These emerging frameworks aim to balance innovation with safety assurance through performance-based rather than prescriptive requirements.
The integration of risk management throughout the product lifecycle represents another evolving aspect of SPA regulation, with increasing emphasis on post-market surveillance and real-world performance data collection to inform iterative safety improvements. This approach acknowledges the relatively limited historical safety data available for these novel technologies compared to traditional pneumatic systems.
Risk assessment methodologies for SPAs typically follow a three-tiered structure: hazard identification, risk estimation, and risk evaluation. The compliant nature of these actuators introduces unique safety considerations not present in rigid systems, particularly regarding unpredictable deformation under varying loads and potential failure modes such as ballooning or rupture under pressure. These characteristics necessitate specialized testing protocols beyond conventional mechanical safety standards.
Failure Mode and Effects Analysis (FMEA) has emerged as a preferred methodology for SPA risk assessment, with particular emphasis on pressure-related failures, material degradation pathways, and environmental sensitivity. The FDA's guidance on benefit-risk determinations for medical devices provides additional structure when SPAs are intended for healthcare applications, requiring quantitative assessment of both probability and severity of harm.
Testing frameworks for SPAs typically include cyclic fatigue testing, pressure boundary testing, and environmental stress testing. These protocols must account for the non-linear material properties and viscoelastic behaviors characteristic of soft materials. The lack of standardized testing methodologies specifically for soft robotics remains a significant regulatory gap, with many manufacturers developing proprietary testing protocols that may not be universally accepted.
Recent collaborative efforts between industry stakeholders and regulatory bodies have begun addressing these gaps through the development of consensus standards. The International Organization for Standardization (ISO) has established working groups focused on soft robotics safety, while the ASTM F42 committee has initiated discussions on testing standards specific to compliant actuators. These emerging frameworks aim to balance innovation with safety assurance through performance-based rather than prescriptive requirements.
The integration of risk management throughout the product lifecycle represents another evolving aspect of SPA regulation, with increasing emphasis on post-market surveillance and real-world performance data collection to inform iterative safety improvements. This approach acknowledges the relatively limited historical safety data available for these novel technologies compared to traditional pneumatic systems.
Cross-Industry Adoption Barriers and Opportunities
The adoption of soft pneumatic actuators across different industries faces several significant barriers despite their promising capabilities. Medical device regulations present a formidable challenge, as these actuators must meet stringent safety and reliability standards before implementation in healthcare settings. The FDA and similar international regulatory bodies require extensive clinical testing and validation, creating lengthy approval timelines that can delay market entry by years. This regulatory burden increases development costs substantially, making it difficult for smaller companies to enter the medical applications market.
Manufacturing industries face different adoption barriers, primarily centered around standardization issues. The lack of established industry standards for soft pneumatic actuators creates uncertainty regarding performance metrics, safety parameters, and quality control processes. This absence of standardization complicates integration with existing systems and hampers interoperability across platforms, limiting widespread industrial adoption.
Intellectual property landscapes present another significant barrier, with complex patent portfolios held by early innovators potentially restricting new market entrants. Companies must navigate this intricate IP environment carefully to avoid costly litigation, often necessitating extensive freedom-to-operate analyses before commercialization.
Despite these challenges, cross-industry opportunities for soft pneumatic actuators remain substantial. The growing emphasis on collaborative robots (cobots) presents a significant opportunity, as these actuators' inherent compliance makes them ideal for human-robot interaction scenarios. Their adaptability to various environments positions them well for deployment in emerging fields like assistive wearable technologies and rehabilitation devices.
The sustainability advantages of soft pneumatic actuators also create adoption opportunities. Their typically lower energy consumption compared to traditional rigid actuators aligns with increasing corporate sustainability initiatives and regulatory pressures for reduced environmental impact. Additionally, their potential for biodegradable material construction could address end-of-life disposal concerns in consumer electronics and temporary medical devices.
Cross-disciplinary collaboration between material scientists, mechanical engineers, and industry specialists is emerging as a catalyst for overcoming adoption barriers. These collaborative ecosystems are developing innovative solutions to technical challenges while simultaneously addressing regulatory concerns through early-stage compliance planning and standardization efforts.
Manufacturing industries face different adoption barriers, primarily centered around standardization issues. The lack of established industry standards for soft pneumatic actuators creates uncertainty regarding performance metrics, safety parameters, and quality control processes. This absence of standardization complicates integration with existing systems and hampers interoperability across platforms, limiting widespread industrial adoption.
Intellectual property landscapes present another significant barrier, with complex patent portfolios held by early innovators potentially restricting new market entrants. Companies must navigate this intricate IP environment carefully to avoid costly litigation, often necessitating extensive freedom-to-operate analyses before commercialization.
Despite these challenges, cross-industry opportunities for soft pneumatic actuators remain substantial. The growing emphasis on collaborative robots (cobots) presents a significant opportunity, as these actuators' inherent compliance makes them ideal for human-robot interaction scenarios. Their adaptability to various environments positions them well for deployment in emerging fields like assistive wearable technologies and rehabilitation devices.
The sustainability advantages of soft pneumatic actuators also create adoption opportunities. Their typically lower energy consumption compared to traditional rigid actuators aligns with increasing corporate sustainability initiatives and regulatory pressures for reduced environmental impact. Additionally, their potential for biodegradable material construction could address end-of-life disposal concerns in consumer electronics and temporary medical devices.
Cross-disciplinary collaboration between material scientists, mechanical engineers, and industry specialists is emerging as a catalyst for overcoming adoption barriers. These collaborative ecosystems are developing innovative solutions to technical challenges while simultaneously addressing regulatory concerns through early-stage compliance planning and standardization efforts.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!