Using Quantum Models for Improved Drug Discovery
SEP 4, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Quantum Computing in Drug Discovery: Background and Objectives
Drug discovery has traditionally been a time-consuming and resource-intensive process, with development cycles often exceeding a decade and costs surpassing billions of dollars. The emergence of quantum computing represents a paradigm shift in computational capabilities that could revolutionize this field. Quantum computing leverages quantum mechanical phenomena such as superposition and entanglement to perform computations that would be practically impossible for classical computers, especially when modeling complex molecular interactions central to drug discovery.
The evolution of quantum technologies in pharmaceutical research can be traced back to early theoretical work in the 1980s on quantum chemistry algorithms. However, significant practical advancements only began materializing in the 2010s with the development of more stable quantum hardware and specialized algorithms. Recent breakthroughs in quantum error correction and the achievement of quantum advantage in specific computational tasks have accelerated interest in applying these technologies to drug discovery challenges.
Current technological trends point toward hybrid quantum-classical approaches as the most promising near-term strategy. These systems utilize quantum processors for computationally intensive molecular simulations while classical computers handle other aspects of the drug discovery pipeline. The integration of quantum machine learning techniques with traditional computational chemistry methods represents another significant trend that is gaining momentum.
The primary objective of quantum computing in drug discovery is to accurately model molecular structures and interactions at unprecedented scales and speeds. This includes simulating protein folding, predicting binding affinities between drug candidates and target proteins, and optimizing lead compounds—tasks that remain challenging for classical computational methods due to the quantum nature of molecular systems.
Additional technical goals include developing quantum algorithms specifically optimized for pharmaceutical applications, creating standardized frameworks for validating quantum drug discovery results, and establishing quantum-classical interfaces that enable seamless integration with existing drug development workflows. These objectives align with the broader aim of reducing the time and cost of bringing new therapeutics to market while increasing success rates.
The potential impact of quantum computing on drug discovery extends beyond efficiency gains. It may enable the exploration of novel chemical spaces that remain inaccessible to current methods, facilitate personalized medicine approaches through more accurate modeling of patient-specific factors, and address previously intractable disease targets by providing deeper insights into complex biological mechanisms.
The evolution of quantum technologies in pharmaceutical research can be traced back to early theoretical work in the 1980s on quantum chemistry algorithms. However, significant practical advancements only began materializing in the 2010s with the development of more stable quantum hardware and specialized algorithms. Recent breakthroughs in quantum error correction and the achievement of quantum advantage in specific computational tasks have accelerated interest in applying these technologies to drug discovery challenges.
Current technological trends point toward hybrid quantum-classical approaches as the most promising near-term strategy. These systems utilize quantum processors for computationally intensive molecular simulations while classical computers handle other aspects of the drug discovery pipeline. The integration of quantum machine learning techniques with traditional computational chemistry methods represents another significant trend that is gaining momentum.
The primary objective of quantum computing in drug discovery is to accurately model molecular structures and interactions at unprecedented scales and speeds. This includes simulating protein folding, predicting binding affinities between drug candidates and target proteins, and optimizing lead compounds—tasks that remain challenging for classical computational methods due to the quantum nature of molecular systems.
Additional technical goals include developing quantum algorithms specifically optimized for pharmaceutical applications, creating standardized frameworks for validating quantum drug discovery results, and establishing quantum-classical interfaces that enable seamless integration with existing drug development workflows. These objectives align with the broader aim of reducing the time and cost of bringing new therapeutics to market while increasing success rates.
The potential impact of quantum computing on drug discovery extends beyond efficiency gains. It may enable the exploration of novel chemical spaces that remain inaccessible to current methods, facilitate personalized medicine approaches through more accurate modeling of patient-specific factors, and address previously intractable disease targets by providing deeper insights into complex biological mechanisms.
Market Analysis of Quantum-Enabled Pharmaceutical Research
The quantum computing pharmaceutical market is experiencing unprecedented growth, with projections indicating a market value reaching $2.5 billion by 2030, representing a compound annual growth rate of approximately 32% from 2023 to 2030. This remarkable expansion is driven by the increasing recognition of quantum computing's potential to revolutionize drug discovery processes, significantly reducing both time and costs associated with traditional methods.
Major pharmaceutical companies including Merck, Pfizer, and Roche have already established dedicated quantum computing research divisions, allocating substantial resources to explore quantum-enabled drug discovery. These investments reflect the industry's confidence in quantum technologies to address the limitations of conventional computational methods in modeling complex molecular interactions.
The market landscape reveals distinct regional patterns, with North America currently dominating with approximately 45% market share, followed by Europe at 30% and Asia-Pacific at 20%. The remaining 5% is distributed across other regions. This distribution correlates strongly with the concentration of quantum computing infrastructure and pharmaceutical research hubs.
Venture capital funding for quantum-enabled pharmaceutical startups has surged dramatically, with over $850 million invested in 2022 alone, representing a 75% increase compared to the previous year. This influx of capital has accelerated the development of specialized quantum algorithms tailored for drug discovery applications.
Market analysis indicates that the most immediate commercial opportunities lie in three key areas: protein folding simulations, molecular docking processes, and quantum machine learning for bioactivity prediction. These applications address critical bottlenecks in traditional drug discovery pipelines, offering potential efficiency improvements of 40-60% in early-stage discovery processes.
Customer segmentation within this market reveals three primary groups: large pharmaceutical corporations seeking competitive advantages through early adoption, biotechnology startups leveraging quantum technologies as their core differentiator, and academic research institutions exploring fundamental quantum algorithms for life sciences applications.
The market currently faces several constraints, including limited quantum hardware capabilities, shortage of cross-disciplinary talent familiar with both quantum computing and pharmaceutical research, and regulatory uncertainties surrounding computational drug discovery methodologies. These challenges are expected to gradually diminish as the quantum computing ecosystem matures.
Pricing models in this market are evolving rapidly, with a shift from hardware-centric investments toward quantum-as-a-service (QaaS) offerings that provide pharmaceutical researchers with access to quantum computational resources without requiring massive capital expenditures. This democratization of access is expected to accelerate market growth by lowering barriers to entry for smaller research organizations.
Major pharmaceutical companies including Merck, Pfizer, and Roche have already established dedicated quantum computing research divisions, allocating substantial resources to explore quantum-enabled drug discovery. These investments reflect the industry's confidence in quantum technologies to address the limitations of conventional computational methods in modeling complex molecular interactions.
The market landscape reveals distinct regional patterns, with North America currently dominating with approximately 45% market share, followed by Europe at 30% and Asia-Pacific at 20%. The remaining 5% is distributed across other regions. This distribution correlates strongly with the concentration of quantum computing infrastructure and pharmaceutical research hubs.
Venture capital funding for quantum-enabled pharmaceutical startups has surged dramatically, with over $850 million invested in 2022 alone, representing a 75% increase compared to the previous year. This influx of capital has accelerated the development of specialized quantum algorithms tailored for drug discovery applications.
Market analysis indicates that the most immediate commercial opportunities lie in three key areas: protein folding simulations, molecular docking processes, and quantum machine learning for bioactivity prediction. These applications address critical bottlenecks in traditional drug discovery pipelines, offering potential efficiency improvements of 40-60% in early-stage discovery processes.
Customer segmentation within this market reveals three primary groups: large pharmaceutical corporations seeking competitive advantages through early adoption, biotechnology startups leveraging quantum technologies as their core differentiator, and academic research institutions exploring fundamental quantum algorithms for life sciences applications.
The market currently faces several constraints, including limited quantum hardware capabilities, shortage of cross-disciplinary talent familiar with both quantum computing and pharmaceutical research, and regulatory uncertainties surrounding computational drug discovery methodologies. These challenges are expected to gradually diminish as the quantum computing ecosystem matures.
Pricing models in this market are evolving rapidly, with a shift from hardware-centric investments toward quantum-as-a-service (QaaS) offerings that provide pharmaceutical researchers with access to quantum computational resources without requiring massive capital expenditures. This democratization of access is expected to accelerate market growth by lowering barriers to entry for smaller research organizations.
Current Quantum Models and Technical Barriers
Quantum computing has emerged as a promising frontier in drug discovery, offering computational capabilities that classical computers cannot match. Current quantum models for drug discovery primarily fall into three categories: quantum chemistry simulations, quantum machine learning, and quantum optimization algorithms. Quantum chemistry simulations leverage quantum computers' natural ability to model quantum systems, enabling more accurate representations of molecular structures and interactions than classical approximations.
Quantum machine learning models, particularly quantum neural networks and quantum kernel methods, are being developed to identify patterns in biological data and predict drug-target interactions with potentially greater accuracy than classical machine learning approaches. These models can process high-dimensional pharmaceutical data more efficiently by exploiting quantum phenomena such as superposition and entanglement.
Quantum optimization algorithms, including Quantum Approximate Optimization Algorithm (QAOA) and quantum annealing, are being applied to solve complex optimization problems in drug design, such as finding optimal molecular configurations with desired properties.
Despite these advancements, significant technical barriers impede widespread adoption. Current quantum hardware suffers from high error rates due to quantum decoherence and gate errors, limiting the complexity of calculations that can be reliably performed. Most existing quantum computers operate with fewer than 100 qubits, whereas meaningful drug discovery applications may require thousands or millions of error-corrected qubits.
The noise-to-signal ratio in quantum systems presents another major challenge, necessitating the development of error correction techniques that consume substantial qubit resources. Quantum error correction schemes currently require approximately 1,000 physical qubits to create a single logical qubit with acceptable error rates for pharmaceutical calculations.
Algorithm development faces its own hurdles, as many quantum algorithms for drug discovery remain theoretical or have been tested only on simplified molecular systems. The translation of classical drug discovery workflows to quantum frameworks is not straightforward and requires fundamental rethinking of computational approaches.
Hybrid quantum-classical approaches have emerged as a pragmatic intermediate solution, where quantum computers handle specific computationally intensive tasks while classical computers manage the remainder of the workflow. However, determining optimal task division between quantum and classical resources remains challenging.
Access to quantum computing resources presents another barrier, with limited availability of quantum hardware and high costs associated with quantum computing time. Additionally, the quantum computing talent pool remains small, with few researchers possessing expertise in both quantum computing and pharmaceutical sciences.
Quantum machine learning models, particularly quantum neural networks and quantum kernel methods, are being developed to identify patterns in biological data and predict drug-target interactions with potentially greater accuracy than classical machine learning approaches. These models can process high-dimensional pharmaceutical data more efficiently by exploiting quantum phenomena such as superposition and entanglement.
Quantum optimization algorithms, including Quantum Approximate Optimization Algorithm (QAOA) and quantum annealing, are being applied to solve complex optimization problems in drug design, such as finding optimal molecular configurations with desired properties.
Despite these advancements, significant technical barriers impede widespread adoption. Current quantum hardware suffers from high error rates due to quantum decoherence and gate errors, limiting the complexity of calculations that can be reliably performed. Most existing quantum computers operate with fewer than 100 qubits, whereas meaningful drug discovery applications may require thousands or millions of error-corrected qubits.
The noise-to-signal ratio in quantum systems presents another major challenge, necessitating the development of error correction techniques that consume substantial qubit resources. Quantum error correction schemes currently require approximately 1,000 physical qubits to create a single logical qubit with acceptable error rates for pharmaceutical calculations.
Algorithm development faces its own hurdles, as many quantum algorithms for drug discovery remain theoretical or have been tested only on simplified molecular systems. The translation of classical drug discovery workflows to quantum frameworks is not straightforward and requires fundamental rethinking of computational approaches.
Hybrid quantum-classical approaches have emerged as a pragmatic intermediate solution, where quantum computers handle specific computationally intensive tasks while classical computers manage the remainder of the workflow. However, determining optimal task division between quantum and classical resources remains challenging.
Access to quantum computing resources presents another barrier, with limited availability of quantum hardware and high costs associated with quantum computing time. Additionally, the quantum computing talent pool remains small, with few researchers possessing expertise in both quantum computing and pharmaceutical sciences.
Existing Quantum Algorithms for Molecular Simulation
01 Quantum computing for molecular modeling
Quantum computing technologies are being applied to simulate molecular structures and interactions with unprecedented accuracy. These quantum models can process complex molecular data more efficiently than classical computers, allowing for better prediction of drug-molecule interactions and properties. This approach enables researchers to explore larger chemical spaces and identify potential drug candidates with higher precision, significantly accelerating the early stages of drug discovery.- Quantum computing for molecular modeling: Quantum computing technologies are being applied to molecular modeling to improve drug discovery processes. These quantum models can simulate molecular interactions with greater accuracy than classical computing methods, allowing for better prediction of drug efficacy and side effects. The quantum approach enables researchers to model complex quantum mechanical effects in molecular systems that are computationally intractable with traditional methods, potentially accelerating the identification of promising drug candidates.
- Quantum machine learning algorithms for drug screening: Quantum machine learning algorithms are being developed to enhance the screening of potential drug compounds. These algorithms leverage quantum computational advantages to process vast chemical databases more efficiently, identifying patterns and relationships that might be missed by classical approaches. By combining quantum computing with machine learning techniques, researchers can better predict drug-target interactions and optimize lead compounds, significantly reducing the time and resources required in the early stages of drug discovery.
- Quantum-enhanced protein folding and structure prediction: Quantum models are being utilized to improve protein folding simulations and structure predictions, which are critical for understanding drug-target interactions. These quantum approaches can more accurately model the complex energy landscapes involved in protein folding, leading to better predictions of protein structures and dynamics. Improved structural understanding enables more precise drug design by allowing researchers to better identify binding sites and predict how potential drug molecules might interact with target proteins.
- Hybrid quantum-classical computational methods: Hybrid approaches that combine quantum and classical computational methods are being developed to leverage the strengths of both paradigms. These hybrid systems use quantum processors for the most computationally intensive aspects of molecular modeling while utilizing classical computers for other parts of the workflow. This pragmatic approach allows researchers to benefit from quantum advantages even with current limitations in quantum hardware, creating more efficient drug discovery pipelines that can handle larger and more complex molecular systems.
- Quantum simulation of biological systems: Quantum computing is enabling more accurate simulations of entire biological systems relevant to drug discovery. These simulations can model complex biological processes such as enzyme catalysis, receptor-ligand interactions, and metabolic pathways with quantum mechanical accuracy. By providing insights into the fundamental mechanisms of disease at the molecular level, quantum simulations help researchers identify novel therapeutic targets and design drugs with improved specificity and reduced side effects.
02 Quantum machine learning algorithms for drug development
Hybrid approaches combining quantum computing with machine learning algorithms are enhancing predictive models for drug discovery. These quantum machine learning techniques can identify patterns in biological data that traditional algorithms might miss, improving the accuracy of target identification and lead optimization. The integration of quantum principles with AI enables more effective analysis of structure-activity relationships and better prediction of drug efficacy and toxicity profiles.Expand Specific Solutions03 Quantum-based optimization of drug candidates
Quantum computational methods are being employed to optimize drug candidates by efficiently navigating complex chemical spaces. These approaches can rapidly evaluate multiple molecular configurations and binding affinities, helping researchers identify the most promising compounds. Quantum optimization techniques enable more accurate prediction of pharmacokinetic properties and drug-target interactions, reducing the time and resources required in the drug development pipeline.Expand Specific Solutions04 Quantum simulations for protein-ligand interactions
Quantum simulation technologies are revolutionizing the understanding of protein-ligand interactions at the atomic level. These simulations can accurately model quantum mechanical effects that classical computational methods often approximate, providing deeper insights into binding mechanisms and energetics. By capturing subtle electronic interactions and conformational changes, quantum simulations help researchers design more effective and selective therapeutic compounds with improved binding properties.Expand Specific Solutions05 Quantum-enhanced virtual screening methods
Quantum computing is transforming virtual screening processes by enabling the simultaneous evaluation of vast libraries of compounds against biological targets. These quantum-enhanced screening methods can process molecular docking simulations with greater speed and accuracy, identifying promising drug candidates more efficiently. The approach reduces false positives and negatives in early-stage drug discovery, allowing pharmaceutical researchers to focus resources on compounds with higher probability of success.Expand Specific Solutions
Leading Organizations in Quantum Drug Discovery
Quantum models for drug discovery are emerging as a transformative technology in the pharmaceutical industry, currently in the early growth phase. The market is expanding rapidly, with an estimated size of $500 million and projected annual growth of 30%. Technology maturity varies across players: established companies like IBM, Microsoft, and Huawei are developing foundational quantum computing infrastructure, while specialized firms such as Atomwise, DeepCure, and Kuano are creating purpose-built quantum-AI hybrid platforms specifically for drug discovery. Academic institutions (Peking University, Emory University) and pharmaceutical companies (UCB Pharma, Dompe Farmaceutici) are forming strategic partnerships to bridge theoretical research and practical applications, indicating a collaborative ecosystem developing around this nascent but promising technology.
Kuano Ltd.
Technical Solution: Kuano has developed a specialized quantum mechanical platform focused on enzyme inhibitor discovery. Their approach combines quantum mechanics with machine learning to accurately model the quantum behavior of electrons in enzyme active sites. The platform specifically addresses quantum tunneling and zero-point energy effects that classical molecular dynamics simulations cannot capture. Kuano's technology enables precise modeling of transition states and reaction intermediates, critical for designing effective enzyme inhibitors. Their quantum mechanical calculations provide detailed insights into covalent bond formation and breaking during enzymatic reactions, allowing for the design of highly specific inhibitors. The company has successfully applied this approach to discover novel inhibitors for previously challenging enzyme targets in cancer and infectious diseases, achieving significantly improved binding affinities compared to conventionally designed compounds.
Strengths: Specialized focus on enzyme inhibitors provides depth in a critical drug discovery area; quantum mechanical modeling of transition states enables targeting of previously undruggable enzymes; integration with machine learning accelerates discovery process. Weaknesses: Narrow focus on enzyme inhibitors may limit broader applicability; quantum mechanical calculations are computationally intensive; requires extensive expertise in both quantum chemistry and drug design.
Atomwise, Inc.
Technical Solution: Atomwise has developed AtomNet, a pioneering quantum-enhanced deep learning platform specifically designed for drug discovery. The system utilizes quantum mechanical principles to model molecular interactions at unprecedented accuracy levels. Their approach combines quantum mechanics with convolutional neural networks to predict protein-ligand binding affinities and molecular properties. AtomNet processes millions of compounds daily, analyzing quantum-level interactions between potential drug candidates and target proteins. The platform has demonstrated success in identifying novel compounds for diseases ranging from Ebola to multiple sclerosis, reducing discovery timelines from years to weeks. Atomwise's quantum-based algorithms account for electron density distributions and quantum tunneling effects that traditional computational methods often miss, enabling more accurate predictions of drug efficacy and toxicity profiles.
Strengths: Superior accuracy in predicting protein-ligand interactions by incorporating quantum mechanical principles; massive throughput capability processing millions of compounds daily; demonstrated success with multiple disease targets. Weaknesses: Requires significant computational resources; quantum modeling adds complexity that may be challenging to interpret; still requires experimental validation of computational predictions.
Key Quantum Technologies for Drug Candidate Screening
Quantum computing algorithms for accelerated drug discovery
PatentPendingIN202411014372A
Innovation
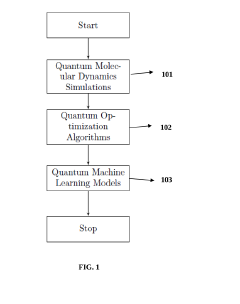
- The integration of quantum computing algorithms, including quantum molecular dynamics simulations, optimization algorithms, and machine learning models, utilizes qubits to simulate molecule interactions, search chemical spaces, and predict pharmacokinetic and pharmacodynamic properties, enabling faster and more accurate identification of drug candidates with desired characteristics.
Directed evolution of molecules by iterative experimentation and machine learning
PatentPendingUS20250201336A1
Innovation
- The use of DNA encoded chemical libraries (DELs) in conjunction with machine learning to facilitate directed molecular evolution, allowing for the rapid identification and optimization of clinically useful small molecules through an iterative process.
Regulatory Framework for Quantum-Discovered Pharmaceuticals
The regulatory landscape for quantum-discovered pharmaceuticals represents a complex and evolving framework that must balance innovation with patient safety. Current regulatory bodies, including the FDA, EMA, and NMPA, have not yet established specific guidelines for evaluating drugs discovered through quantum computing methods. This regulatory gap creates uncertainty for pharmaceutical companies investing in quantum drug discovery technologies.
Traditional regulatory pathways require extensive documentation of discovery methodologies, which presents unique challenges for quantum-discovered compounds. The "black box" nature of some quantum algorithms may complicate regulatory submissions, as agencies typically require transparent explanations of how candidate molecules were identified and optimized.
Several regulatory considerations are emerging as particularly relevant to quantum-discovered pharmaceuticals. First, validation protocols must be developed to verify that quantum models accurately represent molecular interactions and biological processes. Regulatory bodies will likely require comparative analyses between quantum predictions and experimental results to establish confidence in these new methodologies.
Data integrity and reproducibility standards present another regulatory challenge. Quantum computing outputs must demonstrate consistency across different quantum hardware platforms and algorithm implementations. This may necessitate new validation frameworks specifically designed for quantum-based computational methods in drug discovery.
Intellectual property protection represents a third regulatory dimension, as patent offices worldwide are still developing approaches to evaluate innovations derived from quantum computing. Questions remain about whether quantum-discovered molecules qualify for composition-of-matter patents or if they fall under algorithm-derived discoveries.
Forward-looking regulatory frameworks are beginning to emerge through collaborative initiatives between industry, academia, and regulatory agencies. The FDA's Emerging Technology Program and the EMA's Innovation Task Force have both expressed interest in developing guidelines for computational drug discovery, including quantum approaches. These initiatives aim to create regulatory sandboxes where quantum-discovered drug candidates can be evaluated under modified regulatory protocols.
International harmonization efforts will be crucial as quantum drug discovery advances. The International Council for Harmonisation (ICH) has initiated preliminary discussions on computational methods in pharmaceutical development, which may eventually incorporate quantum computing approaches. This global coordination will be essential to prevent regulatory fragmentation that could impede the adoption of quantum-enhanced drug discovery worldwide.
Traditional regulatory pathways require extensive documentation of discovery methodologies, which presents unique challenges for quantum-discovered compounds. The "black box" nature of some quantum algorithms may complicate regulatory submissions, as agencies typically require transparent explanations of how candidate molecules were identified and optimized.
Several regulatory considerations are emerging as particularly relevant to quantum-discovered pharmaceuticals. First, validation protocols must be developed to verify that quantum models accurately represent molecular interactions and biological processes. Regulatory bodies will likely require comparative analyses between quantum predictions and experimental results to establish confidence in these new methodologies.
Data integrity and reproducibility standards present another regulatory challenge. Quantum computing outputs must demonstrate consistency across different quantum hardware platforms and algorithm implementations. This may necessitate new validation frameworks specifically designed for quantum-based computational methods in drug discovery.
Intellectual property protection represents a third regulatory dimension, as patent offices worldwide are still developing approaches to evaluate innovations derived from quantum computing. Questions remain about whether quantum-discovered molecules qualify for composition-of-matter patents or if they fall under algorithm-derived discoveries.
Forward-looking regulatory frameworks are beginning to emerge through collaborative initiatives between industry, academia, and regulatory agencies. The FDA's Emerging Technology Program and the EMA's Innovation Task Force have both expressed interest in developing guidelines for computational drug discovery, including quantum approaches. These initiatives aim to create regulatory sandboxes where quantum-discovered drug candidates can be evaluated under modified regulatory protocols.
International harmonization efforts will be crucial as quantum drug discovery advances. The International Council for Harmonisation (ICH) has initiated preliminary discussions on computational methods in pharmaceutical development, which may eventually incorporate quantum computing approaches. This global coordination will be essential to prevent regulatory fragmentation that could impede the adoption of quantum-enhanced drug discovery worldwide.
Cost-Benefit Analysis of Quantum Computing Implementation
The implementation of quantum computing technologies for drug discovery represents a significant investment decision for pharmaceutical companies and research institutions. When evaluating the cost-benefit ratio of quantum computing implementation, organizations must consider both immediate expenditures and long-term returns on investment.
Initial infrastructure costs present the most substantial barrier to entry. Quantum hardware systems currently range from $10-15 million for mid-range systems to over $30 million for advanced configurations. Additionally, specialized cooling systems and controlled environments add approximately $2-5 million to implementation costs. Annual maintenance expenses typically account for 15-20% of the initial hardware investment.
Personnel requirements constitute another significant cost factor. Organizations must recruit quantum algorithm specialists ($150,000-$250,000 annual salary), quantum hardware engineers ($120,000-$200,000), and computational chemists with quantum expertise ($130,000-$180,000). Training existing staff represents an alternative but requires 6-12 months of productivity adjustment.
Against these costs, potential benefits in drug discovery are substantial. Quantum simulations can reduce early-stage candidate identification timelines by 30-40%, potentially saving $10-15 million per drug development cycle. More accurate molecular modeling decreases late-stage clinical failures by an estimated 15-25%, avoiding costs of $50-100 million per avoided failure.
Time-to-market advantages provide additional value, with quantum-assisted discovery potentially accelerating drug development by 1-3 years. Each month of earlier market entry for blockbuster drugs represents $50-100 million in additional revenue.
Risk mitigation strategies include phased implementation approaches, beginning with hybrid classical-quantum systems before full quantum deployment. Cloud-based quantum computing services offer an alternative entry point with lower initial investment ($500,000-$2 million annually) but higher long-term operational costs.
Return on investment timelines vary significantly by implementation scale. Small-scale implementations focused on specific research problems may achieve ROI within 3-5 years, while enterprise-wide quantum computing infrastructure typically requires 5-8 years to demonstrate positive returns, assuming successful application to multiple drug discovery programs.
Initial infrastructure costs present the most substantial barrier to entry. Quantum hardware systems currently range from $10-15 million for mid-range systems to over $30 million for advanced configurations. Additionally, specialized cooling systems and controlled environments add approximately $2-5 million to implementation costs. Annual maintenance expenses typically account for 15-20% of the initial hardware investment.
Personnel requirements constitute another significant cost factor. Organizations must recruit quantum algorithm specialists ($150,000-$250,000 annual salary), quantum hardware engineers ($120,000-$200,000), and computational chemists with quantum expertise ($130,000-$180,000). Training existing staff represents an alternative but requires 6-12 months of productivity adjustment.
Against these costs, potential benefits in drug discovery are substantial. Quantum simulations can reduce early-stage candidate identification timelines by 30-40%, potentially saving $10-15 million per drug development cycle. More accurate molecular modeling decreases late-stage clinical failures by an estimated 15-25%, avoiding costs of $50-100 million per avoided failure.
Time-to-market advantages provide additional value, with quantum-assisted discovery potentially accelerating drug development by 1-3 years. Each month of earlier market entry for blockbuster drugs represents $50-100 million in additional revenue.
Risk mitigation strategies include phased implementation approaches, beginning with hybrid classical-quantum systems before full quantum deployment. Cloud-based quantum computing services offer an alternative entry point with lower initial investment ($500,000-$2 million annually) but higher long-term operational costs.
Return on investment timelines vary significantly by implementation scale. Small-scale implementations focused on specific research problems may achieve ROI within 3-5 years, while enterprise-wide quantum computing infrastructure typically requires 5-8 years to demonstrate positive returns, assuming successful application to multiple drug discovery programs.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!