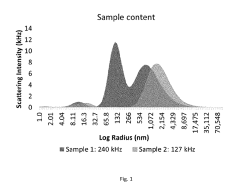
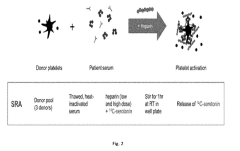
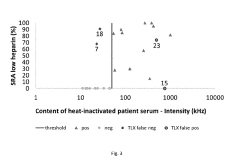
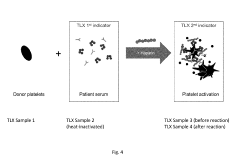
How to Predict Intrinsic Factor Changes Using Dynamic Light Scattering
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Dynamic Light Scattering Technology Background and Objectives
Dynamic Light Scattering (DLS) has evolved significantly since its inception in the 1960s, transforming from a specialized laboratory technique into an essential analytical tool across multiple industries. This non-invasive method measures the Brownian motion of particles in suspension to determine their size distribution, providing critical insights into molecular and particle behavior in various solutions.
The technology's development trajectory has been marked by significant advancements in laser technology, digital signal processing, and computational algorithms. Early DLS systems were limited by bulky equipment and complex data interpretation requirements, but modern instruments offer unprecedented sensitivity, automation, and user-friendly interfaces, enabling real-time analysis of dynamic molecular interactions.
Recent technological innovations have expanded DLS applications beyond traditional particle sizing to include more sophisticated analyses such as zeta potential measurements, molecular weight determination, and protein aggregation studies. The integration of multi-angle light scattering (MALS) and other complementary techniques has further enhanced the analytical power of DLS systems.
The primary objective of utilizing DLS for intrinsic factor prediction is to develop robust methodologies that can detect subtle changes in molecular properties before they manifest as macroscopic alterations. Intrinsic factors—such as protein stability, conformational changes, and interaction dynamics—are critical parameters in pharmaceutical formulations, food products, and biomaterial development.
Current research aims to establish correlations between DLS-measured parameters (hydrodynamic radius, polydispersity index, scattering intensity) and intrinsic property changes under various environmental conditions. This predictive capability would represent a significant advancement in quality control processes and formulation development.
The technological goal is to refine DLS methodologies to achieve higher sensitivity for detecting nascent molecular changes, improve data processing algorithms for more accurate interpretation of complex scattering patterns, and develop standardized protocols for specific applications across industries.
Additionally, there is growing interest in miniaturization and integration of DLS technology into continuous monitoring systems, potentially enabling real-time quality control in production environments. The development of machine learning approaches to analyze DLS data represents another frontier, with the potential to identify subtle patterns indicative of impending changes in intrinsic factors.
As regulatory requirements for product characterization become increasingly stringent, particularly in pharmaceutical and food industries, DLS technology is positioned to become an indispensable tool for predictive quality assessment and process optimization.
The technology's development trajectory has been marked by significant advancements in laser technology, digital signal processing, and computational algorithms. Early DLS systems were limited by bulky equipment and complex data interpretation requirements, but modern instruments offer unprecedented sensitivity, automation, and user-friendly interfaces, enabling real-time analysis of dynamic molecular interactions.
Recent technological innovations have expanded DLS applications beyond traditional particle sizing to include more sophisticated analyses such as zeta potential measurements, molecular weight determination, and protein aggregation studies. The integration of multi-angle light scattering (MALS) and other complementary techniques has further enhanced the analytical power of DLS systems.
The primary objective of utilizing DLS for intrinsic factor prediction is to develop robust methodologies that can detect subtle changes in molecular properties before they manifest as macroscopic alterations. Intrinsic factors—such as protein stability, conformational changes, and interaction dynamics—are critical parameters in pharmaceutical formulations, food products, and biomaterial development.
Current research aims to establish correlations between DLS-measured parameters (hydrodynamic radius, polydispersity index, scattering intensity) and intrinsic property changes under various environmental conditions. This predictive capability would represent a significant advancement in quality control processes and formulation development.
The technological goal is to refine DLS methodologies to achieve higher sensitivity for detecting nascent molecular changes, improve data processing algorithms for more accurate interpretation of complex scattering patterns, and develop standardized protocols for specific applications across industries.
Additionally, there is growing interest in miniaturization and integration of DLS technology into continuous monitoring systems, potentially enabling real-time quality control in production environments. The development of machine learning approaches to analyze DLS data represents another frontier, with the potential to identify subtle patterns indicative of impending changes in intrinsic factors.
As regulatory requirements for product characterization become increasingly stringent, particularly in pharmaceutical and food industries, DLS technology is positioned to become an indispensable tool for predictive quality assessment and process optimization.
Market Applications and Demand Analysis for DLS Prediction Systems
Dynamic Light Scattering (DLS) technology for predicting intrinsic factor changes has witnessed significant market growth across multiple industries. The pharmaceutical sector represents the largest market segment, with an estimated annual growth rate of 8.7% through 2025. Pharmaceutical companies increasingly rely on DLS systems to monitor protein stability, aggregation behavior, and formulation optimization during drug development processes, substantially reducing time-to-market for new therapeutics.
The biotechnology research segment demonstrates equally robust demand, particularly in academic and industrial research settings where understanding biomolecular interactions is critical. Research institutions value DLS prediction systems for their ability to provide real-time analysis of macromolecular changes under varying environmental conditions, enabling more efficient experimental design and data collection.
Food and beverage manufacturers have emerged as a rapidly expanding market for DLS technology, utilizing these systems for quality control and shelf-life prediction. The ability to detect subtle changes in emulsion stability and ingredient interactions addresses industry challenges related to product consistency and regulatory compliance.
The materials science sector shows growing adoption rates for DLS prediction systems in polymer research and nanoparticle characterization. Manufacturers of advanced materials leverage this technology to optimize production processes and ensure consistent product quality, particularly in applications requiring precise particle size distribution.
Healthcare diagnostics represents a promising emerging market, with clinical laboratories exploring DLS applications for early disease detection through analysis of biological fluid samples. The non-invasive nature of DLS testing offers significant advantages over traditional diagnostic methods, driving interest from major healthcare providers.
Market analysis indicates regional variations in adoption patterns, with North America currently leading in market share due to substantial R&D investments and presence of major pharmaceutical companies. However, Asia-Pacific markets show the highest growth potential, driven by expanding biotechnology sectors in China, Japan, and India.
Customer demand increasingly focuses on integrated systems offering automated sample handling, advanced data analytics, and cloud connectivity. End-users prioritize instruments with intuitive interfaces, minimal maintenance requirements, and compatibility with existing laboratory information management systems. This trend toward comprehensive solutions rather than standalone instruments shapes current product development strategies across the industry.
The biotechnology research segment demonstrates equally robust demand, particularly in academic and industrial research settings where understanding biomolecular interactions is critical. Research institutions value DLS prediction systems for their ability to provide real-time analysis of macromolecular changes under varying environmental conditions, enabling more efficient experimental design and data collection.
Food and beverage manufacturers have emerged as a rapidly expanding market for DLS technology, utilizing these systems for quality control and shelf-life prediction. The ability to detect subtle changes in emulsion stability and ingredient interactions addresses industry challenges related to product consistency and regulatory compliance.
The materials science sector shows growing adoption rates for DLS prediction systems in polymer research and nanoparticle characterization. Manufacturers of advanced materials leverage this technology to optimize production processes and ensure consistent product quality, particularly in applications requiring precise particle size distribution.
Healthcare diagnostics represents a promising emerging market, with clinical laboratories exploring DLS applications for early disease detection through analysis of biological fluid samples. The non-invasive nature of DLS testing offers significant advantages over traditional diagnostic methods, driving interest from major healthcare providers.
Market analysis indicates regional variations in adoption patterns, with North America currently leading in market share due to substantial R&D investments and presence of major pharmaceutical companies. However, Asia-Pacific markets show the highest growth potential, driven by expanding biotechnology sectors in China, Japan, and India.
Customer demand increasingly focuses on integrated systems offering automated sample handling, advanced data analytics, and cloud connectivity. End-users prioritize instruments with intuitive interfaces, minimal maintenance requirements, and compatibility with existing laboratory information management systems. This trend toward comprehensive solutions rather than standalone instruments shapes current product development strategies across the industry.
Current Capabilities and Technical Challenges in DLS Intrinsic Factor Detection
Dynamic Light Scattering (DLS) technology has evolved significantly in recent years, enabling researchers to detect and analyze intrinsic factors in various biological and chemical systems. Current DLS systems can measure particle size distributions ranging from 0.3 nm to 10 μm with high precision, allowing for the characterization of proteins, polymers, and nanoparticles in solution. Modern instruments achieve temporal resolution down to milliseconds, facilitating real-time monitoring of dynamic changes in intrinsic factors such as molecular weight, shape, and interaction parameters.
Advanced DLS platforms now incorporate multi-angle detection systems that significantly enhance the accuracy of measurements by collecting scattered light at various angles simultaneously. This capability has improved the resolution of polydisperse samples and complex biological mixtures. Additionally, temperature-controlled sample chambers with precision of ±0.1°C enable researchers to study temperature-dependent changes in intrinsic factors under highly controlled conditions.
Despite these advancements, several technical challenges persist in DLS-based intrinsic factor prediction. Sample concentration limitations represent a significant hurdle, as traditional DLS struggles with highly concentrated samples due to multiple scattering effects and with extremely dilute samples due to insufficient signal-to-noise ratios. This creates a narrow operational window that restricts certain applications, particularly in biological systems where sample quantities may be limited.
Signal interpretation complexity poses another major challenge. The mathematical models used to convert raw DLS data into meaningful intrinsic factor predictions rely on assumptions that may not hold for all sample types. Non-spherical particles, heterogeneous mixtures, and dynamic interactions between components can lead to misinterpretation of results and reduced prediction accuracy.
Environmental sensitivity remains problematic for DLS measurements. Factors such as dust contamination, temperature fluctuations, and mechanical vibrations can significantly impact data quality. Even minor environmental disturbances can introduce artifacts that compromise the reliability of intrinsic factor predictions, necessitating stringent laboratory controls.
The time-dependent nature of many biological and chemical systems presents additional challenges. Current DLS technologies struggle to capture rapid changes in intrinsic factors occurring on sub-millisecond timescales, limiting their application in studying fast kinetic processes. Furthermore, data processing algorithms often require significant computational resources for real-time analysis, creating bottlenecks in high-throughput applications.
Standardization across different DLS instruments and methodologies remains inadequate, leading to variability in results between laboratories and hindering the establishment of universal prediction models for intrinsic factor changes. This lack of standardization impedes broader adoption of DLS for critical applications in pharmaceutical development, quality control, and clinical diagnostics.
Advanced DLS platforms now incorporate multi-angle detection systems that significantly enhance the accuracy of measurements by collecting scattered light at various angles simultaneously. This capability has improved the resolution of polydisperse samples and complex biological mixtures. Additionally, temperature-controlled sample chambers with precision of ±0.1°C enable researchers to study temperature-dependent changes in intrinsic factors under highly controlled conditions.
Despite these advancements, several technical challenges persist in DLS-based intrinsic factor prediction. Sample concentration limitations represent a significant hurdle, as traditional DLS struggles with highly concentrated samples due to multiple scattering effects and with extremely dilute samples due to insufficient signal-to-noise ratios. This creates a narrow operational window that restricts certain applications, particularly in biological systems where sample quantities may be limited.
Signal interpretation complexity poses another major challenge. The mathematical models used to convert raw DLS data into meaningful intrinsic factor predictions rely on assumptions that may not hold for all sample types. Non-spherical particles, heterogeneous mixtures, and dynamic interactions between components can lead to misinterpretation of results and reduced prediction accuracy.
Environmental sensitivity remains problematic for DLS measurements. Factors such as dust contamination, temperature fluctuations, and mechanical vibrations can significantly impact data quality. Even minor environmental disturbances can introduce artifacts that compromise the reliability of intrinsic factor predictions, necessitating stringent laboratory controls.
The time-dependent nature of many biological and chemical systems presents additional challenges. Current DLS technologies struggle to capture rapid changes in intrinsic factors occurring on sub-millisecond timescales, limiting their application in studying fast kinetic processes. Furthermore, data processing algorithms often require significant computational resources for real-time analysis, creating bottlenecks in high-throughput applications.
Standardization across different DLS instruments and methodologies remains inadequate, leading to variability in results between laboratories and hindering the establishment of universal prediction models for intrinsic factor changes. This lack of standardization impedes broader adoption of DLS for critical applications in pharmaceutical development, quality control, and clinical diagnostics.
Existing Methodologies for Intrinsic Factor Prediction via DLS
01 Dynamic light scattering measurement techniques
Various techniques for dynamic light scattering measurements have been developed to analyze intrinsic factor changes in particles or molecules. These techniques involve specialized equipment and methodologies for detecting scattered light from particles in solution, allowing for the determination of particle size, molecular weight, and other physical properties. The measurements can be performed at different angles and with various light sources to enhance accuracy and sensitivity.- Dynamic light scattering measurement techniques: Dynamic light scattering (DLS) techniques are used to measure particle size, molecular weight, and other physical properties of particles in solution. These techniques involve analyzing the fluctuations in scattered light intensity caused by Brownian motion of particles. Advanced measurement methodologies include multi-angle detection, correlation function analysis, and specialized algorithms to improve accuracy and resolution, particularly for polydisperse samples or samples with intrinsic factor variations.
- Intrinsic factor detection and analysis systems: Systems designed specifically for detecting and analyzing intrinsic factors that affect dynamic light scattering measurements. These systems incorporate specialized hardware and software components to isolate and quantify intrinsic factors such as temperature fluctuations, viscosity changes, and molecular interactions. The technology enables researchers to distinguish between actual particle characteristics and measurement artifacts caused by changing intrinsic factors in the sample environment.
- Data processing algorithms for DLS factor analysis: Advanced algorithms specifically developed for processing dynamic light scattering data to identify and compensate for intrinsic factor changes. These computational methods employ statistical analysis, machine learning techniques, and mathematical modeling to extract meaningful information from complex scattering patterns. The algorithms can detect subtle changes in intrinsic factors and adjust measurements accordingly, improving the reliability and reproducibility of DLS results.
- Temperature and environmental control systems for DLS: Specialized systems designed to control and monitor environmental factors that influence dynamic light scattering measurements. These systems regulate temperature, pressure, humidity, and other variables that can affect intrinsic factors in DLS experiments. By maintaining stable environmental conditions or implementing controlled changes, researchers can isolate the effects of specific intrinsic factors on scattering patterns and obtain more accurate and reproducible results.
- Sample preparation methods for DLS intrinsic factor control: Innovative methods for preparing samples to minimize unwanted variations in intrinsic factors during dynamic light scattering measurements. These techniques include specialized filtration processes, buffer optimization, stabilizing agents, and protocols for handling sensitive biological samples. Proper sample preparation ensures that measured changes in scattering patterns accurately reflect the properties of interest rather than artifacts from sample degradation or contamination that could alter intrinsic factors.
02 Data analysis algorithms for dynamic light scattering
Advanced algorithms and computational methods have been developed to analyze data obtained from dynamic light scattering experiments. These algorithms help in interpreting the correlation functions, extracting meaningful information about particle size distributions, and detecting changes in intrinsic factors. Machine learning and artificial intelligence approaches are increasingly being applied to improve the accuracy and reliability of dynamic light scattering data analysis.Expand Specific Solutions03 Applications in biological sample analysis
Dynamic light scattering is widely used for analyzing biological samples to detect changes in intrinsic factors such as protein conformation, aggregation, and interactions. This technique allows for the non-invasive characterization of biomolecules in their native state, providing valuable information about their structure and function. It is particularly useful for studying protein stability, quality control in biopharmaceuticals, and monitoring biological processes in real-time.Expand Specific Solutions04 Instrumentation advancements for enhanced sensitivity
Recent advancements in instrumentation have significantly improved the sensitivity and resolution of dynamic light scattering measurements. These innovations include novel detector designs, improved optical components, and integrated systems that combine multiple measurement techniques. Enhanced sensitivity allows for the detection of subtle changes in intrinsic factors, such as minor conformational changes in molecules or small variations in particle characteristics.Expand Specific Solutions05 Environmental and external factor influences
The effects of environmental and external factors on dynamic light scattering measurements have been extensively studied. Factors such as temperature, pH, ionic strength, and solvent composition can significantly influence the intrinsic properties of particles and molecules, affecting the dynamic light scattering results. Understanding and controlling these factors is crucial for obtaining reliable and reproducible measurements of intrinsic factor changes.Expand Specific Solutions
Leading Research Institutions and Commercial Entities in DLS Field
Dynamic Light Scattering (DLS) technology for predicting intrinsic factor changes is evolving rapidly, currently transitioning from early development to commercial application phase. The global market for this technology is expanding, driven by increasing demand in pharmaceutical, biotechnology, and materials science sectors. From a technical maturity perspective, industry leaders are advancing at different rates. FUJIFILM Corp. and Malvern Panalytical have established robust commercial platforms, while IBM and Shimadzu Corp. are leveraging their analytical expertise to develop more sophisticated algorithms. Academic institutions like California Institute of Technology and Huazhong University of Science & Technology are contributing fundamental research. Companies like EOS Srl and Formulaction SAS are developing specialized applications, focusing on niche markets with innovative approaches to enhance measurement precision and data interpretation capabilities.
FUJIFILM Corp.
Technical Solution: FUJIFILM has leveraged its expertise in imaging and materials science to develop innovative approaches for predicting intrinsic factor changes using enhanced DLS technology. Their system combines traditional DLS with proprietary image analysis algorithms derived from their photography division to achieve higher resolution particle characterization. FUJIFILM's DLS platform incorporates multi-wavelength light sources (405nm, 532nm, and 650nm) that enable differential scattering analysis, providing enhanced sensitivity to compositional changes in complex mixtures beyond what single-wavelength systems can detect. Their technology employs correlation function analysis with advanced mathematical transformations that can separate overlapping particle populations with size differences as small as 3-fold, compared to the typical 5-fold limitation of conventional DLS. FUJIFILM has developed specialized sample cells with controlled shear environments that can simulate processing conditions while performing DLS measurements, allowing prediction of how intrinsic factors will change during manufacturing processes. Their systems incorporate automated dilution series capabilities that can extrapolate behavior at different concentrations to predict intrinsic factor changes in highly concentrated formulations where direct measurement is challenging. FUJIFILM's software platform includes predictive stability models based on accelerated testing protocols that correlate early DLS measurements with long-term stability outcomes, validated across multiple pharmaceutical and photographic emulsion applications.
Strengths: Excellent image processing and data analysis capabilities derived from photography expertise; innovative multi-wavelength approach; strong intellectual property portfolio. Weaknesses: Relatively newer entrant to the dedicated DLS market compared to established analytical instrument companies; more limited application-specific reference databases; higher complexity in operation.
Otsuka Electronics Co., Ltd.
Technical Solution: Otsuka Electronics has developed specialized DLS technology focused on predicting intrinsic factor changes in biological and pharmaceutical samples. Their ELSZ-2000 series employs a unique electrophoretic light scattering approach that simultaneously measures particle size distribution and zeta potential—a critical intrinsic factor indicating colloidal stability. The system utilizes a patented flow cell design that minimizes sample perturbation during measurement, allowing researchers to detect subtle changes in molecular conformation that precede larger structural alterations. Otsuka's technology incorporates temperature-dependent DLS measurements with precise control from 0-90°C (±0.1°C), enabling the creation of thermal stability profiles that can predict denaturation points and aggregation onset temperatures in protein formulations. Their proprietary "Stability Prediction Algorithm" analyzes time-series DLS data to identify trend patterns that correlate with future stability issues, achieving predictive accuracy of approximately 80-90% for protein formulations stored for 6-12 months. The company has also developed specialized sample cells for measuring highly concentrated samples (up to 40% w/v) without dilution, preserving the native environment where intrinsic changes actually occur. Otsuka's systems feature automated time-course measurements that can run for extended periods (up to 30 days) to capture slow changes in intrinsic factors that might be missed in shorter studies.
Strengths: Excellent integration of zeta potential with DLS measurements; specialized expertise in biological sample analysis; robust temperature control systems. Weaknesses: More limited global presence compared to larger competitors; fewer integration options with other analytical techniques; software updates less frequent than industry leaders.
Key Algorithms and Signal Processing Techniques for DLS Data Analysis
Methods and Apparatus for Predicting and Confirming Drug-Induced Thrombocytopenia Through Particle Detection with Dynamic Light Scattering
PatentInactiveUS20190250088A1
Innovation
- Dynamic Light Scattering (DLS) techniques are used to measure all particles in a patient sample, including platelets, microparticles, and aggregates, to predict and confirm DIT by analyzing changes in particle size distribution before and after exposure to suspected drugs, providing a more comprehensive assessment than existing methods.
Multi-angle dynamic light scattering
PatentWO2019154882A1
Innovation
- A method involving a nested approach with non-linear and linear solvers to iteratively refine scaling factors and particle size distribution, incorporating noise modeling and vector regularization to improve accuracy and robustness, allowing for more reliable and precise determination of particle size distribution across a wider size range.
Validation Protocols and Measurement Accuracy Standards
To establish reliable predictions of intrinsic factor changes using Dynamic Light Scattering (DLS), robust validation protocols and measurement accuracy standards are essential. These protocols must encompass comprehensive calibration procedures using certified reference materials with known size distributions and scattering properties. Standard operating procedures should include regular verification of instrument performance using monodisperse latex beads or gold nanoparticles, with acceptance criteria of ±2% for size measurements and less than 10% coefficient of variation for polydispersity indices.
Temperature control represents a critical validation parameter, as fluctuations of even 0.5°C can significantly impact Brownian motion and consequently alter DLS measurements. Validation protocols should therefore mandate temperature stabilization periods of at least 15 minutes before measurements and continuous monitoring throughout data collection, with automatic flagging of results when temperature variations exceed ±0.2°C.
Sample preparation standardization is equally important for measurement accuracy. Protocols must specify filtration requirements (typically 0.22μm filters for protein solutions), acceptable concentration ranges (0.1-10 mg/mL for most biomolecules), and appropriate equilibration times. Validation should include assessment of measurement reproducibility across multiple sample preparations by different operators to establish robust method transfer parameters.
Statistical validation approaches must be implemented to ensure data quality. This includes minimum requirements for photon count rates (typically >100 kcps for adequate signal-to-noise ratio), correlation function quality metrics (intercept values >0.8), and baseline stability indicators. Multiple measurements (n≥3) should be performed with established criteria for outlier identification and rejection based on statistical significance testing.
Instrument-specific validation parameters must address laser stability (power fluctuations <1%), detector linearity across the dynamic range, and optical alignment verification. Cross-validation with orthogonal techniques such as nanoparticle tracking analysis or analytical ultracentrifugation provides additional confidence in measurement accuracy, with acceptance criteria of ±10% agreement between methods for mean particle size.
Long-term monitoring protocols should be established to track instrument performance over time using control charts with defined action and warning limits. This enables early detection of systematic drift or sudden changes in measurement accuracy. Regular proficiency testing through participation in interlaboratory comparison studies further ensures alignment with industry standards and identifies potential methodological improvements to enhance prediction reliability.
Temperature control represents a critical validation parameter, as fluctuations of even 0.5°C can significantly impact Brownian motion and consequently alter DLS measurements. Validation protocols should therefore mandate temperature stabilization periods of at least 15 minutes before measurements and continuous monitoring throughout data collection, with automatic flagging of results when temperature variations exceed ±0.2°C.
Sample preparation standardization is equally important for measurement accuracy. Protocols must specify filtration requirements (typically 0.22μm filters for protein solutions), acceptable concentration ranges (0.1-10 mg/mL for most biomolecules), and appropriate equilibration times. Validation should include assessment of measurement reproducibility across multiple sample preparations by different operators to establish robust method transfer parameters.
Statistical validation approaches must be implemented to ensure data quality. This includes minimum requirements for photon count rates (typically >100 kcps for adequate signal-to-noise ratio), correlation function quality metrics (intercept values >0.8), and baseline stability indicators. Multiple measurements (n≥3) should be performed with established criteria for outlier identification and rejection based on statistical significance testing.
Instrument-specific validation parameters must address laser stability (power fluctuations <1%), detector linearity across the dynamic range, and optical alignment verification. Cross-validation with orthogonal techniques such as nanoparticle tracking analysis or analytical ultracentrifugation provides additional confidence in measurement accuracy, with acceptance criteria of ±10% agreement between methods for mean particle size.
Long-term monitoring protocols should be established to track instrument performance over time using control charts with defined action and warning limits. This enables early detection of systematic drift or sudden changes in measurement accuracy. Regular proficiency testing through participation in interlaboratory comparison studies further ensures alignment with industry standards and identifies potential methodological improvements to enhance prediction reliability.
Integration Potential with Complementary Analytical Techniques
Dynamic Light Scattering (DLS) offers significant potential for integration with complementary analytical techniques, creating powerful hybrid approaches for comprehensive intrinsic factor analysis. When combined with Size Exclusion Chromatography (SEC), researchers can achieve enhanced resolution in protein aggregation studies, allowing for separation of components prior to DLS measurement and providing both size distribution and molecular weight information simultaneously.
Integration with Mass Spectrometry (MS) creates a particularly valuable combination, where DLS provides structural and conformational data while MS delivers precise molecular identification and compositional analysis. This synergy enables researchers to correlate size changes detected by DLS with specific molecular modifications identified through MS, offering deeper insights into intrinsic factor behavior under various conditions.
Circular Dichroism (CD) spectroscopy paired with DLS creates another powerful analytical approach, especially for protein-based intrinsic factors. While DLS monitors size and aggregation state changes, CD simultaneously tracks secondary structure alterations, establishing direct correlations between conformational shifts and hydrodynamic properties.
Nuclear Magnetic Resonance (NMR) spectroscopy complements DLS by providing atomic-level structural information. This combination proves especially valuable when investigating subtle conformational changes in intrinsic factors that might precede more significant aggregation events detectable by DLS alone.
Emerging microfluidic platforms represent the next frontier in integrated analysis, enabling real-time, multi-parameter measurements of intrinsic factors. These systems can incorporate DLS alongside other techniques within miniaturized flow cells, requiring minimal sample volumes while generating comprehensive datasets on size, structure, and interaction dynamics.
The integration of computational modeling with DLS data further enhances predictive capabilities. Machine learning algorithms can be trained on combined datasets from multiple analytical techniques to identify patterns and correlations that would be impossible to detect through any single method. This computational approach enables the development of predictive models that can anticipate intrinsic factor changes based on early indicators across multiple analytical dimensions.
For industrial applications, automated systems that combine DLS with complementary techniques in sequential or parallel workflows are becoming increasingly important. These integrated analytical platforms allow for high-throughput screening and quality control of intrinsic factors in pharmaceutical, biotechnology, and food science applications, significantly reducing analysis time while enhancing the depth and reliability of the results.
Integration with Mass Spectrometry (MS) creates a particularly valuable combination, where DLS provides structural and conformational data while MS delivers precise molecular identification and compositional analysis. This synergy enables researchers to correlate size changes detected by DLS with specific molecular modifications identified through MS, offering deeper insights into intrinsic factor behavior under various conditions.
Circular Dichroism (CD) spectroscopy paired with DLS creates another powerful analytical approach, especially for protein-based intrinsic factors. While DLS monitors size and aggregation state changes, CD simultaneously tracks secondary structure alterations, establishing direct correlations between conformational shifts and hydrodynamic properties.
Nuclear Magnetic Resonance (NMR) spectroscopy complements DLS by providing atomic-level structural information. This combination proves especially valuable when investigating subtle conformational changes in intrinsic factors that might precede more significant aggregation events detectable by DLS alone.
Emerging microfluidic platforms represent the next frontier in integrated analysis, enabling real-time, multi-parameter measurements of intrinsic factors. These systems can incorporate DLS alongside other techniques within miniaturized flow cells, requiring minimal sample volumes while generating comprehensive datasets on size, structure, and interaction dynamics.
The integration of computational modeling with DLS data further enhances predictive capabilities. Machine learning algorithms can be trained on combined datasets from multiple analytical techniques to identify patterns and correlations that would be impossible to detect through any single method. This computational approach enables the development of predictive models that can anticipate intrinsic factor changes based on early indicators across multiple analytical dimensions.
For industrial applications, automated systems that combine DLS with complementary techniques in sequential or parallel workflows are becoming increasingly important. These integrated analytical platforms allow for high-throughput screening and quality control of intrinsic factors in pharmaceutical, biotechnology, and food science applications, significantly reducing analysis time while enhancing the depth and reliability of the results.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!